What’s the diagnosis?
Acral lesions of recent onset and aphthous mouth ulcers
Case presentation
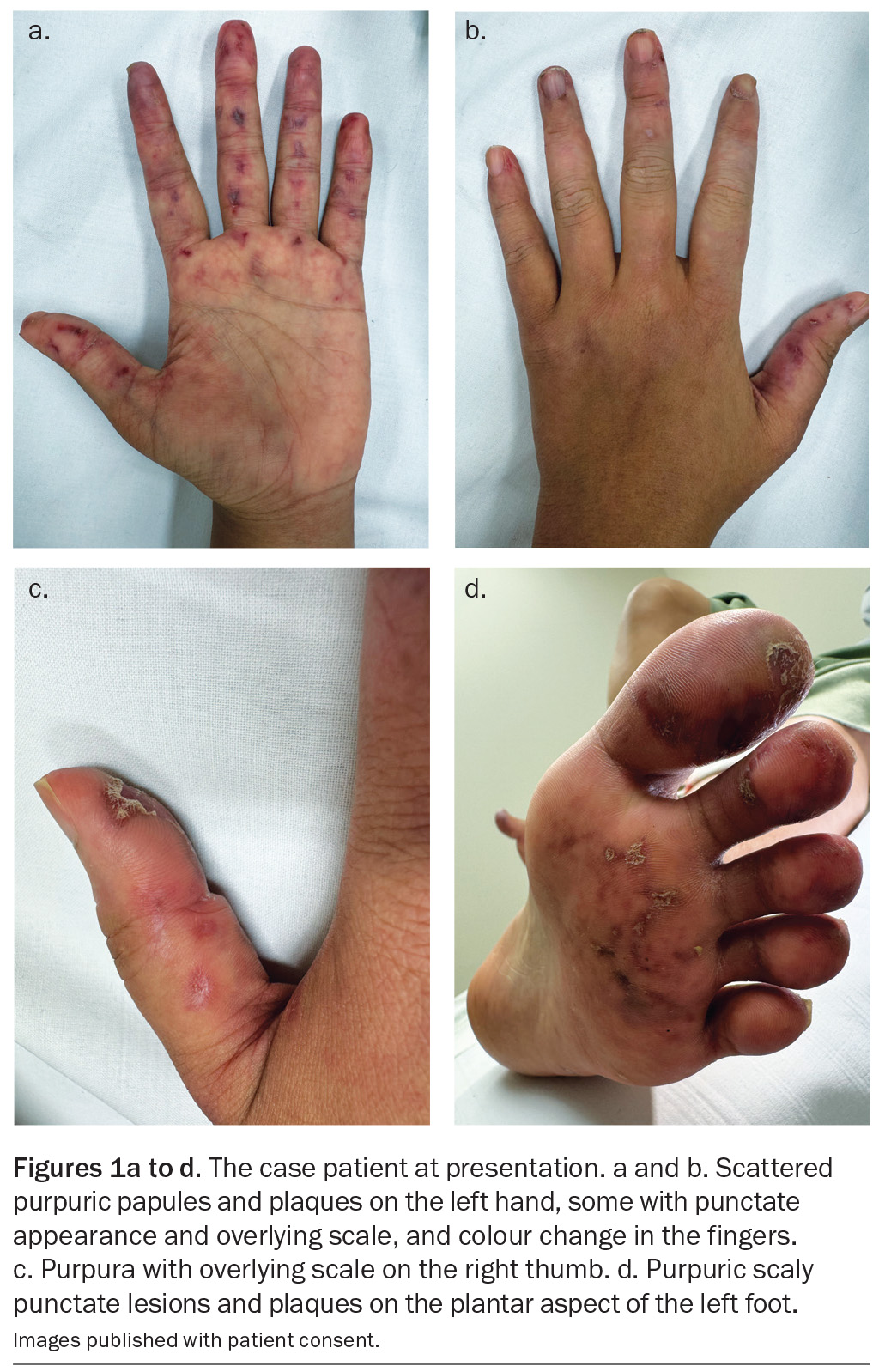
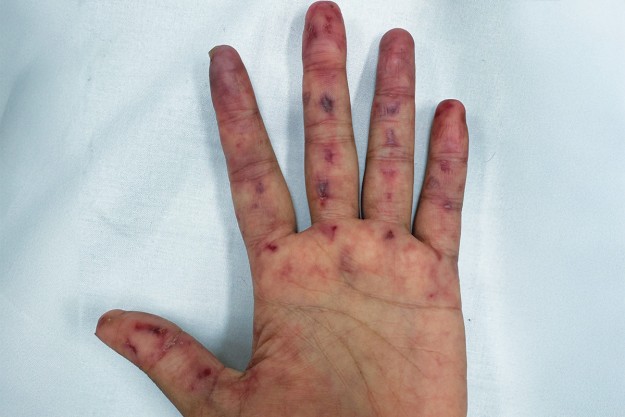
A 34-year-old woman presents with acral lesions of recent onset involving her hands and feet as well as colour change in her fingers (Figures 1a to d). She also reports two episodes of painless mouth ulcers.
