Childhood pneumococcal vaccinations: what’s behind the new schedule?

Pneumococcal vaccines effectively prevent disease and are recommended in Australia for young children, older adults, Aboriginal and Torres Strait Islander people and others at risk. Updated September 2025 childhood guidance under the National Immunisation Program aims to optimise protection. It is important for clinicians to remain up to date with current recommendations.
- 20vPCV replaces 13vPCV in Australia’s National Immunisation Program for children starting vaccination after 1 September 2025.
- Expanded serotype coverage in 20vPCV is expected to reduce invasive pneumococcal disease in children, providing broader protection than 13vPCV.
- 23vPPV is no longer part of the childhood pneumococcal schedule because only a small proportion of invasive pneumococcal disease cases were caused by serotypes included in 23vPPV but not in 20vPCV.
- A four-dose schedule now applies nationally for Aboriginal and Torres Strait Islander children, improving equity and simplifying administration.
- Non-Indigenous children without a risk condition follow a three-dose (2+1) schedule at 2, 4 and 12 months of age.
- Catch-up schedules are available for children who have missed doses, tailored to age, risk factors, Indigenous status and jurisdiction.
Pneumococcal infection can lead to a range of diseases, including invasive pneumococcal disease (IPD), a severe form that can be life-threatening in children. Although rates of pneumococcal disease declined substantially during COVID-19 pandemic restrictions, they have since returned to pre-pandemic levels. In Australia, pneumococcal vaccination is recommended for young children, older adults, Aboriginal and Torres Strait Islander individuals and at-risk cohorts, and is funded for most of these groups under the National Immunisation Program (NIP).
In September 2025, the childhood pneumococcal vaccine recommendations were updated, with the 13-valent pneumococcal conjugate vaccine (13vPCV) replaced with the 20-valent pneumococcal conjugate vaccine (20vPCV). The 23-valent pneumococcal polysaccharide vaccine (23vPPV), previously offered to eligible Aboriginal and Torres Strait Islander children and children with risk conditions, is no longer part of the recommended schedule. This article describes the new pneumococcal vaccine program for children and the rationale behind the introduced changes.
Pneumococcal disease
Pneumococcal disease is caused by the encapsulated Gram-positive diplococcus Streptococcus pneumoniae. In 2019, it was responsible for an estimated 14% of all deaths in children under 5 years old globally.1 IPD is defined as the presence of S. pneumoniae in sterile body sites, including the blood and cerebrospinal, pleural, peritoneal and joint fluid.2 In children, S. pneumoniae resides in the nasopharynx, often causing asymptomatic carriage, which is a precursor to disease and an important factor in transmission.3 More than 100 distinct pneumococcal serotypes have been identified, based on the polysaccharide capsule of S. pneumoniae, a key virulence factor.4 Vaccines target the serotypes most commonly associated with disease.5
Noninvasive pneumococcal disease is more common and typically presents as localised mucosal infections such as acute otitis media, often seen in children.7 The most common adult presentation of IPD is bacteraemic pneumonia; however, most community-acquired pneumococcal pneumonia cases are noninvasive.8
Pneumococcal disease in Australia
IPD became nationally notifiable in Australia in 2001. In 2025, there were 2639 notifications of IPD, 14% of which were in children aged under 5 years and 37% in adults aged 65 years or older.9 High rates of IPD are seen in Aboriginal and Torres Strait Islander individuals.10 Globally, rates of IPD decreased during the COVID-19 pandemic, both in adults and children, mainly due to public health restrictions resulting in reduced circulation of S. pneumoniae and other respiratory infections. However, the easing of COVID-19 restrictions led to a resurgence of IPD to pre-pandemic levels, with broadly similar serotype epidemiology but increased severity of cases in children.
