How predictable will seasonal influenza be in 2026? What Australia can expect and what we can do

Seasonal influenza remains one of the most significant infectious disease threats in Australia, with substantial annual impacts on morbidity, hospitalisation and health system demand. For the 2026 season, all seasonal influenza vaccines are now trivalent following the removal of the B/Yamagata lineage component, and an intranasal, live attenuated influenza vaccine becomes available nationally through the private market with additional state-funded programs for young children in selected jurisdictions.
- The 2025 influenza season in Australia was unusually long and intense; 2026 planning should assume ‘late-season surprises’ remain plausible.
- Influenza virus A(H3N2) subclade K (formerly J.2.4.1) emerged rapidly in Australia and is antigenically distinct from the prior vaccine strain – an explicit rationale for the 2026 A(H3N2) strain update.
- All 2026 seasonal influenza vaccines available in Australia are now trivalent (i.e. no B/Yamagata component).
- The intranasal, live attenuated influenza vaccine is available in Australia in 2026 through the private market nationally with additional state-funded programs in New South Wales, Queensland, South Australia and Western Australia.
- States and territories differ in funded eligibility and timing; therefore, clinicians should default to National Immunisation Program eligibility and then overlay local programs and time windows.
- Early testing, especially for high-risk patients, coupled with prompt antiviral use, particularly in cases of severe or progressive disease, is crucial in reducing complications and transmission.
Australia enters the 2026 influenza season with a warning label attached. The 2025 season was an astonishing 33 weeks long (May to December) and delivered a record number of laboratory-confirmed notifications.1 This was driven, in part, by a second ‘surprise peak’ of A(H3N2) viruses belonging to the newly recognised subclade K (formerly J.2.4.1), building from mid-October and reaching a crescendo the week before Christmas.1,2 Subclade K matters because it is not simply more influenza; it represents a genetically and antigenically drifted A(H3N2) lineage that showed poor inhibition in testing with the 2025 vaccine strain, implying a plausible pathway to lower vaccine effectiveness when K viruses circulate widely.
Against that background, the 2026 program introduces two practical changes: a trivalent-only vaccine market (with B/Yamagata removed) and, for the first time in Australia, an intranasal, live attenuated influenza vaccine (LAIV) option, with several state-funded deployment programs.
National influenza picture
Influenza surveillance tells two complementary stories: disease burden (notifications, hospitalisations, deaths) and preventive effort (vaccination uptake and effectiveness).
Disease burden
In 2025, Australia recorded 502,938 laboratory-confirmed notifications, the highest annual count since influenza became nationally notifiable in 2001.1 This level of activity, likely to be an underestimate of the true magnitude of influenza-related disease, was accompanied by substantial pressure on general practice, emergency departments and hospitals across multiple jurisdictions.
The Australian Bureau of Statistics’ mortality report for acute respiratory illness in 2025 identified 1701 influenza-related deaths, 1423 of which were directly attributed to influenza and a further 278 deaths associated with influenza infection, surpassing previous high levels of mortality seen in 2017 and 2019 (1656 and 1314 influenza-related deaths, respectively).3 Sentinel surveillance site data from the Australian Centre for Disease Control showed that across general practice, influenza-like illness consultation rates from June to December 2025 exceeded historical trends.4 Similarly, sentinel hospital admission data for severe acute respiratory infections in 2025 showed rates were largely driven by increased influenza presentations, especially to intensive care.4 These findings clearly illustrate that influenza remains a major cause of preventable illness despite the availability of effective vaccines and antiviral treatments.
Vaccination uptake
Vaccination data reported to the Australian Immunisation Register (AIR) show substantial vaccine delivery early in each season, but also highlight persistent gaps, particularly in young children and across some jurisdictions. By 5 October 2025, 9.0 million influenza vaccination episodes had been reported to the AIR (1 March to 5 October window). The equivalent early- season totals were 11.1 million (2022), 9.3 million (2023) and 8.8 million (2024).5
Comparisons across years should be interpreted cautiously, given vaccine supply timing, seasonal timing and pandemic-era effects. Nonetheless, the pattern highlights a concerning plateau in vaccination coverage following the unusually high uptake seen during the coronavirus disease 2019 (COVID-19) pandemic period. Uptake remains particularly suboptimal among children younger than 5 years of age, working-age adults and some geographically disadvantaged populations.
