Tackling comorbidities in your patient with heart failure: practical management strategies for GPs

Heart failure most commonly affects older adults with multiple comorbidities. These coexisting conditions substantially increase symptom burden, polypharmacy, hospitalisation risk and mortality, while adding significant complexity to care. As most patients with heart failure are managed in the community, GPs play a central role in early recognition, longitudinal monitoring, treatment optimisation and care co-ordination.
- Comorbidities are common in heart failure (HF) and substantially worsen symptoms, hospitalisation risk and mortality.
- Many HF therapies provide prognostic benefits across multiple comorbidities commonly managed in general practice.
- Early recognition and proactive management of comorbidities in primary care can prevent HF decompensation and hospitalisation.
- Co-ordinated shared care between GPs and specialist services is essential for managing complex HF and supporting patient transition back to the community.
Heart failure (HF) is a clinical syndrome characterised by breathlessness, fatigue, fluid retention and reduced exercise tolerance resulting from structural or functional cardiac disease, supported by elevated natriuretic peptides and objective evidence of pulmonary or systemic congestion.1
Over recent decades, the clinical complexity of HF has increased because of population ageing, a rising incidence of multimorbidity and improved survival with guideline-directed medical therapy (GDMT).2 In Australia, nearly three-quarters of people living with HF are aged 65 years or older, and many have multiple chronic conditions that contribute to polypharmacy, functional decline and frequent hospitalisation.3 Most patients with HF have regular contact with their GP; data from the Study of Heart failure in the Australian Primary carE setting (SHAPE) study indicate that patients with HF visit their GP an average of 14 times per year.4 Consequently, GPs play a central role in early diagnosis, recognition of decompensation, treatment optimisation and care co-ordination. This article provides a practical primary care-focused framework to support GPs in identifying and managing common cardiac and noncardiac comorbidities in HF, prioritising therapies with shared prognostic benefit, recognising red flags requiring escalation and integrating shared care with specialist services to reduce preventable hospitalisation and mortality. Red flags requiring urgent same-day hospital assessment or specialist review are outlined in the Box.
Atrial fibrillation
Atrial fibrillation (AF) commonly coexists with HF, affecting 35 to 50% of patients.5 AF should be suspected in patients with new palpitations, worsening breathlessness, reduced exercise tolerance or unexplained functional decline. Initial assessment should include a 12-lead ECG and blood tests to evaluate electrolytes, renal function and thyroid function.
Management should prioritise early rate control and stroke prevention. In patients with heart failure with reduced ejection fraction, cardioselective beta blockers and digoxin are preferred, whereas beta blockers, diltiazem, verapamil or digoxin may be used in patients with heart failure with preserved ejection fraction (HFpEF).6 Anticoagulation should follow the updated CHA₂DS₂-VA score (Congestive heart failure or left ventricular dysfunction; Hypertension; Age 75 years and older; Diabetes mellitus; Stroke, transient ischaemic attack or thromboembolism history; Vascular disease history; and Age 65 to 74 years).6 Direct oral anticoagulants are preferred over vitamin K antagonists, except in patients with mechanical heart valves, moderate-to-severe mitral stenosis (valvular AF) or severe renal impairment (creatinine clearance <15 mL/min).5,6 Anticoagulant dose adjustments should follow established criteria.6 A cardiology referral is appropriate for cases involving complex anticoagulation decisions, inadequate rate or rhythm control or consideration of rhythm control strategies, including catheter ablation.
Coronary artery disease
Coronary artery disease is a common contributor to HF and may present either as acute coronary syndrome complicated by HF, or as progressive ischaemic cardiomyopathy due to chronic myocardial ischaemia.7 Initial investigations include ECG and transthoracic echocardiography to assess left ventricular function and the presence of regional wall motion abnormalities. Further assessment may involve functional exercise testing or CT coronary angiography. Persistent angina, recurrent HF decompensation and otherwise unexplained left ventricular dysfunction should prompt cardiology referral for further assessment, including the consideration of coronary angiography and revascularisation where appropriate.7
Lipid-lowering therapy remains an important component of care in patients with HF and concomitant atherosclerotic cardiovascular disease (ASCVD) or high cardiovascular risk. LDL-cholesterol (LDL-C) targets should be individualised according to the patient’s overall cardiovascular risk. Patients with moderate chronic kidney disease (CKD; estimated glomerular filtration rate [eGFR] 30–59mL/min/1.73 m2) are considered high risk, with an LDL-C target of less than 1.8 mmol/L. Those with established ASCVD or type 2 diabetes with target organ damage are considered to be at very high risk, with an LDL-C target of less than 1.4 mmol/L. Patients with recurrent ASCVD events despite maximal statin- based therapy, or polyvascular arterial disease, are considered to be at extreme risk with a recommended LDL-C target of less than 1 mmol/L.8
Hypertension
Hypertension frequently precedes the development of HF and remains a major modifiable driver of disease progression.9 Chronic pressure overload promotes maladaptive ventricular remodelling and neurohormonal activation.10-12 Initial investigations and screening may be guided by recommendations outlined in the 2024 European Society of Cardiology Guidelines for the Management of Elevated Blood Pressure and Hypertension.13
