Heavy menstrual bleeding and iron deficiency: breaking the vicious cycle

Heavy menstrual bleeding is a common cause of chronic iron loss and iron deficiency, which may be symptomatic even when haemoglobin level is normal. Early recognition of low iron levels, plus treatment of both iron stores and menstrual blood loss, helps prevent recurrence and improves wellbeing.
- Heavy menstrual bleeding (HMB) is a common, under-recognised cause of iron deficiency in women of reproductive age, even before anaemia develops.
- Iron deficiency should not be excluded by a normal haemoglobin level alone, as many women have symptomatic nonanaemic iron deficiency. Ferritin testing is essential, with a ferritin level below 30 microg/L supporting the diagnosis in adults.
- Assessment of HMB should include evaluation of quality of life and consideration of underlying causes, rather than relying only on blood loss estimates. Investigations should be guided by symptoms, examination findings and risk factors.
- Management of HMB must both restore iron stores and reduce ongoing menstrual blood loss to prevent recurrence. Oral iron is often first line, but intravenous iron may be appropriate earlier when symptoms or losses are significant.
- Effective medical treatment options for HMB include tranexamic acid, NSAIDs and hormonal therapies, with the 52 mg levonorgestrel intrauterine device offering the greatest reduction in blood loss.
- Early treatment is important for current wellbeing and future pregnancy outcomes, as untreated iron deficiency is linked to poorer maternal and neonatal outcomes.
Iron is an essential micronutrient required for cellular energy metabolism, oxygen transport, and optimal neurological and immune function throughout life. Iron deficiency is the most prevalent micronutrient deficiency globally and a major contributor to disease burden across the female life course. It is estimated that more than two billion people have iron deficiency, iron deficiency anaemia or both, and almost one-third of women of reproductive age worldwide are affected by anaemia.1-4 In women of reproductive age, heavy menstrual bleeding (HMB) is a common presentation in general practice, a leading cause of chronic iron loss and, therefore, a major contributor to iron deficiency in this group.
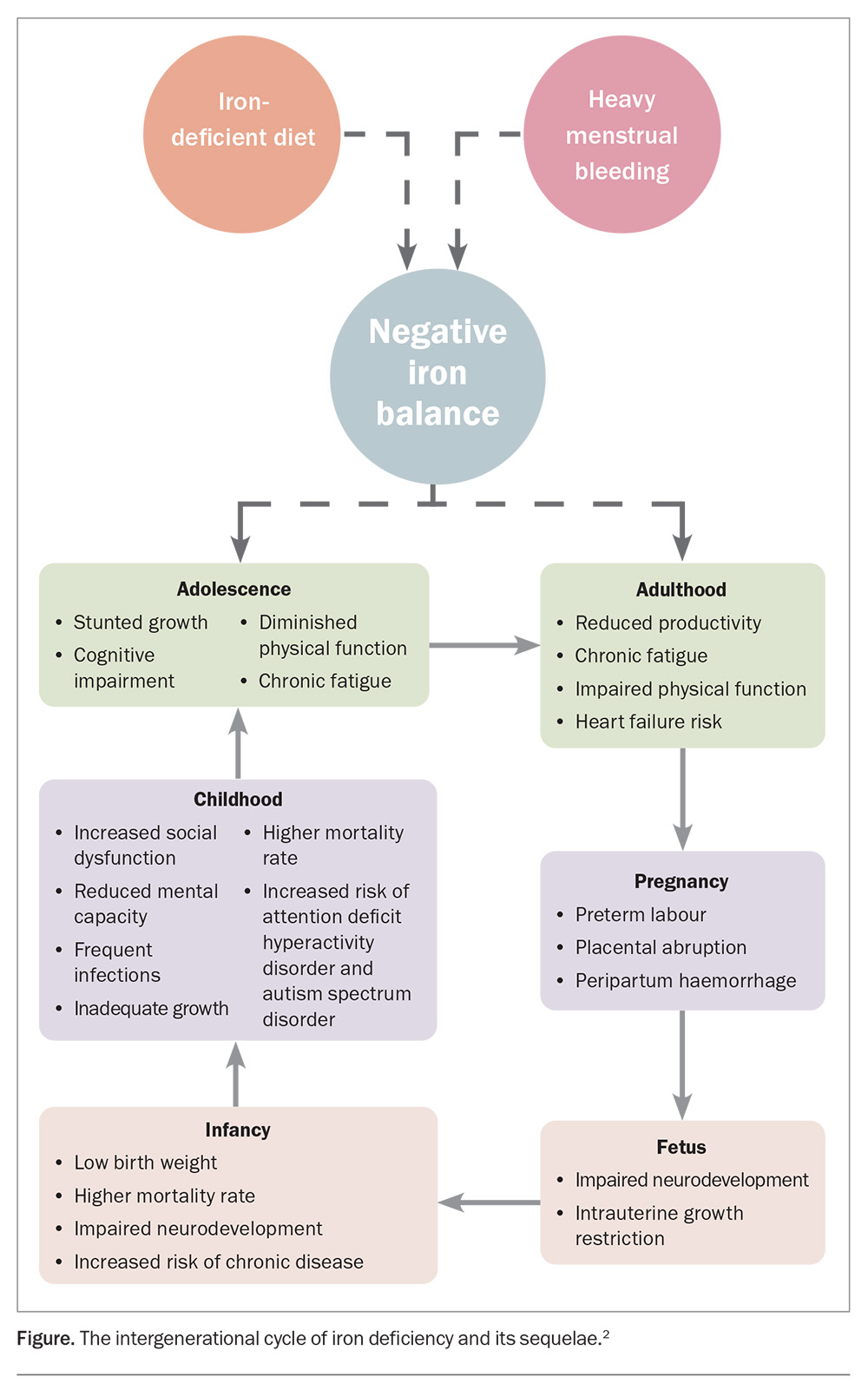
The impact of iron deficiency anaemia is amplified in pregnancy, when iron requirements increase substantially. Worldwide, about 37% of pregnant women are anaemic, mostly because of iron deficiency, and in some countries, the prevalence is as high as 63%.4-6 Crucially, many women enter pregnancy with suboptimal iron reserves, often because of years of unmanaged high-volume menstrual blood loss, placing them at heightened risk of iron deficiency anaemia as gestational demands increase. Maternal iron deficiency anaemia has well-documented adverse outcomes, including preterm birth, low birthweight and impaired neonatal neurodevelopment, contributing to a vicious intergenerational cycle of iron deficiency and its sequelae (Figure).2,4