Delivery channels
GPs and pharmacists together deliver most influenza vaccinations in Australia. AIR data demonstrate that community pharmacies now represent a major delivery channel across jurisdictions, particularly for adults aged 18 to 64 years. The accessibility of pharmacy vaccination services allows for opportunistic vaccination and improves access for individuals who may not regularly attend general practice.
However, general practice remains central to influenza vaccination, especially for high-risk populations including young children, older adults, pregnant women and people with chronic medical conditions. Co-ordination between these delivery channels is therefore essential to maximising coverage.
Why subclade K deserves your attention
Influenza viruses evolve fast enough to embarrass our intuitions. Most year-to-year change is attributed to antigenic drift through incremental mutations, especially in the haemagglutinin surface protein, that alter antibody binding and progressively erode existing immunity.6 A(H3N2) viruses are particularly prone to clinically important drift and have repeatedly driven seasons with lower vaccine effectiveness when antigenic mismatch occurs.7
The ‘clade’ and ‘subclade’ labels used in influenza genomics are bookkeeping tools that track these evolutionary branches. In 2025, Australian and New Zealand laboratories observed a striking late-season replacement of previously dominant A(H3N2) viruses by a newly recognised lineage termed subclade K (formerly J.2.4.1).2 The WHO subsequently described a rapid global rise in these viruses from August 2025, with detection across multiple regions and no clear signal of increased intrinsic severity.8
Genetically, K viruses differ from earlier J.2.4.1 viruses by additional haemagglutinin substitutions and a distinct neuraminidase change.9 These changes sit in or near antigenic sites and glycosylation motifs that can shield epitopes from neutralising antibodies. In plain language, the virus changed in precisely the areas that can help it sidestep existing antibody responses.
In haemagglutination inhibition assays, 204 of 205 (99.5%) Australian K virus isolates showed eight-fold or greater reductions in reactivity to ferret antiserum raised against the 2025 vaccine cell-grown A/Croatia/10136RV/2023 (a J.2 virus).2 By contrast, when tested against antisera to J.2.4 viruses included in the 2026 Southern Hemisphere vaccine (egg based: A/Singapore/GP20238/2024; cell based: A/Sydney/1359/2024), only seven of 205 isolates (3.4%) showed eight-fold or greater reductions. These laboratory signals do not equal clinical vaccine effectiveness, but they are exactly the kind of early warning that informs strain selection.
These data also help explain why subclade K matters clinically even if intrinsic virulence is not clearly greater. A drifted virus can still cause more trouble by extending transmission, increasing reinfection risk and reducing how well prior vaccination or infection protects against future infection, even if vaccines continue to blunt severe outcomes.
Prevention starts with personal measures, no matter the variant
Effective public health campaigns spearheaded by targeted education and motivational behavioural strategies, are essential components in limiting influenza (and other respiratory virus) transmission.10 Personal protective measures include voluntary home isolation when unwell, appropriate respiratory etiquette, frequent hand hygiene and mask use in community settings when symptomatic.
Routine cleaning of frequently touched surfaces in homes, childcare facilities, schools and workplaces can further reduce the spread of viral particles. These simple measures became widely adopted during the COVID-19 pandemic and continue to provide meaningful protection against seasonal respiratory viruses including influenza. Importantly, these behaviours may provide additional protection for individuals, especially those at highest risk of complications.
Furthermore, these behaviours are particularly relevant when transmission occurs outside the classic winter peak because people stop expecting influenza and start behaving as though the virus has politely checked the calendar before arriving. The late 2025 Australian experience is a useful reminder that seasonality remains real but not perfectly obedient.