The patient has a three-month history of fatigue, intermittent low-grade fevers and migratory pains affecting the metacarpophalangeal and interphalangeal joints. She is not taking any new medications. She has recently travelled overseas.
On examination, scattered purpuric papules and plaques are observed, some of which have a punctate appearance, with overlying scale. The hands appear cyanotic with erosions. There are shallow aphthous ulcers on her hard palate and she has mild ankle oedema. There is nonscarring alopecia along the hairline. No malar erythema, ragged cuticles or periungual telangiectasia are present. She is afebrile.
Differential diagnoses
Conditions to consider among the differential diagnoses include the following.
Cutaneous small vessel vasculitis
Cutaneous small vessel vasculitis (CSVV) is an immune-complex mediated inflammation of postcapillary venules in the skin.1 Patients characteristically present with nonblanching palpable purpura, sometimes with petechiae, haemorrhagic bullae, superficial ulcerations and associated pain or oedema.2,3 There is a predilection for the lower limbs, largely reflecting the effects of gravity on hydrostatic pressure and venous return in these dependent regions.2,4 Systemic features are usually absent.
CSVV predominantly affects adults.2 Common precipitants include medications (particularly penicillins, cephalosporins, sulfonamides, phenytoin, allopurinol, NSAIDs and immune-checkpoint inhibitors) and infections (such as hepatitis B or C, chronic bacteraemias such as endocarditis, HIV and, occasionally, SARS-CoV-2 infection).1,3 Onset typically occurs seven to 10 days after exposure to a new drug or infectious antigen; persistent antigens can drive a chronic, waxing–waning course, but most cases subside within two or three weeks.2 Temporal linkage to a new medication or recent infection is a practical clue to diagnosis. However, most cases are idiopathic.
For the case patient, CSVV would not explain her systemic features or the subacute nature of the presentation. This was not the correct diagnosis.
Hand, foot and mouth disease
Hand, foot and mouth disease (HFMD) is an acute enteroviral vesicular stomatitis characterised by painful oral ulcerations and a papulovesicular exanthem on the palms and soles.5 It occurs worldwide, with seasonal peaks in late spring and frequent outbreaks in childcare and school settings.5 Most cases are seen in children under 10 years of age, but
during community outbreaks a higher number of cases may be observed among adolescents and adults (especially caregivers).6-8
The causative agents of HFMD are human enteroviruses, most frequently coxsackievirus A6 and enterovirus 71.9 Transmission occurs via the faecal–oral and oral–oral routes and by respiratory droplets. The incubation period is typically three to six days and patients are most infectious in the first week. However, stool shedding may persist for four to eight weeks.9 The disease typically begins with a low-grade fever and sore throat, followed by tender vesicles (2 to 6 mm) on the palms and soles that evolve from erythematous macules. Oral ulcers are commonly seen on the posterior soft palate. The combination of a brief prodrome, highly contagious course with household contacts, acral vesicles plus painful oral enanthem favours acute viral exanthem. Patients usually experience resolution in seven to 10 days and no chronic systemic symptoms.
This was not the correct diagnosis for the case patient, who was still experiencing systemic symptoms after three months.
