Invasive protozoal infections in Australia
Protozoal infections remain a significant global health concern, and although uncommon in Australia, they are increasingly seen in returned travellers, migrants and immunosuppressed individuals. Invasive amoebiasis, toxoplasmosis, malaria and leishmaniasis may be encountered in primary care. Subtle or delayed presentations make diagnosis challenging, requiring careful assessment of travel history, exposures and immune status. Improved clinician awareness supports earlier detection, timely management and reduced morbidity.
- Returned travellers, migrants and immunosuppressed patients are groups in Australia at higher risk of protozoal infections.
- People with protozoal infections can present with vague or delayed symptoms, creating diagnostic uncertainty in primary care.
- Accurate diagnosis relies on targeted history taking, appropriate investigations and infection-specific clinical features.
- Conditions such as malaria, toxoplasmosis, amoebiasis and leishmaniasis require prompt recognition to prevent complications.
- Prevention strategies, including travel advice and risk mitigation, remain essential in reducing imported infections.
Protozoa are unicellular organisms that may live freely or within a human or other animal host, often causing infection in humans through mechanisms such as antigenic variation or intracellular concealment. Clinically, infections may be broadly classified into intestinal protozoal, free-living amoeba or blood and tissue protozoal infections. These infections pose a significant global health burden, particularly in low- and middle-income countries. Invasive protozoal infections are not often encountered in primary care in Australia, but they are being increasingly observed in returned travellers, migrants and immunosuppressed patients.
Primary health care providers face diagnostic challenges because of subtle or nonspecific clinical presentations that may arise well after exposure to protozoa, compounded by limited familiarity with ordering and interpreting specialised investigations. Although malaria and leishmaniasis are not acquired in Australia, their incidence is rising in the context of global travel and immunosuppression. Entamoeba histolytica is more common in travellers but may be locally acquired. Toxoplasmosis is endemic and has important implications in pregnancy and for immunocompromised individuals.
Accurate diagnosis requires a careful clinical approach, incorporating a detailed history of travel, exposures, prophylactic measures, immunological status and a focused physical examination. This article reviews key protozoal infections relevant to primary care and aims to support GPs in their clinical assessment and investigation of these conditions.
Malaria
Malaria should be regarded as a medical emergency, and timely diagnosis is crucial to avoid serious complications. Malaria is a protozoal infection caused by Plasmodium species. Delays in treatment can result in death, particularly from Plasmodium falciparum malaria. It is a vector-borne disease, spread via the bite of an infected female Anopheles mosquito.1 Five major Plasmodium parasite species cause disease in humans: P. vivax, P. falciparum, P. malariae, P. ovale and P. knowlesi.1,2 Global elimination efforts primarily focus on P. falciparum and P. vivax because of their high prevalence and potential to cause severe disease.3 Malaria is not endemic in Australia; however, 700 to 800 malaria cases are diagnosed annually in returned travellers.4
Epidemiology
In 2023, there were an estimated 263 million new malaria cases in 83 countries worldwide. Although 94% of all global cases occurred in Africa, in Australia most cases were acquired in Oceania and Asia, including Papua New Guinea, Indonesia, the Solomon Islands, the Philippines, Cambodia and Vanuatu. This reflects Australia’s geographic proximity, common travel patterns and its composite migrant population.5-7
The WHO declared Australia malaria-free in 1981.8 However, the tropical north of the country remains malaria-receptive because of the presence of Anopheles mosquito vectors.9 Cases reported in Australia are usually travel-related; P. falciparum is most frequently acquired from sub-Saharan Africa, whereas P. vivax is more often associated with travel to Oceania and Asia and P. knowlesi with Malaysia.8 Of note, although malaria is endemic in many parts of Indonesia, the risk of transmission in Bali is considered low to negligible.10,11
Life cycle
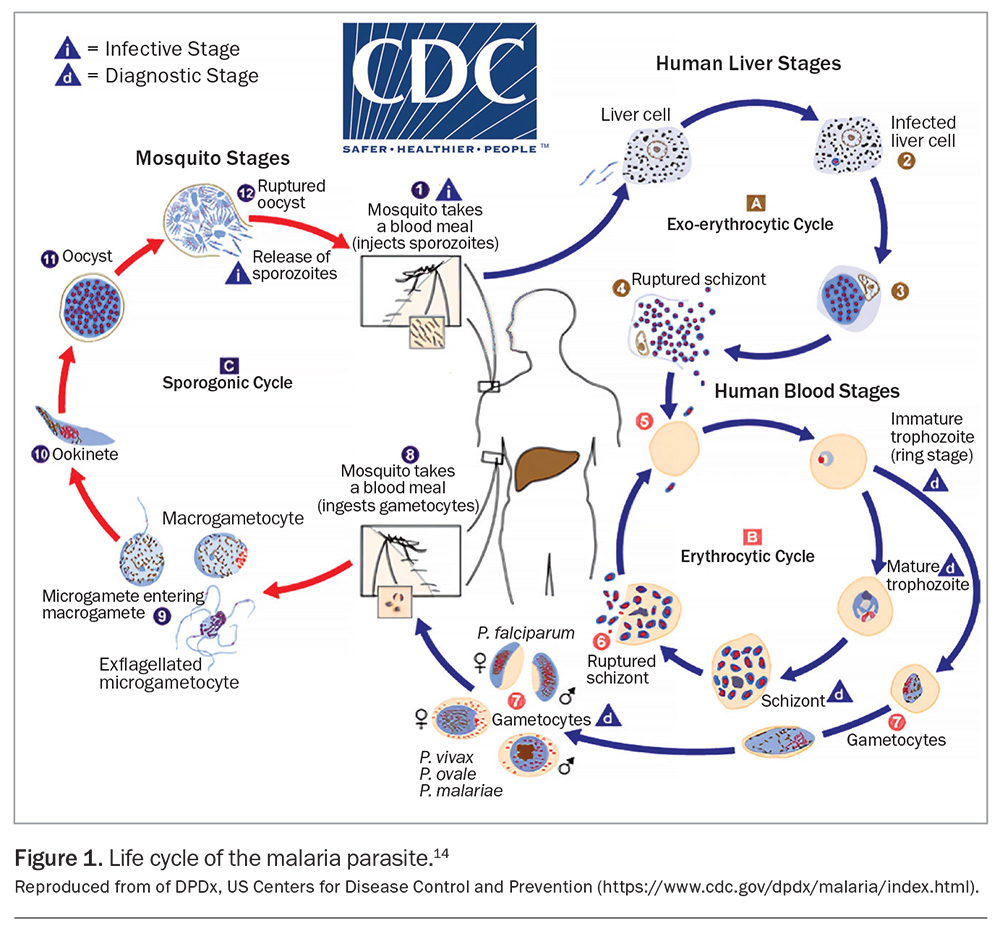
Plasmodium sporozoites are injected into the skin when the female Anopheles mosquito takes a blood meal. Sporozoites migrate through the blood to the liver, where they multiply asexually within hepatocytes, maturing into schizonts. The schizonts rupture and release merozoites. Fevers coincide with schizont rupture. Merozoites invade and multiply in erythrocytes, which then burst, releasing more merozoites. P. vivax and P. ovale have a dormant hepatic stage (hypnozoites), which can be responsible for relapses weeks or even years later.
Some parasites differentiate into sexual erythrocytic stages (gametocytes). These gametocytes circulate in the bloodstream and are taken up by Anopheles mosquitoes during a blood meal before undergoing sexual reproduction in the mosquito.12-14 The life cycle of malaria parasites is summarised in Figure 1.14 Key Plasmodium life cycle stages and terms used in this article are summarised in Box 1.
Clinical manifestations
The incubation period of malaria is usually 10 to 14 days. However, it can be up to 18 months if the patient has acquired infection with P. vivax or P. ovale.15 The most common presentation of malaria is fever in a returned traveller from an endemic region. Other associated symptoms include chills, headache, myalgia, nausea and vomiting. Some patients may develop the classic malaria paroxysm of fevers, chills and sweats as the disease progresses. These periodic fever responses are caused by rupture of the mature schizonts. Fevers classically occur every 48 hours in cases of P. vivax, P. falciparum or P. ovale, and every 72 hours in cases of P. malariae.16,17 Fevers in P. knowlesi can occur daily. Other associated signs include splenomegaly and jaundice.
