Buruli ulcer: coming to a suburb near you?

Buruli ulcer, caused by Mycobacterium ulcerans, is a curable yet often misunderstood skin infection now emerging in Victoria. Evidence increasingly identifies mosquitoes as the key vector, with possums as a major environmental reservoir. Understanding its transmission, recognition and prevention is crucial as the disease continues to expand its range.
- Buruli ulcer, caused by Mycobacterium ulcerans, is a toxin-mediated necrotising condition of the skin and subcutaneous tissue.
- The disease is endemic in Victoria, with a year-on-year increase in infections acquired in coastal regions and, most recently, the inner suburbs of Melbourne and Geelong.
- Research shows that a major transmission pathway of M. ulcerans infection is from possums to humans via mosquitoes.
- Rapid diagnosis of Buruli ulcer is available via polymerase chain reaction testing of samples obtained on swabs from the undermined edge of ulcers or biopsies from the subcutaneous layer in patients with preulcerative plaque and cellulitic presentations.
- Rifampicin-based oral antibiotic regimens, particularly rifampicin and clarithromycin, are highly effective treatments for Buruli ulcer, although complete healing may take many weeks and requires patience and close clinical follow up.
- Mosquito bite prevention for individuals living in Victoria is likely to be the single most effective way to reduce the risk of acquiring Buruli ulcer.
Buruli ulcer is a necrotising infection of the skin and subcutaneous tissue caused by Mycobacterium ulcerans. This neglected tropical disease is endemic in more than 30 countries and has had a variety of other names including Bairnsdale ulcer and Daintree ulcer in Australia, and Kumusi ulcer and sik bilong wara Sepik in Papua New Guinea.1-4 ‘Buruli ulcer’ is the unifying term preferred by the WHO.5
Clinically, Buruli ulcer is a slowly progressive infection of subcutaneous fat and overlying skin. It is not fatal but can leave individuals with permanent cosmetic and functional disability.6 The feared destructive nature of Buruli ulcer is explained by the diffusible polyketide toxin mycolactone, the production of which is encoded by a unique plasmid.7-9 Mycolactone causes tissue necrosis in the subcutaneous fat layer beneath the dermis, allowing the disease to spread laterally from the site of initial inoculation and undermining and destroying the overlying skin as it progresses.
Geographical distribution of Buruli ulcer
In most parts of the world, the primary risk factor for acquiring Buruli ulcer is residence in a Buruli-endemic area. Traditionally, these areas have been low-lying river valleys, especially in tropical West and Central Africa.6 In tropical Australia, there is a persistent small endemic focus of Buruli ulcer between Mossman and the Daintree River in Far North Queensland, and occasionally cases appear along the Capricorn Coast in Central Queensland and near Darwin in the Northern Territory.10-12
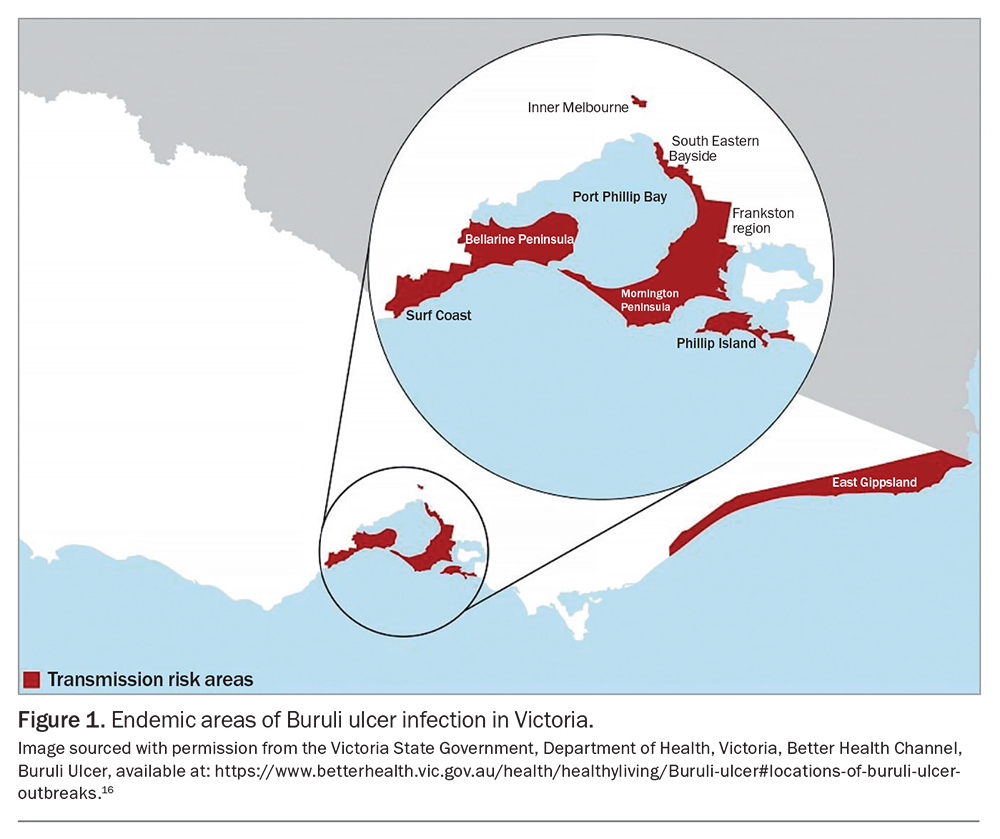