Nontuberculous mycobacterial
infection
Cutaneous nontuberculous mycobacterial infection refers to skin and soft tissue disease caused by environmental mycobacteria other than the Mycobacterium tuberculosis complex or M. leprae. Cutaneous disease usually follows direct inoculation and is most often caused by slow growing organisms, such as M. marinum and M. ulcerans, or rapidly growing organisms, such as M. abscessus group, M. chelonae and M. fortuitum.10
Nontuberculous mycobacteria are ubiquitous in tap and surface water, soil and biofilms, with infections reported worldwide.11 Skin disease arises across age groups but clusters occur after specific procedures, with outbreaks being described after tattooing, cosmetic and surgical procedures, and exposure to contaminated water sources.11M. ulcerans occurs in defined geographic foci, such as parts of Australia (coastal Victoria and smaller foci reported in far north Queensland) and West and Central Africa, whereas M. marinum is linked to aquatic environments and domestic aquaria.12
Cutaneous nontuberculous mycobacteria infections typically present as indolent violaceous papules or nodules that may ulcerate, abscess or track along lymphatics in a sporotrichoid pattern, often on hands or forearms, elbows, knees or feet.11 Systemic symptoms are usually absent or mild in patients with localised disease; disseminated involvement occurs mainly in patients with marked immunosuppression.
This is not the correct diagnosis for the case patient. Although she had recently travelled abroad, she had not visited known outbreak regions and she had not undergone any cosmetic or surgical procedures. In addition, her lesions did not have a sporotrichoid pattern.
Dermatomyositis
Dermatomyositis is an idiopathic inflammatory myopathy defined by characteristic cutaneous disease with or without muscle involvement.13 Skin findings may precede, accompany, or persist without myositis.14 It is uncommon, with an annual incidence of 0.1 to 6 per 100,000 population.15 Dermatomyositis affects women more than men, with peak incidence between 50 and 60 years of age.15 Adult-onset dermatomyositis is strongly associated with malignancy, with up to about 25% of patients having an unsuspected cancer at diagnosis, most often adenocarcinoma.15
Dermatomyositis is thought to reflect an autoimmune microangiopathy of skin and muscle on a background of genetic susceptibility (human leukocyte antigen associations).15 Reported triggers include malignancy, viral infections (such as influenza, enteroviral and parvovirus B19 infections) and medications (such as penicillamine, lipid-lowering agents, NSAIDs, cyclophosphamides, anticonvulsants, immune-checkpoint inhibitors and tumour necrosis factor inhibitors) as well as exposure to silica dust.15 Photosensitivity contributes to lesion development, with phototesting studies showing that many patients with dermatomyositis develop erythema after a lower dose of ultraviolet B radiation than healthy controls.16
Distinguishing clinical features of dermatomyositis include heliotrope eruption (violaceous periocular discoloration), Gottron papules located over the metacarpophalangeal and interphalangeal joints, photosensitive poikiloderma on the chest or shoulders, and pruritic scalp dermatitis. The patient may or may not have proximal weakness or be clinically amyopathic.13
For the case patient, the absence of heliotrope rash, Gottron papules or proximal myopathy argue against a diagnosis of dermatomyositis. In addition, a creatine kinase test was performed and returned a normal result.
Systemic lupus erythematosus
This is the correct diagnosis. Systemic lupus erythematosus (SLE) is a chronic multisystem autoimmune disease characterised by loss of immune tolerance to nuclear antigens, which leads to diverse clinical and serological manifestations. In Australia, the prevalence of SLE is roughly 13 to 89 per 100,000, with higher prevalence and worse outcomes reported in Aboriginal, Torres Strait Islanders, Māori and Pacific peoples.17 Women of childbearing age are most affected.
SLE can affect almost any organ, with a relapsing-remitting course. Severity ranges from mild, with only cutaneous and musculoskeletal involvement, to organ-threatening, with renal, neuropsychiatric, pulmonary or haematological disease. The typical initial complaints are fatigue, arthralgia or inflammatory arthritis, and a photosensitive rash. Mucocutaneous signs include malar erythema, subacute or discoid lesions, oral or nasopharyngeal ulcers and nonscarring alopecia.18
Skin involvement can occur in SLE, and there also can be cutaneous disease without systemic involvement.19 There are three main types of cutaneous lupus erythematosus (CLE): acute, subacute and chronic. Acute CLE presents as a typical malar (‘butterfly’) rash. Subacute CLE, which is almost always drug-induced, is less commonly associated with SLE than acute CLE and the skin changes are typically more persistent, often involving the trunk and upper limbs. Chronic CLE is the most common variant and includes discoid lupus erythematosus, lupus profundus, chilblain lupus erythematosus and lupus tumidus (intermittent lupus), each with distinct presentation.20,21 Some authors include lupus tumidus as a fourth distinct type.18,21
The pathogenesis of SLE is thought to involve a combination of genetic susceptibility, immune response and environmental factors. Triggers for disease activity include ultraviolet light exposure, smoking and certain medications (e.g. calcium channel blockers, ACE inhibitors, terbinafine and NSAIDs).20,21 Photosensitivity is seen in all three types of CLE and therefore the rash is most commonly seen on sun-exposed areas – the face, neck, scalp, upper torso and upper limbs.