Severe malaria is caused by P. falciparum, P. vivax and P. knowlesi. Major complications include pulmonary oedema, acute renal failure, severe anaemia, haemorrhage and cerebral malaria, the last of which may manifest as delirium, confusion, seizures and coma.18 Risk factors for severe malaria and death include being older than 65 years of age, female sex, pregnancy, immunocompromised status (including splenectomy), coexisting medical conditions, delay in treatment, no antimalarial prophylaxis and severity of illness at presentation.19
Diagnosis
The gold standard for laboratory diagnosis of malaria is microscopic examination of thick and thin blood films performed on whole blood. Thick smears are used to quantify parasitaemia, whereas thin smears aid in species identification. If suspicion of malaria is high, three thick and thin films are recommended at different times to increase sensitivity.16
Microscopy assesses the number of parasites relative to the number of red or white blood cells. The term percentage parasitaemia, or percentage of red cells parasitised, is often used to report results and can assist in determining clinical severity. In low-transmission areas, mortality from acute P. falciparum malaria begins to increase with parasite levels over 100,000 per microlitre (about 2.5% parasitaemia).19,20
A full blood count may reveal lymphopenia, thrombocytopenia and occasional atypical lymphocytes. Renal function, liver function, blood glucose levels and venous blood gas should also be assessed to determine the severity of malaria.
Additionally, laboratories may use rapid diagnostic tests. These include rapid antigen detection tests that detect antigens produced by P. falciparum, P. vivax, P. ovale and P. malariae. Tests detecting histidine-rich protein 2 have a high sensitivity for P. falciparum. Some assays combine histidine-rich protein 2 detection with reagents that detect a ‘pan-malaria’ group of antigens produced by all four species, which enhances malaria diagnosis, particularly in mixed infections. Rapid immunochromatography assays detect parasite-specific lactate dehydrogenase and perform better for P. vivax than tests using the ‘pan-malaria’ antigen.21 Nucleic acid amplification testing methods, including polymerase chain reaction (PCR) and loop-mediated isothermal amplification on whole blood, may be used to detect low levels of parasitaemia, especially if clinical suspicion is high despite negative thick and thin blood smears.21,22 However, these methods are not widely available and still require blood films for species identification and parasitaemia levels.
Management
Treatment choice for patients with malaria is guided by clinical risk factors and infection severity. Expert advice is recommended for children, pregnant women, immunocompromised people and those returning from the Greater Mekong Subregion due to antimalarial resistance in this area. Those with signs of severe disease should be referred to hospital. Severe disease is defined as the presence of at least one of the following:
- impaired consciousness (Glasgow Coma Scale <11)
- convulsions
- severe anaemia (haemoglobin <70 g/L in adults or <50 g/L in children younger than 12 years of age)
- acute kidney injury
- hypoglycaemia
- acute respiratory distress syndrome
- shock
- disseminated intravascular coagulation
- acidosis
- coma
- liver dysfunction
- parasite density greater than 2% of erythrocytes (in areas of low endemicity).19,23
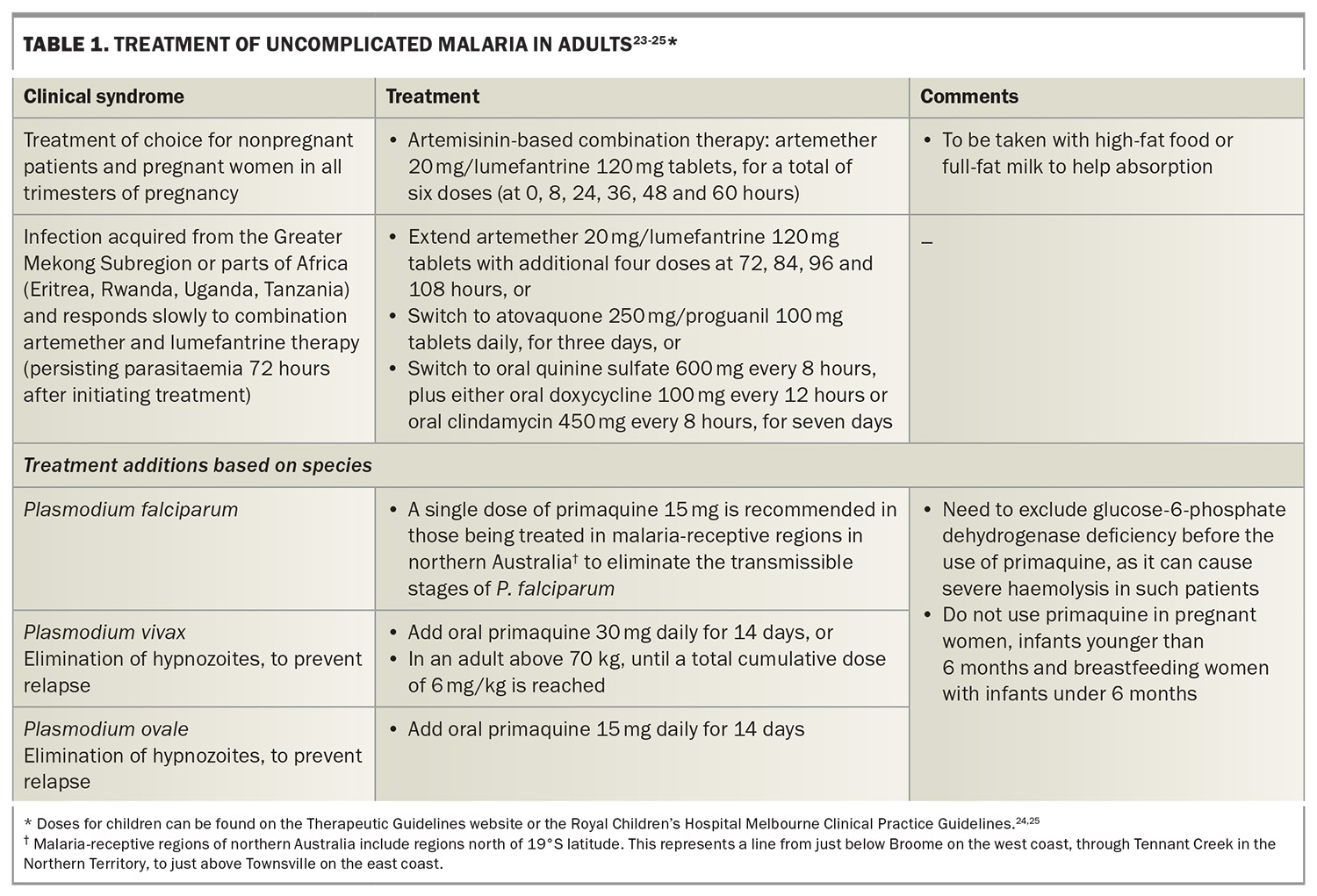
Treatment of uncomplicated malaria is summarised in Table 1.23-25 It usually involves artemisinin-based combination therapy (artemether/lumefantrine). Additionally, oral primaquine is added to avoid relapse in P. vivax or P. ovale infection, after glucose-6-phosphate dehydrogenase testing. Tafenoquine can be used for the radical cure of P. vivax malaria, but specialised quantitative glucose-6-phosphate dehydrogenase testing is required before starting; specialist advice should be sought.26
Monitoring treatment effect
The malaria parasite count should be monitored daily during treatment until it is negative. A full blood count and malaria microscopy at seven and 28 days after completion of therapy are recommended to assess for recrudescence.1
It is important to be aware of mixed infections; for example, P. falciparum may occur within the usual incubation period and P. vivax may occur later if primaquine has not been prescribed to eradicate hypnozoites. Patients should be counselled to monitor for recurrence of symptoms.
Prophylaxis
Malaria prophylaxis should include mosquito avoidance in addition to chemoprophylaxis. Chemoprophylaxis is complicated because of multidrug-resistant strains of P. falciparum worldwide, particularly in Southeast Asia. Clinicians should ask about planned travel to specific geographical locations. Information on malaria risk, drug resistance and prophylaxis is available from the US Centers for Disease Control and Prevention (CDC) Yellow Book 2026 Health Information for International Travel.10 Referral to a travel medicine clinic is strongly recommended.