Pneumococcal vaccination program for Australian children
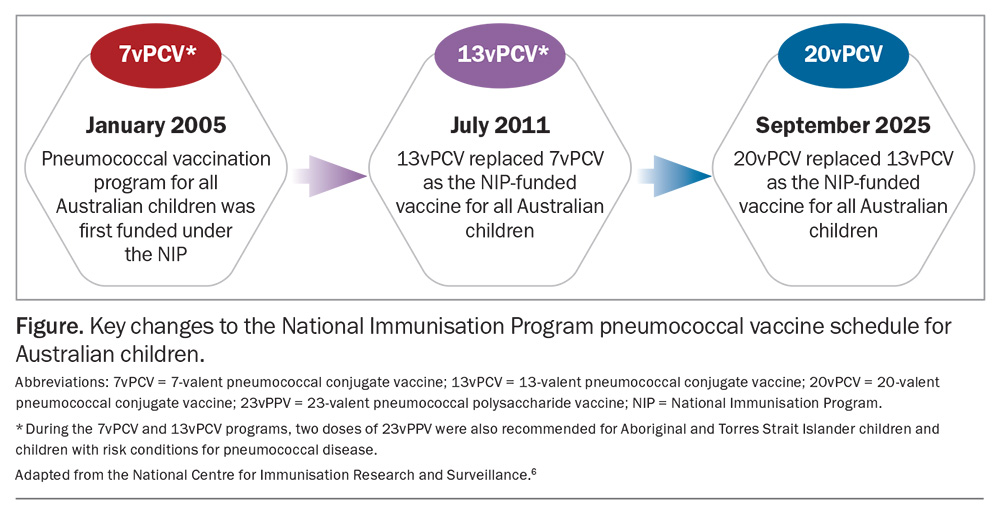
Pneumococcal vaccination for all Australian children was first funded under the NIP in 2005 with the 7-valent pneumococcal conjugate vaccine (7vPCV). In pneumococcal conjugate vaccines (PCVs), multiple selected pneumococcal polysaccharides are conjugated to one or more carrier proteins, converting the polysaccharide to a T cell-dependent antigen. This induces immune memory and results in a robust, high-quality immune response that is sufficient to prevent pneumococcal disease in infants and also reduces carriage of the vaccine serotypes.11,12
In 2011, 7vPCV was replaced by 13vPCV to cover an additional six serotypes that were causing the majority of the residual disease at the time. During both the 7vPCV and 13vPCV periods, additional doses of 23vPPV were also given to children with increased risk of pneumococcal disease to extend the number of serotypes they were protected against. However, pneumococcal polysaccharide vaccines (PPVs) generate antibodies against pneumococcal disease alone, without immune memory, producing relatively short-lived immunity. The 23vPPV has no effect on pneumococcal carriage and therefore does not elicit herd benefit.13
The latest PCV change in September 2025 was the replacement of 13vPCV with the new high valency 20vPCV on the NIP, providing protection against seven additional serotypes. In addition to 13vPCV and 20vPCV, 15-valent pneumococcal conjugate vaccine (15vPCV) and 21-valent pneumococcal conjugate vaccine (21vPCV) are also registered for use in Australia by the TGA.6 These higher‑valency PCVs are designed to address the dynamic serotype shifts that occur as vaccine‑induced selective pressure suppresses covered serotypes, enabling nonvaccine serotypes to become more prominent in residual disease.14 The key changes to the NIP pneumococcal vaccine schedule for Australian children are shown in the Figure and the risk conditions recommended for pneumococcal vaccination are presented in the Box.6,15
Changes to the childhood pneumococcal vaccine recommendations
Children who start their pneumococcal vaccination after 1 September 2025
20vPCV is the only vaccine recommended for children who start their pneumococcal vaccine schedule after 1 September 2025 (Flowchart).15 For non-Indigenous children, this will involve a three-dose (2+1) schedule of 20vPCV at 2, 4 and 12 months of age. For Aboriginal and Torres Strait Islander children and children aged under 12 months with a risk condition, this will involve a four-dose schedule of 20vPCV at 2, 4, 6 and 12 months of age. This schedule now includes Aboriginal and Torres Strait Islander children in all states and territories. It is more inclusive than the previous jurisdiction-based program and simplifies delivery for providers, particularly in border regions.
