Heart failure with reduced ejection fraction: a 2026 update on management

Heart failure with reduced ejection fraction is a common, progressive condition with high morbidity and mortality. However, patient outcomes can be dramatically improved with early recognition and rapid initiation and up-titration of guideline-directed therapy. A structured, primary care-focused approach to diagnosis, risk stratification and optimisation of pharmacological and nonpharmacological management is essential to slow disease progression and reduce hospitalisations.
- Prevention of heart failure progression is crucial in the overall management of heart failure, which involves addressing modifiable risk factors and simultaneous and rapid up-titration of evidence-based therapies, including angiotensin receptor–neprilysin inhibitors, beta blockers, mineralocorticoid receptor antagonists and sodium-glucose cotransporter-2 inhibitors.
- Close monitoring for side effects of therapy and signs of progression, such as fluid overload and hypotension, are required, especially during periods of medication titration.
- Addressing barriers to medication adherence is particularly important in this chronic condition that disproportionately affects vulnerable populations.
- The burden of heart failure with reduced ejection fraction will likely increase in years to come, which makes preventive strategies and a dedicated effort to implement evidence-based therapies imperative. These strategies must be carefully delivered with cultural sensitivity, tools to address barriers to medication adherence and an awareness of the need for multidisciplinary input, including cardiac rehabilitation and palliative care at an appropriate time.
Heart failure is a clinical syndrome with several cardinal symptoms that are due to a functional or structural abnormality of the heart. It results in elevated intracardiac pressures, inadequate cardiac output, or both, at rest or during exercise.1 These elevated filling pressures can be assessed based on natriuretic peptide levels, echocardiography or invasive measures through cardiac catheterisation. Typical symptoms include breathlessness, ankle swelling, orthopnoea, reduced exercise tolerance and fatigue, which are often associated with signs on clinical examination that include a raised jugular venous pressure, pulmonary crackles and peripheral oedema. Less commonly, but still importantly, heart failure can present with nocturnal cough, wheeze, bloating, loss of appetite, dizziness and syncope. Weight gain, cachexia, tachycardia, a third heart sound, a laterally displaced apex beat and oliguria are other important signs on examination. It is commonly associated with a history of myocardial infarction, coronary artery disease, diabetes mellitus, alcohol misuse, chronic kidney disease, cardiotoxic chemotherapy and a family history of cardiomyopathies or sudden cardiac death.1,2
Heart failure is typically classified according to phenotype based on left ventricular systolic function, measured by the left ventricular ejection fraction (LVEF). This is because original treatment trials demonstrated improved outcomes in patients with an LVEF of 40% or less. However, heart failure can occur across a spectrum of left ventricular function, and the diagnosis is based on clinical findings and investigations. Other ways of classifying heart failure include by New York Heart Association (NYHA) class, aetiology and disease progression according to the presence of symptoms.3 Distinction between categories is important because medical therapy recommendations differ. The classifications of heart failure as per European guidelines are described in Box 1.11 This article focuses on heart failure with reduced ejection fraction (HFrEF).
Right ventricular dysfunction is a related but separate entity; it is generally the result of pressure or volume overload of the right ventricle.3 The main aetiology for right ventricular failure is left ventricular dysfunction-induced pulmonary hypertension. Other causes may be myocardial infarction, arrhythmogenic right ventricular cardiomyopathy, valvular disease, chronic lung disease, severe obstructive sleep apnoea, primary thromboembolic disease and congenital heart disease. This is beyond the scope of this article.
In the GP setting, heart failure can present acutely or as a state of chronic heart failure.1 Chronic heart failure embodies those individuals with an established diagnosis of heart failure who have a more gradual onset or progression of symptoms. Deteriorations or progression of symptoms may be labelled as ‘decompensated’ heart failure and manifest with the aforementioned symptoms and signs of congestion and reduced cardiac output. Heart failure can also present more acutely and may require admission to the hospital, especially if there is concern for significant congestion needing intravenous diuretics, vasodilators or inotropes, as well as high symptom burden, reduced cardiac output or likelihood of deterioration.
Heart failure symptoms are graded according to the NYHA functional classification system (Box 2).1,2 It is imperative to be aware that even those individuals with mild heart failure symptoms may still be at high risk of hospitalisation and mortality.1
Some noncardiovascular conditions can present similarly to heart failure; however, in the absence of cardiac dysfunction, they do not meet the criteria for a diagnosis of heart failure.1 They can coexist and exacerbate heart failure symptoms. These conditions may include anaemia and pulmonary, renal, thyroid or hepatic disease.
On presentation in the GP setting, the key features of the history will be eliciting symptoms of heart failure, their impact on function and whether there have been symptoms of angina or ischaemia. In addition to this, the following features are relevant:
- any recent viral illness, neoplastic disease or endocrine disease
- constitutional symptoms
- their past medical history, social history including substance use and family history of heart disease or sudden death
- recent stressors.