Toxoplasmosis
Toxoplasmosis, caused by the intracellular protozoal Toxoplasma gondii, is a common infection that is often asymptomatic but may present with flu-like symptoms and lymphadenopathy. Cats and other felines serve as definitive hosts, with other mammals and birds acting as intermediate hosts.27,28 Although generally benign in immunocompetent individuals, toxoplasmosis has important clinical implications in pregnancy and immunosuppression.27,29
Epidemiology
T. gondii is found worldwide. The estimated seroprevalence in women of childbearing age in Australia is 20 to 40%.15,16 Disease incidence is likely underestimated due to asymptomatic presentation and because toxoplasmosis is not a notifiable condition.30
Life cycle
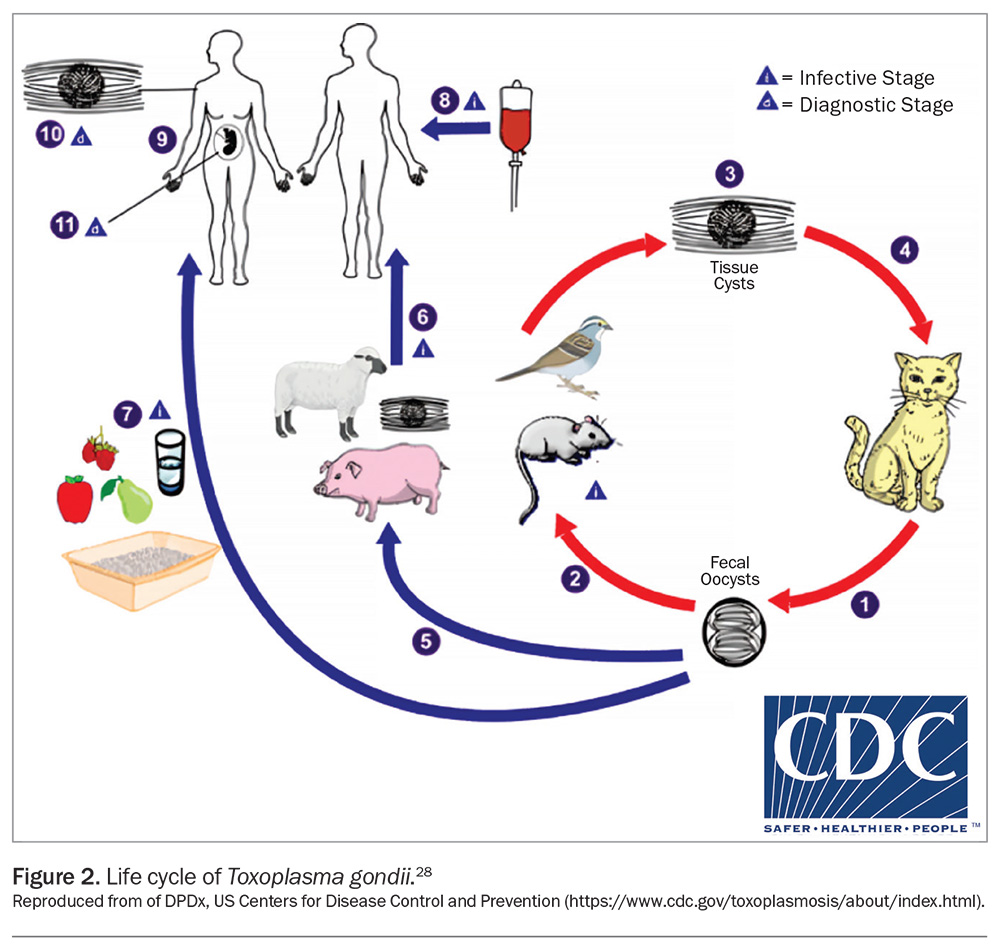
Felines ingest T. gondii oocysts from contaminated environmental material or animal remains containing T. gondii bradyzoite cysts, which are released in their intestine and undergo sexual reproduction. This produces oocysts that are shed in faeces, which become infective after one to five days.
Oocysts can survive for extended periods in the environment. When a person or nonfeline animal ingests oocysts, usually from contaminated food, water or cat litter, the oocysts undergo excystation in the intestines, releasing tachyzoites that spread through the body, potentially causing disseminated disease with a trophism for the brain or eyes. The host immune system eliminates most of the protozoa, but residual tachyzoites return to bradyzoites after seven to 10 days, remaining latent as tissue cysts. Immunosuppression may reactivate these cysts, causing widespread infection. Infection can also occur through organ transplantation or vertical transmission during acute maternal infection.27-29 The life cycle of T. gondii is summarised in Figure 2.28 Key T. gondii life cycle stages and terms used are summarised in Box 1.
Clinical manifestations
Infection with T. gondii is usually asymptomatic, although acute infection may be mononucleosis-like, with flu-like symptoms, lymphadenopathy, pharyngitis and hepatosplenomegaly with maculopapular rash. Rare manifestations include myocarditis, pericarditis, hepatitis or chorioretinitis. Severe disease can occur, and this depends on host factors and parasite genotype, which appears to influence virulence.27,28
Immunocompromised individuals and the developing fetus are most vulnerable to T. gondii infection, and can present with central nervous system involvement ranging from encephalopathy to mass lesions with a typical ring-enhancing appearance. Congenital infection may cause hydrocephalus, chorioretinitis, intracranial calcifications and even fetal death. Infection in the first trimester poses a small risk of transmission to the placenta, with transmission rates rising with gestational age and ranging between 60 and 81% in the third trimester. Although first-trimester infection rates are lower, they carry the highest risk of severe neonatal infection.30 Nonpregnant women with acute toxoplasmosis should use contraception for six months post-infection to prevent this occurrence.27-29
Diagnosis
Unlike in some countries, routine serological screening for T. gondii during pregnancy and of newborns is not performed in Australia, because of low neonatal infection incidence, difficulties interpreting positive results and variable evidence of treatment efficacy. Instead, the focus is on prevention such as avoiding eating raw meat and unpasteurised milk products, as well as cleaning cat litter trays daily to remove oocysts before they become infective.29,31
Acute infection is primarily diagnosed using serology. Toxoplasma-specific immunoglobulin G (IgG) antibodies are produced several weeks after initial exposure and usually remain lifelong. Toxoplasma-specific immunoglobulin M (IgM) antibodies produced within a week or two in response to T. gondii may indicate recent infection but can persist for months, so require interpretation alongside IgG avidity testing. Low IgG avidity suggests infection within the past three months, whereas high avidity indicates infection later than this.32 Results are best clarified with a repeat serological sample after two to three weeks to show rising Toxoplasma-specific IgG titres (seroconversion). False-positive IgM test results can occur, and this may be due to interfering antibodies. If IgM is positive but IgG remains negative on follow-up serology, and determining acute infection is important (e.g. in pregnancy), discussion with the microbiology laboratory regarding confirmatory IgM testing should be considered.32 The Australasian Society for Infectious Diseases Management of Perinatal Infections guideline provides further information on toxoplasmosis in pregnancy.33