The guidelines recommend blood pressure (BP) targets of 120/70 to 129/79 mmHg, provided treatment is tolerated.13 In HFpEF, sodium-glucose cotransporter-2 (SGLT-2) inhibitors reduce HF hospitalisation and should be initiated irrespective of the baseline BP.13 Additional BP lowering may be achieved with mineralocorticoid receptor antagonists, including finerenone, which has demonstrated reductions in cardiovascular mortality and HF hospitalisation.14 ACE inhibitors or angiotensin II receptor blockers may also be used if further BP control is required.1,13,15
In heart failure with reduced ejection fraction, GDMT should be prioritised. This includes ACE inhibitors, angiotensin II receptor blockers or angiotensin receptor-neprilysin inhibitors, HF-selective beta blockers (bisoprolol, metoprolol extended-release, carvedilol or nebivolol), mineralocorticoid receptor antagonists and SGLT-2 inhibitors.13,15,16 Low BP alone should not prompt dose reduction or cessation unless accompanied by symptomatic hypotension or evidence of end-organ hypoperfusion.17
Type 2 diabetes
Type 2 diabetes and HF share a complex bidirectional relationship, whereby the presence of type 2 diabetes increases the risk of HF, and established HF promotes insulin resistance and worsens glycaemic control.18-20 About 40% of patients with HF have coexisting type 2 diabetes, and the presence of type 2 diabetes is associated with worse prognosis, higher rates of hospitalisation and increased mortality.21,22 Conversely, patients with type 2 diabetes who develop HF experience poorer outcomes compared with those without HF.
Glycaemic targets in patients with HF should be individualised, with less stringent goals appropriate for older or frail patients. Preference should be given to glucose-lowering therapies with a proven HF benefit, particularly SGLT-2 inhibitors, which reduce HF hospitalisation and cardiovascular mortality irrespective of baseline glycaemic control.23-34 Incretin-based therapies have also demonstrated benefit in selected populations, with recent trials showing improvements in symptoms, reductions in neurohormonal activation and fewer HF hospitalisations in patients with overweight or obesity and HFpEF.35-37
Obesity
Obesity increases the risk of HF both indirectly through associated conditions such as hypertension, diabetes and coronary artery disease, and directly via obesity-related cardiomyopathy.38 In patients with established HF, obesity is associated with a greater symptom burden and reduced functional capacity.
Assessment should extend beyond the body mass index to include the waist circumference, waist-to-hip ratio or waist-to-height ratio, which better reflect an individual’s cardiometabolic risk.39 N-terminal pro B-type natriuretic peptide (NT-proBNP) concentrations may be suppressed in obesity and should be interpreted cautiously.
Sustained weight loss remains the cornerstone of management. Weight reductions greater than 10% improve functional capacity and cardiovascular outcomes, particularly in patients with HFpEF.40-42 Glucagon-like peptide-1 receptor agonists can achieve significant weight loss and have demonstrated improvements in symptoms and reductions in HF hospitalisations, although a definitive survival benefit has yet to be established.36,43,44
Chronic kidney disease
CKD frequently coexists with HF and accelerates disease progression in both organs.45 Routine monitoring of the eGFR and albuminuria is essential, as even mild abnormalities are associated with worse prognosis.46-53
CKD should not delay the initiation or continuation of GDMT, provided appropriate monitoring is in place. In patients with an eGFR greater than 30 mL/min/1.73 m2, GDMT reduces mortality and HF hospitalisation and slows renal decline.54-60 A modest early reduction in eGFR after therapy initiation is common and usually transient. A nephrology referral is appropriate for cases of advanced CKD, rapid renal decline, refractory hyperkalaemia or uncertainty regarding GDMT continuation, or consideration of renal replacement therapy.
Pulmonary disease
Chronic obstructive pulmonary disease (COPD) and sleep-disordered breathing commonly coexist with HF and contribute to dyspnoea and functional limitations. Distinguishing HF decompensation from pulmonary exacerbations is challenging, and both may coexist.
Initial investigations should include a chest x-ray and assessment of NT-proBNP levels. Low NT-proBNP levels make HF unlikely, although elevations may occur during COPD exacerbations, particularly with renal dysfunction or AF.61,62 Management priorities include smoking cessation, optimisation of inhaler therapy and the prevention of exacerbations.63,64 In patients requiring systemic corticosteroids, careful monitoring for fluid retention and HF deterioration is essential. Cardioselective beta blockers should not be withheld in patients with COPD, given their established prognostic benefits in HF.65-67 Vaccination against influenza, severe acute respiratory syndrome coronavirus 2, pneumococcus, pertussis, respiratory syncytial virus and shingles is recommended. Respiratory referral should be considered for persistent or worsening dyspnoea despite optimised HF therapy, frequent exacerbations, unexplained hypoxia or recent hospitalisation.
Sleep-disordered breathing includes obstructive sleep apnoea and central sleep apnoea. The former results from upper airway collapse, whereas the latter results from delayed circulatory feedback between pulmonary capillaries and carotid chemoreceptors, leading to cyclical hyperventilation and apnoea.68 Management of obstructive sleep apnoea includes optimisation of HF therapy, weight management and positive airway pressure therapy. Positive airway pressure is not recommended in central sleep apnoea.