In conjunction with cardiovascular examination, the focus should be on establishing the severity of symptoms, initiating investigations into the causes of heart failure, referral of the patient to a cardiologist and commencing guideline-directed medical therapy (GDMT). Common causes of acute decompensation of chronic heart failure include myocardial ischaemia or infarction, arrhythmias, infection, anaemia, thyroid disease, increased sympathetic drive (e.g. Takotsubo cardiomyopathy or acute hypertension), acute renal failure, mechanical emergencies (e.g. ruptured septum, acute valvular regurgitation), aggravating drugs and nonadherence.4
Causes of HFrEF
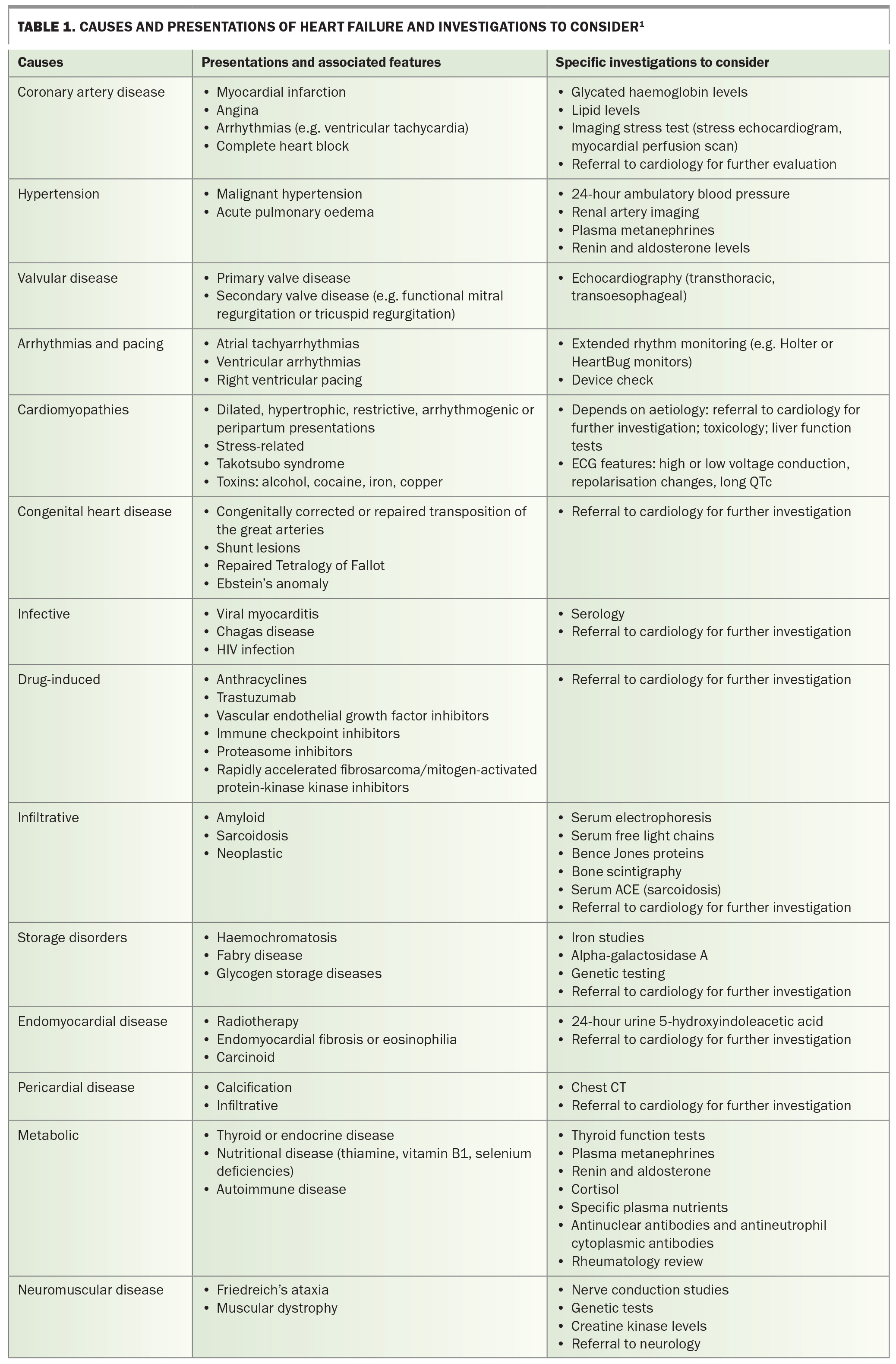
A hallmark characteristic of heart failure is its diverse profile of underlying aetiologies. Careful identification of aetiology is critical because it can shape the approach to treatment and prognosis. The most common underlying substrate is myocardial dysfunction, which is typically systolic in HFrEF, although most patients also have a degree of diastolic dysfunction. Pathology of the valves, pericardium and endocardium, as well as abnormalities of heart rhythm and conduction can contribute. The causes of HFrEF and subsequent investigation to identify and risk stratify are summarised in Table 1.1 The most common causes are ischaemic heart disease, myocardial infarction, hypertension and valvular heart disease.
Investigations and workup for suspected HFrEF
In the GP setting, blood tests (including full blood count; urea, electrolytes and creatinine; liver function tests; thyroid function tests; and a B-type natriuretic peptide [BNP] or N-terminal pro-BNP [NT-proBNP]) form part of the initial workup alongside 12-lead ECG, chest x-ray and referral for transthoracic echocardiography. Importantly, if the BNP or NT-pro-BNP and transthoracic echocardiography findings are normal (i.e. BNP <35 pg/mL or NT-proBNP ≤125 pg/mL), heart failure is unlikely in the nonacute setting and other diagnoses should be considered. Twenty-four-hour ambulatory blood pressure monitoring may be considered to assess hypertension. ECG features that raise suspicion of heart failure or underlying cardiac disease include atrial fibrillation, the presence of ST-segment changes, q waves, features of left ventricular hypertrophy or broad QRS complexes. Chest x-ray can help assess an individual’s likelihood of having pulmonary oedema but does not rule out heart failure.
Further investigations for heart failure are typically performed in the specialist cardiology setting and are dependent on the suspected cause and severity of disease, which may include a CT coronary angiogram, cardiac MRI, invasive coronary angiography, left and right heart catheterisation and cardiopulmonary exercise testing.
