Vulval lichen sclerosus: not uncommon, serious and often missed

Vulval lichen sclerosus is a chronic, scarring dermatosis affecting genital skin in women of all ages. This article outlines the latest evidence-based approach to diagnosis, management, surveillance and follow up, highlighting practical strategies to improve long-term outcomes.
- Vulval lichen sclerosus is a chronic, underdiagnosed condition requiring lifelong treatment and surveillance.
- Untreated disease may result in anatomical distortion and carries a 2 to 6% lifetime risk of malignancy.
- Potent topical corticosteroids are safe, effective and remain the gold standard of treatment.
- Regular follow up and patient education are critical to ensuring adherence and preventing complications.
Vulval lichen sclerosus (VLS) is an uncommon skin disease with a predilection for genital skin. Although exact prevalence is unknown, estimates suggest it affects around 3% of women over the age of 50 years.
VLS is likely underdiagnosed. This reflects both inadequate awareness among health professionals and patient reluctance to present due to embarrassment. Although severe itch typically prompts women to seek help, about 10 to 15% of patients are asymptomatic, especially early in the course of the disease. As regular genital self-examination is uncommon, the condition is often missed by patients themselves.
VLS can occur at any age, with incidence peaking in late childhood and again in the peri- and postmenopausal years. It is relatively rare in girls. In clinical practice, about 5% of cases begin before 18 years of age, 20% between 18 and 50 years of age and 75% after 50 years of age. The mean age of onset is about 55 years.
Accurate VLS diagnosis is crucial for two key reasons. Firstly, without adequate aggressive treatment, at least half of VLS cases result in significant scarring, shrinkage and distortion of the vulva, as well as stenosis of the introitus. If left untreated, the labia minora eventually becomes reabsorbed, and the clitoris becomes entrapped and buried.1 Secondly, untreated VLS carries a 2 to 6% lifetime risk of developing squamous cell neoplasia of the vulva, including vulval intraepithelial neoplasia (VIN) and invasive squamous cell carcinoma.2
Adequate treatment can prevent or significantly reduce the risk of both complications and, in patients with prior malignancy, lower the risk of recurrence, thereby altering the course of the disease.1,3
Although there are no data on how often VLS genuinely remits, it is safest to assume that treatment should be lifelong, with ongoing observation and individually titrated treatment regimens, as would be done for any chronic condition.
Aetiology
The true aetiology of VLS remains unknown; however, there is a well-documented association with autoimmune disease, particularly Hashimoto’s thyroiditis and vitiligo.4 Cases of severe VLS have also been reported in patients treated with checkpoint inhibitors for cancers, particularly melanoma, further supporting the autoimmune theory of aetiology.5
Clinical presentation in adult women
VLS may occur on any part of the skin, but it is almost always primarily a genital condition, involving the vulva and, at times, extending to the perineum and perianal skin. The most common presenting symptom is a severe itch, often disrupting sleep and daily life. Pain resulting from excoriations or fissures, distressing clitoral hyperaesthesia and dyspareunia are also common.
In asymptomatic cases of VLS, diagnosis may occur incidentally during examination by the patient themselves or by their GP (e.g. during a routine smear test). These cases may present late, and thus be at advanced stages of disease or complicated by malignancy.
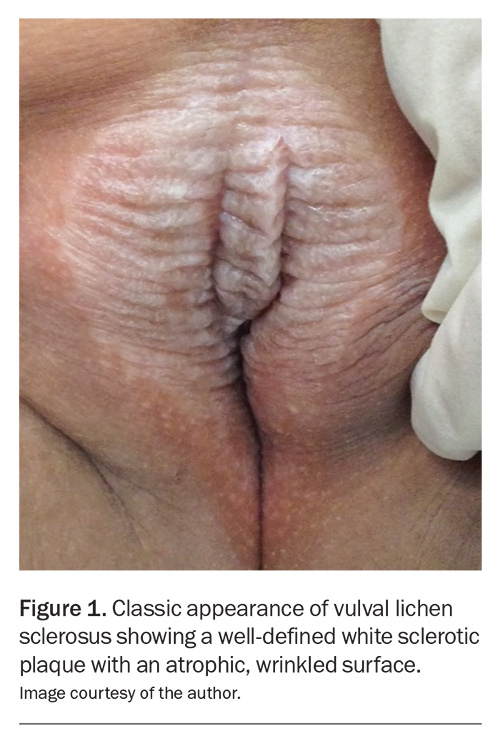
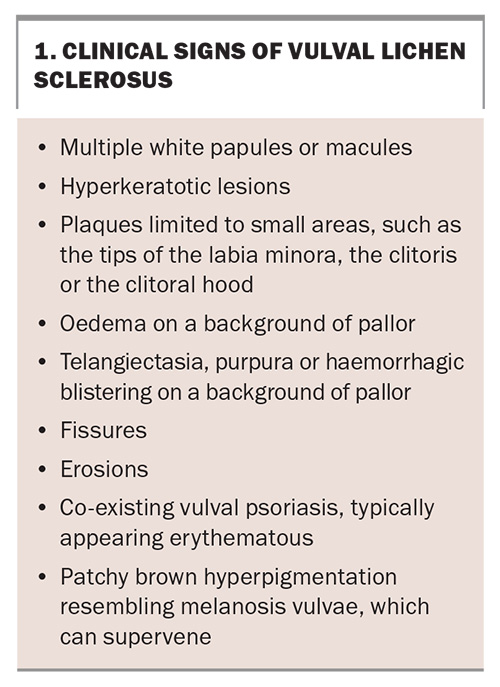
The hallmark clinical sign is a well-defined white sclerotic plaque with an atrophic, wrinkled surface (Figure 1). Although purpura and erosions may occur, they are less common. However, VLS can present with a wide range of clinical signs (Box 1).
VLS distribution is highly variable. The classic textbook description is of a figure of eight encircling the vulva, perineum and perianal skin. However, it can affect only the perianal region, clitoris, internal surface of the labia majora or labia minora, and the vaginal opening (i.e. introitus). VLS does not involve the vagina proper (that is, beyond the hymen) unless a patient has a vaginal prolapse that protrudes beyond the hymen.
In transgender women, VLS affects the same sites as in cisgender women, even when the labia majora are formed from scrotal skin, which is not typically involved in biological males. This suggests a possible role for oestrogen, although this has not been proven.