Heavy menstrual bleeding
Although menstruation is a normal biological process, excessive monthly blood loss over time can deplete iron stores, initially resulting in iron deficiency with normal haemoglobin levels and progressing to iron deficiency anaemia if left untreated. HMB affects about one in four women of reproductive age.7,8 Many women with HMB also report coexisting pelvic pain, and it is a key contributor to the development of iron deficiency and iron deficiency anaemia.2
In the past, HMB was termed menorrhagia and defined as menstrual blood loss exceeding 80 mL per cycle, based on early menstrual collection studies.9 Although objective, this metric proved impractical in day-to-day clinical care, as women are often not able to measure the total blood loss during their cycle. Contemporary definitions therefore recognise HMB as excessive menstrual bleeding that adversely interferes with a woman’s physical, emotional, social or material quality of life. This definition is endorsed by the Australian Commission on Safety and Quality in Health Care’s Heavy Menstrual Bleeding Clinical Care Standard.8
This patient-centred approach has clear advantages: it validates women’s experiences and enables earlier recognition. However, it is also a double-edged sword. Because quality-of-life impact is more subjective, some clinicians perceive it as a vague yardstick. HMB-associated iron deficiency remains underdiagnosed for multiple reasons, including uncertainty about clinical thresholds for investigation, normalisation of significant menstrual bleeding by patients and their clinicians, deprioritisation of women’s health needs and barriers related to cost and access to healthcare. This is particularly important because a range of effective medical, surgical and radiological treatment options is available for women with HMB.2
In this context, a structured approach to assessing abnormal uterine bleeding can help clinicians progress from recognising HMB to identifying potential underlying causes and selecting appropriate investigations. The PALM–COEIN classification system, developed by the International Federation of Gynaecology and Obstetrics (FIGO), provides a useful framework for considering causes of abnormal uterine bleeding. It classifies causes into structural (PALM: polyp, adenomyosis, leiomyoma, malignancy and hyperplasia), nonstructural (COEI: coagulopathy, ovulatory dysfunction, endometrial and iatrogenic) and not yet classified (N) categories (Table 1).