Indications for referral to cardiologist and a care plan
Where possible, all patients with suspected new-onset heart failure should be referred for a cardiology opinion. Additional indications for referral include:
- a persistently reduced LVEF of 30% or less despite GDMT
- when a second opinion is needed regarding aetiology or contributing pathology
- chest pain or concerns for ischaemia
- chronic heart failure with high-risk features such as previous inotrope use, persistent NYHA class III to IV symptoms, elevated creatinine, atrial fibrillation, palpitations or syncope suspicious for a ventricular arrhythmia, repeated implantable cardioverter defibrillator (ICD) shocks, multiple hospitalisations or clinical deterioration.
Management of HFrEF
Preventing heart failure progression based on trajectory
Epidemiology: why we need to prevent progression
Heart failure is a major cause of morbidity and mortality both in Australia and globally.1,2 The overall incidence is rising because of advancing age and it consumes a substantial volume of healthcare resources.6,7 In 2022, an estimated 144,000 people in Australia older than 18 years of age (0.7%) were living with heart failure.8 Almost 75% of those with heart failure are aged older than 65 years.8 In 2021 to 2022, 173,300 hospitalisations due to heart failure or cardiomyopathy were recorded.8 It was the underlying cause of death in 5000 people in 2022 and associated with another 28,600 deaths.8 Importantly, the incidence is 2.8 times higher in Aboriginal and Torres Strait Islander people and the disparity was greater for females.8 The heart failure incidence is also higher in people from lower socioeconomic backgrounds and in those living in rural or remote areas.8 The mortality rates from heart failure are higher in those who identify as Aboriginal and Torres Strait Islander people, and those from rural or lower socioeconomic backgrounds.8 These figures highlight the great reliance our healthcare system has on primary care providers in managing heart failure and preventing its progression.
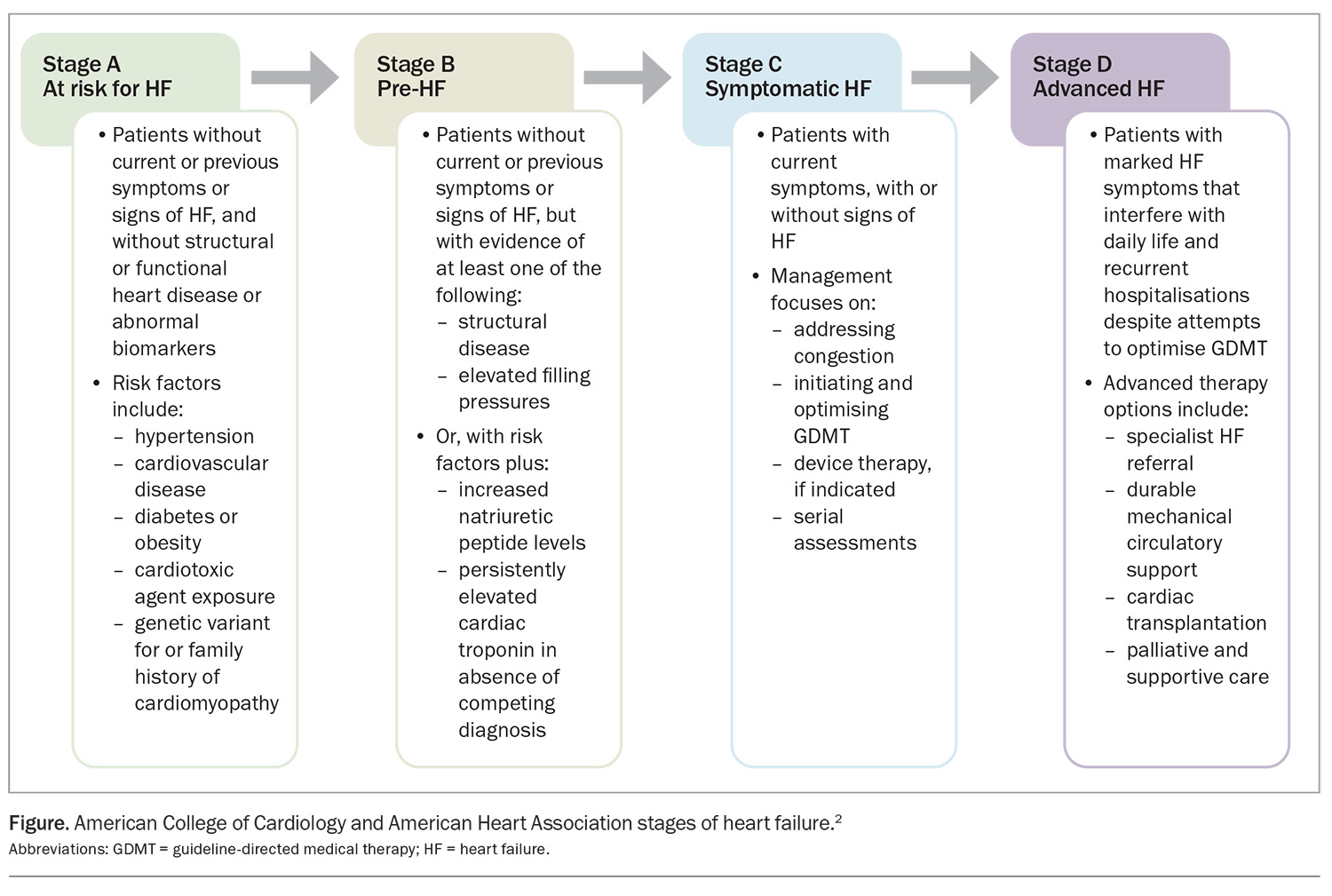
The progression of heart failure can range from those at risk of disease through to those with advanced heart failure. This means there is a real need for improved prevention strategies in the primary care setting and early detection of heart failure to prevent progression to clinically significant heart failure. The clinical trajectory and progression of heart failure is summarised in the Figure.2
The trajectory and prognosis of heart failure is variable and associated with the underlying aetiology. Some individuals, particularly those with viral myocarditis, stress cardiomyopathy, peripartum cardiomyopathy or a tachycardiomyopathy, may demonstrate excellent recovery.1 Some patients with left ventricular systolic dysfunction may even demonstrate substantial or complete recovery in LVEF after receiving optimal GDMT and device therapy. However, on the other end of the spectrum, some patients can demonstrate aggressive disease that drives them towards advanced heart failure therapies, such as left ventricular assist devices or cardiac transplantation, whether that is acute or over a period of years. The prognosis has improved but remains poor, with mortality rates estimated to be between 20% at one year and 53% at five years.1