Investigations
In 2019, the European League Against Rheumatism and the American College of Rheumatology proposed new classification criteria for SLE.20,22 A positive antinuclear antibody (ANA) titre greater than or equal to 1:80 (on HEp-2 cells) is the entry criterion, followed by weighted clinical and immunological items; a total score of at least 10 points (including at least one clinical criterion), classifies a patient as having SLE.20,22 In day-to-day care, these criteria can help prompt consideration of SLE and structure investigations, but diagnosis still rests on clinical judgement, usually in conjunction with specialist input.20
The diagnosis of SLE is supported by serological testing. Initial laboratory assessment includes ANA by immunofluorescence, which is the hallmark immunological test for SLE. Tests for other disease markers include anti-double-stranded (anti-ds)DNA, an extractable nuclear antigen panel and complement (C3/C4) levels. A full blood count and renal and liver function tests should also be performed.23
In addition, patients should be actively screened at baseline for organ-threatening disease. Tests include urine microscopy for haematuria or casts, serial urine protein-creatinine ratio, estimated glomerular filtration rate (eGFR) and blood pressure measurement. Symptom-guided evaluation for serositis, neuropsychiatric SLE and haematological cytopenias is important.23 Antiphospholipid antibodies are tested when clinically indicated – for example, in patients with unexplained venous or arterial thrombosis and women who are pregnant or planning pregnancy and those who have experienced recurrent pregnancy loss.
Decisions regarding imaging and tissue biopsy are guided by symptoms. For example, renal biopsy may be appropriate for suspected lupus nephritis; skin biopsy with direct immunofluorescence testing for new skin lesions.23 Imaging, such as chest x-ray or echocardiography, may be requested for suspected serositis or cardiopulmonary involvement.
Management
The overall goals of management for patients with SLE are achieving remission or low disease activity and preventing organ damage, while minimising corticosteroid use and drug toxicities.24 All patients should be educated about the importance of photoprotection and those who smoke should be informed about smoking cessation strategies. Other core components of management include vaccinations according to immunosuppression status and addressing cardiovascular risk, bone health and pregnancy planning.
All patients should be managed with specialist input. Patients with suspected systemic SLE should be co-managed with a rheumatologist. Nephrology referral is indicated for patients with renal involvement. Dermatology referral is indicated for diagnostic biopsy and management of cutaneous disease. Most patients benefit from review every three to four months, with more frequent monitoring in active disease.24,25
Hydroxychloroquine is first-line therapy for SLE and ideally continued long term. Typical adult dosing is 200 to 400 mg daily (not exceeding 5 mg/kg body weight). Benefits include fewer flares, reduced serositis and improvement in lipid profile and thrombotic risk. Hydroxychloroquine is generally safe in pregnancy. Ophthalmic monitoring is recommended to mitigate retinal toxicity.23
Corticosteroids are used to treat flares and severe organ-threatening disease, with the aim of using the lowest dose for the shortest duration. Mild features (e.g. rash, arthralgia) may be treated with prednisolone 5 to 15 mg daily with a planned taper, aiming for chronic maintenance of 7.5 mg per day, or less, to limit harms. Severe organ-threatening disease may be treated with intravenous methylprednisolone then switched to oral prednisolone, tapering as control is achieved. It is important to recognise and prevent corticosteroid toxicities, including infection, osteoporosis, cardiometabolic effects, mood disturbance and ocular complications.23,25 Patients should be reviewed every four to six weeks during a corticosteroid taper for renal indices, complement levels and anti-dsDNA, as well as for side effects of corticosteroid therapy.