Anaemic versus nonanaemic iron deficiency
Anaemia represents a later stage of iron deficiency; many women develop symptomatic iron deficiency before haemoglobin levels decline. In general practice, women presenting with fatigue and heavy periods are often treated symptomatically or reassured if their haemoglobin level is within the normal range. Without ferritin testing, nonanaemic iron deficiency – which affects energy, cognition, mood, stamina and pregnancy outcomes – is frequently missed. Breaking this pattern requires early recognition of iron deficiency, timely correction of iron stores and proactive management of menstrual blood loss in line with the updated Heavy Menstrual Bleeding Clinical Care Standard.8
Case study. Presentation of a 29-year-old woman with heavy menstrual bleeding
Sally, a 29-year-old woman (gravida 2, para 2), presents with fatigue, impaired concentration and reduced stamina while managing a full-time job and caring for two young children. She reports HMB since the birth of her second child two years ago, with regular menstrual cycles lasting an average of eight to nine days, passage of clots and two-hourly tampon changes on heavy days. During her most recent delivery, she experienced a postpartum haemorrhage (cumulative per vaginam blood loss estimated at just under one litre). She had been prescribed oral iron on discharge from hospital but her adherence has not been consistent because of both forgetfulness and concerns about it causing constipation. She is worried about her symptoms, as her mother was diagnosed with endometrial cancer several years ago. Her current weight is 65 kg.
Investigations
In women with HMB, investigations should be selected and interpreted in conjunction with the clinical assessment (including an understanding of the impact on the individual’s quality of life), to help identify an underlying cause and guide management. A pregnancy test should be offered if pregnancy is possible, along with blood tests for iron deficiency and anaemia. With the patient’s consent, a pelvic examination is usually performed to assess for uterine enlargement, tenderness or masses. Additional investigations are guided by the clinical picture and may include further blood tests, cervical screening (if due) and pelvic ultrasound.
Assessing iron status
Serum ferritin is the most reliable marker of iron stores in the absence of significant inflammation. The WHO defines iron deficiency as serum ferritin levels less than 15 microg/L, a threshold derived from bone marrow iron studies showing absent iron stores at a mean ferritin level of 15 microg/L.10
An Australian cohort study demonstrated that nearly 25% of iron deficiency diagnoses would be missed using this serum ferritin threshold.11 Accordingly, the Royal College of Pathologists Australasia (RCPA) recommends a ferritin threshold of less than 30 microg/L for diagnosing iron deficiency in all adults. Although ferritin reference thresholds may vary between laboratories in Australia, GPs should keep this RCPA recommendation in mind when evaluating possible iron deficiency.12
Ferritin is an acute-phase reactant and may be falsely normal or elevated in the presence of inflammation (e.g. infection, chronic inflammatory disease, liver disease or obesity), which can mask underlying iron deficiency. The RCPA acknowledges serum ferritin may be normal (up to 100 microg/L) when iron deficiency coexists with an acute phase response.12 In this setting, assessment of iron availability is helpful; a low transferrin saturation supports iron deficiency despite a nonlow ferritin level, and should be interpreted alongside the clinical context.13
A broader clinical approach is emerging across multiple disciplines, including cardiology and geriatrics, that emphasises maintaining treatment targets towards the middle of the ferritin reference range rather than simply remaining above the lower limit. Because oral treatments are often used as initial therapy at first presentation, and are generally less effective at reducing menstrual blood loss than the 52 mg levonorgestrel intrauterine device (IUD), many women with HMB may remain at risk of ongoing cumulative iron loss. In the author’s opinion, a target-based approach to maintaining iron stores (e.g. ferritin level 80 to 100 microg/L) may improve longer-term wellbeing and reduce recurrent iron deficiency; however, further research is needed.