Management of modifiable risk factors for heart failure
For patients at risk of heart failure in the primary care setting, primary prevention is imperative to prevent progression. Elevated systolic and diastolic blood pressure is a major risk factor for the development of symptomatic heart failure. Treatment of hypertension, achieving good glycaemic control and, in patients with diabetes and high cardiovascular risk, considering a sodium-glucose cotransporter-2 (SGLT-2) inhibitors are the key approaches of management.2 Encouraging regular physical activity, achieving and maintaining a healthy weight and avoiding smoking are important for blood pressure control and avoidance of obesity. Wholegrain, plant-based and Mediterranean diets have been shown in some studies to be effective for controlling weight.9,10
Pharmacological management: guideline-directed medical therapies
Rationale for guideline-directed medical therapies
Targeted treatment for heart failure usually begins with addressing the underlying cause. For example, this could mean treating atrial fibrillation if the cardiac dysfunction is due to tachyarrhythmia, or coronary revascularisation if the heart failure is due to ischaemia. Certain specific therapies are available for certain conditions, such as tafamidis for transthyretin cardiac amyloidosis.1,2
Beyond relieving congestion, the goals of pharmacotherapy in HFrEF are to:
- reduce mortality
- prevent recurrent hospitalisations for worsening heart failure
- improve clinical status, functional capacity and quality of life.
This is achieved via the combined effect of a well-established four-drug regimen consisting of an angiotensin receptor–neprilysin inhibitor (ARNI), angiotensin receptor blocker (ARB) or ACE inhibitor, beta blocker, mineralocorticoid receptor antagonist (MRA) and SGLT-2 inhibitor.2,7,11,12 These four drugs should be initiated as soon as possible with simultaneous (rather than stepwise) rapid up-titration to maximally tolerated doses, which has been shown to be safe and effective.13,14 Modulation of the renin–angiotensin–aldosterone system and sympathetic nervous system via an ARNI, beta blocker and MRA has been shown to improve survival in clinical trials.2,7,11,12 In recent years since our last update, there is further strong evidence for using an SGLT-2 inhibitor regardless of diabetes status.15
Important drugs to avoid in HFrEF include nondihydropyridine calcium channel blockers, such as diltiazem and verapamil, thiazolidinediones (glitizones), the dipeptidyl peptidase-4 (DPP-4) inhibitors saxagliptin and alogliptin, NSAIDs and flecainide, unless with specialist cardiology input. Several additional medications can also increase the risk of worsening heart failure. These include tricyclic antidepressants, corticosteroids, tyrosine kinase inhibitors, clozapine and minoxidil.4
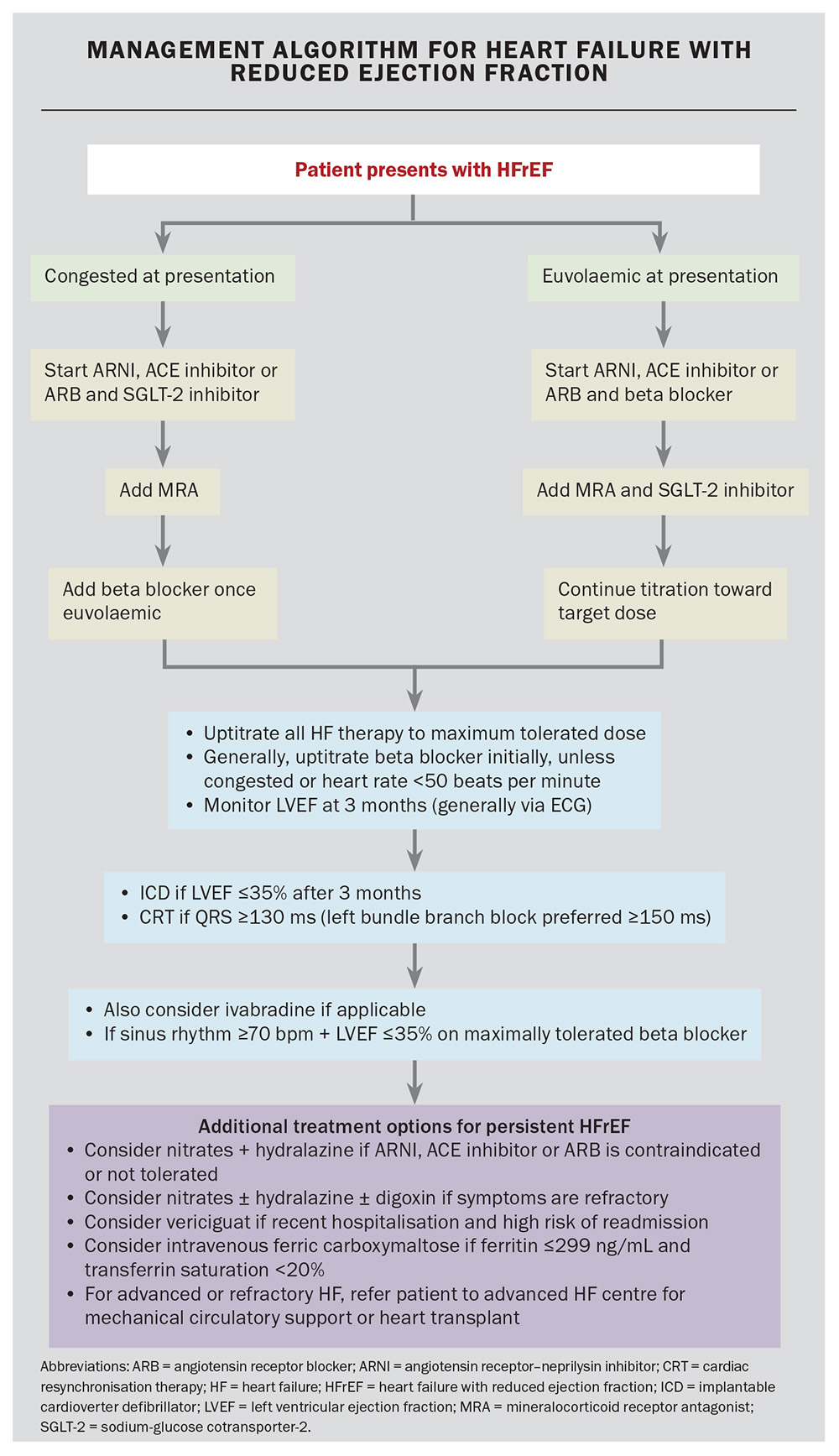
An approach to commencing GDMT in HFrEF is illustrated in the Flowchart.