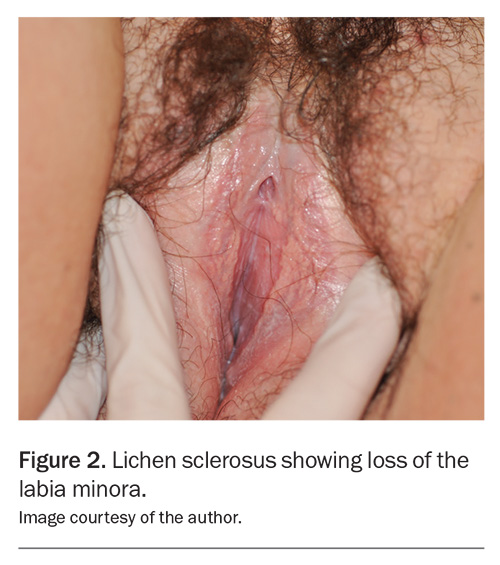
A key diagnostic clue for VLS, particularly in the later stages (but sometimes within six to 12 months from onset), is that the vulval shape is abnormal. Virtually all women develop labia minora. These are present, although diminutive, in prepubertal girls and enlarge around puberty. The size of the labia minora is highly variable, but their absence – or a clitoris that has shrunk or is buried under scar tissue – is very suggestive of VLS (Figure 2). The only other condition that will produce this is lichen planus, which can be distinguished from VLS as is not white but erythematous and often eroded.
VLS obeys the Koebner phenomenon, which means it localises to areas of friction and trauma. This possibly explains why it is usually most recalcitrant on the perineum and the inner surfaces of the labia minora.
Extragenital lesions of lichen sclerosus may be found on any part of the skin, but are most common on the neck, buttocks, inner thigh, shoulders and wrists. Extragenital lichen sclerosus can also occur as multiple small ‘confetti’ lesions.
Scarring
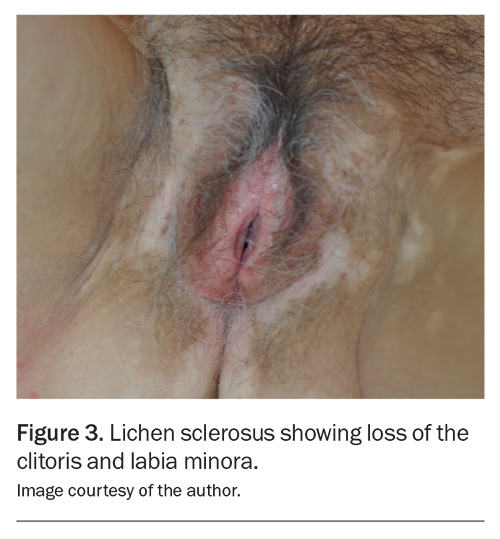
The tendency for VLS to gradually change the shape of the vulva is well recognised but underreported, and is a far more common complication than malignancy.1 The labia minora may completely resorb or fuse beneath the clitoris. The resulting fusion line is brittle and prone to tearing during intercourse. The clitoris itself may disappear; although the nerve supply is usually preserved and most patients can still achieve orgasm, the external structure may be entirely lost (Figure 3).
The end result is a vulva with completely absent normal anatomy. In rare, severely neglected cases, the introitus may become significantly stenosed, leading to pooling of urine within the vagina and mimicking urinary incontinence. In end-stage disease, epithelial changes may no longer be visible, and gross distortion of the vulva may be the only remaining sign.
Clinical presentation in girls
When VLS occurs in girls, it typically presents during the middle years of primary education. Late diagnosis is not uncommon. The most common presenting symptoms are itching and soreness. Other symptoms and signs at presentation may include purpura, dysuria, constipation, genital erosions and extragenital lesions. As in women, about 10% of girls are asymptomatic.6
Dysuria and pain with defaecation leading to constipation are presentations that differ from those in women, who more commonly present with itch and dyspareunia. It is not uncommon for girls with VLS to be referred to urologists or gastroenterologists. If purpura is present, they may be referred to child protection units.
The clinical appearance of VLS in girls is similar to that in women, with atrophy, fusion of the labia and loss of vulval architecture. Although prepubertal girls naturally have diminutive labia minora, a well-defined clitoris should be visible if the clitoral hood is deliberately retracted during examination. Clitoral phimosis is common in girls with VLS, but it may go unnoticed unless specifically looked for.
In girls who have not received adequate treatment, the labia minora may be lost or fail to develop during puberty. The risk of irreversible architectural loss in undertreated or untreated girls is significant.7
Diagnosis and investigation
VLS is often a clinical diagnosis, but it is not always possible to be certain. The symptoms of VLS – itching, soreness and dyspareunia – are common to many vulval conditions. Signs include white macules, papules and plaques; thickening of the vulval skin; swelling; fissures; and scarring.
The definitive diagnostic test is a vulval biopsy. The histology is distinctive and consistent across ages and genders. It typically shows an atrophic epidermis with hydropic degeneration of basal cells and a homogenous pale zone in the upper dermis. A lichenoid infiltrate, composed mainly of mononuclear cells, may also be present in the dermis.
A biopsy should be performed to confirm the diagnosis if there is any doubt about the clinical presentation or a concern about malignancy. All patients should be offered the option of a histopathological diagnosis. A definite tissue diagnosis provides patients with the certainty that long-term maintenance treatment is necessary.
Diagnosis requires only a very small skin biopsy, taken with a 2 to 4 mm punch biopsy from the most densely white area of involvement. It is important to note that prior treatment with topical corticosteroids may render the histological findings nonspecific.
Histopathological false negatives are rare. If biopsy is not feasible or is declined by the patient, a photographic record should be kept. This can be useful if the patient relocates or changes medical practitioners. Treated disease may appear clinically normal, so it is important to have a clear histopathological record of the diagnosis before treatment commences, to help prevent future clinicians from discontinuing treatment because clinical examination findings are now normal.
In girls, a clinical diagnosis is almost always sufficient due to the difficulties of biopsy, the limited differential diagnoses and the fact that neoplastic transformation has never been reported in paediatric VLS.
Should we test for associated diseases?
When patients are diagnosed with VLS and begin researching it on the internet, they invariably encounter frightening accounts of associated diseases and may become concerned that their immune system is compromised. Several conditions have been found to occur in association with VLS (Box 2). It is reasonable to test for thyroid function and thyroid autoantibodies; however, even when autoantibodies are present, thyroid function is often normal. Nevertheless, their presence is a useful marker and warrants ongoing monitoring for the development of frank thyroid disease. Other nondirected testing is unlikely to yield clinically significant findings.
Genetic factors
VLS can run in families, and several attempts have been made to identify a genetic association. Although no link has been found with the autoimmune-associated human leukocyte antigens (HLA) antigens HLA-A1, -B8 or -DR3, an association has been shown with the HLA class II antigen HLA-DQ7.
Although these documented HLA associations are of interest, the current evidence base is limited, and the strength of these associations remains inconclusive.
Differential diagnoses
Lichenified skin often appears white, particularly in Caucasian women. In women, the differential diagnosis includes lichenification of any origin, such as dermatitis, lichen planus and psoriasis. These conditions, although typically inflammatory and red, may appear white due to a thickened stratum corneum. Other conditions that may present with a white surface include extramammary Paget’s disease, genital warts, nonpigmented seborrhoeic keratosis and VIN. Vitiligo, which can co-occur with VLS, presents as sharply demarcated white patches but lacks symptoms and textural changes. Vitiligo lesions are fluorescent on ultraviolet light examination.