However, Buruli ulcer in Australia is most common in the temperate southeast.13,14 In Victoria, more than 350 cases are currently being reported annually, compared with only five to 10 cases annually in the rest of Australia combined.12 Over the past 35 years in Victoria, transmission of Buruli ulcer has moved from the original focus area around Bairnsdale to the main centres of population.13-15 It has become endemic in coastal towns on the Bellarine Peninsula and along the Surf Coast as far as Aireys Inlet, as well as on the Mornington Peninsula, extending up to the southeastern bayside suburbs of Melbourne (Figure 1; Box).16
Most recently, locally acquired Buruli ulcer has been identified in the inner northern suburbs of Melbourne and Geelong, leaving both the coast and coastal theories of causation behind.14 Buruli ulcer infection appears to be expanding not just westward from the Bairnsdale region but also to the north, with the first report from southern coastal New South Wales in 2007 and a small cluster of cases in 2024.17,18
Importantly, nearly half of Buruli ulcer cases in Victoria occur in summer visitors to the Mornington and Bellarine Peninsulas, rather than in residents of endemic areas.14 Some exposures are extremely brief; I have treated patients who had been in an endemic area for only one to four hours before developing Buruli ulcer many months later.
Transmission of Buruli ulcer
Infection with M. ulcerans occurs through contact with the environment and is not transmitted from person to person. For many years, researchers have sought to identify the elusive environmental reservoir and the precise mode of transmission. In Victoria, various hypotheses have been proposed and subsequently dismissed, ranging from aerosol transmission via recycled water on golf courses to associations with certain coastal plants or alkaline soils.19-21
Although it has been possible to detect the DNA of M. ulcerans in the environment since the mid-1990s, it took more than 50 years of unsuccessful attempts to obtain the first positive culture directly from any environmental sample, and that was from an African insect.19,22,23 Furthermore, with the completion and analysis of the whole M. ulcerans genome in 2007, it became clear that M. ulcerans may not be free-living in water and soil after all.24,25 A new paradigm was required to explain the unexpectedly high number of individuals from Victoria presenting with Buruli ulcer in clinics.
Possums
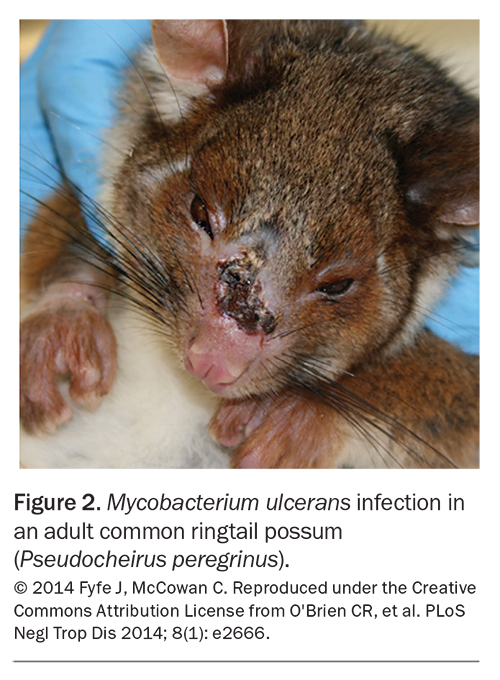
After almost 30 years of environmental research, it is now clear that Buruli ulcer in Victoria is a zoonosis, transmitted indirectly from possums via mosquitoes.26-28 The disease initially appears in local possum populations, which both develop Buruli ulcer themselves and excrete M. ulcerans in high concentrations in their faeces (Figure 2).28 The possum outbreak develops silently for some time before spilling over to humans living nearby.29-31
Humans, possums and mosquitoes are ubiquitous, yet human Buruli ulcer is restricted to certain areas. This is because Buruli ulcer in possums is also restricted. Methods are now available to survey possums in trees by collecting and testing their excreta on the ground below.28,29,32 Possum excreta samples in nonendemic areas reliably test negative. How M. ulcerans spreads to new possum populations that are sometimes too far apart to be explained by natural possum or mosquito movement remains a mystery.