Optimising pharmacotherapy in practice
The goals of pharmacotherapy titration include:
- simultaneous and rapid initiation and up-titration of GDMT
- early post-discharge assessment and ongoing close monitoring of complications associated with dose up-titration
- commencement of more than one drug at a time (in many individuals, this is safe and effective)
- diligent management of volume status will reduce patient symptoms
- addressing clinical, psychosocial and financial barriers to achieving GDMT.
The practical approach to commencing GDMT depends on the patient’s fluid status, their other comorbidities, renal function and electrolyte stability, heart rate and blood pressure. As highlighted in the Flowchart, once the diagnosis of HFrEF has been established with clinical assessment and echocardiography, any fluid overload or congestion should be treated with loop diuretics, such as furosemide. Addressing volume status response to diuretics can occur with one- to two-weekly follow up as an example. Failure to achieve decongestion with high doses of furosemide might warrant trialling a thiazide diuretic (e.g. hydrochlorothiazide), renal function and serum sodium level permitting.7 If there is a failed response to oral diuresis in the community, referral for inpatient intravenous therapy may be required.
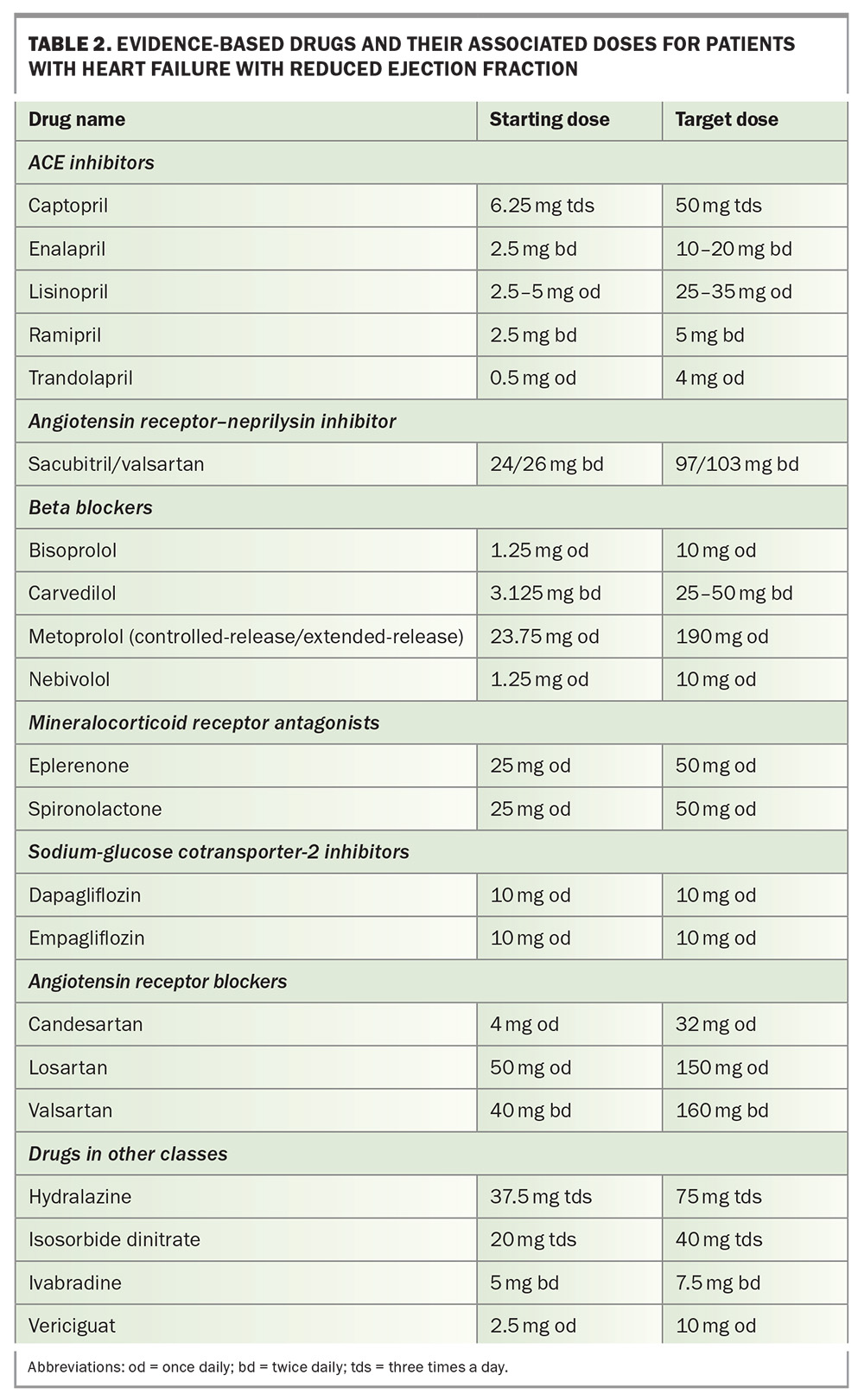
GDMT can be commenced before complete euvolaemia in the primary care setting. An ACE inhibitor, ARB or ARNI may be commenced first in this case. If there are no features of fluid overload and the patient is clinically stable, then a beta blocker, such as bisoprolol, carvedilol, metoprolol succinate or nebivolol, can be initiated alongside an ACE inhibitor, ARB or ARNI. An MRA, such as spironolactone or eplerenone, can then be initiated in addition to the above. Similarly, some emerging studies are looking at finerenone in patients with both diabetes mellitus and albuminuric chronic kidney disease with estimated glomerular filtration rate greater than 25 mL/min/1.73 m2.16 Optimal and target doses for GDMT are listed in Table 2. Monitoring and up-titration are recommended to occur with follow up every couple of weeks to assess side effects and potential complications. Importantly, it is not necessary to achieve maximal target doses of one drug class before commencing another. Once the patient is stable on maximally tolerated doses of GDMT, regular assessment should occur ideally every three to six months. If recovery to LVEF higher than 40% occurs, it is essential to continue therapies and not cease them.
Aside from diuretics and GDMT, ivabradine, an inhibitor of the If channel in the sinus node, can be used in symptomatic patients with an LVEF of 35% or less who are in sinus rhythm with a resting heart rate of higher than 70 beats per minute despite treatment with the maximally tolerated dose of a beta blocker; ACE inhibitor, ARB or ARNI; and MRA. Practically, this can be introduced at a dose of 5 mg twice daily if the patient is younger than 75 years of age, or 2.5 mg twice daily if they are older. The heart rate can then be reassessed in two to four weeks and up-titrated as per guidelines.