PCR testing for Toxoplasma DNA in blood, CSF or amniotic fluid is useful in immunocompromised patients or when congenital infection is suspected. Amniotic fluid PCR testing is usually performed between 18 and 20 weeks with supplementary imaging to assess complications. Less frequently, diagnosis can be made through microscopy of biopsied tissue (e.g. lymph nodes, placenta) and PCR analysis of the same material.29,32,34
Management
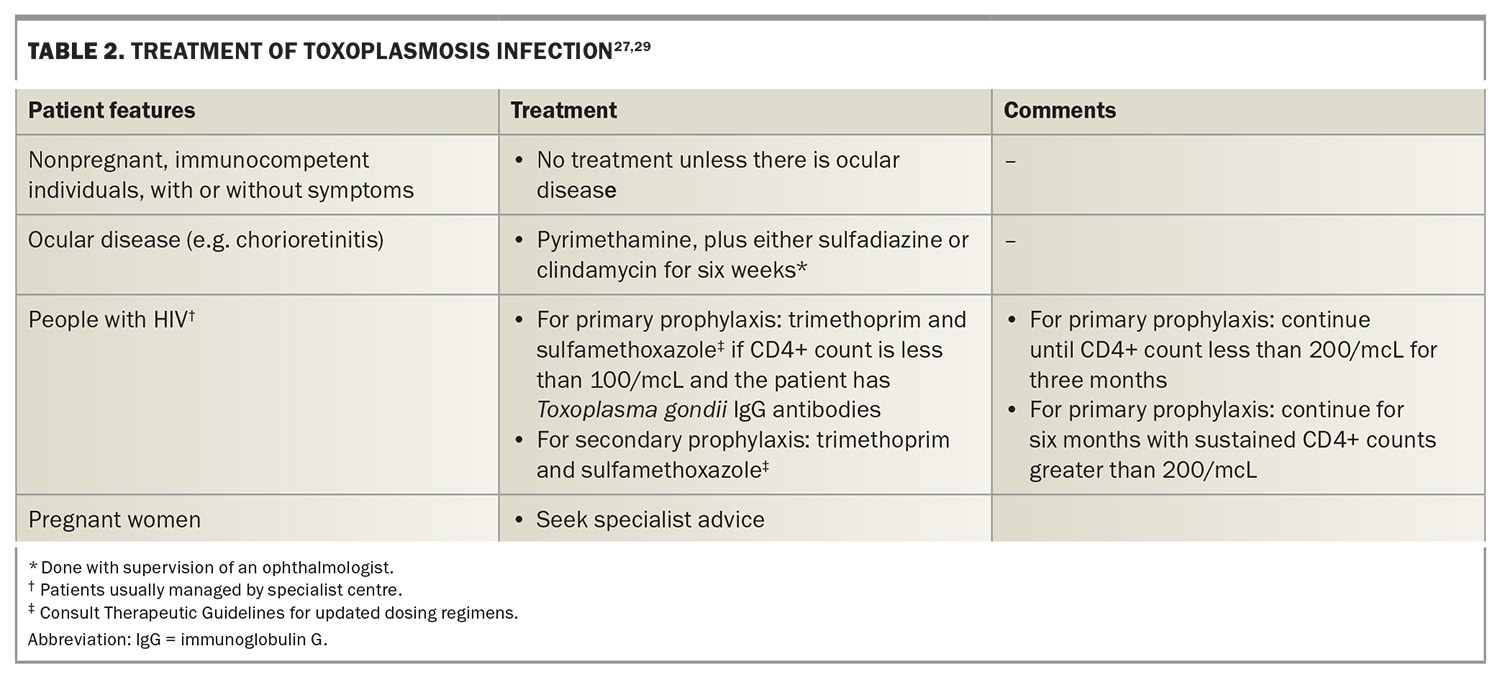
The management of infection with T. gondii is summarised in Table 2.27,29 Pyrimethamine, plus either sulfadiazine or clindamycin, is the treatment of choice for those with ocular disease. Trimethoprim and sulfamethoxazole are used as prophylaxis for patients with HIV, depending on their CD4+ count. Suspected infection with T. gondii during pregnancy warrants referral to a high-risk pregnancy clinic with consideration of input from maternal-fetal medicine and infectious diseases specialists. Although treatment during pregnancy is controversial and lacks proven efficacy in preventing congenital infection, it is believed to reduce severity.34
Amoebiasis
Entamoeba species are the causative agent of amoebiasis. About 500 million people are infected worldwide, with 40,000 to 74,000 deaths occurring annually because of infection.35,36 Prompt diagnosis and treatment are crucial to prevent complications and onward transmission.
Epidemiology
In Australia, most cases of amoebiasis are acquired following travel to regions of high endemicity; however, sexual exposure to an infected individual, particularly involving faecal contact, and colonic irrigation are recognised risk factors.37 Higher risk groups include men who have sex with men and people of Aboriginal or Torres Strait Islander background.38 Cases have also been reported in northern Australia in individuals without traditional risk factors, suggesting possible endemicity.39
Life cycle
More than seven species of Entamoeba have been isolated from humans.36 Of these, E. histolytica is a clear pathogen, the pathogenicity of several other microscopically indistinguishable species (E. dispar, E. moshkovskii and E. bangladeshi) is the subject of ongoing debate and others (including E. coli and E. hartmanni) are nonpathogenic.36,40Entamoeba species exist in two main forms: cysts and trophozoites.41 Mature cysts are ingested from contaminated food or water, or through sexual contact (e.g. oral-anal sex), and pass to the caecum, where they excyst to trophozoites. Trophozoites may remain within the intestinal lumen, invade the colonic mucosa or disseminate to extraintestinal sites. Some trophozoites undergo binary fission and re-encyst, with both forms being passed in faeces; however, only cysts can survive outside of the body.41 Key Entamoeba life cycle stages and terms used are summarised in Box 1.
Clinical manifestations
Over 90% of patients infected with Entamoeba species remain asymptomatic, but these patients can shed infectious cysts. In symptomatic patients, intestinal amoebiasis may present as diarrhoea, dysentery or lower abdominal pain mimicking appendicitis. Severe cases can cause colitis and are part of the differential diagnosis for inflammatory bowel disease. Potential complications include dehydration, colonic perforation, peritonitis and formation of amoebic granulomas (amoebomas).42,43 Patients with severe dysentery may pass trophozoites rapidly, causing infection of perianal skin and the development of necrotic skin lesions.
Extraintestinal manifestations of amoebiasis have also been described; hepatic abscess is a relatively common late presentation and may or may not be preceded by colitis.37 Other extraintestinal manifestations are rare, but include cerebral, pericardial and pleuropulmonary abscesses.43-45
Diagnosis
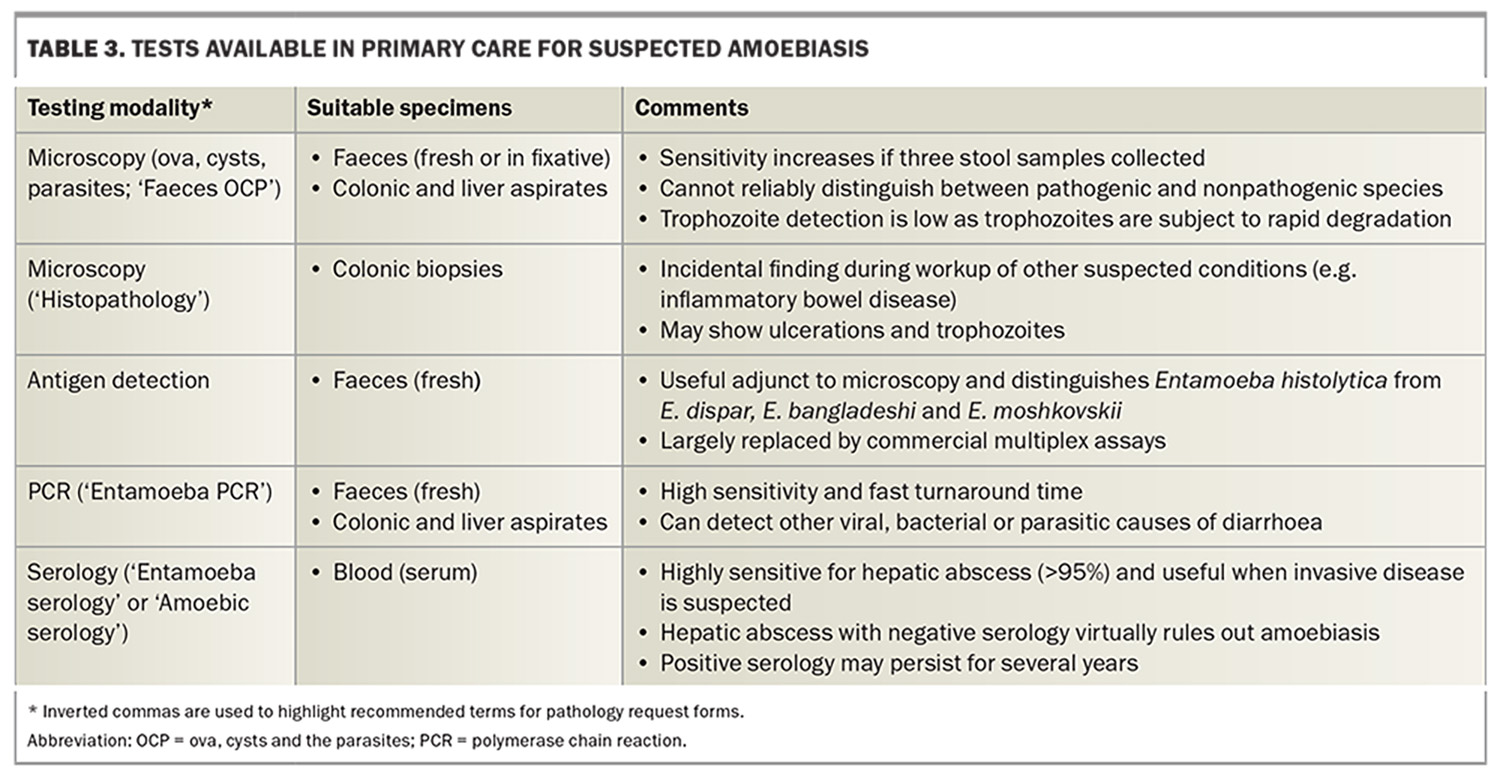
Diagnostic modalities for amoebiasis include microscopy, PCR and serological tests; the most appropriate test depends on the clinical syndrome and should be interpreted alongside travel history and sexual exposure. These are detailed in Table 3.
