The immunocompromised traveller

People who are immunocompromised are more vulnerable to infectious diseases, often experience more severe illness and may have a reduced response to immunisation. Careful risk assessment, preventive strategies and specialist collaboration can support safe and rewarding international travel.
- Immunocompromised travellers are more susceptible to disease, often more severely affected and may have a reduced response to immunisation.
- In general, avoid live vaccines in immunocompromised individuals, or use only following consultation with a specialist.
- Administer live vaccines at least four weeks before commencing immunosuppressive therapy.
- Consider both the risk of disease at the destination and the class of immunosuppressive medication.
- Provide preventive advice for respiratory, gastrointestinal and arboviral diseases.
- Advise travellers to review travel insurance, including treatment and evacuation cover.
- Encourage travellers to identify medical facilities at their destination.
An estimated 500,000 people in Australia are severely immunocompromised,1 consistent with US prevalence estimates of 2 to 3%.2 These individuals are generally more susceptible to infectious diseases, often more severely affected, and may also have a reduced response to immunisation. The degree of immunocompromise cannot be measured quantitatively, so assessment relies on careful clinical judgement. This article provides an overview of how to assess and advise people who are immunocompromised and plan to travel internationally.
The terms immunocompromised or immunodeficient refer to a general state of weakened immunity, regardless of cause. Immunosuppressed describes an impaired immune system due to medications or therapies designed to reduce immune activity.
The Australian Immunisation Handbook (AIH) provides detailed guidance, including a dedicated chapter on vaccination for people who are immunocompromised.3
Causes of immunodeficiency
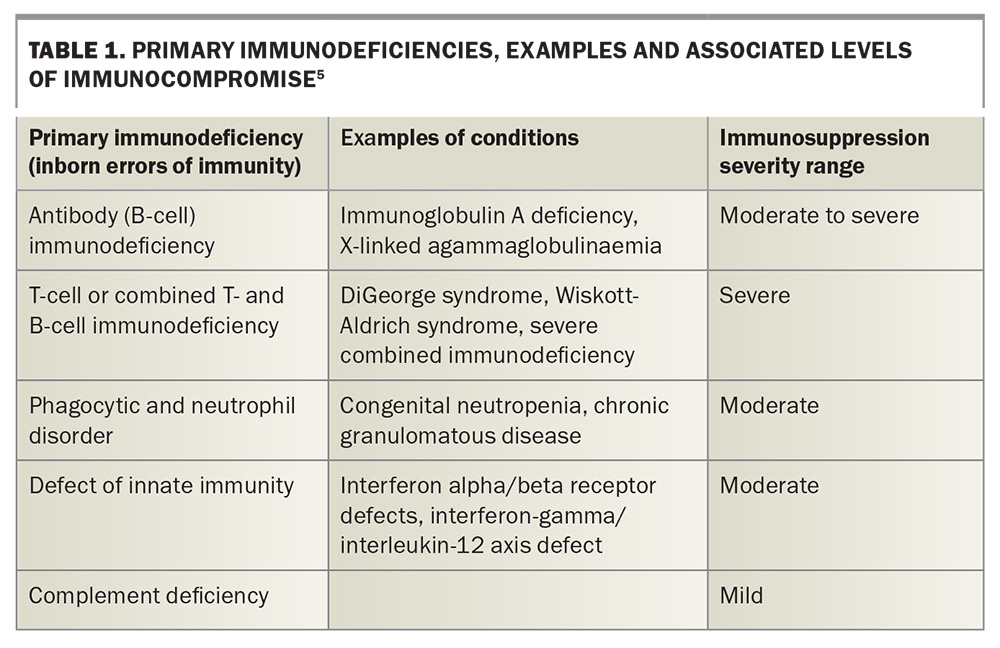
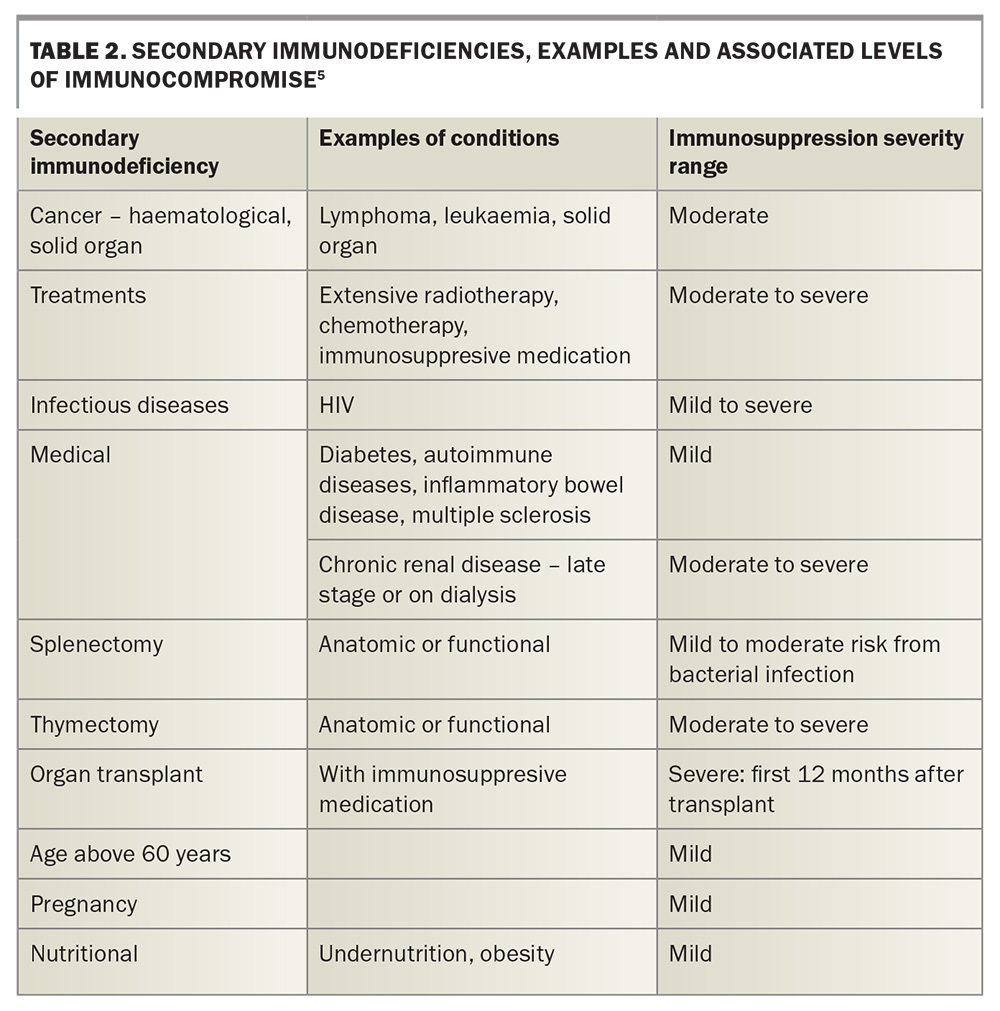
Immunodeficiency can be primary or secondary, and severity is classified as mild, moderate or severe. Primary immunodeficiencies comprise numerous inborn errors of immunity, which may be humoral, cellular or combined.4 The primary and secondary immunodeficiencies, examples of conditions and associated levels of immunocompromise are outlined in Table 1 and Table 2.5 The AIH also provides a detailed classification of immunocompromise by medical condition and medication.5
Assessing immune status
Risk assessment involves reviewing the patient’s medical history, underlying conditions, use of immunosuppressive medications and relevant results of laboratory investigations (Box 1). The type of immune disorder will affect susceptibility to disease and which vaccines are indicated or contraindicated. A full blood examination may be useful for white cell counts, and lymphocyte subset testing can clarify T- and B-cell status. CD4 T-lymphocyte counts are particularly relevant when considering yellow fever vaccination in travellers with HIV.6 Best practice includes discussion with the treating specialist, and if appropriate, haematology or immunology input.