Vericiguat has modest evidence for reducing cardiovascular death and heartfailure hospitalisation in individuals with HFrEF and worsening symptoms with recent decompensation (NYHA class II–IV) despite optimal treatment.1,2 The initial dose can start at 2.5 mg daily, then double every two weeks until a target dose of 10 mg is achieved. Hydralazine and isosorbide dinitrate similarly have some evidence when the LVEF is 35% or less, or less than 45% with a dilated left ventricle in NYHA class III to IV failure despite GDMT.2
More recently, emerging evidence has shown that digitoxin may lead to a lower risk of death from any cause or hospitalisation from heart failure among patients with HFrEF who received GDMT, although more research is required. Caution should also be exercised in elderly, female and frail individuals; those in sinus rhythm; and those who have hypokalaemia.17
Omecamtiv mecarbil is a selective cardiac myosin activator shown to decrease cardiovascular death and hospitalisation in patients with HFrEF but is thought to be most suitable for patients with the lowest LVEF. It is not currently available in Australia.18
Precautions and considerations
There are several precautions to be aware of. Sacubitril/valsartan should not be commenced within 36 hours of an ACE inhibitor. Caution with sacubitril/valsartan should also be exercised if the estimated glomerular filtration rate is less than 30 mL/min/1.73 m2 and if the systolic blood pressure is lower than 100 mmHg. At present, SGLT-2 inhibitor use in individuals with type 1 diabetes remains off label, mainly due to concerns of diabetic ketoacidosis. Patients commencing SGLT-2 inhibitors should be counselled on the risk of mycotic genital infections, ketoacidosis and instructions for fasting. Ivabradine and beta blockers should not be used when there is evidence of acute decompensated heart failure, or significant conduction disease with a risk of causing bradyarrhythmia. Beta blockers can be recommenced when the patient is euvolaemic. Considerations for specific populations with heart failure are listed in Box 3.1
Monitoring and managing complications in practice
Common side effects of GDMT include symptomatic hypotension, bradycardia, dizziness and hypovolaemia. There are some strategies for managing symptomatic hypotension. Symptoms tend to occur more with ARNIs; if the patient’s systolic blood pressure is lower than 100 mmHg, possible solutions include reducing the dose of sacubitril/valsartan 24/26 mg to half a tablet twice daily, changing to an ACE inhibitor or ARB monotherapy and reducing loop diuretic doses to allow room to up-titrate the ARNI if not clinically congested. However, if the complications are persistent, medications are poorly tolerated or there is progression of symptoms, expediting specialist review is important.
Follow-up visits in the weeks following therapy up-titration should focus on physical examination to assess for symptoms of congestion, assessment of blood pressure and heart rate and laboratory evaluation that includes serum potassium levels and renal function. Specifically, blood pressure, electrolytes and kidney function should be assessed within one to two weeks after dose initiation or titration of an ARNI, ACE inhibitor or ARB. Ongoing assessment depends on kidney stability. Typically, reassessment of ventricular function with echocardiography should occur within three to six months after target or maximally tolerated doses of GDMT are achieved to determine the need for device therapies or referral to advanced heart failure services. This will need to be via specialist review.