Fresh stool or stool collected in fixative is suitable for detecting Entamoeba cysts and occasionally trophozoites, with sensitivity increasing if three stool samples are collected. However, microscopy cannot reliably differentiate between Entamoeba species. By contrast, PCR can distinguish E. histolytica from the nonpathogenic Entamoeba species, and several commercial multiplexed PCR assays are available for testing stool samples from patients presenting with diarrhoeal symptoms.46 PCR has increased sensitivity and can potentially detect co-pathogens causing diarrhoea, including viral (e.g. norovirus, rotavirus), bacterial (Salmonella spp., Campylobacter spp.) and parasitic causes (e.g. Giardia).
Serological assays are useful to confirm invasive disease; Entamoeba antibodies are usually positive in more than 95% of patients with extraintestinal disease, 70% of those with amoebic colitis and 10% of asymptomatic cyst carriers.43 Specificity is high (95%), although positive results may be present in patients from endemic areas with infection within the past 10 years. Direct antigen detection kits exist but are not routinely available in Australia and are mostly supplanted by PCR. Both microscopy and PCR can be performed on colonic and liver aspirates.43,47 Histopathological examination of colonic tissue may show ulcerations or trophozoites; this may occur if patients undergo workup for another condition, such as inflammatory bowel disease.43
Management and prevention
Treatment is recommended for all patients infected with E. histolytica, regardless of symptoms. Asymptomatic patients should be treated with a luminal-active agent, such as paromomycin 500 mg every eight hours for seven days (or 10 mg/kg up to 500 mg for children, per dose), available through the Special Access Scheme in Australia. Paromomycin is active against the cyst form of E. histolytica, reducing the risk of persistence and secondary transmission. Patients with acute amoebic colitis or extraintestinal disease are recommended to receive a tissue-active agent (e.g. metronidazole 600 to 800 mg every eight hours for seven days) first, followed by paromomycin to eradicate intestinal carriage.48,49 Hand hygiene is recommended during travel. No vaccines are available, although research is ongoing; prophylaxis with antimicrobials is not routinely recommended.50
Leishmaniasis
Leishmaniasis is a vector-borne disease caused by protozoa of the genus Leishmania transmitted by the bite of an infected female phlebotomine sandfly. Leishmania species cause a diverse range of disease, with three main clinical forms: visceral, cutaneous and mucocutaneous. Each form’s clinical presentation is influenced by the infecting species and the host’s immune response.51
Cutaneous leishmaniasis is the most common form, typically presenting with one or more chronic skin lesions at the site of inoculation. While often self-limiting, lesions can be slow to heal and may result in disfiguring scars. Visceral leishmaniasis, also known as kala-azar, is a more severe systemic illness affecting the liver, spleen and bone marrow, presenting with prolonged fever, weight loss, hepatosplenomegaly and pancytopenia. Visceral leishmaniasis is usually fatal without treatment and requires urgent inpatient assessment and management, often in specialist settings. Mucocutaneous leishmaniasis is less common, usually arising from cutaneous infection, and involves destructive lesions of the nose, mouth or throat. It is most often caused by species from Central and South America.51
This article focuses on cutaneous leishmaniasis, as patients with this form are most likely to present to primary care.
Epidemiology
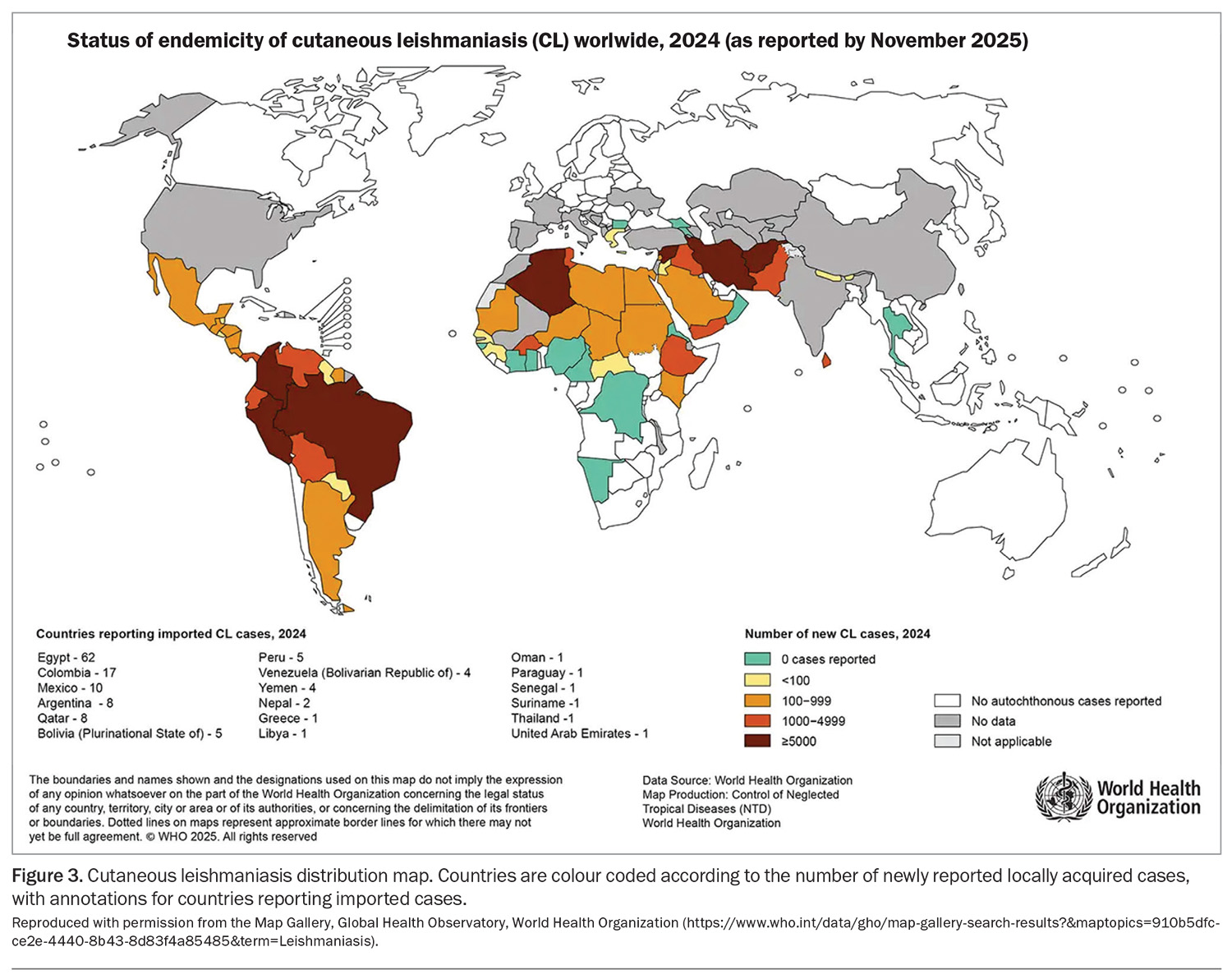
More than one billion people live in regions endemic for leishmaniasis, putting them at risk of infection, with about 700,000 to 1,000,000 new cases annually.51,52 Many cases go unreported, so global incidence is likely higher. It is prevalent in more than 90 countries, primarily in tropical and subtropical regions. More than 20 species of Leishmania cause disease in humans.51 Geographical origin, classified as Old World (Europe, Middle East, Asia) or New World (Americas), can help infer the likely species causing infection.53Figure 3 shows the global status of endemicity and new cases of cutaneous leishmaniasis in 2023. Climate change is expected to expand the range of sandfly vectors, potentially increasing global spread.
