Topical therapies for psoriasis: selection, education, escalation of therapy
Topical therapies remain central to the management of psoriasis and include emollients, corticosteroids, vitamin D and A derivatives, calcineurin inhibitors, coal tar, dithranol and novel nonsteroidal options. Appropriate selection, patient education and escalation strategies can optimise outcomes, improve adherence and reduce adverse effects, allowing tailored treatment across disease severities and anatomical sites.
- Topical therapies remain first-line treatment for mild-to-moderate psoriasis and are often used as adjunctive therapy in more severe disease.
- Treatment selection should be guided by disease severity, plaque thickness and anatomical site.
- Topical corticosteroids and vitamin D analogues form the cornerstone of therapy and are often used in combination.
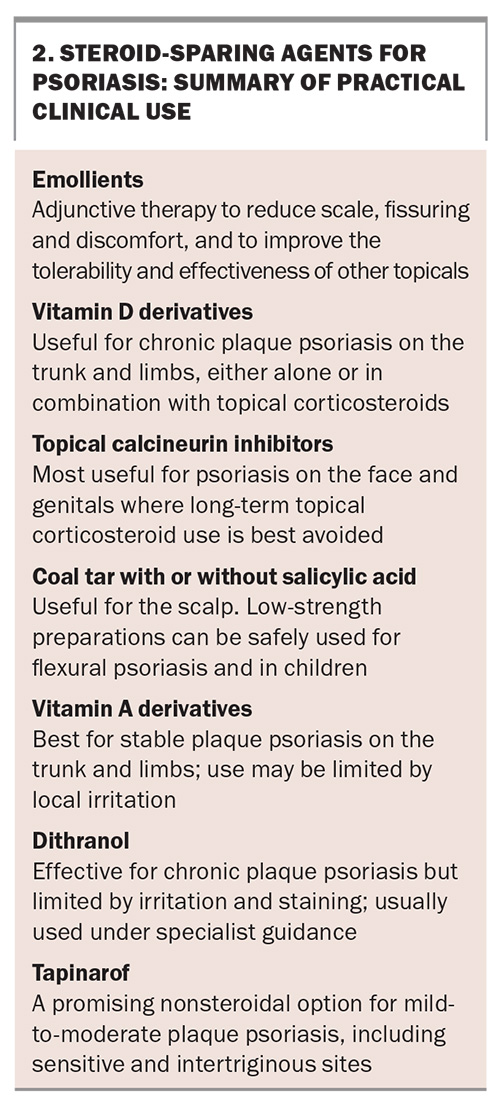
- Steroid-sparing options such as calcineurin inhibitors can be useful in selected situations, particularly on sensitive sites.
- The use of treatments such as coal tar, dithranol and retinoids may be considered but their use is waning as other options become available.
- Emerging nonsteroidal agents, including tapinarof and roflumilast, may expand future topical treatment options for psoriasis.
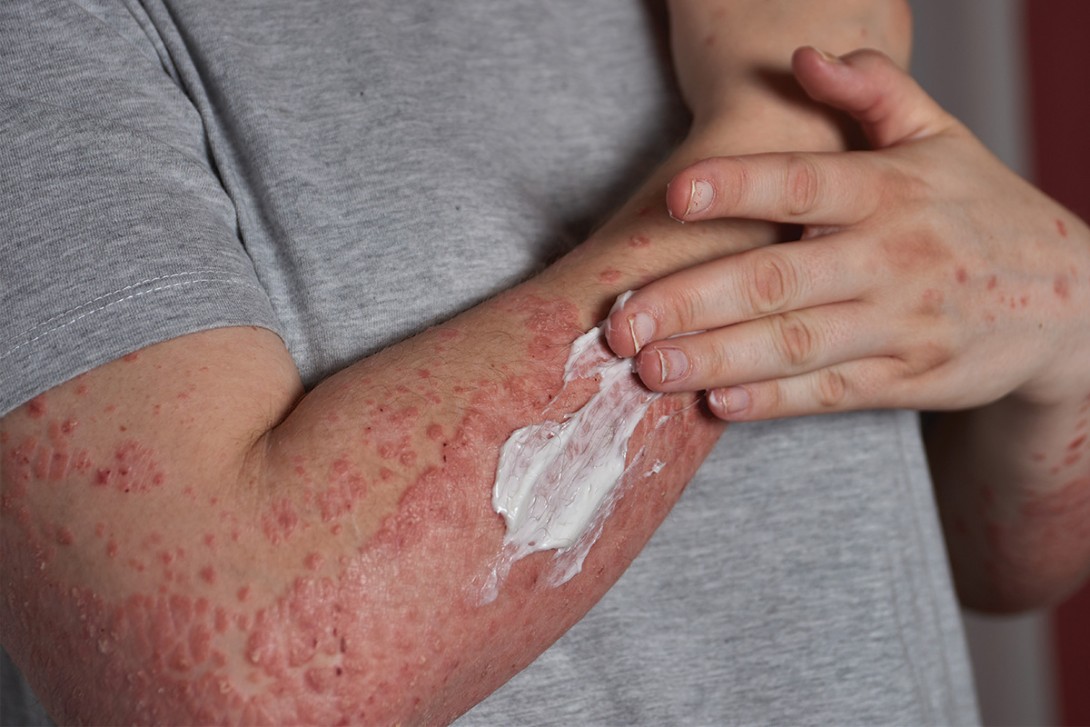
Psoriasis is a chronic, immune-mediated, multisystem disorder affecting 2.3 to 6.6% of people in Australia.1 Psoriasis is thought to be initiated by an inflammatory trigger that leads to a self-perpetuating disease cycle, in which activated immune cells promote keratinocyte activation via a positive feedback loop involving proinflammatory cytokines (including tumour necrosis factor-alpha and interleukins-1, -12, -17, -23 and -36), chemokines and antimicrobial peptides.2,3 This results in excessive keratinocyte proliferation, abnormal epidermal differentiation and the development of persistent, scaly, erythematous plaques. Histologically, this manifests as acanthosis (epidermal thickening), parakeratosis (retention of nuclei in the stratum corneum) and a dermal inflammatory infiltrate.