Nonpharmacological management
Iron optimisation
Iron optimisation improves heart failure symptoms, exercise capacity and quality of life. Although it is controversial, no mortality benefit has been demonstrated to date, and there is uncertainty regarding the effect on hospitalisations, although a meta-analysis revealed reductions in HF hospitalisation.19 Screening for anaemia and iron deficiency with full blood count, serum ferritin concentration and transferrin saturation is recommended every three to six months in all patients with HFrEF.1 Intravenous iron replacement is currently recommended in symptomatic patients with HFrEF.11
Implantable cardioverter defibrillator therapy
For primary prevention, ICD therapy should be considered to reduce the risk of sudden death and all-cause mortality in patients with symptomatic ischaemic cardiomyopathy (NYHA class II–III) and an LVEF of 35% or less despite three or more months of GDMT, provided the long-term prognosis is greater than 12 months.1 Weaker evidence exists for those with nonischaemic cardiomyopathy. ICD is also indicated for secondary prevention with recovered ventricular arrhythmia and haemodynamic instability.20,21 Different options exist, including transvenous ICDs and subcutaneous ICDs.
Cardiac resynchronisation therapy
Cardiac resynchronisation therapy (CRT) is an important strategy for symptomatic patients with HFrEF who have evidence of cardiac dyssynchrony. It is considered for individuals with heart failure in sinus rhythm with broad QRS durations of 130 to 149 ms, left bundle branch block on ECG and an LVEF of 35% or less despite optimal medical therapy to improve symptoms and reduce morbidity.1 Patients who have a conventional permanent pacemaker or ICD, who subsequently develop worsening heart failure symptoms despite GDMT and have a significant burden of right ventricular pacing should be considered for an upgrade to CRT. CRT over right ventricular pacing is recommended for patients with HFrEF (regardless of NYHA status or QRS width) who have an indication for ventricular pacing for high-degree atrioventricular block to reduce morbidity. This is because right ventricular pacing leads to dyssynchrony and worsening symptoms. This can occur in patients with atrial fibrillation. Symptomatic patients with heart failure with an LVEF of 35% or less, sinus rhythm with QRS 150 ms or greater and non-left bundle branch block morphology also derive benefits from CRT.22
Management of valvular disease
Patients with valvular disease have a very poor prognosis in HFrEF.23,24 Aortic valve intervention with transcatheter or surgical aortic valve replacement is recommended in patients with HFrEF and severe high-gradient aortic stenosis to improve symptoms and reduce mortality.1 For individuals with severe mitral regurgitation (MR), surgical treatment is a first-line recommendation for severe primary chronic MR resulting in HFrEF. For severe functional MR, mitral valve transcatheter edge-to-edge repair can be considered in selected patients not eligible for surgery and not requiring coronary revascularisation who are symptomatic despite GDMT. The decision to perform valvular repair or replacement is made in conjunction with local structural heart teams.
Referral for advanced heart failure therapy
Patients being considered for long-term mechanical support must have good adherence to therapy, appropriate capacity for device handling and psychological support.1 Heart transplantation is recommended for patients with advanced heart failure that is refractory to medical and device therapy who do not have absolute contraindications. In Australia, referral for advanced heart failure therapies is typically performed by the treating cardiologist; however, GPs are incredibly important in the co-ordination of care and psychosocial support for these individuals.
Prognosis and long-term planning
Implementing psychosocial support strategies and referral to cardiac rehabilitation or heart failure disease management services are important longer-term objectives. These are helpful to support drug titration, monitor symptoms and increase exercise tolerance, empowering individuals to employ self-management strategies and understanding the appropriate suitability and timing for the integration of palliative care support. The frequency of medical follow up is recommended to be every three to six months once the patient has attained optimal dosing of medical therapy and is minimally symptomatic. Virtual care or telehealth services can be useful for GPs providing care in rural and remote locations. Home-based and clinic-based heart failure disease management programs involving heart failure nurses supported by allied health staff improve outcomes and are recommended to reduce the risk of hospitalisation and mortality.1 Wearable activity monitors and mobile technologies may be used for activity tracking, recording dietary trends, weight management and communicating with the heart failure team. They can also assist with prompts for medication and lifestyle adherence, although privacy issues may be difficult to circumvent in some cases.
It is also important to address reasons for poor adherence, such as access to medical therapy, follow up and cardiac rehabilitation services; mental health disorders; cognitive impairment; polypharmacy; and homelessness. This is especially important as low socioeconomic status and remoteness in Australia is linked with an increased incidence of heart failure. Some simple strategies in the primary care setting could include:
- simplifying medication regimens with pill packets
- creating patient goals or care plans
- providing culturally sensitive patient materials
- addressing mental health concerns
- planning pharmacist visits for complex medication regimens
- referring to home-based nursing visits.
If patients are not suitable for referral for advanced heart failure therapies, clarifying the goals of care and communicating prognosis are especially important. Introducing palliative care can reduce distressing symptoms and integrate important psychological and spiritual components of care. As heart failure progresses, GDMT should continue as long as it does not contribute to discomfort.
Conclusion
HFrEF is a major cause of morbidity and mortality both in Australia and globally. Management is centred around fluid management and the rapid up-titration of four key drug classes: ARNIs, beta blockers, MRAs and SGLT-2 inhibitors. Together, these medications reduce cardiovascular mortality and heart failure hospitalisation by over 60%. Device therapy and transcatheter valve repair strategies have emerged as effective strategies in parallel to GDMT. Early referral to a cardiologist and close monitoring, particularly at times of medication titration, are crucial. The burden of HFrEF will likely increase in the years to come, which makes preventive strategies and a dedicated effort to implement evidence-based therapies imperative. Supporting these strategies must be carefully delivered with cultural sensitivity, tools to address barriers to medication adherence and an awareness of the need for multidisciplinary input, including cardiac rehabilitation and palliative care at an appropriate time. MT
COMPETING INTERESTS: None.