Although not endemic to Australia, cases (especially cutaneous forms) are increasingly reported among migrants, travellers and immunocompromised individuals.51,54
Life cycle
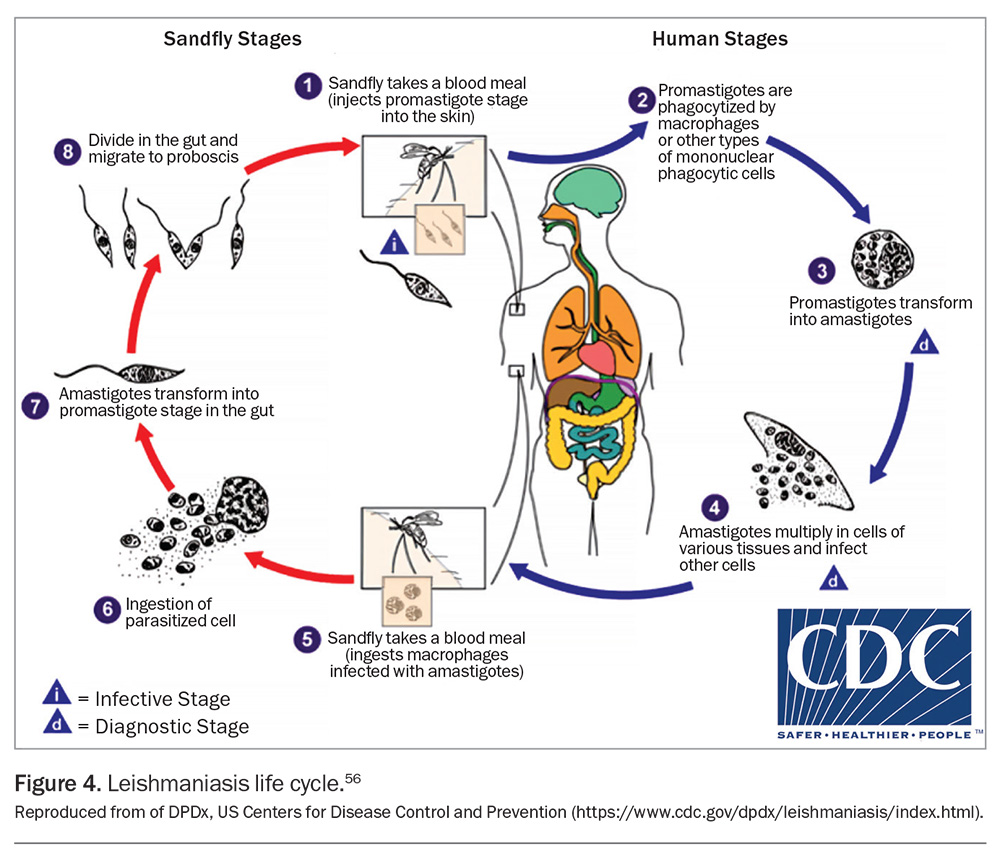
Transmission of Leishmania occurs when the infective promastigote stage is transmitted to humans via sandfly bites, followed by a second (amastigote) stage, during which the protozoa replicate within monocytes and macrophages, evading host immune defences.55,56 The disease presents significant diagnostic and treatment challenges because of its varied clinical manifestations and complex intracellular life cycle, as shown in Figure 4.56 Its intracellular persistence also makes treating the amastigote stage difficult.51 Key Leishmania life cycle stages and terms used are summarised in Box 1.
Clinical syndromes
Leishmaniasis has a variable incubation period of two weeks to several years, necessitating review of travel over this time frame. Cutaneous disease typically begins at the sandfly bite site as a firm, painless papule, which may ulcerate and scar, a characteristic feature of all three forms of leishmaniasis.51
Sandfly mouth parts cannot penetrate clothing, so cutaneous leishmaniasis typically presents as nodules, plaques or nonhealing ulcers on exposed body parts. Ulcers often have an indurated border, are painless and may be associated with local lymphadenopathy. Satellite lesions may occur along lymphatics and may be palpable without ulceration.57 These lesions develop weeks to months after a sandfly bite. Although not life threatening, disfiguring scars can result in social stigma and psychological distress.58
Mucocutaneous leishmaniasis often develops as a progression from initial cutaneous leishmaniasis, with mucosal lesions appearing months to years after the primary skin lesion heals. This can contribute to difficulty in diagnosis.56
Visceral leishmaniasis should be considered in cases of prolonged fever, cytopenia (due to bone marrow invasion) and relevant epidemiology.59,60
Diagnosis
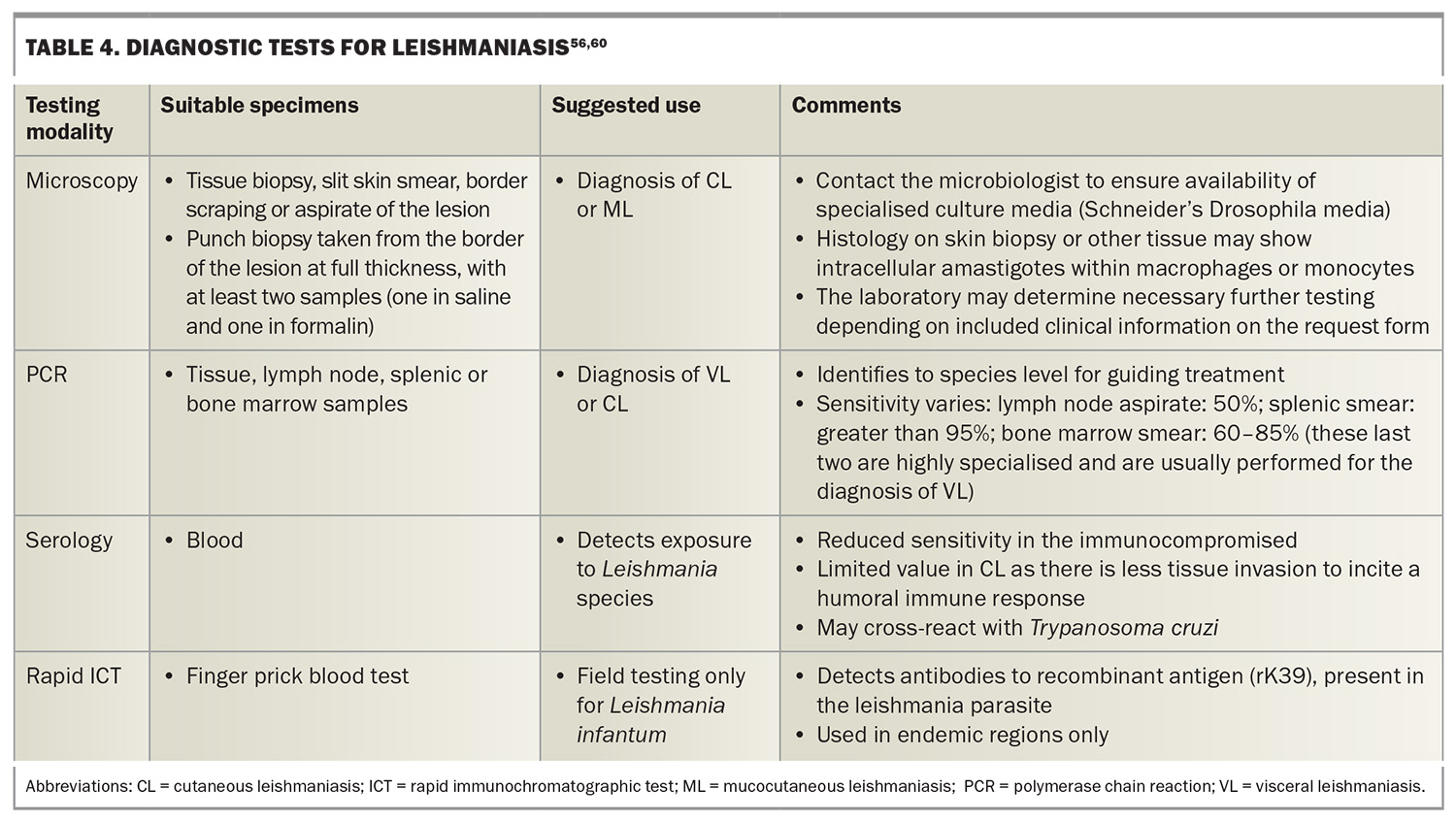
Leishmania species differ in drug sensitivity and treatment response. Traditional diagnostics include direct smear, histopathology and culture, although species are morphologically indistinguishable. Molecular methods, such as PCR on cultured promastigotes (the extracellular form), provide accurate species differentiation and can be combined with epidemiological information (e.g. geographic exposure risk) to guide appropriate treatment.51,59 Testing for Leishmania species should be considered in patients with atypical ulcers, systemic symptoms and travel to endemic regions. A summary of the various diagnostic tests is shown in Table 4.56,60 Serology is of limited value in the diagnosis of cutaneous leishmaniasis, as there is less tissue invasion to incite a humoral immune response. Additionally, the rapid immunochromatographic test is only used in endemic regions.