Haemoglobin thresholds are lower in premenopausal women (120 g/L) than in men (130 g/L), which is important to keep in mind. By contrast, there is no sex-based difference in haemoglobin thresholds in children or postmenopausal populations.
Pelvic ultrasound
Pelvic ultrasound is indicated when structural pathology is suspected or when symptoms, signs or bleeding pattern warrant further investigation. A woman with HMB generally requires a pelvic ultrasound (preferably transvaginal) if there is:
- an increased risk of malignancy, based on the patient’s history
- suspicion of structural pathology, such as fibroids, polyps or adenomyosis
- abnormal findings on physical examination, such as a bulky uterus, adnexal or uterine masses, or tenderness
- symptoms such as deep dyspareunia, severe dysmenorrhoea or an atypical bleeding pattern (e.g. intermenstrual, irregular or postcoital bleeding)
- bleeding that has not responded to initial treatment.8
If indicated in the investigation of HMB, a high-quality pelvic ultrasound is ideally performed between days five and 10 of the menstrual cycle, when the endometrium is thinnest and structural causes, such as endometrial polyps or submucosal fibroids, are easiest to identify.8 However, a normal ultrasound does not diminish a woman’s experience of HMB. Up to 50% of women with HMB have no structural cause identified, yet still experience clinically significant quality-of-life impairment and iron deficiency.14
Case continued: investigations
Sally’s investigations reveal the following:
- haemoglobin level: 123 g/L (normal range for women: 120–165 g/L)
- transferrin saturation: 18% (normal range: 20–50%)
- serum ferritin level: 21 microg/L (<30 microg/L for an adult is diagnostic of iron deficiency)
- normal thyroid stimulating hormone level.
These findings are consistent with nonanaemic iron deficiency.
Sally is referred for a transvaginal pelvic ultrasound given her increased risk of malignancy (a first-degree relative with endometrial cancer). Ultrasound reveals a normal-sized anteverted uterus with a homogeneous myometrial echotexture and no focal lesions. The endometrium measures 7 mm and has a smooth, regular endometrial–myometrial interface, which is appropriate for day seven of her menstrual cycle. Both ovaries are normal in size and demonstrate mobile premenopausal morphology, with no cysts or adnexal masses. No free fluid is seen in the pouch of Douglas.
Management
Management of nonanaemic iron deficiency and iron deficiency anaemia requires both correction of iron deficiency and reduction of ongoing losses.
Iron repletion
Iron is crucial for oxygen transport via haemoglobin and for mitochondrial electron transport and adenosine triphosphate production. Nonanaemic iron deficiency can impair muscle function, cognitive performance and quality of life, even in the absence of anaemia. Fatigue in nonanaemic iron deficiency can be multifactorial and is often influenced by overlapping biological, psychological and social determinants, including stress, sleep quality and caregiving burden.
Population studies demonstrate that women with a history of low iron report lower vitality and a higher prevalence of fatigue compared with women without such a history.15 However, psychosocial factors may also contribute to fatigue. Proactive correction of iron deficiency is important to restore iron stores and establish the patient’s symptom baseline once iron status is normal.