The schedule for Aboriginal and Torres Strait Islander children and children with a risk condition has been further simplified by the removal of 23vPPV from the program. This means children who receive at least one dose of 20vPCV in their schedule no longer require any 23vPPV doses. Children and infants can receive their doses of 20vPCV coadministered with other routine childhood vaccines.
Children who have already commenced their pneumococcal vaccination following previous recommendations
All children who commenced their 2+1 or 3+1 schedule before 1 September 2025 with 13vPCV (or 15vPCV) should complete it with 20vPCV. If at least one dose in this schedule is 20vPCV, then their schedule is complete and no additional doses of 20vPCV are required. The only exception may be children who were recommended a 2+1 schedule (i.e. all non-Indigenous children without risk conditions and Aboriginal and Torres Strait Islander children born before 1 March 2025 living in the Australian Capital Territory, New South Wales, Victoria or Tasmania) and who later develop a risk condition. These children are recommended an additional dose of PCV, and 20vPCV should be used.
If a child has already completed their schedule and they were previously recommended a dose of 23vPPV, then a dose of 20vPCV should replace this 23vPPV dose at the same schedule point. For example, if an Aboriginal child aged 3 years living in Queensland received four doses of 13vPCV, then at 4 years of age they should receive a dose of 20vPCV instead of the previously recommended 23vPPV. This child’s schedule is now complete, and no further doses are required. Another example is a child born with a risk condition now aged 8 years who received four doses of 13vPCV and a single dose of 23vPPV. At age 9 years, this child would have received a dose of 23vPPV according to previous recommendations but is now recommended a dose of 20vPCV instead.
For Aboriginal and Torres Strait Islander children living in the Australian Capital Territory, New South Wales, Victoria or Tasmania, only those born on or after 1 March 2025 will be eligible for the four-dose schedule of 20vPCV. Children born in these jurisdictions before 1 March 2025 were still recommended to receive the three-dose schedule and do not require a supplementary dose of 20vPCV if they have already completed the 2+1 PCV schedule.
Children who have missed their pneumococcal vaccinations
Children who have missed a due dose or doses of pneumococcal vaccine should follow a catch-up schedule. Their catch-up schedule will depend on when they were born, which state or territory they reside in, if they have a risk condition and if they identify as Aboriginal or Torres Strait Islander. There are two main resources in the Australian Immunisation Handbook that provide guidance on individualised catch-up schedules:15
- Pneumococcal vaccine catch-up tables: https://immunisationhandbook.health.gov.au/contents/catch-up-vaccination
- National Immunisation Catch-up Calculator: https://immunisationhandbook.health.gov.au/catch-up-calculator/calculator
Key considerations behind the changes to the childhood pneumococcal vaccination program
The key areas considered in the changes to the childhood pneumococcal vaccination program were the epidemiology of IPD in Australian children, vaccine characteristics from clinical trials, equity and feasibility.
Benefits of 20vPCV in broadening protection against more serotypes
When the current serotype epidemiology of IPD cases in Australia was examined, it was clear that significant benefit would be derived from the seven additional serotypes (8, 10A, 11A, 12F, 15B, 22F and 33F) in the 20vPCV compared with the 13vPCV. Data from mandatory case reporting captured in the National Notifiable Diseases Surveillance System showed that for 2023 to 2024, around a third of all IPD cases in children aged under 5 years were caused by the extra serotypes included in 20vPCV. In total, the serotypes in 20vPCV accounted for 63% of IPD in children aged under 5 years, based on 2023 and 2024 data.16
Overall, the main benefit of 20vPCV is its broader serotype coverage, which is expected to provide better protection than 13vPCV. Although efficacy data from clinical trials are not available, immunogenicity data indicate that 20vPCV is likely to provide good protection against pneumococcal disease caused by the additional serotypes it contains. In trials in which 20vPCV was compared with 13vPCV in children, immunogenicity after at least three doses was similar for all shared serotypes, except serotype 3. Notably, 13vPCV has not been associated with reductions in serotype 3 disease. These immunogenicity data are used to infer protection and for licensure of PCVs such as 20vPCV.