Immunosuppressive medication
The use of immunosuppressive therapy has expanded beyond conventional agents to biologics and small-molecule therapies (Box 2). The degree of immune suppression depends on both the underlying disease and the class of medication. The AIH provides detailed tables on immunosuppressive therapies and the recommended timing of live vaccine administration.7,8
The American College of Rheumatology has also published guidelines for vaccination in patients with rheumatological and musculoskeletal conditions.9 However, these are broad and may not apply to all live vaccines, particularly yellow fever. In the absence of robust evidence, AIH recommendations should generally be prioritised. Examples of lower immunosuppressive therapies in which live viral vaccines may generally be given, if indicated, are listed in Box 3.
Before commencing immunosuppressive therapy
In general, lifelong immunity is conferred by illness, a completed course of immunisations or positive serology. However, verbal histories are not always accurate and immunity to some vaccines may decrease with time. If vaccine or illness history is uncertain, serology should be requested for hepatitis A and B, measles, mumps, rubella and varicella (Table 3). Pretravel screening may also include serology for HIV and syphilis, as well as an interferon-gamma release assay for tuberculosis. Positive serology avoids unnecessary vaccination, reducing patient discomfort and cost to the patient and the PBS.
After ceasing immunosuppressive therapy
People who have no active disease and have completed chemotherapy or radiotherapy more than three months earlier are generally considered sufficiently immunocompetent for live vaccines and international travel. For those taking immunosuppressive therapy, it may be possible to temporarily pause treatment to allow live vaccine administration. Typical intervals are four weeks for some agents, three to six months for biologics, and one to two years following an organ transplant.3,6,9
Vaccinations
Nonlive vaccines are generally safe for immunocompromised people, although immune responses may be reduced. If vvaccines were given during a period of immune suppression or following bone marrow transplant, they may need to be repeated. Routine vaccinations, as per the Australian vaccination schedule, should be reviewed and updated. The recombinant zoster vaccine is recommended before commencing immunosuppressive therapy.
Splenectomised travellers should receive meningococcal conjugate vaccine against serogroups A, C, W and Y, meningococcal B and Haemophilus influenzae type b vaccines, with boosters as appropriate.10 There are currently inadequate data to provide advice on administration of Q fever vaccine, dengue vaccine, the tick-borne encephalitis vaccine or chikungunya vaccine.
Close contacts of people who are immunocompromised should be advised to keep all their routine vaccinations up to date. Live vaccines can generally be given safely to contacts, although strict hand hygiene should follow oral live typhoid and cholera vaccines. In view of rare cases of person-to-person transmission, the author recommends avoiding any type of body fluid transfer for about one week after vaccination.
Live vaccines should be administered at least four weeks before immunosuppressive therapy. Measles, mumps, rubella and varicella vaccination is indicated in seronegative travellers. Live vaccines are generally contraindicated in immunocompromised individuals (Box 4).
Yellow fever vaccine is a key exception. For mildly immunocompromised travellers to high-risk regions, the benefits of vaccination may outweigh the risks. Adverse events, including fatalities, are rare but more frequent in immunocompromised populations.6 For example, a pregnant woman or a 70-year-old person travelling to a village in rural Nigeria for two weeks would, following careful discussion of risks and benefits, generally be offered vaccination because the risk of disease significantly outweighs the risk of vaccination. HIV-positive travellers to high-risk destinations with CD4 counts of more than 200 to 500 cells/mm³ (or >16–24% of total lymphocytes in children under 6 years of age) may safely receive yellow fever vaccine (Box 5).11 Travellers wishing to avoid any risk should be advised against travel to yellow fever-endemic areas, which currently include countries in tropical South America and sub-Saharan Africa. All travellers should be counselled on risks, and waiver certificates may be issued if vaccination is contraindicated.
Other considerations
Preventive advice should be provided for respiratory, gastrointestinal and arboviral diseases.
- Respiratory infections: recommend influenza, coronavirus disease 2019, pertussis, pneumococcal and respiratory syncytial virus vaccines. N95 masks are recommended especially in crowded settings. Antibiotics and antivirals may be useful.
- Gastrointestinal infections: provide detailed food and hand hygiene advice, and advise travellers to take self-treatment kits with antibiotics.
- Arboviruses: emphasise mosquito avoidance, including the use of physical barriers and appropriate repellents. Check for specific drug–drug interactions before prescribing antimalarial medications to immunocompromised travellers.