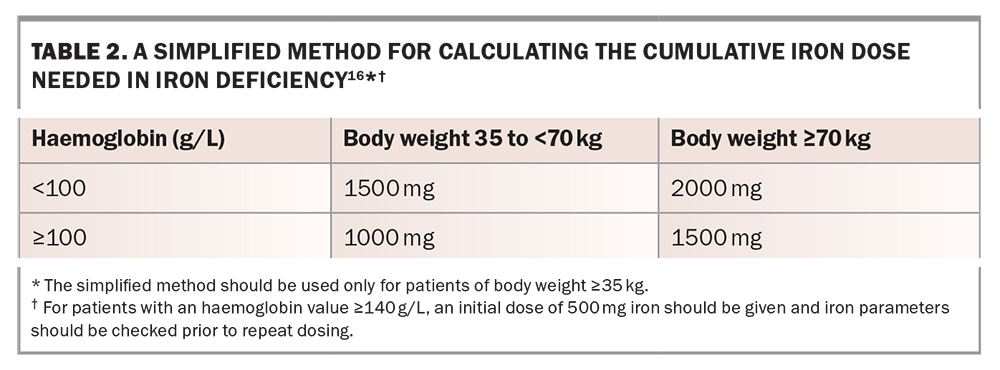
The cumulative dose of iron required to replenish body iron reserves can be calculated using Ganzoni formula or a simplified method based on haemoglobin level and body weight (Table 2).16 Oral iron supplementation is considered first-line therapy for iron supplementation because of accessibility, ease of administration and perceived lower upfront costs. However, adherence is often suboptimal because of gastrointestinal side effects, and fractional iron absorption is limited, particularly in the setting of ongoing HMB.17 Thus, it may be insufficient to restore iron reserves in women with significant ongoing iron losses. Alternate-day dosing may improve absorption and tolerability.18
A recent reproductive-lifespan economic evaluation compared first-line intravenous iron (iron dextran, ferumoxytol or iron sucrose) with oral ferrous sulfate for women aged 18 to 51 years with HMB-associated iron deficiency anaemia, using a Markov model. First-line intravenous iron dextran was the most cost-effective strategy, with a lifetime cost of US$157,500 and 19.26 quality-adjusted life years versus US$152,900 and 19.10 quality-adjusted life years for oral ferrous sulfate. This corresponded to an incremental cost-effectiveness ratio of US$28,600 per quality-adjusted life year gained. Intravenous iron dextran remained cost-effective across scenarios of varying menstrual blood losses and sensitivity analyses, supporting its value relative to oral iron over the reproductive lifespan.19
These findings support consideration of earlier intravenous iron use for women with significant iron loss and symptomatic deficiency, rather than delaying escalation until prolonged oral therapy has failed. Therefore, early patient education and a contingency plan for intravenous iron when oral therapy is not tolerated or is ineffective are essential.
Intravenous iron infusions are generally safe, but they can cause mild, short-lived adverse effects such as headache, nausea, myalgia or a metallic taste. Rarely, more serious complications occur, including severe hypersensitivity reactions (including anaphylaxis), permanent skin staining from extravasation, iron overload with repeated dosing or a potential increase in infection risk. For this reason, patients are routinely monitored during infusion.
Addressing heavy menstrual bleeding
Effective iron repletion must be paired with management of menstrual blood loss to prevent recurrent deficiency. Hormonal therapies and antifibrinolytics are established strategies for reducing menstrual blood loss, and include:
- tranexamic acid: an antifibrinolytic agent taken during menstruation. Has an expected reduction in menstrual blood loss of 40 to 50%20
- 52 mg levonorgestrel IUD: suppresses the endometrium’s response to oestrogen, leading to a thinner, less proliferative endometrium. It has an expected reduction in menstrual blood loss of more than 90% by six months after insertion and can also be used as contraception21
- combined oral contraceptive pill: regulates the menstrual cycle and can reduce menstrual blood loss; it is appropriate for women who also want contraception and have no contraindications to oestrogen
- progestogen-only pill: may reduce bleeding in some women but can be associated with irregular bleeding; it is an option when oestrogen is contraindicated or not preferred
- NSAIDs (e.g. naproxen, mefenamic acid): works by reducing prostaglandin production in the uterine lining, taken during menstruation. Often combined with tranexamic acid.
The 19.5 mg levonorgestrel IUD, although a contraceptive option, is not recommended for symptomatic control of HMB. The copper IUD is also not recommended because it may worsen HMB.
Several of these therapies can be used in combination, depending on symptom burden and patient preference. When counselling, it is also important to discuss expected adverse effects and practical drawbacks. Tranexamic acid is generally well tolerated but can cause gastrointestinal upset and should be used with caution in women with HMB and a recent thrombotic event. There has been some concern that use of the antifibrinolytic tranexamic acid for HMB may increase the risk of venous thromboembolism; however, a Cochrane review found insufficient evidence to determine whether treatment is associated with increased thromboembolic risk.20 NSAIDs may reduce bleeding and dysmenorrhoea but can cause gastrointestinal adverse effects and are not suitable for all women (e.g. those with renal impairment or peptic ulcer disease). Hormonal options can be associated with unscheduled bleeding, breast tenderness and other hormone-related effects, particularly in the first few months after initiation or insertion, and these should be anticipated and normalised as part of shared decision-making.