Mosquitoes
The evidence for the last link in the transmission chain – mosquitoes – is now also very strong. M. ulcerans was first detected in mosquitoes in 2007. Research went on to show that the risk of acquiring Buruli ulcer in coastal towns is closely correlated with the proportion of locally trapped polymerase chain reaction (PCR)-positive mosquitoes.26,33 Since then, human body maps of Buruli lesions have indicated that the disease is most likely to occur in the location of the body where mosquitoes bite. Analysis of Victoria Department of Health notification data has shown that transmission of Buruli ulcer aligns closely with the Victorian mosquito season, once the very long incubation period is taken into account.34,35
Two Victorian case control studies have produced slightly different results, but a finding in common was that insect repellent reduces the risk of Buruli ulcer.27,36 Most recently, sophisticated molecular techniques have confirmed that not only do backyard mosquitoes feed on both possums and humans, but also the strains of M. ulcerans recoverable from mosquitoes, possums and humans are the same. ‘Backyard mosquitoes’ refers to Aedes notoscriptus in this research.37
Transmission outside of Victoria
Whether an animal reservoir and insect vector are involved in transmission outside of Victoria is not known, but there is preliminary evidence linking Buruli ulcer to possums in southern New South Wales and bandicoots and mosquitoes in tropical Far North Queensland.17,38-40
Back to the clinic … diagnosis of Buruli ulcer
Common scenarios in clinicians unfamiliar with the disease
There are two scenarios that commonly challenge doctors who are inexperienced with Buruli ulcer. Firstly, patients may have visited an endemic area by the coast for a summer holiday. The median incubation period is 4.8 months, so individuals may not develop a skin lesion until many months later, in winter, and often do not associate it with their summer holiday.41
The second challenging scenario is if a new group of infected possums and human infections develop in a local suburb. Unfortunately, the initial cases in a newly endemic area typically have delayed diagnoses, which can result in more severe disease.14,42 This diagnostic uncertainty can be frustrating, leading patients to consult multiple clinicians before their condition is correctly identified.
Fortunately, the same PCR test used for environmental research is widely available in clinical practice and can identify or rule out Buruli ulcer with a high degree of confidence, provided it is used correctly.
Presentation
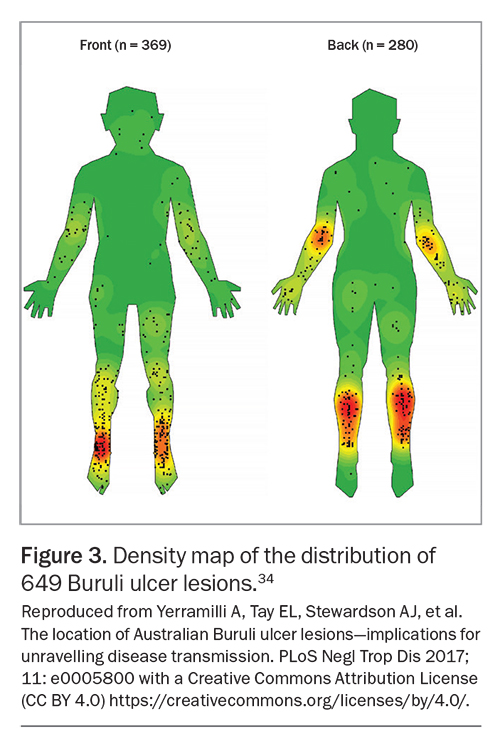
Buruli ulcer can present in all ages. Lesions are usually single on an exposed part of the body but can occur anywhere (Figure 3). Common lesion locations are around the ankles and elbows and the backs of the legs and thighs. It is rare for patients to report systemic symptoms, and the lesion is relatively painless in its early stages compared with more rapidly progressive skin infections caused by staphylococci or streptococci.