Anaemia and iron deficiency
Anaemia and iron deficiency affect up to 80% of patients with HF and are associated with reduced exercise capacity, increased hospitalisation and higher mortality.69,70 Iron deficiency frequently occurs independently of anaemia and may be overlooked without targeted testing.
Anaemia is defined by haemoglobin levels less than 130 g/L in men and less than 120 g/L in women.71 In HF, absolute iron deficiency is defined as ferritin levels less than 100 mcg/L, and functional iron deficiency as ferritin levels of 100 to 299 mcg/L with transferrin saturation less than 20%.16
Iron studies should be included in routine HF blood monitoring. Early studies have demonstrated that intravenous iron therapy, including ferric carboxymaltose and ferric derisomaltose, improves symptoms, exercise capacity and quality of life, and reduces HF hospitalisations irrespective of anaemia status.72-79 Although recent individual randomised controlled trials did not demonstrate a clear clinical benefit when considered individually, a contemporary meta-analysis suggests that intravenous iron supplementation reduces HF hospitalisations and, to a lesser extent, all-cause mortality.80 Oral iron supplementation, blood transfusion and erythropoietin-stimulating agents are not recommended and may increase adverse events.81-84
Frailty, sarcopenia and falls
Frailty is highly prevalent in HF and is associated with increased hospitalisation, disability and mortality, contributing to symptom burden and medication intolerance.85 Recognition of frailty supports individualised care planning and timely consideration of supportive or palliative approaches.
Frailty screening should be integrated into chronic disease management plans and medication reviews, using validated tools including the Fried phenotype, Rockwood Clinical Frailty Scale, Barthel Index, Edmonton Frail Scale and Heart Failure Association Frailty Score.86-94
Frailty alone should not result in withholding GDMT, as the prognostic benefits persist in older and frail populations.95-99 Careful dose titration, close monitoring and referral to cardiac rehabilitation can improve functional outcomes.85,100-102
Depression, anxiety and cognitive impairment
Depression, anxiety and cognitive impairment are common in HF and increase with disease severity.103 Reduced cardiac output may impair cerebral perfusion, whereas depression is independently associated with increased healthcare utilisation, HF hospitalisation and mortality.104-108
Persistent symptoms, difficulty with self-care or poor treatment adherence should prompt assessment using validated screening tools, including the Patient Health Questionnaire-9, Beck Depression Inventory, Cardiac Depression Scale, Geriatric Depression Scale, Mini-Mental State Exam or Montreal Cognitive Assessment.109-113
Referral to a psychologist, geriatrician or psychiatrist should be considered for patients with persistent symptoms, safety concerns or impaired self-management, with involvement of carers to support adherence and shared decision-making.
Conclusion
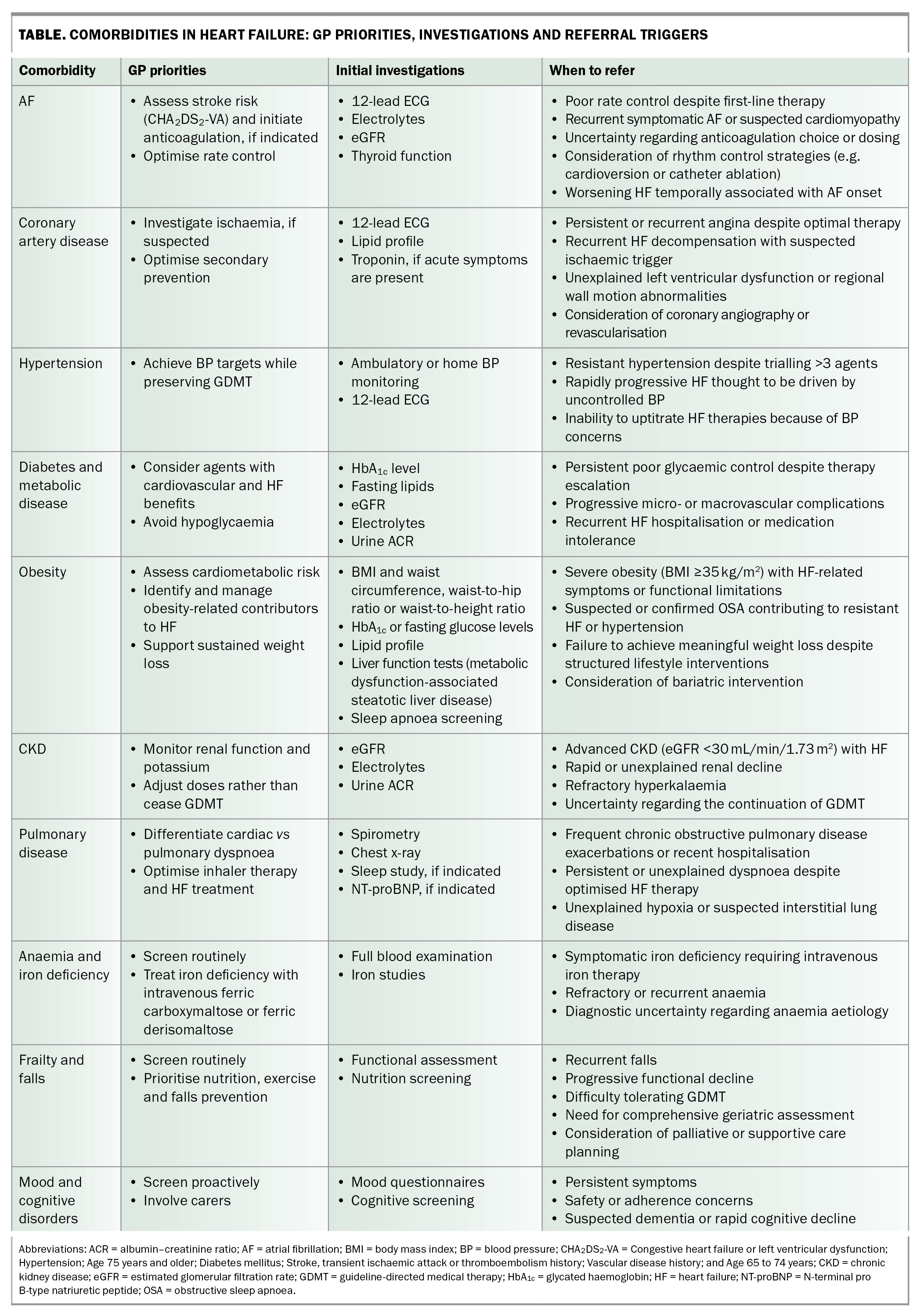