Vaccine changes for 2026
Vaccination remains the cornerstone preventive strategy against influenza.11 Annual vaccination is recommended for all people aged 6 months and older, with the Australian Immunisation Handbook specifying several groups for which influenza vaccination is particularly recommended (Box 1).12
In 2026, all seasonal influenza vaccines available in Australia are trivalent, containing two influenza A strains and one influenza B (Victoria lineage) strain. The B/Yamagata lineage component has been removed, consistent with WHO advice and broader expert consensus that naturally circulating B/Yamagata viruses may no longer be in meaningful global circulation.13,14
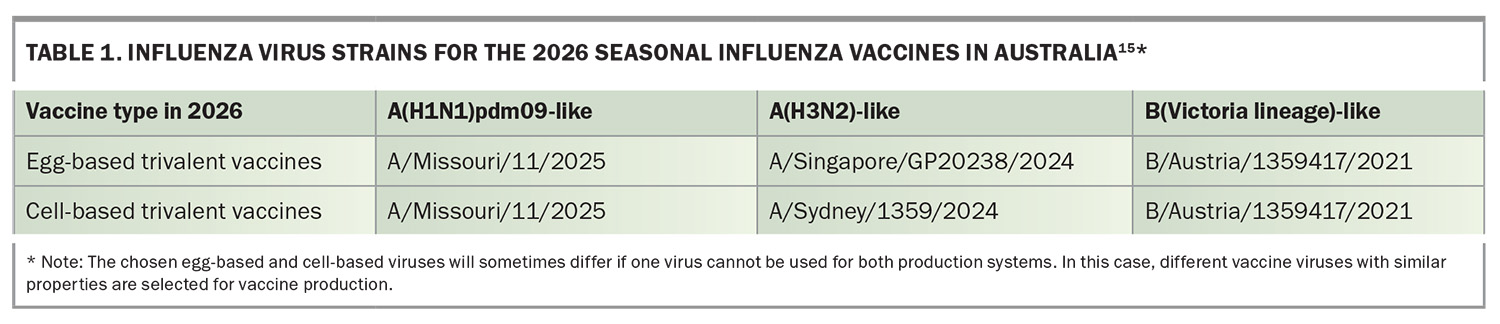
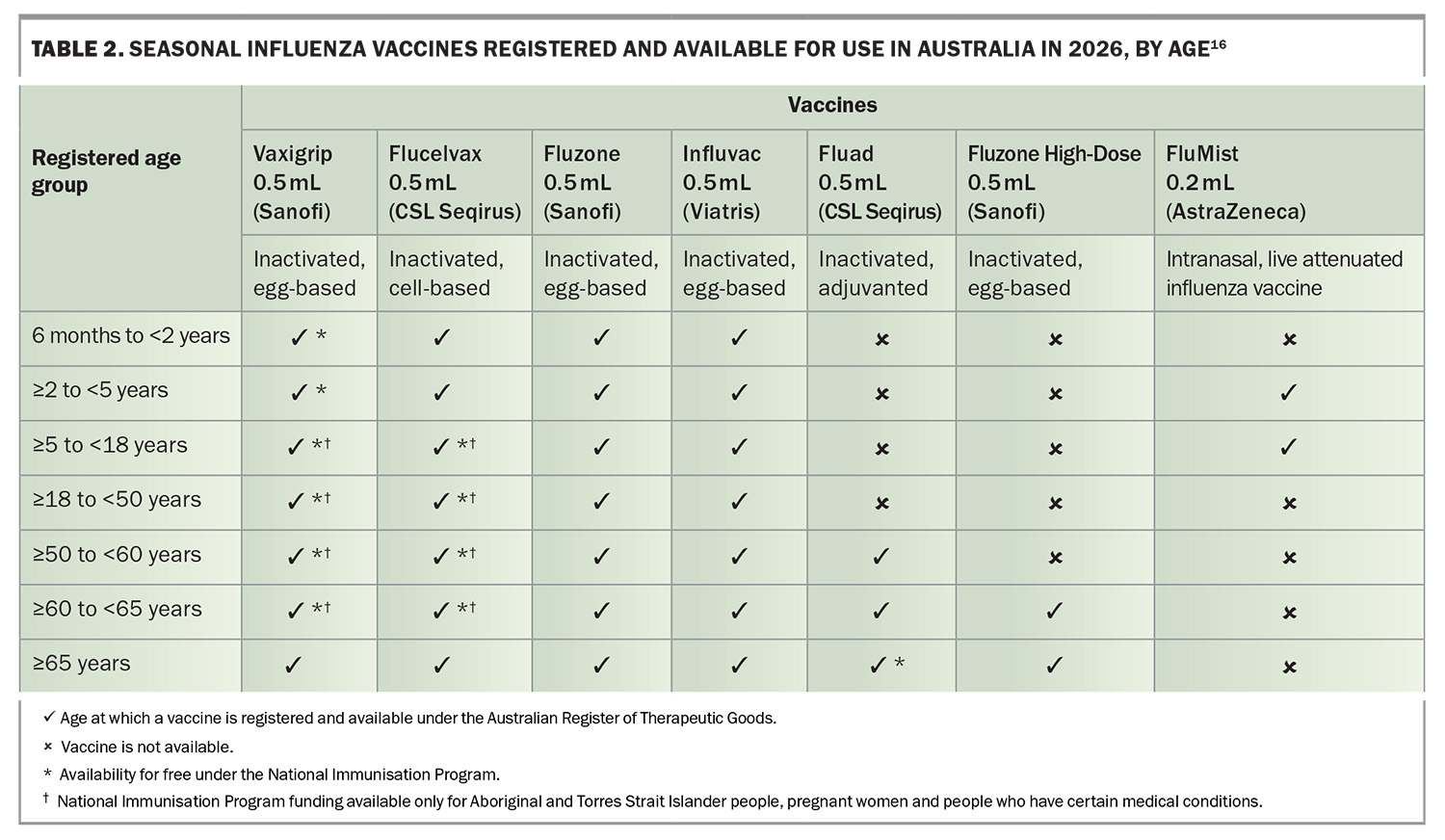
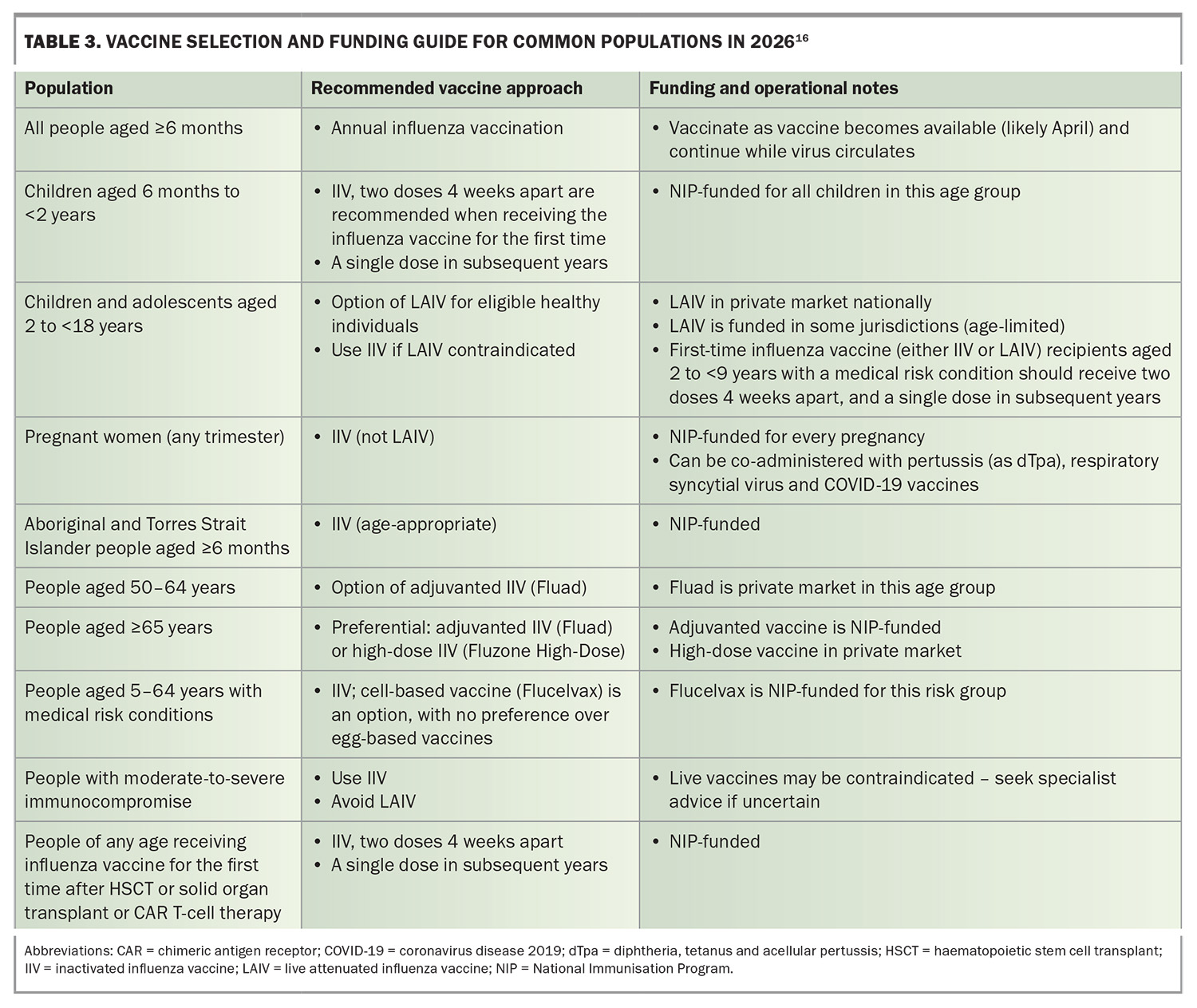
Vaccine strains for the 2026 Australian season are shown in Table 1.15 Notably, the A(H3N2) component has been updated to a J.2.4-like virus, directly reflecting the antigenic drift observed with emergent A(H3N2) lineages such as subclade K in late 2025. Registered influenza vaccine products available in Australia in 2026 and their age indications are summarised in Table 2.16 A pragmatic selection guide aligned to Australian Technical Advisory Group on Immunisation and National Immunisation Program (NIP) advice is provided in Table 3. 6,17