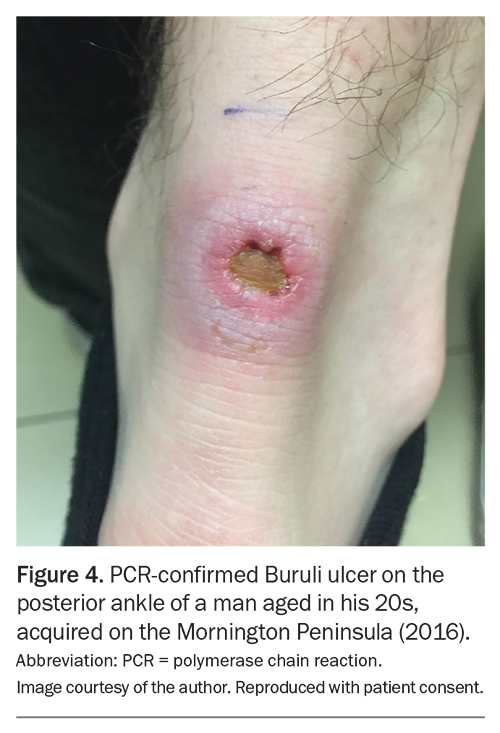
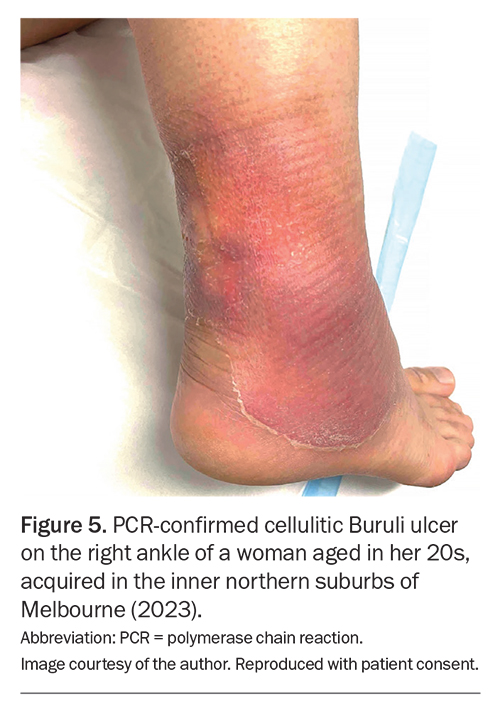
In about 80% of cases, individuals present with an existing ulcer (Figure 4), and about 20% of cases have plaques or subacute areas of cellulitis with associated oedema (Figure 5).14 Patients frequently delay seeking a diagnosis (median: three weeks), reflecting the indolent natural history of Buruli ulcer compared with more typical bacterial skin infections.14
Nondirected bacterial swabs can be misleading, as they may yield positive results for normal skin flora including Staphylococcus aureus. Empirical treatment with usual antibiotics, such as flucloxacillin or cefalexin, has little effect.
Approach to testing
Successful testing requires an understanding that Buruli ulcer is a subcutaneous infection, and that diagnostic samples must be collected from the appropriate tissue layer to give reliable results. When an ulcer is present, this is easy. A standard bacterial swab rubbed around the undermined edge of the ulcer and observed to ensure there is visible biological material on the swab is an excellent specimen. It is essential to order the correct tests: PCR specific for Buruli ulcer, mycobacterial smear and culture. For plaque and cellulitis presentations, a swab is not useful and a biopsy that samples down into the subcutaneous layer is required.
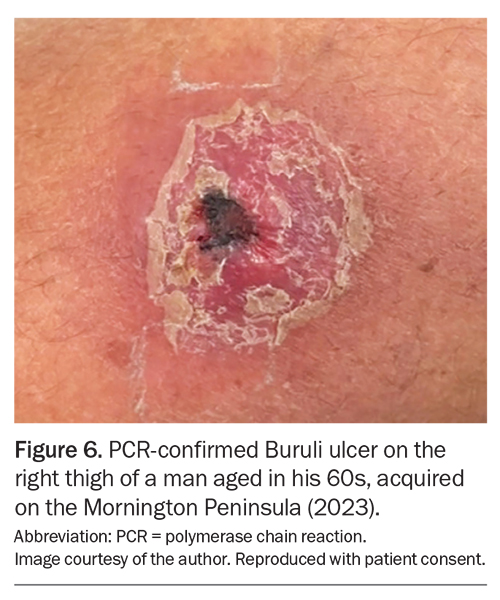
Many patients present with lesions that are intermediate between an ulcer and a plaque, often resembling a small volcano with a central gelatinous plug (Figure 6). Working on this with a saline soaked swab can often produce a diagnostically useful sample even though a definitive ulcer has not yet become established.
Buruli ulcer can progress at different rates in different people and can be alarming at times, particularly in children. However, once the diagnosis is made, highly effective therapy is available. Nowadays, most severe Buruli ulcer cases are caused by delayed diagnosis.14
Differential diagnoses
Differential diagnoses include community- acquired staphylococcal infections (typically more rapidly progressing, painful and sometimes associated with systemic features), skin cancers in older patients with chronically damaged skin, cutaneous leishmaniasis (notably in patients with a history of overseas travel, with ulcers that are not undermined) and vascular, diabetic or venous ulcers. In rural or tropical practice, cutaneous anthrax, melioidosis and infected tick bites could resemble Buruli ulcer initially.
Treatment of Buruli ulcer
Previous and current treatments
Previously, there was a strong emphasis on surgery with wide excision and grafting as the principal approach to treatment of Buruli ulcer. However, the huge increase in cases has led to innovation from necessity, both in Australia and in Africa.43-50 The treatment of Buruli ulcer has been transformed by recognising the effectiveness of antibiotics and the importance of patience during treatment. Surgical intervention is now reserved for select cases.