HF is a chronic multisystem condition managed predominantly in the community. Comorbidities are highly prevalent and are major determinants of symptom burden, treatment tolerance, hospitalisation and survival. Early identification and proactive management of these conditions are therefore central to improving outcomes. Common comorbidities in HF and associated GP management priorities are summarised in the Table.
GPs play a pivotal role in delivering this care. Many HF therapies provide benefit across multiple comorbidities, allowing cardiovascular, metabolic, renal and functional risk to be addressed in parallel. Co-ordinated shared care between general practice and specialist services remains essential to delivering sustainable, high-quality care for patients living with HF. MT
COMPETING INTERESTS: Dr Eng-Frost has received honoraria from Boehringer-Ingelheim, Novartis, Menarini and Bayer; support for attending meetings from Novo Nordisk, Astra Zeneca, Boehringer-Ingelheim and Novartis; and research support from Medtronic. Dr Chan has received honoraria from Astra Zeneca, Boehringer-Ingelheim, Eli Lilly, Novo Nordisk, Medtronic, Abbott and Biotronik; has received support for attending meetings from Novo Nordisk, Astra Zeneca, Boehringer-Ingelheim, Phebra, Medtronic, Abbott and Biotronik; has participated in advisory boards for Novo Nordisk, Boehringer-Ingelheim, Eli Lilly, Astra Zeneca and Phebra; is a Member of the Heart Foundation Advisory Board; and has received items or services from Astra Zeneca.
References
1. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2022; 145: e895-e1032.
2. Wong CY, Chaudhry SI, Desai MM, Krumholz HM. Trends in comorbidity, disability and polypharmacy in heart failure. Am J Med 2011; 124: 136-143.
3. Australian Institute of Health and Welfare (AIHW). Heart, stroke and vascular disease: Australian facts. Canberra: AIHW; 2025. Available online at: https://www.aihw.gov.au/reports/heart-stroke-vascular-diseases/hsvd-facts/contents/about (accessed February 2026).
4. Audehm R, Neville AM, Piazza P, et al. Healthcare services use by patients with heart failure in Australia. Aust J Gen Pract 2022; 51: 713-720.
5. Gopinathannair R, Olshansky B, Sullivan RM, et al. Managing atrial fibrillation in patients with heart failure and reduced ejection fraction: a scientific statement from the American Heart Association. Circ Arrhythm Electrophysiol 2021; 14: e0000078.
6. Van Gelder IC, Rienstra M, Crijns HJGM, et al. 2024 ESC guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2024; 45: 3314-3414.
7. Flaherty JD, Bax JJ, De Luca L, et al. Acute heart failure syndromes in patients with coronary artery disease: early assessment and treatment. J Am Coll Cardiol 2009; 53: 254-263.
8. Mach F, Baigent C, Catapano AL, et al. 2025 focused update of the 2019 ESC/EAS guidelines for the management of dyslipidaemias. Eur Heart J 2025; 46: 4359-4378.
9. Levy D, Larson MG, Vasan RS, Kannel WB, Ho KK. The progression from hypertension to congestive heart failure. JAMA 1996; 275: 1557-1562.
10. Messerli FH, Rimoldi SF, Bangalore S. The transition from hypertension to heart failure: contemporary update. JACC Heart Fail 2017; 5: 543-551.