Management
Antimicrobial treatment of leishmaniasis can be challenging because of limited availability and the complexity of therapy, which depends on clinical syndrome, illness stage and the species of Leishmania. Given these complexities, referral to an infectious diseases physician is recommended for systemic or complicated cases.
For uncomplicated cutaneous leishmaniasis, local therapies such as cryotherapy may be appropriate. Other treatments include intralesional antimonials or topical azoles, which are not readily available and may require specialist consultation or liaison with a compounding pharmacy.58 Systemic therapy is required for diffuse or complex lesions. Although lesions may self-remit, treatment helps prevent scarring.56 Leishmaniasis prevention for travellers should focus on minimising sandfly exposure using protective clothing, insect repellents and bed nets, as no vaccine is available.52
Conclusion
A high degree of clinical suspicion alongside a thorough travel and exposure history is imperative when evaluating suspected invasive or systemic protozoal infections such as malaria, toxoplasmosis, amoebiasis and leishmaniasis. This is particularly important with returned travellers and immunocompromised patients. If uncertain, discussion with infectious diseases or microbiology specialists can help in appropriate test selection and treatment. Box 2 provides a quick reference for GPs on the diagnosis and management of these four protozoal infections. MT
COMPETING INTERESTS: Dr Bywater, Dr Ung, Dr Baumgart and Ms Phan: None. Dr McKew is a Member of Strongyloides Australia Inc., a not-for-profit advocacy group aimed at improving the diagnosis and treatment of strongyloidiasis in Australia and making it a notifiable disease with the aim of eradication.
References
1. World Health Organization (WHO). Malaria. Geneva: WHO; 2024. Available online at: https://www.who.int/news-room/fact-sheets/detail/malaria (accessed November 2025).
2. Lee WC, Cheong FW, Amir A, et al. Plasmodium knowlesi: the game changer for malaria eradication. Malar J 2022; 21: 140.
3. Chaturvedi R, Biswas S, Bisht K, Sharma A. The threat of increased transmission of non-knowlesi zoonotic malaria in humans: a systematic review. Parasitology 2023; 150: 1167-1177.
4. Victorian Government, Department of Health. Malaria. Melbourne: Victorian Government, Department of Health; 2024. Available online at: https://www.health.vic.gov.au/infectious-diseases/malaria (accessed November 2025).
5. Charles DM, Hart J, Davis WA, Sullivan E, Dowse GK, Davis TME. Notifications of imported malaria in Western Australia, 1990-2001: incidence, associated factors and chemoprophylaxis. Med J Aust 2005; 182: 164-167.
6. World Health Organization (WHO). World malaria report 2024. Geneva: WHO; 2024. Available online at: https://www.who.int/publications/i/item/9789240104440 (accessed November 2025).
7. Sohail A, Barry A, Auburn S, et al. Imported malaria into Australia: surveillance insights and opportunities. J Travel Med 2024; 31: taad164.
8. Gray TJ, Trauer JM, Fairley M, Krause VL, Markey PG. Imported malaria in the Northern Territory, Australia - 428 consecutive cases. Commun Dis Intell Q Rep 2012; 36: 107-113.
9. Lover AA, Baird JK, Gosling R, Price RN. Malaria elimination: time to target all species. Am J Trop Med Hyg 2018; 99: 17.
10. O’Laughlin K, Leidel L, Ridpath AD. Yellow fever vaccine and malaria prevention information, by country. Atlanta: US Centers for Disease Control and Prevention; 2025. Available online at: https://www.cdc.gov/yellow-book/hcp/preparing- international-travelers/yellow-fever-vaccine-and-malaria-prevention-information-by-country.html (accessed November 2025).
11. Bin Said I, Kouakou YI, Omorou R, et al. Systematic review of Plasmodium knowlesi in Indonesia: a risk of emergence in the context of capital relocation to Borneo? Parasites Vectors 2022; 15: 258.
12. PATH. Malaria parasite life cycle infographic. Seattle: PATH; 2024. Available online at: https://www.malariavaccine.org/tools-resources/malaria-parasite-life-cycle (accessed November 2025).
13. Gouagna LC, Mulder B, Noubissi E, Tchuinkam T, Verhave JP, Boudin C. The early sporogonic cycle of Plasmodium falciparum in laboratory-infected Anopheles gambiae: an estimation of parasite efficacy. Trop Med Int Health 1998; 3: 21-28.
14. US Centers for Disease Control and Prevention (CDC). DPDx – laboratory identification of parasites of public health concern. Malaria. Atlanta: CDC; 2024. Available online at: https://www.cdc.gov/dpdx/malaria/index.html (accessed November 2025).
15. Croft AM. Malaria: prevention in travellers (non-drug interventions). BMJ Clin Evid 2014; 2014: 0903.
16. Marie C, William AP Jr. MSD manual consumer version. Malaria. Rahway: Merck & Co.; 2025. Available online at: https://www.msdmanuals.com/home/infections/parasitic-infections-extraintestinal-protozoa/malaria (accessed November 2025).
17. World Health Organization. Severe falciparum malaria. World Health Organization, Communicable Diseases Cluster. Trans R Soc Trop Med Hyg 2000; 94 Suppl 1: S1-90
18. Trampuz A, Jereb M, Muzlovic I, Prabhu RM. Clinical review: severe malaria. Crit Care 2003; 7: 315-323.
19. Severe Malaria Observatory (SMO). Severe malaria criteria, features & definition. Geneva: SMO. Available online at: https://www.severemalaria.org/severe-malaria/severe-malaria-criteria-features-definition (accessed November 2025).
20. US Centers for Disease Control and Prevention (CDC). Clinical features of malaria. Atlanta: CDC; 2024. Available online at: https://www.cdc.gov/malaria/hcp/clinical-features/index.html (accessed November 2025).
21. Australian Government, Department of Health, Disability and Ageing. Malaria – laboratory case definition. Canberra: Australian Government, Department of Health, Disability and Ageing; 2025. Available online at: https://www.health.gov.au/resources/publications/malaria-laboratory-case-definition?language=en (accessed November 2025).