Overall, these interventions not only improve quality of life but may also reduce ongoing iron loss and the need for repeated iron repletion. Treatment should be individualised according to patient preference, bleeding severity and reproductive goals. It is concerning that many women continue to experience substantial, preventable morbidity from HMB and its sequelae despite the availability of effective medical options.
Case continued: management
Sally’s GP is able to offer an intravenous iron infusion within the week of her nonanaemic iron deficiency diagnosis. Both ferric carboxymaltose and ferric derisomaltose are available, and the final choice is based on patient preference, hypophosphataemia risk, adverse effects, hypersensitivity risk and the number of doses required. Sally receives a 1000 mg intravenous dose of ferric carboxymaltose, a dose supported by a haemoglobin level between 100 and less than 140 g/L and a body weight between 35 and 70 kg.
Sally is counselled about HMB management options, including NSAIDs (such as mefenamic acid), tranexamic acid and hormonal treatments. She leaves the initial consultation with a prescription for tranexamic acid and a 52 mg levonorgestrel IUD to be inserted during day four of her next menstrual cycle.
With counselling framed around relative efficacy, Sally is able to make an informed choice from the available HMB management options. She had previously been unaware that several effective treatment options were available to improve her quality of life.
Obstetric considerations
Optimal iron repletion is essential for women contemplating pregnancy, given that each pregnancy increases iron requirements by about 1 g to support maternal and fetal erythropoiesis, placental development and the establishment of adequate neonatal iron stores. Early maternal iron deficiency is associated with suboptimal neonatal iron status and may adversely affect offspring neurodevelopment.22 FIGO recognises iron deficiency as the most common micronutrient deficiency worldwide and emphasises that women of reproductive age, particularly those with HMB, carry a disproportionate burden because of cumulative menstrual- and pregnancy-related iron loss. Crucially, FIGO highlights that iron deficiency anaemia represents a late stage of deficiency, and that iron deficiency alone can adversely affect maternal wellbeing and pregnancy outcomes long before anaemia develops.4
In Australia, current antenatal guidance differs on the role of routine ferritin screening. The Royal Australian and New Zealand College of Obstetricians and Gynaecologists recommends routine full blood count testing (with particular attention to haemoglobin, mean corpuscular volume and platelet count) at the booking visit and at 28 weeks’ gestation, with ferritin testing in women with signs of anaemia (e.g. low mean corpuscular volume), rather than universal ferritin screening in early pregnancy.23 The Living Evidence for Australian Pregnancy and Postnatal Care Australian Pregnancy Care Guidelines (approved by the National Health and Medical Research Council) recommends haemoglobin concentration testing for all pregnant women at the first visit and at 28 weeks’ gestation, and that ferritin testing at the first visit should be considered in areas where prevalence of iron-deficiency anaemia is high.24 By contrast, the Australian Haematology in Obstetric and Women’s Health Collaborative issued consensus recommendations in 2025 calling for routine ferritin screening in all pregnant women at the booking visit and again at 24 to 28 weeks, with repeat testing at 36 weeks if clinically indicated. They define iron deficiency in pregnancy as a ferritin level of less than 30 microg/L and recommend oral iron as first-line therapy, with alternate day dosing to improve tolerability, and intravenous iron for women who do not respond to or cannot tolerate oral iron, or who require treatment in the third trimester.11
Despite their differences, these positions reflect an international shift toward proactive detection and treatment of early iron deficiency.