11. Gallo G, Savoia C. Hypertension and heart failure: from pathophysiology to treatment. Int J Mol Sci 2024; 25: 6661.
12. Slivnick J, Lampert BC. Hypertension and heart failure. Heart Fail Clin 2019; 15: 531-541.
13. McEvoy JW, McCarthy CP, Bruno RM, et al. 2024 ESC guidelines for the management of elevated blood pressure and hypertension. Eur Heart J 2024; 45: 3912-4018.
14. Solomon SD, McMurray JJV, Claggett B, et al. Finerenone in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med 2024; 391: 1475-1485.
15. McDonagh TA, Metra M, Adamo M, et al. 2023 focused update of the 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2023; 44: 3627-3639.
16. McDonagh TA, Metra M, Adamo M, et al. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2021; 42: 3599-3726.
17. Oh GC, Cho HJ. Blood pressure and heart failure. Clin Hypertens 2020; 26: 1.
18. Gottdiener JS, Arnold AM, Aurigemma GP, et al. Predictors of congestive heart failure in the elderly: the Cardiovascular Health Study. J Am Coll Cardiol 2000; 35: 1628-1637.
19. Chadalavada S, Sathian K, Blaha MJ, et al. Women with diabetes are at increased relative risk of heart failure compared to men: insights from UK Biobank. Front Cardiovasc Med 2021; 8: 658726.
20. Ohkuma T, Komorita Y, Peters SAE, et al. Diabetes as a risk factor for heart failure in women and men: a systematic review and meta-analysis of 47 cohorts including 12 million individuals. Diabetologia 2019; 62: 1550-1560.
21. Dei Cas A, Khan SS, Butler J, et al. Impact of diabetes on epidemiology, treatment and outcomes of patients with heart failure. JACC Heart Fail 2015; 3: 136-145.
22. Win TT, Davis HT, Laskey WK. Mortality among patients hospitalized with heart failure and diabetes mellitus: results from the National Inpatient Sample 2000 to 2010. Circ Heart Fail 2016; 9: e003023.
23. Vermes E, Ducharme A, Bourassa MG, et al. Enalapril reduces the incidence of diabetes in patients with chronic heart failure: insight from the Studies of Left Ventricular Dysfunction (SOLVD). Circulation 2003; 107: 1291-1296.
24. MacDonald MR, Petrie MC, Hawkins NM, et al. Impact of diabetes on outcomes in patients with low and preserved ejection fraction heart failure: an analysis of the CHARM programme. Eur Heart J 2008; 29: 1377-1385.
25. Yusuf S, Ostergren JB, Gerstein HC, et al. Effects of candesartan on the development of a new diagnosis of diabetes mellitus in patients with heart failure. Circulation 2005; 112: 48-53.
26. Maggioni AP, Anand I, Gottlieb SO, et al. Effects of valsartan on morbidity and mortality in patients with heart failure not receiving angiotensin-converting enzyme inhibitors. J Am Coll Cardiol 2002; 40: 1414-1421.
27. Konstam MA, Neaton JD, Dickstein K, et al. Effects of high-dose versus low-dose losartan on clinical outcomes in patients with heart failure (HEAAL study): a randomised, double-blind trial. Lancet 2009; 374: 1840-1848.
28. Ryden L, Armstrong PW, Cleland JGF, et al. Efficacy and safety of high-dose lisinopril in chronic heart failure patients at high cardiovascular risk, including those with diabetes mellitus: results from the ATLAS trial. Eur Heart J 2000; 21: 1967-1978.
29. Deedwania PC, Gottlieb S, Ghali JK, et al. Efficacy, safety and tolerability of metoprolol CR/XL in patients with diabetes and chronic heart failure: experiences from MERIT-HF. Am Heart J 2005; 149: 159-167.
30. Packer M, Coats AJS, Fowler MB, et al. Effect of carvedilol on survival in severe heart failure. N Engl J Med 2001; 344: 1651-1658.
31. Torp-Pedersen C, Metra M, Spark P, et al. Effects of metoprolol and carvedilol on pre-existing and new-onset diabetes in patients with chronic heart failure: data from the COMET trial. Heart 2007; 93: 968-973.
32. Erdmann E, Lechat P, Verkenne P, et al. Results from post-hoc analyses of the CIBIS II trial: effect of bisoprolol in high-risk patient groups with chronic heart failure. Eur J Heart Fail 2001; 3: 469-479.
33. Packer M, Fowler MB, Roecker EB, et al. Effect of carvedilol on the morbidity of patients with severe chronic heart failure: results of the COPERNICUS study. Circulation 2002; 106: 2194-2199.
34. Seferovic JP, Claggett B, Seidelmann SB, et al. Effect of sacubitril/valsartan versus enalapril on glycaemic control in patients with heart failure and diabetes: a post-hoc analysis from the PARADIGM-HF trial. Lancet Diabetes Endocrinol 2017; 5: 333-340.
35. Kosiborod MN, Abildstrom SZ, Borlaug BA, et al. Semaglutide in patients with obesity-related heart failure and type 2 diabetes. N Engl J Med 2024; 390: 1394-1407.
36. Packer M, Anker SD, Butler J, et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N Engl J Med 2025; 392: 427-437.
37. Packer M, Anker SD, Butler J, et al. Influence of type 2 diabetes on the effects of tirzepatide in patients with heart failure with preserved ejection fraction and obesity: a prespecified stratification-based analysis. J Am Coll Cardiol 2025; 86: 696-707.