Vulval lichen sclerosus and associated malignancy
Before it was realised that VLS could be effectively treated, about 60% of all vulval keratinocyte carcinomas showed histological evidence of adjacent VLS, and it was well established that affected women had a 2 to 6% lifetime risk of developing vulval cancer. Any degree of VLS, even mild disease, carries this non-negligible malignancy risk. It is not possible to predict which patients will develop malignancy, and the interval from disease onset to cancer development has not been determined.
In adult women, but not girls, VLS is associated with vulval squamous cell carcinoma and VIN. A recently described entity, vulval aberrant maturation, may also be associated with VLS. Carcinoma in situ, known as differentiated VIN, as well as invasive carcinoma, can occur.8 Interestingly, extragenital lichen sclerosus is not associated with malignancy.
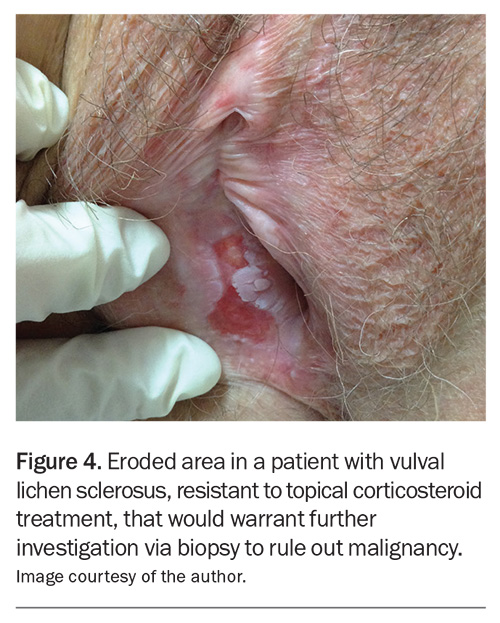
The clinical appearance of vulval squamous cell carcinoma can include nodules, persistent fissures, hyperkeratotic plaques, nonhealing ulcers and fungating tumours. Any change in an area affected by VLS that does not resolve promptly with topical treatment must be biopsied (Figure 4).
Although vulval malignancy has not been reported in girls, squamous cell carcinoma of the vulva has been described prior to 40 years of age in patients with childhood-onset VLS. The association with genital malignancy has important implications for management: patients must be made aware of the risk, educated about warning signs and provided with regular treatment and follow up.
Malignancy after treatment
Large cohorts of patients with adequately treated VLS appear to have a malignancy rate much lower than 6%. This has led to the suggestion that the risk of malignancy is significantly reduced when VLS is diagnosed early and treated appropriately. A prospective study conducted at The University of Sydney, published in 2015, followed 507 women with VLS and compared outcomes in those who adhered to treatment and those who did not.1 The study demonstrated that topical corticosteroid treatment that maintained objectively normal skin greatly reduced the risk of cancer. These findings confirmed earlier retrospective studies and the observations by other authors, supporting the conclusion that adequate control of VLS minimises the risk of subsequent malignancy.
Over the 10 years since the publication of The University of Sydney study, none of the patients in the original cohort who have remained under observation and on regular topical corticosteroid treatment have developed a cancer. The study also found that regular treatment significantly reduces the risk of developing new scarring and the progression of scarring existing at the time of first presentation – an observation supported by long-term follow up of the cohort.1
These findings suggest that the best outcomes for women with VLS are achieved through consistent efforts to obtain complete disease suppression and ongoing, careful surveillance.
Management
An Australian consensus statement on the diagnosis and management of VLS was published in 2021.9 In adult women, VLS is a lifelong disease that is unlikely to remit. It is unknown how many patients relapse upon ceasing treatment, but given the potential consequences of untreated disease, it is safest to assume that permanent remission is unlikely. Relapse may take many months and is often silent.
This reality can be difficult for some patients to accept. It is important to reinforce the chronic nature of VLS at every visit until it is clear the patient understands and accepts the need for long-term treatment. Drawing a comparison to diabetes, a chronic but manageable condition widely understood to be incurable, can be a helpful analogy.
Even in cases where the disease appears to have remitted, long-term observation is crucial, as VLS can reactivate after many years of dormancy.
No universally accepted severity scale for VLS exists. However, a practical approach combines patient-reported quality of life impact – such as that measured by the vulval quality of life index, which was validated in Australia – with clinical observation of the degree of whiteness and scarring.10
The principles of management are set out in Box 3.10
Topical therapy
There are two phases of treatment for VLS: induction of remission and maintenance treatment.
Potent topical corticosteroids are widely accepted as the gold standard for obtaining remission in VLS. The first report of this approach, published in 1991, used clobetasol propionate 0.05%, a superpotent topical corticosteroid.11 Prior to this, it was considered inappropriate to apply such strong corticosteroids to genital skin. As a result, treatment regimens relied on weaker topical corticosteroids, testosterone and progesterone, and VLS was considered very difficult to manage.
The 1991 study demonstrated that potent corticosteroids were both effective and safe, shifting the paradigm of VLS treatment. Since then, VLS has become one of the easiest vulval conditions to manage. Numerous subsequent studies using potent and superpotent topical corticosteroids have confirmed the safety and efficacy of this approach.
VLS is so responsive to topical corticosteroids that a lack of improvement should prompt reconsideration of the diagnosis, concerns about treatment adherence or evaluation of confounding factors such as allergy or superinfection.
Most subsequent studies have also used clobetasol propionate, although more recent trials have employed mometasone furoate 0.1%.12 Until recently, clobetasol was only available in Australia as a compounded, unapproved medication, whereas it was widely available in New Zealand. However, in practice, any potent topical corticosteroid can achieve the desired outcome. The main focus of treatment should not be on the specific product used, but on the treatment goal: to attain and maintain normal skin.
There is no single regimen that suits all patients, and clinicians should use their judgement, taking into account disease severity and patient preference for daily versus intermittent treatment. It appears, however, that regimens that work best are generally used at least three to four times per week; very intermittent regimens are less effective and more likely to be forgotten.