Follow up and broader considerations
Follow up provides an opportunity to confirm treatment response, reassess symptoms and ensure appropriate safety-netting for less common but important causes of abnormal uterine bleeding. Although most cases of HMB bleeding are benign, clinicians should remain alert to red flags and patient-specific risk factors that warrant further investigation or referral, particularly to exclude endometrial pathology (Box).8 The lifetime risk of endometrial cancer is about one in 44 women in Australia, and risk increases with age, particularly after menopause.25 Although malignancy is uncommon in younger women such as Sally, it should remain part of the differential diagnosis when investigating abnormal bleeding patterns.
Follow up with reassessment of ferritin and transferrin saturation allows confirmation of iron repletion. Persistent fatigue despite correction of iron status should prompt broader reassessment, including consideration of mental health screening, sleep assessment and other contributing factors to low energy levels.
Case continued: follow up
Sally is reviewed four months later and reports significant improvement in her quality of life and fatigue. She had experienced mild bilateral breast tenderness and per vaginam spotting during the first month after the insertion, but these symptoms have resolved. On repeat testing, her ferritin levels are 113 microg/L. She is pleased to have reliable contraceptive support in place until she is ready to plan another pregnancy in about three years. Importantly, she is no longer entering a future pregnancy with depleted iron stores, because her GP has proactively managed both her iron deficiency and HMB, restoring iron reserves and reducing ongoing menstrual blood loss.
Conclusion
Iron deficiency, even in the absence of anaemia, is a clinically significant condition among women with HMB. Breaking the vicious cycle of chronic iron loss requires timely diagnosis using appropriate ferritin thresholds, restoration of iron stores including consideration of intravenous iron where appropriate, and active management of excessive menstrual blood loss to minimise ongoing losses and support sustained wellbeing. MT
COMPETING INTERESTS: Associate Professor Uppal is a Bayer media spokesperson for HMB on The Period Perspective Survey, CSL Vifor speaker, Ambassador for Heidi Health and RANZCOG media spokesperson and is part of the Bayer APAC Digital Thought Leader Academy program. She has also received payment from Bayer, Orion, Organon and Besins for GP Education and Implanon trainer sessions.
References
1. National Institute for Health and Care Excellence (NICE). Heavy menstrual bleeding: assessment and management 2018. Manchester: NICE; 2018. Available online at: https://www.nice.org.uk/guidance/ng88/resources/heavy-menstrual-bleeding-assessment-and-management-pdf-1837701412549 (accessed March 2026).
2. Munro MG, Mast AE, Powers JM, et al. The relationship between heavy menstrual bleeding, iron deficiency, and iron deficiency anemia. Am J Obstet Gynecol 2023; 229: 1-9.
3. World Health Organization (WHO). Anaemia in women and children. Geneva: WHO; 2025. Available online at: https://www.who.int/data/gho/data/themes/topics/anaemia_in_women_and_children (accessed March 2026).
4. International Federation of Gynecology and Obstetrics (FIGO). Iron deficiency and anaemia in women and girls. London: FIGO; 2023. Available online at: https://www.figo.org/sites/default/files/2023-09/FIGO-20Statement-20IDA-20in-20women-20and-20girls-202023_EN.pdf (accessed March 2026).
5. Frayne J, Pinchon D. Anaemia in pregnancy. Aust J Gen Pract 2019; 48: 125-129.
6. Araujo Costa E, de Paula Ayres-Silva J. Global profile of anemia during pregnancy versus country income overview: 19 years estimative (2000-2019). Ann Hematol 2023; 102: 2025-2031.
7. NSW Government. Heavy menstrual bleeding and hysterectomy. Canberra: NSW Government; 2025. Available online at: https://aci.health.nsw.gov.au/__data/assets/pdf_file/0006/984642/ACI-Heavy-Menstrual-bleeding-and-hysterectomy-evidence-series.pdf (accessed March 2026).
8. Australian Commission on Safety and Quality in Health Care. The heavy menstrual bleeding clinical care standard. Sydney: Australian Commission on Safety and Quality in Health Care; 2024. Available online at: https://www.safetyandquality.gov.au/publications-and-resources/resource-library/heavy-menstrual-bleeding-clinical-care-standard-2024 (accessed March 2026).