Additional immunomodulatory drugs are available and are started early for their corticosteroid-sparing effects – these include methotrexate, azathioprine and mycophenolate mofetil or mycophenolate sodium. The calcineurin inhibitor tacrolimus and ciclosporin may also be used. Pulsed intravenous regimens of cyclophosphamide are used in life- or organ-threatening presentations.23,24 For patients with refractory or moderate-to-severe disease, there is a role for biologic therapies such as belimumab, rituximab and anifrolumab.23,26
It can take months to stabilise the disease. An important element of management for SLE is distinguishing disease flares from infection. CRP often rises disproportionately in infection, whereas rising anti-dsDNA antibodies and falling C3/C4 complement levels may accompany disease flares.23,24
For cutaneous disease, the general management approach involves a combination of strict sun protection and topical therapy. The Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI), a tool for scoring the severity and distribution of CLE based on distinct anatomical areas, can be helpful for informing damage assessment and response.27 Potent topical corticosteroids are used to treat localised lesions, while intralesional corticosteroids may be considered for hypertrophic plaques, and short courses of systemic corticosteroids used for flares. Hydroxychloroquine is first-line systemic therapy for most patients with CLE when disease is widespread or insufficiently controlled with topical agents. In the longer term, topical calcineurin inhibitors such as pimecrolimus and tacrolimus can be used if required. Treatment may be escalated to agents such as methotrexate, mycophenolate or azathioprine if refractory.25 There is emerging evidence for selective TYK2 inhibitors such as deucravacitinib for controlling inflammation for patients with refractory CLE, which will be an important area to consider as regulatory approvals evolve.28
Patients need to be managed for increased thrombosis risk and antiphospholipid syndrome where necessary. If antiphospholipid syndrome is present, long-term anticoagulation therapy should be managed per standard guidance and oestrogen-containing therapies avoided.23
Outcome
For the case patient, a diagnosis of SLE was suspected based on her history and presentation, and investigations were arranged. Results revealed: an ANA titre of 1:320 with a homogeneous pattern; anti-dsDNA antibody 1284 IU/mL; erythrocyte sedimentation rate 69 mm/h; C-reactive protein less than 4 mg/L; and creatine kinase 54 U/L. Normal results were returned for an extractable nuclear antigen panel, liver function test, full blood count, thyroid function test and iron studies. Urine protein-creatinine ratio was high at 22.3 mg/mmol. Her complement C3 level was low at 0.55 g/L and her C4 was 0.20 g/L. Overall, these findings were consistent with a diagnosis of active SLE with renal involvement. The cutaneous findings, while in keeping with SLE, were not entirely typical.
Two distinct skin biopsies were performed. Histopathology showed a band-like superficial and deep inflammatory infiltrate with periadnexal involvement. Mucin stain was positive. The results of the biopsies, together with the pathology results and clinical presentation, confirmed a diagnosis of SLE with cutaneous involvement. The patient commenced treatment with 50 mg prednisone and hydroxychloroquine 200 mg twice daily, with mycophenolate as a corticosteroid-sparing agent. Patient monitoring is ongoing. MT
COMPETING INTERESTS: None.
References
1. Gota C. Overview of cutaneous small vessel vasculitis [updated 2025]. In: UpToDate, Connor RF (Ed), Wolters Kluwer. Available online at: https://www.uptodate.com/contents/overview-of-cutaneous-small-vessel-vasculitis (accessed November 2025).
2. Coulson I. Cutaneous small vessel vasculitis [Based on: Stanway A. Cutaneous vasculitis, 2003]. DermNet NZ; 2024. Available online at: https://dermnetnz.org/topics/cutaneous-small-vessel-vasculitis (accessed November 2025).
3. Goeser MR, Laniosz V, Wetter DA. A practical approach to the diagnosis, evaluation, and management of cutaneous small-vessel vasculitis. Am J Clin Dermatol 2014; 15: 299-306.
4. Micheletti RG. Cutaneous small vessel vasculitis: a practical guide to diagnosis and management. Am J Clin Dermatol 2023; 24: 89-95.
5. de Crom SCM, Rossen JWA, van Furth AM, Obihara CC. Enterovirus and parechovirus infection in children: a brief overview. Eur J Pediatr 2016; 175: 1023-1029.
6. Second J, Velter C, Calès S, Truchetet F, Lipsker D, Cribier B. Clinicopathologic analysis of atypical hand, foot, and mouth disease in adult patients. J Am Acad Dermatol 2017; 76: 722-729.
7. Russell NG, Kessler R. A hand-foot-and-mouth disease outbreak in an atypical population of college students. J Prim Care Community Health 2024; 15: 21501319241266506.
8. Yin XG, Yi HX, Shu J, Wang XJ, Wu XJ, Yu LH. Clinical and epidemiological characteristics of adult hand, foot, and mouth disease in northern Zhejiang, China, May 2008 – November 2013. BMC Infect Dis 2014; 14: 251.