Travellers should carry spare medications in both checked and cabin baggage, together with appropriate emergency supplies. Insurance should be checked for treatment and evacuation cover, and local medical facilities identified in advance.
Travellers should be advised to report any symptoms within four to six weeks of return, including fever, diarrhoea, respiratory illness, skin lesions or neurological deficits. Early investigation guided by travel history is essential and may include full blood examination and relevant serology.
Conclusion
It can be challenging to assess the degree of immunocompromise and how this may relate to the proposed destination and itinerary. It is important to discuss potential risks with the traveller, consider their need or desire to travel and provide them with a sufficiently balanced view to allow them to make appropriate decisions about their trip. With tailored risk assessment, implementation of preventive strategies and specialist collaboration, most immunocompromised travellers are able to undertake safe and rewarding international travel. MT
COMPETING INTERESTS: Dr Cohen has received financial support for provision of and attendance at educational events from Sanofi Pasteur, CSL Seqirus and Biocelect.
References
1. Australian Government Department of Health, Disability and Ageing. Booster shot for severely immunocompromised Australians. Canberra: Australian Government Department of Health, Disability and Ageing; 2021 Available online at: https://www.health.gov.au/ministers/the-hon-greg-hunt-mp/media/booster-shot-for-severely-immunocompromised-australians (accessed October 2025).
2. Martinson ML, Lapham J. Prevalence of immunosuppression among US adults. JAMA 2024; 331: 880-882.
3. Australian Technical Advisory Group on Immunisation. The Australian immunisation handbook. Canberra: Australian Government Department of Health and Aged Care; 2025. Available online at: https://immunisationhandbook.health.gov.au/contents/vaccination-for-special-risk-groups/vaccination-for-people-who-are-immunocompromised (accessed October 2025).
4. Immune Deficiency Foundation (IDF). Patient and family handbook for primary immunodeficiency diseases. 6th ed. Towson, IDF; 2019. Available online at: https://primaryimmune.org/understanding-primary-immunodeficiency/diagnosis/laboratory-tests (accessed October 2025).
5. Australian Technical Advisory Group on Immunisation. Types of medical conditions and immunosuppressive therapy and associated levels of immunocompromise. Canberra: Australian Government Department of Health and Aged Care; 2025. Available online at: https://immunisationhandbook.health.gov.au/resources/tables/table-types-of-medical-conditions-and-immunosuppressive-therapy-and-associated-levels-of-immunocompromise (accessed October 2025).
6. Kotton CN, Chen LH, Freedman DO, et al. Immunocompromised travelers. In: Brunette GW, ed. CDC Yellow Book: Health Information for International Travel. New York: Oxford University Press, 2025. Available online at: https://www.cdc.gov/yellow-book/hcp/travelers-with-additional-considerations/immunocompromised-travelers.html (accessed October 2025).
7. Australian Technical Advisory Group on Immunisation. Immunosuppressive potential of biological therapies. Canberra: Australian Government Department of Health and Aged Care; 2025. Available online at: https://immunisationhandbook.health.gov.au/resources/tables/table-immunosuppressive-potential-of-biological-therapies (accessed October 2025).
8. Australian Technical Advisory Group on Immunisation. Recommended timing of live vaccine administration for people receiving immunosuppressive therapies. Canberra: Australian Government Department of Health and Aged Care; 2025. Available online at: https://immunisationhandbook.health.gov.au/resources/tables/table-recommended-timing-of-live-vaccine-administration-for-people-receiving-immunosuppressive-therapies (accessed October 2025).
9. Bass AR, Perin J, Calabrese C, et al. 2022 American College of Rheumatology guideline for vaccinations in patients with rheumatic and musculoskeletal diseases. Arthritis Care Res 2023; 75: 449-464.
10. Spleen Australia. Clinical guidelines for splenectomised patients. Melbourne: Spleen Australia; 2023. Available online at: https://spleen.org.au/clinicians/ (accessed October 2025).
11. Australian Technical Advisory Group on Immunisation. Yellow fever. In: The Australian Immunisation Handbook [Internet]. Canberra: Australian Government Department of Healthand Aged Care; 2023. Available online at: https://immunisationhandbook.health.gov.au/contents/vaccine-preventable-diseases/yellow-fever (accessed October 2025).
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.