Induction of remission
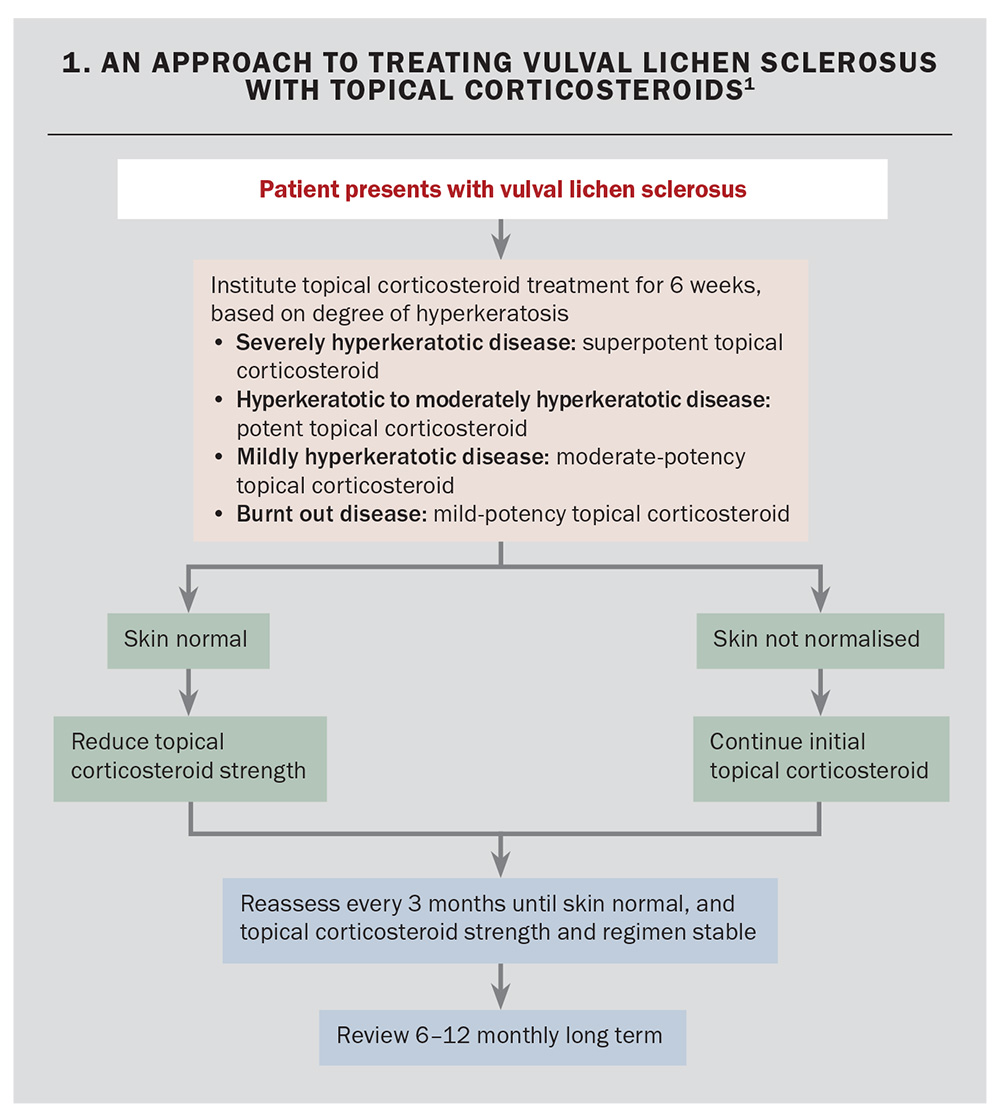
The potency of topical corticosteroid should be selected based on the degree of hyperkeratosis (thickening) of the vulval skin. Although existing scarring at presentation may influence symptom severity, it cannot be reversed by topical treatment. In select cases, surgery may be appropriate.
Suggested regimens based on severity are listed below.
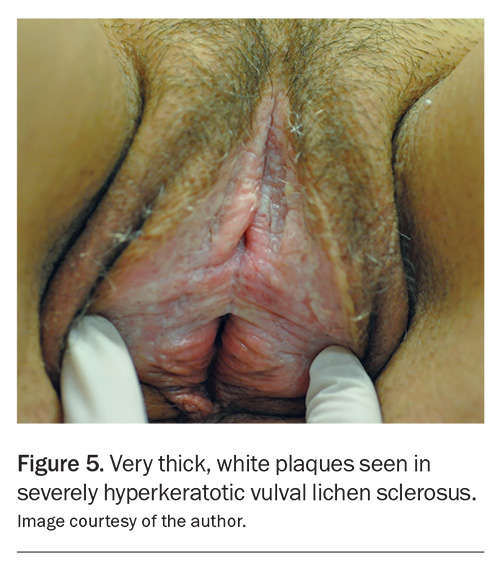
- Severely hyperkeratotic disease (Figure 5): superpotent topical corticosteroid (e.g. clobetasol propionate 0.05% ointment or betamethasone dipropionate 0.05% in optimised vehicle containing propylene glycol) applied twice daily until itching ceases (usually one to two weeks), then once daily until review at six weeks.
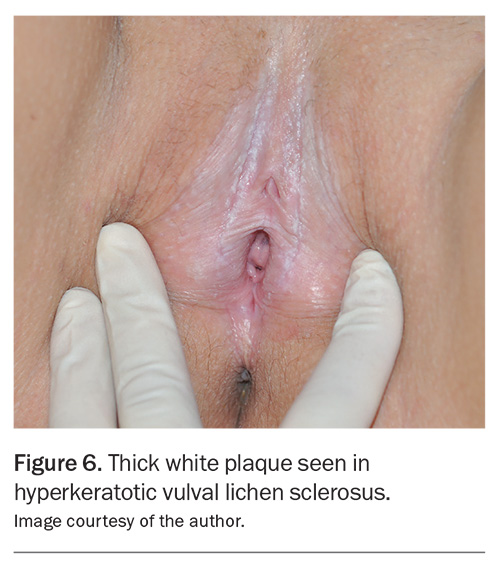
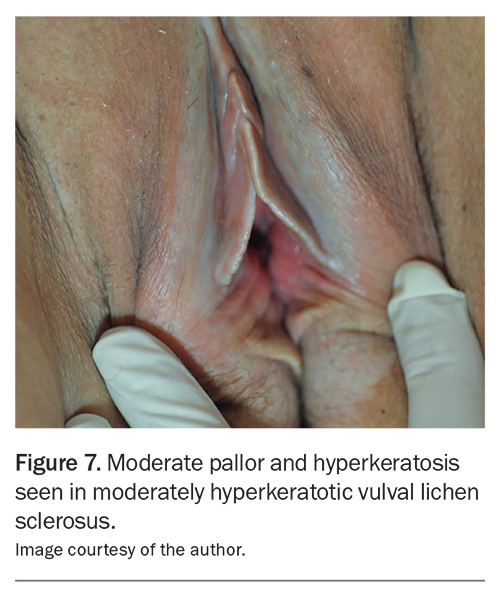
- Hyperkeratotic to moderately hyperkeratotic disease (Figure 6 and Figure 7): potent topical corticosteroid (e.g. betamethasone dipropionate 0.05% ointment or mometasone furoate 0.1% ointment) applied twice daily until itching ceases, then once daily until review at six weeks. Mometasone furoate may sting, and is particularly poorly tolerated by girls.