38. Ebong IA, Goff DC Jr, Rodriguez CJ, et al. Mechanisms of heart failure in obesity. Obes Res Clin Pract 2014; 8: e540-e548.
39. Kittleson MM, Shah SJ, Vaduganathan M, et al. 2025 ACC scientific statement on the management of obesity in adults with heart failure: a report of the American College of Cardiology. J Am Coll Cardiol 2025; 86: 1953-1975.
40. Patel KV, Bahnson JL, Gaussoin SA, et al. Optimal cardiometabolic health and risk of heart failure in type 2 diabetes: an analysis from the Look AHEAD trial. Eur J Heart Fail 2022; 24: 2037-2047.
41. El Hajj EC, Pibarot P, Despres JP, et al. Pragmatic weight management program for patients with obesity and heart failure with preserved ejection fraction. J Am Heart Assoc 2021; 10: e022930.
42. Look AHEAD Research Group; Gregg E, Jakicic J, Blackburn G, et al. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: a post-hoc analysis of the Look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol 2016; 4: 913-921.
43. Kosiborod MN, Borlaug BA, Abildstrom SZ, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med 2023; 389: 1069-1084.
44. Butler J, Packer M, Filippatos G, et al. Semaglutide versus placebo in people with obesity-related heart failure with preserved ejection fraction: a pooled analysis of the STEP-HFpEF and STEP-HFpEF DM randomised trials. Lancet 2024; 403: 1635-1648.
45. Beldhuis IE, Streng KW, Ter Maaten JM, et al. Evidence-based medical therapy in patients with heart failure with reduced ejection fraction and chronic kidney disease. Circulation 2022; 145: 693-712.
46. Kobayashi M, Girerd N, McMurray JJV, et al. Urinary markers in heart failure: types, timing and thresholds. Eur J Heart Fail 2025; 27: 2372-2396.
47. Jackson CE, Solomon SD, Gerstein HC, et al. Albuminuria in chronic heart failure: prevalence and prognostic importance. Lancet 2009; 374: 543-550.
48. Masson S, Latini R, Milani V, et al. Prevalence and prognostic value of elevated urinary albumin excretion in patients with chronic heart failure: data from the GISSI-Heart Failure trial. Circ Heart Fail 2010; 3: 65-72.
49. Anand IS, Bishu K, Rector TS, et al. Proteinuria, chronic kidney disease and the effect of an angiotensin receptor blocker in addition to an angiotensin-converting enzyme inhibitor in patients with moderate to severe heart failure. Circulation 2009; 120: 1577-1584.
50. Lathem W, Maddox DA, Brenner BM, et al. Urinary protein excretion and renal haemodynamic adjustments during orthostasis in patients with acute and chronic renal diseases. J Clin Invest 1954; 33: 1457-1465.
51. Boorsma EM, Ter Maaten JM, Damman K, et al. Albuminuria as a marker of systemic congestion in patients with heart failure. Eur Heart J 2023; 44: 368-380.
52. Oka T, Ter Maaten JM, Damman K, et al. Mineralocorticoid receptor antagonist use and hard renal outcomes in real-world patients with chronic kidney disease. Hypertension 2022; 79: 679-689.
53. McCausland FR, Claggett BL, Jhund PS, et al. Finerenone and kidney outcomes in patients with heart failure: the FINEARTS-HF trial. J Am Coll Cardiol 2025; 85: 159-168.
54. Khan MS, Butler J, Gheorghiade M, et al. Albuminuria and heart failure: a JACC state-of-the-art review. J Am Coll Cardiol 2023; 81: 270-282.
55. Kotecha D, Holmes J, Krum H, et al. Impact of renal impairment on beta-blocker efficacy in patients with heart failure. J Am Coll Cardiol 2019; 74: 2893-2904.
56. Damman K, Gori M, Claggett B, et al. Renal effects and associated outcomes during angiotensin–neprilysin inhibition in heart failure. JACC Heart Fail 2018; 6: 489-498.
57. Packer M, Anker SD, Butler J, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med 2020; 383: 1413-1424.
58. Zannad F, Ferreira JP, Pocock SJ, et al. Cardiac and kidney benefits of empagliflozin in heart failure across the spectrum of kidney function: insights from EMPEROR-Reduced. Circulation 2021; 143: 310-321.
59. Heerspink HJL, Stefansson BV, Correa-Rotter R, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020; 383: 1436-1446.
60. Jhund PS, Solomon SD, Docherty KF, et al. Efficacy of dapagliflozin on renal function and outcomes in patients with heart failure with reduced ejection fraction: results of DAPA-HF. Circulation 2021; 143: 298-309.
61. Cuthbert JJ, Pellicori P, Clark AL. Optimal management of heart failure and chronic obstructive pulmonary disease: clinical challenges. Int J Gen Med 2022; 15: 7961-7975.
62. de Miguel-Diez J, Jimenez-Garcia R, Hernandez-Barrera V, et al. Multidisciplinary management of patients with chronic obstructive pulmonary disease and cardiovascular disease. Arch Bronconeumol 2024; 60: 226-237.