New Australian consensus guidelines for the treatment of Buruli ulcer are available online.51 Nevertheless, some hard-earned insights drawn from my clinical experience are described below.
Antibiotic therapy and paradoxical reactions
Combination antibiotic therapy, usually with rifampicin combined with clarithromycin, effectively eradicates M. ulcerans. However, progress is very slow. Paradoxically, the lesion may expand, become red and painful and copiously discharge during or even after completion of antibiotic therapy. This is not what patients and doctors usually expect during successful antibiotic treatment and is one of the reasons behind the historical preference for surgery.
Paradoxical reactions are explained by declining levels of the immunosuppressive toxin, meaning that the immune system is suddenly confronted with large numbers of highly immunogenic dying mycobacteria.52,53 Occasionally, corticosteroids are indicated to reduce these reactions in conjunction with antibiotics.51,54,55 Repeating PCR on swabs to monitor progress is not recommended. PCR detects bacterial DNA, not live bacteria, and may remain positive for months or even years after successful treatment.
Patience
Above all, patience from both the doctor and the patient is extremely important. The usual duration of antibiotic therapy is eight weeks. However, the ulcer is unlikely to have fully healed by this time and typically takes many more weeks. Dressings may need to be used for weeks or months thereafter.51
Experience
Experience and confidence help, and for this reason many patients in Victoria are managed in infectious disease clinics with Buruli ulcer experience or with a shared model of care. However, experienced GPs in endemic areas have become excellent diagnosticians, and some confidently manage Buruli ulcer without referral. Helpfully, rifampicin has recently been assigned a Buruli ulcer indication on the PBS.
Early recognition and testing for Buruli ulcer by clinicians can prevent progression to severe disease. Importantly, even in its most advanced form, Buruli ulcer is a curable infection.
Prevention of Buruli ulcer
Traditionally, M. ulcerans was considered a free-living environmental pathogen, and most authorities still attribute Buruli ulcer infection to direct contact with contaminated environments. General advice has therefore been to cover up when outside all year, avoid contact with soil and water, clean and cover any cuts and scratches, and check for new skin lesions. When evidence first emerged that mosquitoes can carry M. ulcerans and that possums excrete the bacterium in their faeces, new recommendations were added to the growing list of precautions: avoid mosquito bites and refrain from handling possums or their excreta.
However, as understanding of Buruli ulcer in Victoria grows, the evidence increasingly points almost exclusively to mosquitoes. M. ulcerans is present in possums and their excreta throughout the year, yet transmission is restricted to the mosquito season.13,29,35 Mosquitoe-bite prevention is likely to be the single best prevention strategy for Buruli ulcer in Victoria.
There have been two trials of Bacille Calmette-Guérin (BCG) vaccine to prevent Buruli ulcer in humans in Africa. Both showed some benefit, although this was short lived.56 A novel strategy could involve using the BCG vaccine to control Buruli ulcer in possums in southeastern Australia, an approach currently under consideration in New Zealand for managing bovine tuberculosis in feral brushtail possums.57,58
Beat the bite
More than 74,000 mosquitoes were tested for M. ulcerans on the Mornington Peninsula in Victoria from 2016 to 2021 as part of the Beating Buruli in Victoria project.59 About five per 1000 mosquitos tested positive and almost all of these were Ae. notoscriptus – the native Australian backyard mosquitoe.37
Measures to avoid mosquito bites include choosing appropriate outdoor clothing, applying personal insect repellent and maintaining functional fly screens. Mosquitoe control in backyards can also be effective, but this can be controversial if the focus is on spraying insecticides.
Mosquitoe control in backyards
Fortunately, there are methods of reducing mosquito numbers in backyards. Anecdotally, I have had success both in inner city Melbourne and at my holiday house in coastal Victoria in a Buruli-endemic area. The steps I took noticeably reduced mosquito numbers, encouraging us to go outside and enjoy our garden more than we used to.
Firstly, locate and remove, or carefully screen off, all breeding sites such as containers of standing water, rainwater and sewerage systems, blocked gutters and discarded tyres. Fine-mesh insect screening secured with adhesive or cable ties to pipes, vents and drain openings makes logical sense. Then, I installed and maintained passive gravid traps (Figure 7); around four are needed for a standard backyard. These use water to attract egg-laying female mosquitoes but prevent the emergence of new adult mosquitos. They work best when other egg-laying locations are unavailable. Gravid traps can be purchased online or made at home. One mistake I made was to try an overnight electric blue light (fan trap); the next morning I found dozens of the most beautiful moths, most species of which I had never seen before.
Where next for Buruli ulcer?