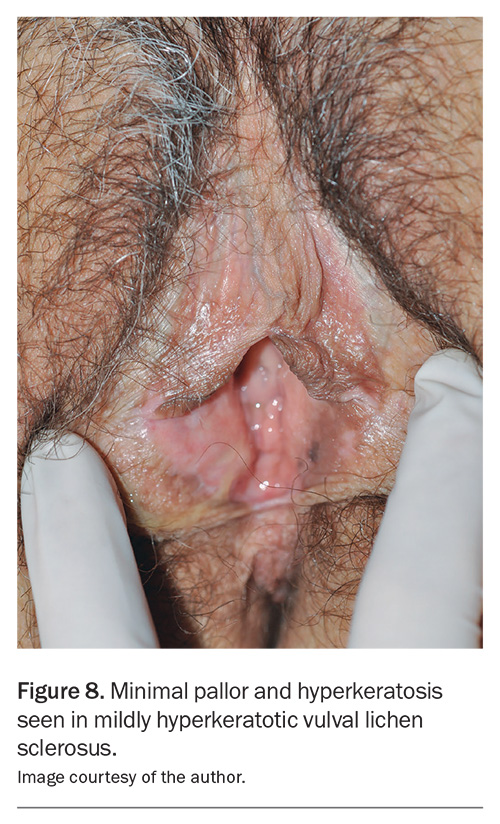
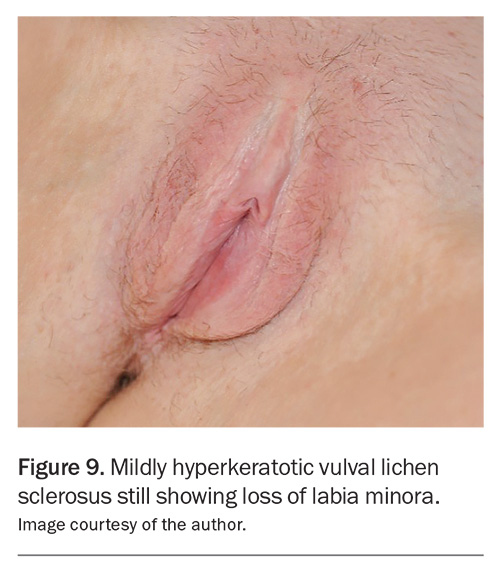
- Mildly hyperkeratotic disease (Figure 8 and Figure 9): moderate-potency topical corticosteroid (e.g. methylprednisolone aceponate 0.1% fatty ointment) applied daily until review at six weeks.
- Very mildly hyperkeratotic (burnt out) disease: mild-potency topical corticosteroid (e.g. hydrocortisone 1.0% ointment) applied once daily until review at six weeks.
The six-week review is critical to assess treatment response, monitor for side effects and provide emotional support. VLS is often a difficult diagnosis for most women to accept, and at this point most feel significantly better and may assume they are cured. It is important to emphasise that long-term treatment is essential to prevent cancer and scarring.
The initial potency of topical corticosteroid should be continued until both skin texture and colour return to normal. Residual hyper- or hypopigmentation may persist but are not signs of active disease. Clinically, the skin surface usually shows marked improvement.
Further reviews should be scheduled at three months and then every six months for the first two years. During this time, topical corticosteroid potency should be slowly titrated down to a moderate to mild formulation for maintenance therapy.
The aim of treatment is both symptom resolution and the disappearance of abnormal clinical signs. Symptoms typically resolve quickly, but abnormal signs – particularly skin thickening and colour changes – may take longer. Patients must therefore continue their regular treatment even after symptom resolution. Decisions regarding management should be guided by objective clinical response. Adherence is best when treatment is incorporated into the patient’s daily routine. Patients who struggle with adherence benefit from regular six-monthly reviews.
The average time to achieve normal skin is four to six months of consistent treatment. Changes such as scarring and loss of structure are not reversible with topical corticosteroid treatment, although clitoral phimosis may resolve, particularly if not severe.
Flowchart 1 shows an approach to the management of VLS.1
Maintenance treatment
Regimens for the long-term treatment of VLS are far less well defined than those for initial disease control, primarily due to a lack of robust data. Although many reviews and published articles state that VLS does not resolve spontaneously and requires ongoing management, there is no consensus on what this long-term treatment should involve. The main limitation of most published studies is their short follow-up duration, typically no more than three years.
There is no single long-term regimen suitable for all patients, as treatment must be tailored to the severity of the disease. Practitioners who limit their prescribing to agents only supported by published data may find their therapeutic options too narrow. It is essential to understand the relative potencies of available topical corticosteroids, noting that their availability differs between countries (Box 4).
In general, ointments are preferable to creams as they adhere better and are less likely to sting. However, patient preference must be considered, as this improves long-term adherence.
The overarching aim of maintenance therapy is to preserve normal vulval skin texture and colour. Treatment should be titrated accordingly. A typical maintenance regimen involves a moderate-potency topical corticosteroid used daily or two to three times per week – but this represents the minimum frequency of use. For forgetful patients, a routine involving a moderate-potency corticosteroid such as methylprednisolone aceponate 0.1%, applied two to three times per week, alternating with a mild-potency corticosteroid such as hydrocortisone 1% on other days, could be helpful in establishing a daily routine.
However, it is important not to follow a rigid, ‘recipe-style’ approach to managing VLS since there is no single regimen that suits all patients. As with any chronic disease, treatment should be titrated according to disease severity. Regimens may range from daily application of a superpotent topical corticosteroid to daily use of a mild-potency corticosteroid, but treatment should not be less frequent than once weekly.
A common technique involves initiating treatment with a potent to superpotent preparation daily until skin normalises, then stepping down: alternating superpotent with moderate-potency, then moderate-potency alone, and finally alternating moderate- with mild-potency topical corticosteroids. Treatment intensity should be adjusted to maintain normal skin colour. If the skin is white, potency should be increased; if red, it should be reduced.
Potential problems with long-term potent topical corticosteroid use on the genital skin include atrophy (evidenced by fragility and striae), corticosteroid-induced erythema and, rarely, superinfection, usually with Candida. However, most studies report that such side effects are rare. Corticosteroid-induced erythema can occur, particularly with stronger agents, but is usually asymptomatic and reversible with dose reduction. Importantly, it is preferable to recurrence of active disease. Therefore, concerns about corticosteroid-induced skin atrophy do not justify undertreatment of VLS.
Treatment should be reassessed every six months until stable remission is achieved, allowing titration to the lowest effective maintenance regimen. Topical corticosteroid potency is guided by the degree of hyperkeratosis, typically reflected by skin whiteness. If whiteness relapses, the strength of treatment should be increased. If atrophy or corticosteroid dermatitis (manifesting as irritability and redness) occurs, potency should be reduced. This tailored approach is successful, safe, inexpensive and outstandingly effective, and appears to reduce the incidence of malignancy to near zero, while significantly limiting disease progression and scarring. More than 90% of patients achieve complete and sustained symptom control, and among those who are sexually active, more than 90% report resolution of dyspareunia. A recent study also demonstrated significant improvement in quality of life with good disease control.13
It is unwise to adopt an ‘as needed’ approached focused solely on symptom control, even though this is supported in some guidelines. Suppressing objective disease activity – not merely relieving symptoms – should be the target outcome, as asymptomatic patients are still at risk of scarring or malignant transformation. Many patients who later developed cancer or progressive disease reported poor treatment adherence, often because symptom resolution led them to believe they were cured.