63. Lipson DA, Barnhart F, Brealey N, et al. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med 2018; 378: 1671-1680.
64. Rabe KF, Martinez FJ, Ferguson GT, et al. Triple inhaled therapy at two glucocorticoid doses in moderate-to-very-severe COPD. N Engl J Med 2020; 383: 35-48.
65. Vestbo J, Hurd SS, Agusti AG, et al. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med 2013; 187: 347-365.
66. Lipworth B, Wedzicha J, Devereux G, et al. Beta-blockers in COPD: time for reappraisal. Eur Respir J 2016; 48: 880-888.
67. Yang YL, Xiang ZJ, Yang JH, et al. Association of beta-blocker use with survival and pulmonary function in patients with chronic obstructive pulmonary and cardiovascular disease: a systematic review and meta-analysis. Eur Heart J 2020; 41: 4415-4422.
68. Cowie MR, Linz D, Redline S, et al. Sleep-disordered breathing and cardiovascular disease: a JACC state-of-the-art review. J Am Coll Cardiol 2021; 78: 608-624.
69. Klip IT, Comin-Colet J, Voors AA, et al. Iron deficiency in chronic heart failure: an international pooled analysis. Am Heart J 2013; 165: 575-582.e3.
70. Jankowska EA, Rozentryt P, Witkowska A, et al. Iron deficiency: an ominous sign in patients with systolic chronic heart failure. Eur Heart J 2010; 31: 1872-1880.
71. Cappellini MD, Motta I. Anemia in clinical practice: definition and classification – does hemoglobin change with aging? Semin Hematol 2015; 52: 261-269.
72. Anker SD, Colet JC, Filippatos G, et al. Ferric carboxymaltose in patients with heart failure and iron deficiency. N Engl J Med 2009; 361: 2436-2448.
73. Beck-da-Silva L, Piardi D, Soder S, et al. IRON-HF study: a randomized trial to assess the effects of iron in heart failure patients with anemia. Int J Cardiol 2013; 168: 3439-3442.
74. Ponikowski P, van Veldhuisen DJ, Comin-Colet J, et al. Beneficial effects of long-term intravenous iron therapy with ferric carboxymaltose in patients with symptomatic heart failure and iron deficiency. Eur Heart J 2015; 36: 657-668.
75. Ponikowski P, Kirwan BA, Anker SD, et al. Ferric carboxymaltose for iron deficiency at discharge after acute heart failure: a multicentre, double-blind, randomised, controlled trial. Lancet 2020; 396: 1895-1904.
76. Graham FJ, Pellicori P, Cleland JGF, et al. Treating iron deficiency in patients with heart failure: what, why, when, how, where and who. Heart 2024; 110: 1201-1207.
77. Jankowska EA, Tkaczyszyn M, Suchocki T, et al. Effects of intravenous iron therapy in iron-deficient patients with systolic heart failure: a meta-analysis of randomized controlled trials. Eur J Heart Fail 2016; 18: 786-795.
78. Anker SD, Kirwan BA, van Veldhuisen DJ, et al. Effects of ferric carboxymaltose on hospitalisations and mortality rates in iron-deficient heart failure patients: an individual patient data meta-analysis. Eur J Heart Fail 2018; 20: 125-133.
79. Kalra PR, Cleland JGF, Petrie MC, et al. Intravenous ferric derisomaltose in patients with heart failure and iron deficiency in the UK (IRONMAN): an investigator-initiated, prospective, randomised, open-label, blinded-endpoint trial. Lancet 2022; 400: 2199-2209.
80. Anker SD, Comin-Colet J, Filippatos G, et al. Systematic review and meta-analysis of intravenous iron therapy for patients with heart failure and iron deficiency. Nat Med 2025; 31: 2640-2646.
81. Grote Beverborg N, van Veldhuisen DJ, van der Meer P. Anemia in heart failure: still relevant? JACC Heart Fail 2018; 6: 201-208.
82. Swedberg K, Young JB, Anand IS, et al. Treatment of anemia with darbepoetin alfa in systolic heart failure. N Engl J Med 2013; 368: 1210-1219.
83. Garty M, Shotan A, Gottlieb S, et al. Blood transfusion for acute decompensated heart failure: friend or foe? Am Heart J 2009; 158: 653-658.
84. Lewis GD, Malhotra R, Hernandez AF, et al. Effect of oral iron repletion on exercise capacity in patients with heart failure with reduced ejection fraction and iron deficiency: the IRONOUT HF randomized clinical trial. JAMA 2017; 317: 1958-1966.
85. McDonagh J, Curnow J, Driscoll A, et al. An expert opinion on the management of frailty in heart failure from the Australian Cardiovascular Alliance National Taskforce. Heart Lung Circ 2025; 34: 693-703.
86. Saum KU, Dieffenbach AK, Muller H, et al. Development and evaluation of a modification of the Fried frailty criteria using population-independent cutpoints. J Am Geriatr Soc 2012; 60: 2110-2115.
87. Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 2001; 56A: M146-M156.