9. Leong S, Mahon C. Hand, foot, and mouth disease (HFMD): enteroviral vesicular stomatitis. DermNet NZ; 2022. Available online at: https://dermnetnz.org/topics/hand-foot-and-mouth-disease (accessed November 2025).
10. Fedrizzi T, Meehan CJ, Grottola A, et al. Genomic characterization of nontuberculous mycobacteria. Sci Rep 2017; 7: 45258.
11. Griffith DE. Overview of nontuberculous mycobacterial infections [updated 2025]. In: UpToDate, Connor RF (Ed), Wolters Kluwer. Available online at: https://www.uptodate.com/contents/overview-of-nontuberculous-mycobacterial-infections (accessed November 2025).
12. Ngan V. Atypical mycobacterial infection. DermNet NZ; 2023. Available online at: https://dermnetnz.org/topics/atypical-mycobacterial-infection (accessed November 2025).
13. Concha JSS, Pena S, Gaffney RG, et al. Developing classification criteria for skin-predominant dermatomyositis: the Delphi process. Br J Dermatol 2020; 182: 410-417.
14. Cobos GA, Femia A, Vleugels RA. Dermatomyositis: an update on diagnosis and treatment. Am J Clin Dermatol 2020; 21: 339-353.
15. Ngan V, Sapsford S. Adult-onset dermatomyositis. DermNet NZ; 2020. https://dermnetnz.org/topics/adult-onset-dermatomyositis (accessed November 2025).
16. Dourmishev L, Meffert H, Piazena H. Dermatomyositis: comparative studies of cutaneous photosensitivity in lupus erythematosus and normal subjects. Photodermatol Photoimmunol Photomed 2004; 20: 230-234.
17. Eades LE, Hoi AY, Liddle R, et al. Systemic lupus erythematosus in Aboriginal and Torres Strait Islander peoples in Australia: addressing disparities and barriers to optimising patient care. Lancet Rheumatol 2024; 6: e713-e726.
18. Filotico R, Mastrandrea V. Cutaneous lupus erythematosus: clinico-pathologic correlation. G Ital Dermatol Venereol 2018; 153: 216-229.
19. Petty AJ, Floyd L, Henderson C, Nicholas MW. Cutaneous lupus erythematosus: progress and challenges. Curr Allergy Asthma Rep 2020; 20: 12.
20. Wallace DJ. Systemic lupus erythematosus in adults: clinical manifestations and diagnosis [updated 2025]. In: UpToDate, Connor RF (Ed), Wolters Kluwer. Available online at: https://www.uptodate.com/contents/systemic-lupus-erythematosus-in-adults-clinical-manifestations-and-diagnosis (accessed November 2025).
21. Oakley A. Cutaneous lupus erythematosus. DermNet NZ; 2024. Available online at: https://dermnetnz.org/topics/cutaneous-lupus-erythematosus (accessed November 2025).
22. Aringer M, Costenbader K, Daikh D, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol 2019; 71: 1400-1412.
23. Systemic lupus erythematosus (SLE) in adults. In: Therapeutic Guidelines: Rheumatology. Melbourne: TG; 2024. Available online at: https://tg.org.au (accessed November 2025).
24. Wallace DJ. Systemic lupus erythematosus in adults: overview of the management and prognosis [updated 2025]. In: UpToDate, Connor RF (Ed), Wolters Kluwer. Available online at: https://www.uptodate.com/contents/systemic-lupus-erythematosus-in-adults-overview-of-the-management-and-prognosis (accessed November 2025).
25. Cutaneous lupus erythematosus. In: Therapeutic Guidelines: dermatology. Melbourne: TG; 2022. Available online at: https://tg.org.au (accessed November 2025).
26. Gordon C, Amissah-Arthur MB, Gayed M, et al. The British Society for Rheumatology guideline for the management of systemic lupus erythematosus in adults. Rheumatology (Oxford) 2018; 57: e1-e45.
27. Albrecht J, Taylor L, Berlin JA, et al. The CLASI (Cutaneous Lupus Erythematosus Disease Area and Severity Index): an outcome instrument for cutaneous lupus erythematosus. J Invest Dermatol 2005; 125: 889-894.
28. Wasserer S, Seiringer P, Kurzen N, et al. Tyrosine kinase 2 inhibition improves clinical and molecular hallmarks in subtypes of cutaneous lupus. British J Dermatol 2025: ljaf293.
Skin conditions
Lupus