Once a patient has been in a stable remission for two years, review can occur annually. Intervals longer than this are associated with poorer outcomes due to lapses in treatment adherence. Recurrence in a patient who has finally achieved remission after years of suffering can be psychologically devastating. Each review is an opportunity to reinforce the importance and safety of maintenance treatment.
Although some authors argue that regular follow up is a burden on the health system, VLS is relatively uncommon, and the cost of even a single case of vulval cancer far outweighs that of ensuring regular follow up in multiple patients. Ideally, competent nurse practitioners could be trained to carry out meaningful follow up and monitoring of VLS patients.
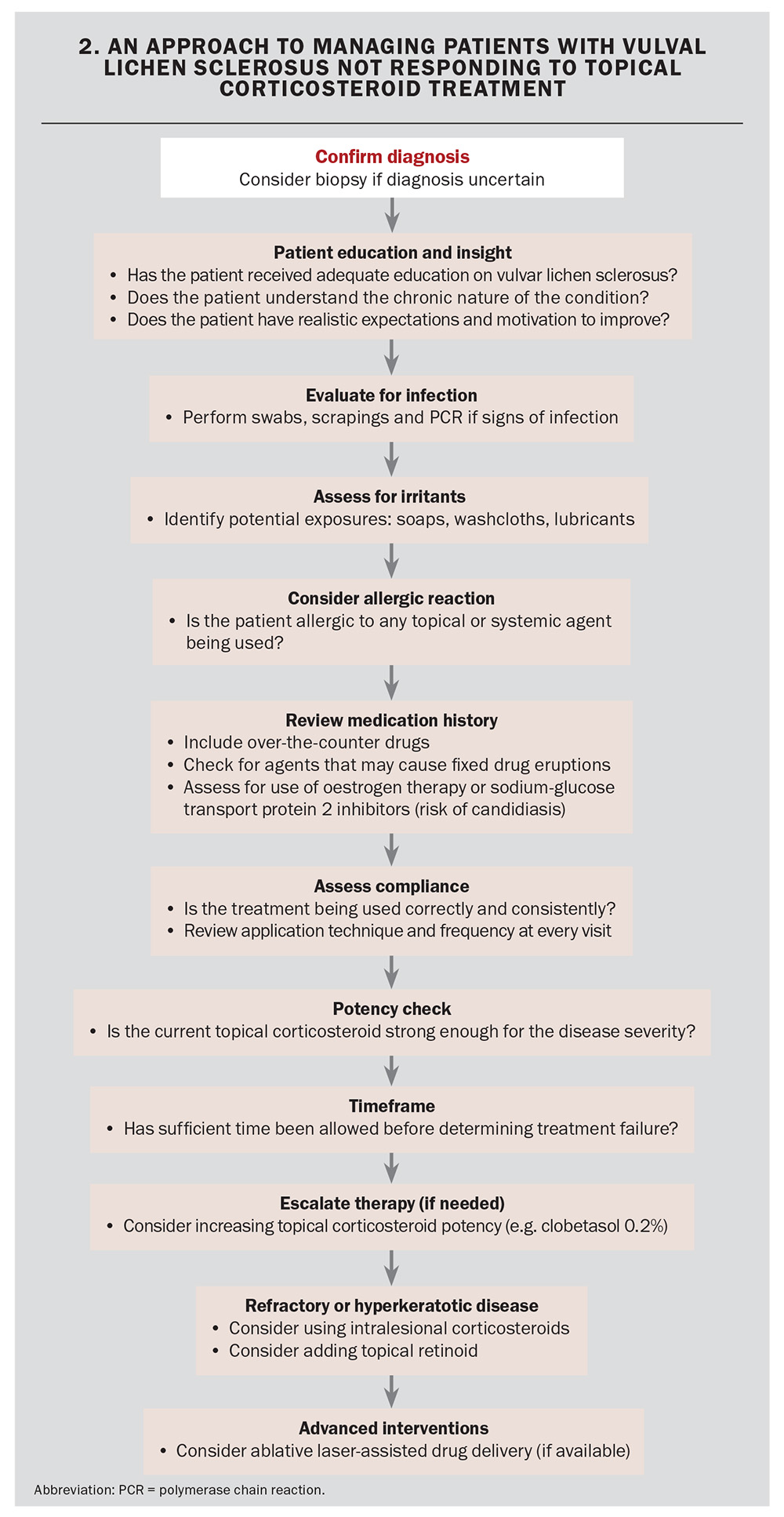
When topical corticosteroids are ineffective, not tolerated or refused
Although resistance to topical corticosteroids is rare and tolerance is generally excellent, some patients decline treatment due to safety concerns. Corticosteroid phobia remains a challenge, often reinforced by misunderstandings regarding the role of corticosteroids in dermatological conditions and risk aversion among health professionals.
Nonresponse is most often due to misdiagnosis, as VLS symptoms are nonspecific and overlap with other vulval conditions. While white plaques with purpura or cigarette-paper wrinkling are pathognomonic, they are not always present. Other lichenified skin conditions, such as lichen planus, can also cause scarring. As such, loss of vulvar structure is very characteristic but not confined to VLS.
Occasionally, normal anatomical variation – such as a small labia minora or an inconspicuous clitoris – may be mistaken for VLS. However, structural loss alongside typical white plaques strongly suggests the diagnosis.