References
1. McDonagh TA, Metra M, Adamo M, et al.; ESC Scientific Document Group. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: developed by the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the European Society of Cardiology (ESC). With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 2021; 42: 3599-3726.
2. Heidenreich PA, Bozkurt B, Aguilar D, et al.; Writing Committee Members; ACC/AHA Joint Committee Members. 2022 AHA/ACC/HFSA guideline for the management of heart failure. J Am Coll Cardiol 2022; 28: e1-e167.
3. Bozkurt B, Coats AJS, Tsutsui H, et al. Universal definition and classification of heart failure: a report of the Heart Failure Society of America, Heart Failure Association of the European Society of Cardiology, Japanese Heart Failure Society and Writing Committee of the Universal Definition of Heart Failure: endorsed by the Canadian Heart Failure Society, Heart Failure Association of India, Cardiac Society of Australia and New Zealand, and Chinese Heart Failure Association. Eur J Heart Fail 2021; 23: 352-380.
4. NHFA CSANZ Heart Failure Guidelines Working Group; Atherton JJ, Sindone A, De Pasquale CG, et al. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand: guidelines for the prevention, detection, and management of heart failure in Australia 2018. Heart Lung Circ 2018; 27: 1123-1208.
5. Galderisi M, Cosyns B, Edvardsen T, et al.; 2016–2018 EACVI Scientific Documents Committee. Standardization of adult transthoracic echocardiography reporting in agreement with recent chamber quantification, diastolic function, and heart valve disease recommendations: an expert consensus document of the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 2017; 18: 1301-1310.
6. Roth GA, Forouzanfar MH, Moran AE, et al. Demographic and epidemiologic drivers of global cardiovascular mortality. N Engl J Med 2015; 372: 1333-1341.
7. Maddox TM, Januzzi JL, Jr., Allen LA, et al. 2024 ACC expert consensus decision pathway for treatment of heart failure with reduced ejection fraction: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol 2024; 83: 1444-1488.
8. Australian Institute of Health and Welfare (AIHW). Heart, stroke and vascular disease: Australian facts. Canberra: AIHW; 2025. Available online at: https://www.aihw.gov.au/reports/heart-stroke-vascular-diseases/hsvd-facts/contents/all-heart-stroke-and-vascular-disease/heart-failure-and-cardiomyopathy (accessed April 2026).
9. Tektonidis TG, Åkesson A, Gigante B, Wolk A, Larsson SC. Adherence to a Mediterranean diet is associated with reduced risk of heart failure in men. Eur J Heart Fail 2016; 18: 253-259.
10. Lara KM, Levitan EB, Gutierrez OM, et al. Dietary patterns and incident heart failure in US adults without known coronary disease. J Am Coll Cardiol 2019; 73: 2036-2045.
11. McDonagh TA, Metra M, Adamo M, et al.; ESC Scientific Document Group. 2023 focused update of the 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 2023; 44: 3627-3639.
12. van Essen BJ, Ceelen DCH, Ouwerkerk W, et al. Pharmacologic treatment of heart failure with reduced ejection fraction: an updated systematic review and network meta-analysis. J Am Coll Cardiol 2025; 86: 2513-2526.
13. McMurray JJV, Solomon SD, Inzucchi SE, et al.; DAPA-HF Trial Committees and Investigators. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 2019; 381: 1995-2008.
14. Packer M, Anker SD, Butler J, et al.; EMPEROR-Reduced Trial Investigators. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med 2020; 383: 1413-1424.
15. Hamid AK, Tayem AA, Al-Aish ST, Al Sakini AS, Hadi DD, Al-Aish RT. Empagliflozin and other SGLT2 inhibitors in patients with heart failure and preserved ejection fraction: a systematic review and meta-analysis. Ther Adv Cardiovasc Dis 2024; 18: 17539447241289067.
16. Sindone AP, De Pasquale C, Amerena J, et al. Consensus statement on the current pharmacological prevention and management of heart failure. Med J Aust 2022; 217: 212-217.
17. Bavendiek U, Großhennig A, Bauersachs J; DIGIT-HF Group. Digitoxin in patients with heart failure and reduced ejection fraction. Reply. N Engl J Med 2026; 394: 98.
18. Teerlink JR, Diaz R, Felker GM, et al.; GALACTIC-HF Investigators. Effect of ejection fraction on clinical outcomes in patients treated with omecamtiv mecarbil in GALACTIC-HF. J Am Coll Cardiol 2021; 78: 97-108.
19. Cheema B, Chokshi A, Orimoloye O, Ardehali H. Intravenous iron repletion for patients with heart failure and iron deficiency: JACC state-of-the-art review. J Am Coll Cardiol 2024; 83: 2674-2689.
20. Kober L, Thune JJ, Nielsen JC, et al.; DANISH Investigators. Defibrillator implantation in patients with nonischemic systolic heart failure. N Engl J Med 2016; 375: 1221-1230.
21. Beggs SAS, Jhund PS, Jackson CE, McMurray JJV, Gardner RS. Non-ischaemic cardiomyopathy, sudden death and implantable defibrillators: a review and meta-analysis. Heart 2018; 104: 144-150.
22. Gage RM, Burns KV, Bank AJ. Echocardiographic and clinical response to cardiac resynchronization therapy in heart failure patients with and without previous right ventricular pacing. Eur J Heart Fail 2014; 16: 1199-1205.
23. Dziadzko V, Clavel MA, Dziadzko M, et al. Outcome and undertreatment of mitral regurgitation: a community cohort study. Lancet 2018; 391: 960-969.
24. Goliasch G, Bartko PE, Pavo N, et al. Refining the prognostic impact of functional mitral regurgitation in chronic heart failure. Eur Heart J 2018; 39: 39-46.