Buruli ulcer is now present in inner Melbourne and Geelong, where there are large contiguous populations of humans and possums and where most gardens have backyard mosquitoes. There is the potential for further expansion and a continued increase in cases. Already, 2025 is tracking ahead of all previous years for Buruli ulcer notifications in Victoria. Other states and territories are also at risk. Cases are now being seen in New South Wales, historically free of Buruli ulcer.17
New endemic areas continue to emerge, but endemicity can also decline over time, perhaps because susceptible possums die out, leaving descendants with greater resistance.13-15 Buruli ulcer is declining in West and Central Africa for unknown reasons and now appears to be rare in East Africa, where Buruli County used to be (now known as the Nakasongola District). Victoria is currently the only jurisdiction in the world with a rising incidence of Buruli ulcer.
Conclusion
Buruli ulcer remains a curable infection that continues to expand its range in Victoria. Evidence now strongly supports transmission from possums to humans via mosquitoes, reshaping understanding of the disease and guiding prevention strategies. Continued public health vigilance, environmental management and early clinical recognition is essential in limiting further spread and reducing the burden of this potentially disabling skin condition. MT
COMPETING INTERESTS: Professor Johnson has received support from the NHMRC, Department of Health Victoria Public Health Grants; and is a Data and Safety Monitoring Board member for TREAT-BU, a phase 2 multicentre, open-label study to evaluate the efficacy, safety and tolerability, and pharmacokinetics of 28 days telacebec treatment in adult participants with Buruli ulcer.
References
1. Korman TM, Johnson PDR, Hayman J. Etymologia: Buruli ulcer. Emerg Infect Dis 2020; 26: 3104.
2. Johnson PDR, Hayman JA. Comment on: Mycobacterium ulcerans infection: an eponymous ulcer by Derek H Meyers. Med J Aust 2007; 187: 63.
3. Roltgen K, Pluschke G. Buruli ulcer: history and disease burden. In: Pluschke G, Roltgen K, eds. Buruli ulcer: Mycobacterium ulcerans disease. Cham (CH): Springer; 2019. p. 1-41.
4. Roltgen K, Pluschke G. Overview: Mycobacterium ulcerans disease (Buruli ulcer). Methods Mol Biol 2022; 2387: 3-6.
5. World Health Organization. Buruli ulcer (Mycobacterium ulcerans infection). Geneva: WHO; 2023. Available online at: https://www.who.int/news-room/fact-sheets/detail/buruli-ulcer-(mycobacterium-ulcerans-infection) (accessed November 2025).
6. Johnson PD, Stinear T, Small PL, et al. Buruli ulcer (M. ulcerans infection): new insights, new hope for disease control. PLoS Med 2005; 2: e108.
7. George KM, Chatterjee D, Gunawardana G, et al. Mycolactone: a polyketide toxin from Mycobacterium ulcerans required for virulence. Science 1999; 283: 854-857.
8. Guenin-Mace L, Ruf MT, Pluschke G, Demangel C. Mycolactone: more than just a cytotoxin. In: Pluschke G, Roltgen K, eds. Buruli ulcer: Mycobacterium ulcerans disease. Cham (CH): Springer; 2019. p. 117-34.
9. Stinear TP, Mve-Obiang A, Small PL, et al. Giant plasmid-encoded polyketide synthases produce the macrolide toxin of Mycobacterium ulcerans. Proc Natl Acad Sci U S A 2004; 101: 1345-1349.
10. Steffen CM, Smith M, McBride WJ. Mycobacterium ulcerans infection in North Queensland: the ‘Daintree ulcer’. ANZ J Surg 2010; 80: 732-736.
11. Steffen CM, Freeborn H. Mycobacterium ulcerans in the Daintree 2009-2015 and the mini-epidemic of 2011. ANZ J Surg 2018; 88: E289-E293.
12. Johnson PDR. Buruli ulcer in Australia. In: Pluschke G, Roltgen K, eds. Buruli ulcer: Mycobacterium ulcerans disease. Cham (CH): Springer; 2019. p. 61-76.
13. Loftus MJ, Tay EL, Globan M, et al. Epidemiology of Buruli ulcer infections, Victoria, Australia, 2011-2016. Emerg Infect Dis 2018; 24: 1988-1997.
14. Ravindran B, Hennessy D, O’Hara M, et al. Epidemiology of Buruli ulcer in Victoria, Australia, 2017-2022. Emerg Infect Dis 2025; 31: 448-457.
15. Johnson PD, Veitch MG, Leslie DE, Flood PE, Hayman JA. The emergence of Mycobacterium ulcerans infection near Melbourne. Med J Aust 1996; 164: 76-78.
16. Victoria State Government, Department of Health, Victoria, Better Health Channel, Buruli Ulcer, available at: https://www.betterhealth.vic.gov.au/health/healthyliving/Buruli-ulcer#locations-of-buruli-ulcer-outbreaks (accessed November 2025).
17. Hossain ME, Keighley C, Buultjens AH, et al. Buruli ulcer in Australia: evidence for a new endemic focus at Batemans Bay, New South Wales. PLoS Negl Trop Dis 2024; 18: e0012702.