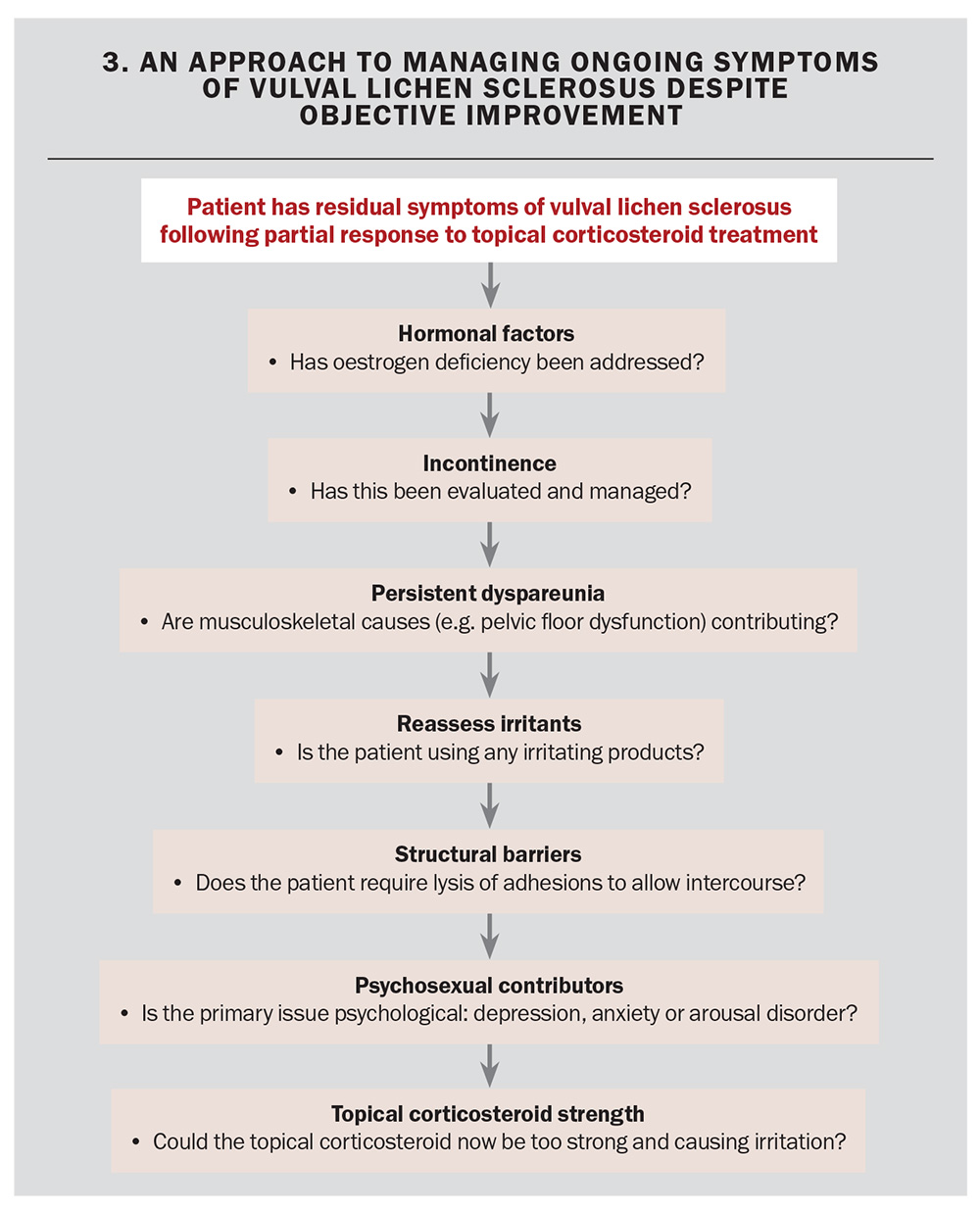
Ongoing symptoms may also result from concurrent infection, allergy to or irritancy caused by medication and other applied substances, continence issues, postmenopausal dryness, scarring and chronic pain conditions associated with pelvic floor dysfunction or neuropathy.14 Nonadherence is another frequent cause, driven by anxiety, denial or misunderstanding. It is therefore important to explore patient beliefs and attitudes towards long-term treatment at every visit.
An approach to managing patients with VLS who do not respond to treatment is shown in Flowchart 2. Flowchart 3 outlines the approach to managing ongoing symptoms despite objective clinical improvement.
Treatment alternatives to topical corticosteroids
Platelet-rich plasma, often misrepresented online as a stem-cell treatment, has been proposed as a treatment for VLS, but lacks evidence, is invasive and expensive. In very hyperkeratotic disease, ablative laser (laser-assisted drug delivery) may help in reducing hyperkeratosis and facilitating corticosteroid absorption, but must be used in conjunction with a topical corticosteroid.
Topical immunosuppressants such as tacrolimus and pimecrolimus have been proposed as a VLS treatment in girls and women, but concerns remain due to the premalignant nature of the condition and the potential risk of promoting malignancy. Cases of squamous cell carcinoma have been reported in women with VLS in association with the use of such topical immunosuppressants.15 These agents offer no advantage over topical corticosteroids, are more expensive, are very likely to sting or burn and lack long-term safety data.
Topical testosterone is no longer used as a VLS treatment, due to ineffectiveness and the risk of androgenisation in girls. Similarly, topical oestrogen has no role in VLS, except for managing hypo-oestrogenic symptoms in sexually active postmenopausal women.
Systemic therapies – including methotrexate, retinoids, hydroxychloroquine and adalimumab – have been used anecdotally but are rarely needed, as most cases respond well to appropriately potent and sustained topical corticosteroid therapy.16,17
Management in girls
The situation in girls is less well documented than in women; however, the principles of treatment with topical corticosteroids are the same. Historically, it was believed that childhood VLS would improve or remit at puberty, but this is incorrect. Although few studies have examined long-term outcomes in adolescent girls who developed VLS in childhood, all available data casts doubt on this assumption.18
It is therefore essential that both parents and patients understand that resolution at puberty is unlikely, and that the condition requires long-term follow up, just as it does in women. A recently completed retrospective study of 46 girls with VLS, comparing those who were adherent with treatment with those who were not, showed that when normal skin is attained and maintained, disease progression ceases and scarring and atrophy do not occur.6 However, any scarring that is present prior to treatment will not reverse. The most common changes are loss of the labia minora and clitoral phimosis. The latter may improve with treatment, but once labia minora have resorbed, they will not regrow.6
More recent research has shown that the risk of scarring is greatly increased in girls who are not adherent with treatment, particularly around puberty, when the genital area is developing adult proportions.7 Follow up during adolescence is difficult, as many teenage girls feel embarrassed about genital examination. To avoid this, some may reassure their parents and doctors that they are asymptomatic, leading to a loss to follow up.
VLS may have a profound psychological impact on both young and adolescent girls. Many develop unvoiced fears around sexuality and reproduction, and often feel isolated or different from their peers. Fear of using tampons is common, and usually stems from concerns about their genital area. Around puberty, when girls should be developing autonomy, many with VLS begin to resist treatment and wish to self-manage. This is a crucial developmental stage, and if control of the condition is lost, there is a risk of long-term physical and psychological consequences.
A recent retrospective study of women with a history of childhood VLS supports these concerns and highlights the need for individualised regimens and regular follow up.19 More work is required in this area of social science to determine how best to support these young women.
Lichen sclerosus and sexual abuse in girls
Concerns about possible sexual abuse frequently arise when girls with VLS are examined, due to the presence of erosions, fissures, purpura, bleeding or scarring. There have been numerous reports of girls with classic VLS features undergoing extensive and inappropriate evaluations for sexual abuse. Although increased awareness of sexual abuse among healthcare professionals has led to more timely referrals for correct diagnosis of VLS, the additional emotional trauma inflicted on the child and family in these cases is entirely unnecessary.
Sexual abuse is unfortunately common, with retrospective studies suggesting that approximately 20 to 25% of females have experienced such abuse in childhood. However, girls who have been sexually abused rarely have clinical signs at the time of examination. A diagnosis of VLS neither supports nor rules out the possibility of abuse, and both conditions must be considered independently.