Topical therapies for psoriasis
Although targeted systemic therapies have reshaped the treatment landscape for moderate-to-severe psoriasis, topical therapies remain first line for mild or mild-to-moderate disease (Box 1) and play an adjunctive role in all disease severities. Topical therapies aim to reduce inflammation, suppress keratinocyte proliferation and restore normal epidermal differentiation.
Emollients
Regular use of fragrance-free emollients is an important adjunct in the management of psoriasis. Emollients, ideally applied at least once daily, help soften scale, reduce fissuring and improve patient comfort. Keratolytic agents such as urea (5 to 40%) or salicylic acid (0.5 to 10%) may be added to a thick cream base or white soft paraffin to enhance scale removal and improve penetration of other topical therapies. Higher-strength keratolytics (e.g. urea 25% or salicylic acid >8%) can be useful for hyperkeratotic psoriasis on the palms and soles, but may cause stinging, irritation or pain if applied to inflamed, fissured or broken skin.4
Topical corticosteroids
Topical corticosteroids remain the cornerstone of treatment for mild-to-moderate plaque psoriasis and are frequently used as first-line therapy in general practice. They act primarily by reducing cutaneous inflammation and immune activation. Topical corticosteroids should be applied to the skin prior to emollients to ensure adequate penetration.5,6
Potency selection should be guided by lesion thickness and anatomical site (Table). Low- to moderate-potency corticosteroids such as hydrocortisone 1%, or methylprednisolone aceponate 0.1% cream or ointment are preferred for sensitive areas such as the face, flexures and genital skin, whereas higher potency agents such as betamethasone dipropionate 0.05% or mometasone furoate 0.1% may be required for the trunk and limbs, and ultrapotent topical corticosteroids, such as clobetasol propionate 0.05%, are often required for the scalp, palms and soles.