Negligible benefit of a 23vPPV dose for children who receive 20vPCV
The previous recommendation of two doses of 23vPPV for Aboriginal Torres Strait Islander children in certain jurisdictions and children with a risk condition was based on the considerable burden of IPD caused by serotypes in 23vPPV but not in 13vPCV. However, most of these additional serotypes responsible for disease in those children are contained in 20vPCV. Recent IPD epidemiological data showed that only a small number of IPD cases in children were caused by the serotypes contained in 23vPPV that were not included in 20vPCV (23v-non-20 serotypes: 2, 9N, 17F and 20).16 This small number meant that 23vPPV no longer provided sufficiently broader coverage beyond 20vPCV, making its continued use of limited value.
Comparable 20vPCV-type IPD burden supports national additional dose for Aboriginal and Torres Strait Islander children
When IPD data were reviewed for Aboriginal and Torres Strait Islander children, the incidence of 20vPCV-type IPD in jurisdictions that recommended the three-dose schedule for the previous 13vPCV program was sufficiently comparable with jurisdictions that recommended the four-dose schedule. This was particularly noted in the border regions of New South Wales–Queensland and Victoria–South Australia. Expanding the four-dose schedule to all jurisdictions improves equity and feasibility by establishing a single national schedule for Aboriginal and Torres Strait Islander children.
Adult pneumococcal vaccination program
The adult pneumococcal vaccination program is under review by the Australian Technical Advisory Group on Immunisation. Pneumococcal vaccines are recommended for all adults aged 70 years and over, Aboriginal and Torres Strait Islander adults aged 50 years and over, and adults with a risk condition aged 18 years and over (Box).15 A single dose of 13vPCV is funded on the NIP for all older adults from those threshold ages and adults with selected risk conditions.17 Aboriginal and Torres Strait Islander adults and adults with selected risk conditions are also still recommended and funded to receive two doses of 23vPPV, with the first dose 12 months after the PCV dose and the second dose at least five years after the first dose of 23vPPV. Further details of the current adult recommendations are available here: https://medicinetoday.com.au/mt/2022/august/regul-update/pneumococcal-vaccinations-in-australia-current-recommendations-and-beyond-update-online-april-2024.
The 15vPCV and 20vPCV are available for use in Australian adults but are not NIP-funded. The 21vPCV is also registered for use in adults in Australia but is not yet available. These vaccines cover the main serotypes that are causing residual IPD following several years of 13vPCV use, noting that some of those serotypes are more prevalent in adult disease compared with in children. PCVs with an even greater number of serotypes, some using novel vaccine platforms, are also in the pipeline, including a 25-valent PCV and a 31-valent PCV candidate.18
Conclusion
In September 2025, Australia’s childhood pneumococcal vaccination recommendations were updated, with key changes including replacement of 13vPCV with 20vPCV as the NIP-funded vaccine, removal of 23vPPV from the schedule and expansion of the four-dose schedule to include all Aboriginal and Torres Strait Islander children. The changes were primarily driven by the additional protection offered by the seven additional serotypes included in 20vPCV. Recommendations for use of pneumococcal vaccines continue to be based on vaccine characteristics and individuals’ IPD risk factors, including age, Aboriginal and Torres Strait Islander status, the presence of risk conditions and previous pneumococcal vaccination history. Understanding the changes and the rationale behind these new recommendations will support clinicians with implementation of pneumococcal vaccination recommendations and practices. MT
COMPETING INTERESTS: Associate Professor Williams and Dr Jayasinghe hold emerging leadership investigator grants from the National Health and Medical Research Council. Ms van Eldik and Dr Quinn: None.
ACKNOWLEDGEMENTS: The authors thank Dr Anna Bachlani, Senior Research Officer at the National Centre for Immunisation Research and Surveillance, for undertaking the analysis of IPD notifications included in this article.