Surgical therapy
Historically, vulvectomies have been performed in women with VLS, but recurrence of the disease following surgery was common. This approach is no longer considered acceptable and is now completely contraindicated.
In girls, surgery is rarely indicated unless there is significant fusion of the labia. A variety of procedures have been employed to treat labial and peri-clitoral adhesions. Simple division of adhesions can provide an excellent outcome, provided that potent topical corticosteroids are applied daily postoperatively until healing is complete. In some cases, the corticosteroid may need to be applied using a dilator to ensure adequate contact with the affected area.20
Can patients with lichen sclerosus resume normal sexual activity?
In most cases, the answer is yes. Physically, particularly in younger women or those treated before significant disease progression, there is usually no barrier to resuming a normal sex life.
Women with a long history of painful intercourse often develop pelvic floor dysfunction and may benefit from physiotherapy. Others may have developed an aversion to sex prior to diagnosis and might require psychological support. Some older women decline intervention due to a lack of interest in sex after menopause. However, those who wish to become sexually active again usually do so successfully.
When to refer
Referral decisions depend on a GP’s experience and confidence in diagnosing and managing VLS.
If VLS is suspected, confirmation with biopsy is ideal, for the reasons discussed above. If the GP is not comfortable per- forming a biopsy, referral to either a dermatologist or a gynaecologist is appropriate. Dermatologists generally have more experience and knowledge of VLS, as it is a skin condition; however, gynaecologists are also capable of performing biopsies. Although male dermatologists are typically well versed in VLS, some female patients may prefer to see a female practitioner, which can lead to deskilling in some male clinicians over time.
It is not acceptable to make a diagnosis of VLS without initiating treatment. If a dermatologist confirms the diagnosis but the patient is referred back without a clear plan, topical corticosteroid therapy should be commenced as outlined above.
GP-led long-term management of VLS could significantly reduce the burden on specialist services and improve access for new patients. Referral is appropriate when a patient is not responding to treatment despite an accurate diagnosis, or if there is suspicion of malignancy.
Some patients prefer regular specialist follow up, and if this supports adherence to treatment and provides reassurance, it is a valid reason for referral. In general, annual review is recommended, or every two years in very stable cases.
Conclusion
VLS is a potentially disfiguring and premalignant condition that responds well to early and consistent treatment. Long-term disease control hinges on regular use of potent topical corticosteroids, careful surveillance and patient education. With an individually tailored approach and ongoing care, clinicians can help patients maintain function, quality of life and avoid long-term complications. MT
COMPETING INTERESTS: None.
References
1. Lee A, Bradford J, Fischer G. Long-term management of adult vulvar lichen sclerosus: a prospective cohort study of 507 women. JAMA Dermatol 2015; 151: 1061-1067.
2. Bleeker MC, Visser PJ, Overbeek LI, van Beurden M, Berkhof J. Lichen sclerosus: incidence and risk of vulvar squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev 2016; 25: 1224-1230.
3. Chin S, Scurry J, Bradford J, Lee G, Fischer G. Association of topical corticosteroids with reduced vulvar squamous cell carcinoma recurrence in patients with vulvar lichen sclerosus. JAMA Dermatol 2020: 156: 813-814.
4. Guttentag A, Wijaya M, Fischer GO, Lee A, Liu K, Saunderson RB. A guide to screening for autoimmune diseases in patients with vulvar lichen sclerosus. Australas J Dermatol 2025; 66: 135-141.
5. Truong K, Jones-Caballero M, Chou S, Carlino MS, Consuegra-Romero G, Araujo RR. Lichen sclerosus and immune checkpoint inhibitors: a case and review of the literature. Australas J Dermatol 2023; 64: 158-161.
6. Ellis E, Fischer G. Prepubertal-onset vulvar lichen sclerosus: the importance of maintenance therapy in long-term outcomes. Pediatr Dermatol 2015; 32: 461-467.
7. Kherlopian A, Fischer G. Does compliance to topical corticosteroid therapy reduce the risk of development of permanent vulvar structural abnormalities in pediatric vulvar lichen sclerosus? A retrospective cohort study. Pediatr Dermatol 2022; 39: 22-30.
8. Heller DS, Day T, Allbritton JI, et al. Diagnostic criteria for differentiated vulvar intraepithelial neoplasia and vulvar aberrant maturation. J Low Genit Tract Dis 2021; 25: 57-70.
9. Yeon J, Oakley A, Olsson A, et al. Vulval lichen sclerosus: an Australasian management consensus. Australas J Dermatol 2021; 62: 292-299.
10. Saunderson RB, Harris V, Yeh R, Mallitt KA, Fischer G. Vulvar quality of life index (VQLI) - a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol 2020; 61: 152-157.
11. Dalziel KL, Millard PR, Wojnarowska F. The treatment of vulval lichen sclerosus with a very potent topical steroid (clobetasol propionate 0.05%) cream. Br J Dermatol 1991; 124:461-464.
12. Virgili A, Borghi A, Minghetti S, Corazza M. Mometasone fuoroate 0.1% ointment in the treatment of vulvar lichen sclerosus: a study of efficacy and safety on a large cohort of patients. J Eur Acad Dermatol Venereol 2014; 28: 943-948.
13. Wijaya M, Lee G, Fischer G, Lee A. Quality of life in vulvar lichen sclerosus patients treated with long-term topical corticosteroids. J Low Genit Tract Dis 2021; 25: 158-165.
14. Wijaya M, Lee G, Fischer G. Why do some patients with vulval lichen sclerosus on long-term topical corticosteroid treatment experience ongoing poor quality of life? Australas J Dermatol 2022; 63: 463-472.
15. Lewis FM, Neill SM. Safety of calcineurin inhibitors in the management of lichen sclerosis. Br J Dermatol 2007; 156: 1389-1390.
16. Manuelpillai N, Saunders H, Veysey E. Management of severe vulval lichen sclerosus with adalimumab. Australas J Dermatol 2022; 63: 248-250.
17. Hargis A, Ngo M, Kraus CN, Mauskar M. Systemic therapy for lichen sclerosus: a systematic review. J Low Genit Tract Dis 2024; 28: 84-90.
18. Smith SD, Fischer G. Childhood onset vulvar lichen sclerosus does not resolve at puberty: a prospective case series. Pediatr Dermatol 2009; 26: 725-729.
19. Morrel B, Kampherbeek HJ, Ten Kate-Booij MJ, et al. Growing up with juvenile vulvar lichen sclerosus: the experiences and care needs of adult women with lichen sclerosus since childhood - a qualitative exploration. Br J Dermatol 2024; 191: 556-567.
20. Bradford J, Fischer G. Surgical division of labial adhesions in vulvar lichen sclerosus and lichen planus. J Low Genit Tract Dis 2013; 17: 48-50.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.



-Fischer-Vulva_0.jpg)