Adverse effects are usually related to inappropriate potency or prolonged use and include skin atrophy, striae and telangiectasia. Sudden cessation can result in rebound flares. For this reason, topical corticosteroids are commonly used intermittently or in combination with other agents such as vitamin D analogues to improve efficacy and minimise toxicity. Fixed-combination products are widely used in clinical practice.5,6
Vitamin D derivatives
Vitamin D analogues, most commonly calcipotriol, are widely used topical agents for psoriasis. They regulate keratinocyte proliferation and differentiation and exert immunomodulatory effects. Calcipotriol is already in its active form and does not require ultraviolet activation. Calcipotriol is generally well tolerated; however, local irritation at the site of application may occur, particularly if applied to the face. Although vitamin D analogues can affect calcium metabolism, systemic absorption is minimal and clinically significant hypercalcaemia is rare. To mitigate this risk, the maximum dose should not exceed 100 g per week. Salicylic acid inactivates calcipotriol and should not be used concurrently in the same anatomical area. Vitamin D derivatives are typically applied once or twice daily and are frequently prescribed in combination with topical corticosteroids, either separately or as fixed-dose combination products (usually foam or ointments) containing calcipotriol and betamethasone dipropionate, which have been shown to improve efficacy and tolerability.4,8
A novel combination cream containing calcipotriol and betamethasone dipropionate formulated using polyaphron dispersion technology is showing promise. This technology allows the active ingredients to be suspended in a cream rather than the traditional ointment, gel or foam, and this improves penetration of the ingredients into the epidermis and dermis. The cream vehicle provides a fast-absorbing, nongreasy formulation suitable for use on both body and scalp lesions. In clinical trials, the polyaphron dispersion-based cream demonstrated greater improvement in psoriasis severity than gel formulations, a rapid onset of effect, and high levels of patient acceptance and treatment satisfaction. Improved tolerability and patient satisfaction may help address poor adherence, which is a common limitation of topical psoriasis therapy. As a result, this therapy, which is expected to be listed on the PBS in early 2026, may represent a valuable treatment option for mild-to-moderate plaque psoriasis and is a useful adjunct in patients with more extensive disease.7
Topical calcineurin inhibitors
Topical calcineurin inhibitors, such as tacrolimus 0.1% and pimecrolimus 1%, are anti-inflammatory agents commonly used in atopic dermatitis that act by inhibiting T-cell activation and downstream proinflammatory cytokine release.9 They are generally well tolerated, with adverse effects largely limited to mild skin irritation at the site of application. In psoriasis, these treatments can be used off label for plaques in sensitive sites such as the face and genital area, where they appear to be safe and effective alternatives to topical corticosteroids.9,10
Coal tar with or without salicylic acid
Coal tar is a long-standing topical therapy for psoriasis and remains a low-cost option for some patients. Although its precise mechanism of action is not fully understood, coal tar is thought to suppress DNA synthesis, thereby reducing keratinocyte proliferation. Coal tar is available in a range of over-the-counter and prescription formulations and is often combined with salicylic acid, which acts as a keratolytic to reduce scale and enhance penetration of other topical therapies. Refined or liquefied coal tar preparations remain useful for psoriasis, particularly affecting the scalp and flexural areas. These preparations can be safely used in children, with low-strength formulations such as 2% liquor picis carbonis in aqueous cream commonly prescribed for flexures. For scalp involvement, tar-based treatments are typically applied for a prescribed duration, and then rinsed out with a tar- or salicylic acid- containing shampoo. Despite its effectiveness, the use of coal tar is often limited by cosmetic acceptability. It can be malodorous, messy, stain clothing and skin, and cause local irritation, which may reduce adherence.8 Despite occupational and animal studies showing an increased risk of cancer after exposure to coal tar, there is no evidence of this in humans exposed to topical tar medicaments.11
Vitamin A derivatives
Vitamin A and its synthetic derivatives (retinoids) normalise epidermal differentiation and reduce inflammation by binding to nuclear retinoic acid receptors. Tazarotene is the most commonly used topical retinoid for psoriasis and is receptor- selective, resulting in a lower risk of systemic side effects; however, topical retinoids are not commonly used in Australia, as no commercially available preparations exist and access is limited to compounded formulations. Topical retinoids commonly cause local irritation, erythema, dryness and photosensitivity, and are therefore often used in combination with topical corticosteroids or emollients to improve tolerability.12
Dithranol
Dithranol (also known as anthralin) is an anthraquinone derivative that reduces epidermal hyperproliferation by inhibiting DNA synthesis and mitotic activity. It also has anti-inflammatory effects and can restore more normal keratinisation. Although effective, dithranol is now rarely used due to practical limitations. It can cause significant local irritation and stain skin, clothing and bathroom surfaces. As a result, its use is generally limited to short-contact regimens or specialist-supervised treatment settings.4,8 In Australia, dithranol is not commercially available in pre-packaged tubes and must be prepared as a compounded formulation.
Novel and emerging agents
Tapinarof 1% cream is a novel, nonsteroidal topical therapy for psoriasis that acts via activation of the aryl hydrocarbon receptor, leading to reduced inflammation, decreased oxidative stress and restoration of skin barrier function. Clinical trials have demonstrated that once-daily use is effective and well tolerated across a range of psoriasis severities, including when used on sensitive anatomical sites. It is currently under review by the TGA for approval in Australia. Adverse effects include folliculitis and are generally mild, making tapinarof a promising steroid-sparing option for the long-term topical management of psoriasis.13 A summary of practical clinical uses for tapinarof and other steroid-sparing agents is outlined in Box 2.