22. World Health Organization (WHO). Nucleic acid amplification-based diagnostics Geneva: WHO; 2021. Available online at: https://www.who.int/teams/global-malaria-programme/case-management/diagnosis/nucleic-acid-amplification-based-diagnostics (accessed November 2025).
23. Shahbodaghi SD, Rathjen NA. Malaria: prevention, diagnosis, and treatment. Am Fam Physician 2022; 106: 270-278.
24. Therapeutic Guidelines. Malaria. Melbourne: Therapeutic Guidelines; 2019. Available online at: https://tgldcdp.tg.org.au/searchAction?appendedinputbuttons=malaria (accessed November 2025).
25. The Royal Children’s Hospital (RCH) Melbourne. Clinical practice guidelines : malaria. Melbourne: RCH Melbourne; 2019. Available online at: https://www.rch.org.au/clinicalguide/guideline_index/malaria/ (accessed November 2025).
26. Therapeutic Goods Administration (TGA). Australian product information kozenis (tafenoquine) tablets. Canberra: TGA; 2018. Available online at: https://www.tga.gov.au/sites/default/files/auspar-tafenoquine-as-succinate-181115-pi.pdf (accessed November 2025).
27. Montoya JG, Remington JS. Management of Toxoplasma gondii infection during pregnancy. Clin Infect Dis 2008; 47: 554-566.
28. US Centers for Disease Control and Prevention (CDC). About toxoplasmosis. Atlanta: CDC; 2025. Available online at: https://www.cdc.gov/toxoplasmosis/about/index.html (accessed November 2025).
29. Contini C. Clinical and diagnostic management of toxoplasmosis in the immunocompromised patient. Parassitologia 2008; 50: 45-50.
30. Chaudhry SA, Gad N, Koren G. Toxoplasmosis and pregnancy. Can Fam Physician 2014; 60: 334-336.
31. Molan A, Nosaka K, Hunter M, Wang W. Seroprevalence and associated risk factors of Toxoplasma gondii infection in a representative Australian human population: the Busselton health study. Clin Epidemiol Glob Health 2020; 8: 808-814
32. Liesenfeld O, Press C, Montoya JG, et al. False-positive results in immunoglobulin M (IgM) toxoplasma antibody tests and importance of confirmatory testing: the Platelia Toxo IgM test. J Clin Microbiol 1997; 35: 174.
33. Palasanthiran P, Starr M, Jones C, Giles M. Management of perinatal infections. 3rd ed. Sydney: Australasian Society for Infectious Diseases; 2022.
34. American College of Obstetricians and Gynecologists. Practice bulletin no. 151: cytomegalovirus, parvovirus B19, varicella zoster, and toxoplasmosis in pregnancy. Obstet Gynecol 2015; 125: 1510-1525.
35. Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2095-2128.
36. Morán P, Serrano-Vázquez A, Rojas-Velázquez L, et al. Amoebiasis: advances in diagnosis, treatment, immunology features and the interaction with the intestinal ecosystem. Int J Mol Sci 2023; 24: 11755.
37. Domazetovska A, Lee R, Adhikari C, et al. A 12-year retrospective study of invasive amoebiasis in Western Sydney: evidence of local acquisition. Trop Med Infect Dis 2018; 3: 73.
38. Hal SJ van, Stark DJ, Fotedar R, Marriott D, Ellis JT, Harkness JL. Amoebiasis: current status in Australia. Med J Aust 2007; 186: 412-416.
39. Smith S, Phillips GE, McBride WJH, Hanson J. Case report: endemic amebiasis in Australia: implications for residents, travelers, and clinicians. Am J Trop Med Hyg 2017; 97: 245-247.
40. Sardar SK, Ghosal A, Haldar T, et al. Prevalence and molecular characterization of Entamoeba moshkovskii in diarrheal patients from Eastern India. PLoS Negl Trop Dis 2023; 17: e0011287.
41. US Centers for Disease Control and Prevention (CDC). DPDx - amebiasis. Atlanta: CDC; 2019. Available online at: https://www.cdc.gov/dpdx/amebiasis/index.html (accessed November 2025).
42. Shirley DAT, Watanabe K, Moonah S. Significance of amebiasis: 10 reasons why neglecting amebiasis might come back to bite us in the gut. PLoS Negl Trop Dis 2019; 13: e0007744.
43. Kantor M, Abrantes A, Estevez A, et al. Entamoeba histolytica: updates in clinical manifestation, pathogenesis, and vaccine development. Can J Gastroenterol Hepatol 2018; 2018: 4601420.
44. Matsuo T, Saito A, Kawai F, et al. Use of PCR in the diagnosis of pericardial amebiasis: a case report and systematic review of the literature. BMC Infect Dis 2021; 21: 960.
45. Victoria-Hernández JA, Ventura-Saucedo A, López-Morones A, et al. Case report: multiple and atypical amoebic cerebral abscesses resistant to treatment. BMC Infect Dis 2020; 20: 669.
46. Calle-Pacheco GL, Jiménez-Chunga JA, Vivas-Ruiz DE. Molecular diagnosis of amoebiasis. Bol Med Hosp Infant Mex 2022; 79: 3-16.
47. Yanagawa Y, Shimogawara R, Endo T, et al. Utility of the rapid antigen detection test E. histolytica Quik Chek for the diagnosis of Entamoeba histolytica infection in nonendemic situations. J Clin Microbiol 2020; 58: e01991-20.
48. Therapeutic Guidelines. Entamoeba histolytica infection (amoebiasis). Melbourne: Therapeutic Guidelines; 2019. Available online at: https://tgldcdp.tg.org.au.acs.hcn.com.au/viewTopic?etgAccess=true&guidelinePage=Antibiotic&topicfile=gastrointestinal-protozoa&guidelinename=Antibiotic§ionId=toc_d1e474#toc_d1e453 (accessed November 2025).
49. Gonzales MLM, Dans LF, Sio‐Aguilar J. Antiamoebic drugs for treating amoebic colitis. Cochrane Database Syst Rev 2019; 2019: CD006085.
50. Abhyankar MM, Orr MT, Kinsey R, et al. Optimizing a multi-component intranasal Entamoeba histolytica vaccine formulation using a design of experiments strategy. Front Immunol 2021; 12: 683157.
51. World Health Organization (WHO). Leishmaniasis. Geneva: WHO; 2023. Available online at: https://www.who.int/news-room/fact-sheets/detail/ leishmaniasis (accessed November 2025).
52. Institute for Health Metrics and Evaluation (IHME). Global Burden of Disease 2023: findings from the GBD 2023 study. Seattle: IHME; 2019. Available online at: https://www.healthdata.org/gbd/2019 (accessed November 2025).
53. Stark D, van Hal S, Lee R, Marriott D, Harkness J. Leishmaniasis, an emerging imported infection: report of 20 cases from Australia. J Travel Med 2008; 15: 351-354.
54. Desjeux P. Leishmaniasis: current situation and new perspectives. Comp Immunol Microbiol Infect Dis 2004; 27: 305-318.
55. Burza S, Croft SL, Boelaert M. Leishmaniasis. Lancet 2018; 392: 951-970.
56. US Centers for Disease Control and Prevention (CDC). DPDx – leishmaniasis. Atlanta: CDC; 2024. Available online at: https://www.cdc.gov/dpdx/leishmaniasis/index.html (accessed November 2025).
57. Sousa Ade Q, Parise ME, Pompeu MM, et al. Bubonic leishmaniasis: a common manifestation of Leishmania (Viannia) braziliensis infection in Ceara, Brazil. Am J Trop Med Hyg 1995; 53: 380.
58. Dowlati Y. Cutaneous leishmaniasis: clinical aspect. Clin Dermatol 1996; 14: 425-431.
59. Jeronimo, SMB, de Queiroz Sousa, A, Pearson, RD. Leishmaniasis. In: Guerrant RL, Walker DH, Weller PF, ed. Tropical infectious diseases: principles, pathogens, and practice. 3rd ed. Philadelphia: Saunders Elsevier; 2011. p. 969.
60. Van Griensven J, Diro E. Visceral leishmaniasis: recent advances in diagnostics and treatment regimens. Infect Dis Clin North Am 2019; 33: 79-99.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.