18. Lavender CJ, Senanayake SN, Fyfe JA, et al. First case of Mycobacterium ulcerans disease (Bairnsdale or Buruli ulcer) acquired in New South Wales. Med J Aust 2007; 186: 62-63.
19. Ross BC, Johnson PD, Oppedisano F, et al. Detection of Mycobacterium ulcerans in environmental samples during an outbreak of ulcerative disease. Appl Environ Microbiol 1997; 63: 4135-4138.
20. Hayman J. Postulated epidemiology of Mycobacterium ulcerans infection. Int J Epidemiol 1991; 20: 1093-1098.
21. Blasdell KR, McNamara B, O’Brien DP, et al. Environmental risk factors associated with the presence of Mycobacterium ulcerans in Victoria, Australia. PLoS One 2022; 17: e0274627.
22. Ross BC, Marino L, Oppedisano F, Edwards R, Robins-Browne RM, Johnson PD. Development of a PCR assay for rapid diagnosis of Mycobacterium ulcerans infection. J Clin Microbiol 1997; 35: 1696-1700.
23. Portaels F, Meyers WM, Ablordey A, et al. First cultivation and characterization of Mycobacterium ulcerans from the environment. PLoS Negl Trop Dis 2008; 2: e178.
24. Stinear TP, Seemann T, Pidot S, et al. Reductive evolution and niche adaptation inferred from the genome of Mycobacterium ulcerans, the causative agent of Buruli ulcer. Genome Res 2007; 17: 192-200.
25. Yip MJ, Porter JL, Fyfe JA, et al. Evolution of Mycobacterium ulcerans and other mycolactone-producing mycobacteria from a common Mycobacterium marinum progenitor. J Bacteriol 2007; 189: 2021-2029.
26. Johnson PD, Azuolas J, Lavender CJ, et al. Mycobacterium ulcerans in mosquitoes captured during outbreak of Buruli ulcer, southeastern Australia. Emerg Infect Dis 2007; 13: 1653-1660.
27. Quek TY, Athan E, Henry MJ, et al. Risk factors for Mycobacterium ulcerans infection, southeastern Australia. Emerg Infect Dis 2007; 13: 1661-1666.
28. Fyfe JA, Lavender CJ, Handasyde KA, et al. A major role for mammals in the ecology of Mycobacterium ulcerans. PLoS Negl Trop Dis 2010; 4: e791.
29. Vandelannoote K, Buultjens AH, Porter JL, et al. Statistical modeling based on structured surveys of Australian native possum excreta harboring Mycobacterium ulcerans predicts Buruli ulcer occurrence in humans. Elife 2023; 12: e84983.
30. McNamara BJ, Cornish J, Blasdell KR, et al. Mycobacterium ulcerans in possum feces before emergence in humans, Australia. Emerg Infect Dis 2025; 31: 569-573.
31. Hobbs EC, Porter JL, Lee JYH, et al. Buruli ulcer surveillance in south-eastern Australian possums: infection status, lesion mapping and internal distribution of Mycobacterium ulcerans. PLoS Negl Trop Dis 2024; 18: e0012189.
32. Carson C, Lavender CJ, Handasyde KA, et al. Potential wildlife sentinels for monitoring the endemic spread of human Buruli ulcer in South-East Australia. PLoS Negl Trop Dis 2014; 8: e2668.
33. Lavender CJ, Fyfe JA, Azuolas J, et al. Risk of Buruli ulcer and detection of Mycobacterium ulcerans in mosquitoes in southeastern Australia. PLoS Negl Trop Dis 2011; 5: e1305.
34. Yerramilli A, Tay EL, Stewardson AJ, et al. The location of Australian Buruli ulcer lesions-Implications for unravelling disease transmission. PLoS Negl Trop Dis 2017; 11: e0005800.
35. Buultjens AH, Tay EL, Yuen A, Friedman ND, Stinear TP, Johnson PDR. Mosquitoes as vectors of Mycobacterium ulcerans based on analysis of notifications of alphavirus infection and Buruli ulcer, Victoria, Australia. Emerg Infect Dis 2024; 30: 1918-1921.
36. McNamara BJ, Blasdell KR, Yerramilli A, et al. Comprehensive case-control study of protective and risk factors for Buruli ulcer, southeastern Australia. Emerg Infect Dis 2023; 29: 2032-2043.
37. Mee PT, Buultjens AH, Oliver J, et al. Mosquitoes provide a transmission route between possums and humans for Buruli ulcer in southeastern Australia. Nat Microbiol 2024; 9: 377-389.
38. Singh A, McBride WJH, Govan B, Pearson M, Ritchie SA. A survey on Mycobacterium ulcerans in mosquitoes and march flies captured from endemic areas of Northern Queensland, Australia. PLoS Negl Trop Dis 2019; 13: e0006745.