9. Warner PE, Critchley HO, Lumsden MA, Campbell-Brown M, Douglas A, Murray GD. Menorrhagia I: measured blood loss, clinical features, and outcome in women with heavy periods: a survey with follow-up data. Am J Obstet Gynecol 2004; 190: 1216-1223.
10. World Health Organization (WHO). WHO guideline on use of ferritin concentrations to assess iron status in individuals and populations. Geneva: WHO; 2020. Available online at: https://www.who.int/publications/i/item/9789240000124 (accessed March 2026).
11. Clarke L, Froessler B, Tang C, et al. Iron optimisation in pregnancy: a Haematology in Obstetric and Women’s Health Collaborative consensus statement. Intern Med J 2025; 55: 300-307.
12. Royal College of Pathologists of Australasia (RCPA). Iron studies standardised reporting protocol. Sydney: RCPA; 2021. Available online at: https://www.rcpa.edu.au/getattachment/554ba672-4d34-4e7c-b812-5741359bca78/Iron-Studies-Standardised-Reporting-Protocol.aspx (accessed March 2026).
13. Balendran S, Forsyth C. Non-anaemic iron deficiency. Aust Prescr 2021; 44: 193-196.
14. The Women’s. What causes heavy periods? Melbourne: The Royal Women’s Hospital. Available online at: https://www.thewomens.org.au/health-information/periods/heavy-periods/what-causes-heavy-periods (accessed March 2026).
15. Patterson AJ, Brown WJ, Powers JR, Roberts DC. Iron deficiency, general health and fatigue: results from the Australian Longitudinal Study on Women’s Health. Qual Life Res 2000; 9: 491-497.
16. Therapeutic Goods Administration (TGA). Australian product information – Ferinject. Canberra: TGA; 2023. Available online at: https://www.tga.gov.au/sites/default/files/2024-12/FOI-200012_1.pdf (accessed March 2026).
17. Tolkien Z, Stecher L, Mander AP, Pereira DI, Powell JJ. Ferrous sulfate supplementation causes significant gastrointestinal side-effects in adults: a systematic review and meta-analysis. PLoS One 2015; 10: e0117383.
18. Zimmermann MB. Iron absorption from oral iron supplements given on consecutive versus alternate days and as single morning doses versus twice-daily split dosing in iron-depleted women: two open-label, randomised controlled trials. Lancet Haematol 2017; 4: e524-e533.
19. Wang D, Sra M, Ito S, et al. Cost-effectiveness of first-line IV vs oral iron for iron-deficiency anemia in women with heavy menstrual bleeding. Blood Adv 2026; 10: 1508-1517.
20. Bryant-Smith AC, Lethaby A, Farquhar C, Hickey M. Antifibrinolytics for heavy menstrual bleeding. Cochrane Database Syst Rev 2018; 4: CD000249.
21. Creinin MD, Barnhart KT, Gawron LM, Eisenberg D, Mabey RG Jr, Jensen JT. Heavy menstrual bleeding treatment with a levonorgestrel 52-mg intrauterine device. Obstet Gynecol 2023; 141: 971-978.
22. Lewkowitz AK, Tuuli MG. Identifying and treating iron deficiency anemia in pregnancy. Hematology Am Soc Hematol Educ Program 2023; 2023: 223-228.
23. The Royal Australian and New Zealand College of Obstetricians and Gynaecologists (RANZCOG). Routine antenatal assessment in the absence of pregnancy complications. Melbourne: RANZCOG; 2019. Available online at: https://ranzcog.edu.au/wp-content/uploads/Routine-Antenatal-Assessment.pdf (accessed March 2026).
24. Living Evidence for Australian Pregnancy and Postnatal Care (LEAPP). Australian pregnancy care guidelines. Melbourne: LEAPP; 2024. Available online at: https://files.magicapp.org/guideline/3aa6b881-4be5-4806-bb3e-e7c53b5b0bbf/published_guideline_7933-2_0.pdf (accessed March 2026).
25. Australian Government. Uterine cancer statistics. Canberra: Australian Government; 2025. Available online at: https://www.canceraustralia.gov.au/cancer-types/endometrial-cancer/uterine-cancer-statistics (accessed March 2026).
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.