References
1. World Health Organization. Pneumonia in children. Geneva: WHO; 2021. Available online at: https://www.who.int/news-room/fact-sheets/detail/pneumonia (accessed February 2026).
2. Tuomanen EI, Austrian R, Masure HR. Pathogenesis of pneumococcal infection. N Engl J Med 1995; 332: 1280-1284.
3. Jochems SP, Weiser JN, Malley R, Ferreira DM. The immunological mechanisms that control pneumococcal carriage. PLoS Pathog 2017; 13: e1006665.
4. Blacklock CB, Weinberger DM, Perniciaro S, Wyllie A. Streptococcus pneumoniae serotypes. New Haven: Yale School of Public Health; 2024. Available online at: https://pneumococcalcapsules.github.io/serotypes/ (accessed February 2026).
5. Ganaie F, Saad JS, McGee L, et al. A new pneumococcal capsule type, 10D, is the 100th serotype and has a large cps fragment from an oral streptococcus. mBio 2020; 11: e00937-20.
6. National Centre for Immunisation Research and Surveillance (NCIRS). Significant events in pneumococcal vaccination practice in Australia. Sydney: NCIRS; 2025. Available online at: https://ncirs.org.au/sites/default/files/2025-09/Pneumococcal-20history_September-2020-2025-update.pdf (accessed February 2026).
7. Eskola J, Kilpi T, Palmu A, et al. Efficacy of a pneumococcal conjugate vaccine against acute otitis media. N Engl J Med 2001; 344: 403-409.
8. Drijkoningen JJ, Rohde GG. Pneumococcal infection in adults: burden of disease. Clin Microbiol Infect 2014; 20(Suppl 5): 45-51.
9. Australian Centre for Disease Control. National Notifiable Disease Surveillance System. Canberra: Commonwealth of Australia; 2026. Available online at: https://nindss.health.gov.au/pbi-dashboard/ (accessed February 2026).
10. Australian Institute of Health and Welfare. Pneumococcal disease in Australia. Canberra: AIHW; 2018. Available online at: https://www.aihw.gov.au/getmedia/ 0e959d27-97c9-419c-8636-ecc50dbda3c1/aihw-phe-236_Pneumococcal.pdf.aspx (accessed February 2026).
11. Pichichero ME. Protein carriers of conjugate vaccines: characteristics, development, and clinical trials. Hum Vaccin Immunother 2013; 9: 2505-2523.
12. Rose MA, Schubert R, Strnad N, Zielen S. Priming of immunological memory by pneumococcal conjugate vaccine in children unresponsive to 23-valent polysaccharide pneumococcal vaccine. Clin Diagn Lab Immunol 2005; 12: 1216-1222.
13. Daniels CC, Rogers PD, Shelton CM. A review of pneumococcal vaccines: current polysaccharide vaccine recommendations and future protein antigens. J Pediatr Pharmacol Ther 2016; 2: 27-35.
14. Jayasinghe S. Pneumococcal conjugate vaccines in children. Microbiol Aust 2024; 45: 179-183.
15. Australian Technical Advisory Group on Immunisation. Pneumococcal disease. In: Australian Immunisation Handbook. Canberra: Australian Government Department of Health; 2018. Available online at: https://immunisationhandbook.health.gov.au/contents/vaccine-preventable-diseases/pneumococcal-disease (accessed February 2026).
16. Australian Centre for Disease Control. NNDSS public dataset – pneumococcal disease (invasive). Canberra: Commonwealth of Australia; 2026. Available online at: https://www.cdc.gov.au/resources/publications/nndss-dataset-pneumococcal (accessed February 2026).
17. Australian Government Department of Health. National Immunisation Program Schedule. Canberra: Commonwealth of Australia; 2022. Available online at: https://www.health.gov.au/health-topics/immunisation/when-to-get-vaccinated/national-immunisation-program-schedule (accessed February 2026).
18. King LM, Andrejko KL, Kobayashi M, et al. Pneumococcal serotype distribution and coverage of existing and pipeline pneumococcal vaccines. J Infect Dis 2025; 232: e609-e620.