Other emerging nonsteroidal topical options for psoriasis include roflumilast 0.3% cream, a topical phosphodiesterase-4 inhibitor, and topical Janus kinase inhibitors such as tofacitinib 2% ointment and ruxolitinib 0.5 to 1% cream, which have demonstrated efficacy in clinical trials. However, larger, longer-term randomised controlled trials are needed to clarify their safety, durability of response and role in routine practice.14,15
Conclusion
Topical therapies remain central to the management of psoriasis, with an expanding range of agents allowing treatment to be tailored across disease severities and anatomical sites. Appropriate selection of therapy, patient education and timely treatment escalation can significantly improve patient outcomes. MT
COMPETING INTERESTS: Dr Larney, Dr Iyengar, Dr Daniel and Professor Shumack: None. Professor Foley has received research grants paid to institution from AbbVie, Amgen, Bristol-Myers Squibb, Eli Lilly, Janssen, LEO Pharma, Novartis, Pfizer, Sanofi, Sun Pharma, UCB Pharma; consulting fees from Apogee, Aslan, Boehringer Ingelheim, Eli Lilly, Galderma, GenesisCare, Janssen, LEO Pharma, Mayne Pharma, MedImmune, Novartis, Oruka, Pfizer, Takeda, UCB Pharma; payment or honoraria from AbbVie, Almirall, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Janssen, Novartis, Sanofi, Sun Pharma, UCB Pharma; payment for expert testimony from Pfizer; is on Advisory board for AbbVie, Amgen, Arrotex/Juniper, Aslan, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Galderma, GlaxoSmithKline, Janssen, LEO Pharma, Mayne Pharma, Novartis, Pfizer, Sanofi, Sun Pharma, Takeda, UCB Pharma; and has received drug samples provided free of charge for patient use from AbbVie, Amgen, Bristol-Myers Squibb, Eli Lilly, Janssen, LEO Pharma, Mayne Pharma, Novartis, Sun Pharma and UCB Pharma.
References
1. Parisi R, Symmons DP, Griffiths CE, Ashcroft DM; Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project team. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol 2013; 133: 377-385.
2. Ben Abdallah H, Johansen C, Iversen L. Key signaling pathways in psoriasis: recent insights from antipsoriatic therapeutics. Psoriasis 2021; 11: 83-97.
3. Hawkes JE, Chan TC, Krueger JG. Psoriasis pathogenesis and the development of novel targeted immune therapies. J Allergy Clin Immunol 2017; 140: 645-653.
4. Chan JJ. Psoriasis: an update on topical and systemic therapies. Aust Prescr 2025; 48: 87-92.
5. Gabros S, Nessel TA, Zito PM. Topical corticosteroids. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. Available online at: https://www.ncbi.nlm.nih.gov/books/NBK532940/ (accessed March 2026).
6. Aung T, Aung ST. Selection of an effective topical corticosteroid. Aust J Gen Pract 2021; 50: 651-655.
7. Torres T, Galván J, Crutchley N, et al. Calcipotriol and betamethasone dipropionate cream based on PAD technology for the treatment of plaque psoriasis: a narrative review. Dermatol Ther (Heidelb) 2023; 13: 2153-2169.
8. DermNet. Topical therapy for psoriasis. Hamilton: DermNet.nz; 2009. Available online at: https://dermnetnz.org/cme/scaly-rashes/topical-therapy-for-psoriasis (accessed March 2026).
9. Amiri D, Schwarz CW, Gether L, Skov L. Safety and efficacy of topical calcineurin inhibitors in the treatment of facial and genital psoriasis: a systematic review. Acta Derm Venereol 2023; 103: adv00890.
10. Wang C, Lin A. Efficacy of topical calcineurin inhibitors in psoriasis. J Cutan Med Surg 2014; 18(1): 8-14.
11. Roelofzen JH, Aben KK, Oldenhof UT, et al. No increased risk of cancer after coal tar treatment in patients with psoriasis or eczema. J Invest Dermatol 2010; 130: 953-961.
12. Motamedi M, Chehade A, Sanghera R, Grewal P. A clinician’s guide to topical retinoids. J Cutan Med Surg 2022; 26: 71-78.
13. Bobonich M, Gorelick J, Aldredge L, et al. Tapinarof, a novel, first-in-class, topical therapeutic aryl hydrocarbon receptor agonist for the management of psoriasis. J Drugs Dermatol 2023; 22: 779-784.
14. de Moraes-Souza R, Chahine Chater R, Pera Calvi I, et al. Efficacy and safety of topical roflumilast for the treatment of psoriasis: a systematic review and meta-analysis of randomized controlled trials. Clin Drug Investig 2024; 44: 655-665.
15. Słuczanowska-Głąbowska S, Ziegler-Krawczyk A, Szumilas K, Pawlik A. Role of Janus kinase inhibitors in therapy of psoriasis. J Clin Med 2021; 10: 4307.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.