88. Rockwood K, Song X, MacKnight C, et al. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005; 173: 489-495.
89. Searle SD, Mitnitski A, Gahbauer EA, et al. A standard procedure for creating a frailty index. BMC Geriatr 2008; 8: 24.
90. Chivite D, Formiga F, Corbella X, et al. Basal functional status predicts one-year mortality after a heart failure hospitalization in elderly patients: the RICA prospective study. Int J Cardiol 2018; 254: 182-188.
91. Formiga F, Chivite D, Conde A, et al. Basal functional status predicts three-month mortality after a heart failure hospitalization in elderly patients: the prospective RICA study. Int J Cardiol 2014; 172: 127-131.
92. Graham MM, Galbraith PD, O’Neill D, et al. Frailty and outcome in elderly patients with acute coronary syndrome. Can J Cardiol 2013; 29: 1610-1615.
93. Sze S, Pellicori P, Zhang J, et al. Identification of frailty in chronic heart failure. JACC Heart Fail 2019; 7: 291-302.
94. Vitale C, Jankowska E, Hill L, et al. Heart Failure Association/European Society of Cardiology position paper on frailty in patients with heart failure. Eur J Heart Fail 2019; 21: 1299-1305.
95. Greene SJ, Fonarow GC, DeVore AD, et al. Clinical effectiveness of sacubitril/valsartan among patients hospitalized for heart failure with reduced ejection fraction. J Am Heart Assoc 2021; 10: e021459.
96. Yaku H, Ozasa N, Morimoto T, et al. Association of mineralocorticoid receptor antagonist use with all-cause mortality and hospital readmission in older adults with acute decompensated heart failure. JAMA Netw Open 2019; 2: e195892.
97. Butt JH, Docherty KF, Jhund PS, et al. Efficacy and safety of dapagliflozin according to frailty in heart failure with reduced ejection fraction: a post hoc analysis of the DAPA-HF trial. Ann Intern Med 2022; 175: 820-830.
98. Hernandez AF, Greiner MA, Fonarow GC, et al. Clinical effectiveness of beta-blockers in heart failure: findings from the OPTIMIZE-HF registry. J Am Coll Cardiol 2009; 53: 184-192.
99. Butt JH, Claggett BL, Jhund PS, et al. Efficacy and safety of dapagliflozin according to frailty in patients with heart failure: a prespecified analysis of the DELIVER trial. Circulation 2022; 146: 1210-1224.
100. Pandey A, Kitzman DW, Brubaker P, et al. Frailty status modifies the efficacy of exercise training among patients with chronic heart failure and reduced ejection fraction: an analysis from the HF-ACTION trial. Circulation 2022; 146: 80-90.
101. Savage PA, Shaw AO, Miller MS, et al. Effect of resistance training on physical disability in chronic heart failure. Med Sci Sports Exerc 2011; 43: 1379-1386.
102. Sun X, Ni Y, Chu Y, et al. Comparative effectiveness of non-pharmacological interventions for frailty: a systematic review and network meta-analysis. Age Ageing 2023; 52: afad004.
103. Angermann CE, Ertl G. Depression, anxiety and cognitive impairment: comorbid mental health disorders in heart failure. Curr Heart Fail Rep 2018; 15: 398-410.
104. van Nieuwkerk AC, Hofman A, Ikram MA, et al. Cognitive impairment in patients with cardiac disease: implications for clinical practice. Stroke 2023; 54: 2181-2191.
105. Ovsenik A, Podbregar M, Fabjan A. Cerebral blood flow impairment and cognitive decline in heart failure. Brain Behav 2021; 11: e02176.
106. Rutledge T, Reis VA, Linke SE, et al. Depression in heart failure: a meta-analytic review of prevalence, intervention effects and associations with clinical outcomes. J Am Coll Cardiol 2006; 48: 1527-1537.
107. Vaccarino V, Kasl SV, Abramson J, et al. Depressive symptoms and risk of functional decline and death in patients with heart failure. J Am Coll Cardiol 2001; 38: 199-205.
108. Moraska AR, Chamberlain AM, Shah ND, et al. Depression, healthcare utilization and death in heart failure: a community study. Circ Heart Fail 2013; 6: 387-394.
109. Cameron J, Worrall-Carter L, Page K, et al. Screening for mild cognitive impairment in patients with heart failure: Montreal cognitive assessment versus mini-mental state examination. Eur J Cardiovasc Nurs 2013; 12: 252-260.
110. Davis KK, Allen JK. Identifying cognitive impairment in heart failure: a review of screening measures. Heart Lung 2013; 42: 92-97.
111. Lahlou-Laforet K, Ledru F, Potard C, et al. Validity of the Beck Depression Inventory for the assessment of depressive mood in chronic heart failure patients. J Affect Disord 2015; 184: 256-260.
112. Hare DL, Davis CR. Cardiac Depression Scale: validation of a new depression scale for cardiac patients. J Psychosom Res 1996; 40: 379-386.
113. Hammash MH, Hall LA, Lennie TA, et al. Psychometrics of the PHQ-9 as a measure of depressive symptoms in patients with heart failure. Eur J Cardiovasc Nurs 2013; 12: 446-453.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.