39. Singh A, William McBride JH, Govan B, Pearson M. Survey of local fauna from endemic areas of Northern Queensland, Australia for the presence of Mycobacterium ulcerans. Int J Mycobacteriol 2019; 8: 48-52.
40. Roltgen K, Pluschke G, Johnson PDR, Fyfe J. Mycobacterium ulcerans DNA in bandicoot excreta in Buruli ulcer-endemic area, Northern Queensland, Australia. Emerg Infect Dis 2017; 23: 2042-2045.
41. Loftus MJ, Trubiano JA, Tay EL, et al. The incubation period of Buruli ulcer (Mycobacterium ulcerans infection) in Victoria, Australia - remains similar despite changing geographic distribution of disease. PLoS Negl Trop Dis 2018; 12: e0006323.
42. Quek TY, Henry MJ, Pasco JA, et al. Mycobacterium ulcerans infection: factors influencing diagnostic delay. Med J Aust 2007; 187: 561-563.
43. Jenkin GA, Smith M, Fairley M, Johnson PD. Acute, oedematous Mycobacterium ulcerans infection in a farmer from far north Queensland. Med J Aust 2002; 176: 180-181.
44. O’Brien DP, Hughes AJ, Cheng AC, et al. Outcomes for Mycobacterium ulcerans infection with combined surgery and antibiotic therapy: findings from a south-eastern Australian case series. Med J Aust 2007; 186: 58-61.
45. Gordon CL, Buntine JA, Hayman JA, et al. All-oral antibiotic treatment for Buruli ulcer: a report of four patients. PLoS Negl Trop Dis 2010; 4: e770.
46. O’Brien DP, McDonald A, Callan P, et al. Successful outcomes with oral fluoroquinolones combined with rifampicin in the treatment of Mycobacterium ulcerans: an observational cohort study. PLoS Negl Trop Dis 2012; 6: e1473.
47. Sarfo FS, Phillips R, Asiedu K, et al. Clinical efficacy of combination of rifampin and streptomycin for treatment of Mycobacterium ulcerans disease. Antimicrob Agents Chemother 2010; 54: 3678-3685.
48. Etuaful S, Carbonnelle B, Grosset J, et al. Efficacy of the combination rifampin-streptomycin in preventing growth of Mycobacterium ulcerans in early lesions of Buruli ulcer in humans. Antimicrob Agents Chemother 2005; 49: 3182-3186.
49. Chauty A, Ardant MF, Marsollier L, et al. Oral treatment for Mycobacterium ulcerans infection: results from a pilot study in Benin. Clin Infect Dis 2011; 52: 94-96.
50. Nienhuis WA, Stienstra Y, Thompson WA, et al. Antimicrobial treatment for early, limited Mycobacterium ulcerans infection: a randomised controlled trial. Lancet 2010; 375: 664-672.
51. Muhi S, Cox VR, O’Brien M, et al. Management of Mycobacterium ulcerans infection (Buruli ulcer) in Australia: consensus statement. Med J Aust 2025; 222: 571-578.
52. O’Brien DP, Robson ME, Callan PP, McDonald AH. "Paradoxical" immune-mediated reactions to Mycobacterium ulcerans during antibiotic treatment: a result of treatment success, not failure. Med J Aust 2009; 191: 564-566.
53. Nienhuis WA, Stienstra Y, Abass KM, et al. Paradoxical responses after start of antimicrobial treatment in Mycobacterium ulcerans infection. Clin Infect Dis 2012; 54: 519-526.
54. O’Brien DP, Huffam S. Pre-emptive steroids for a severe oedematous Buruli ulcer lesion: a case report. J Med Case Rep 2015; 9: 98.
55. Trevillyan JM, Johnson PD. Steroids control paradoxical worsening of Mycobacterium ulcerans infection following initiation of antibiotic therapy. Med J Aust 2013; 198: 443-444.
56. Muhi S, Stinear TP. Systematic review of M. Bovis BCG and other candidate vaccines for Buruli ulcer prophylaxis. Vaccine 2021; 39: 7238-7252.
57. O’Brien DP, Blasdell K, Muhi S, et al. Is BCG vaccination of possums the solution to the Buruli ulcer epidemic in south-eastern Australia? Med J Aust 2023; 219: 520-522.
58. Buddle BM, Vordermeier HM, Chambers MA, de Klerk-Lorist LM. Efficacy and safety of BCG vaccine for control of tuberculosis in domestic livestock and wildlife. Front Vet Sci 2018; 5: 259.
59. Victoria State Government, Department of Health. Beating Buruli in Victoria. Melbourne: Victoria State Government, Department of Health; 2024. Available online at: https://www.health.vic.gov.au/infectious-diseases/beating-buruli-in-victoria (accessed November 2025).
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.