Cell-based and egg-based vaccines are both safe and effective, but they are not biologically identical. Egg-based manufacturing can select egg-adaptive mutations in haemagglutinin, which may alter antigenicity, particularly for A(H3N2) viruses.18,19 This is one reason cell-based vaccines attract interest when the mismatch risk is elevated.
In Australia, the cell-based formulation is funded under the NIP for people aged 5 to 64 years with specified medical risk conditions that increase the risk of influenza complications, with no preferential recommendation over standard egg-based vaccines for that group. For adults aged 65 years and older, both the adjuvanted trivalent inactivated vaccine (NIP-funded) and trivalent inactivated vaccine (private market) are preferentially recommended over standard high-dose influenza vaccines.
This preference reflects the well-recognised phenomenon of immunosenescence – the age-related decline in immune function that reduces the robustness of the antibody response to standard influenza vaccines in older adults. Suboptimal seroconversion in this group means that standard-dose formulations may not reliably generate protective antibody titres. The adjuvanted trivalent inactivated vaccine addresses this through the inclusion of MF59C.1, an oil-in-water adjuvant that enhances immunogenicity and has been shown to improve vaccine effectiveness against influenza-related outcomes in adults aged 65 years and older compared with standard unadjuvanted vaccines. The high-dose trivalent inactivated vaccine, which contains four times the standard antigen load, achieves a similar goal through antigenic mass. Both strategies are designed to overcome the immunological limitations of ageing and provide better protection in this high-risk group.
A notable change for 2026 is that the adjuvanted trivalent inactivated influenza vaccine is now licensed and available on the private market for adults aged 50 to 64 years – an expansion of its previous indication, which was restricted to those aged 65 years and older. Clinicians should be aware that this group is not NIP-eligible for this vaccine and will need a private prescription, but for adults in this age range who wish to access an enhanced vaccine formulation, this represents a new option worth discussing.
National program with jurisdictional overlays
The NIP provides consistent funded eligibility nationally for:
- children aged 6 months to younger than 5 years
- Aboriginal and Torres Strait Islander people aged 6 months and older
- pregnant women
- people aged 65 years or older
- people aged 6 months or older with specified medical risk conditions.20
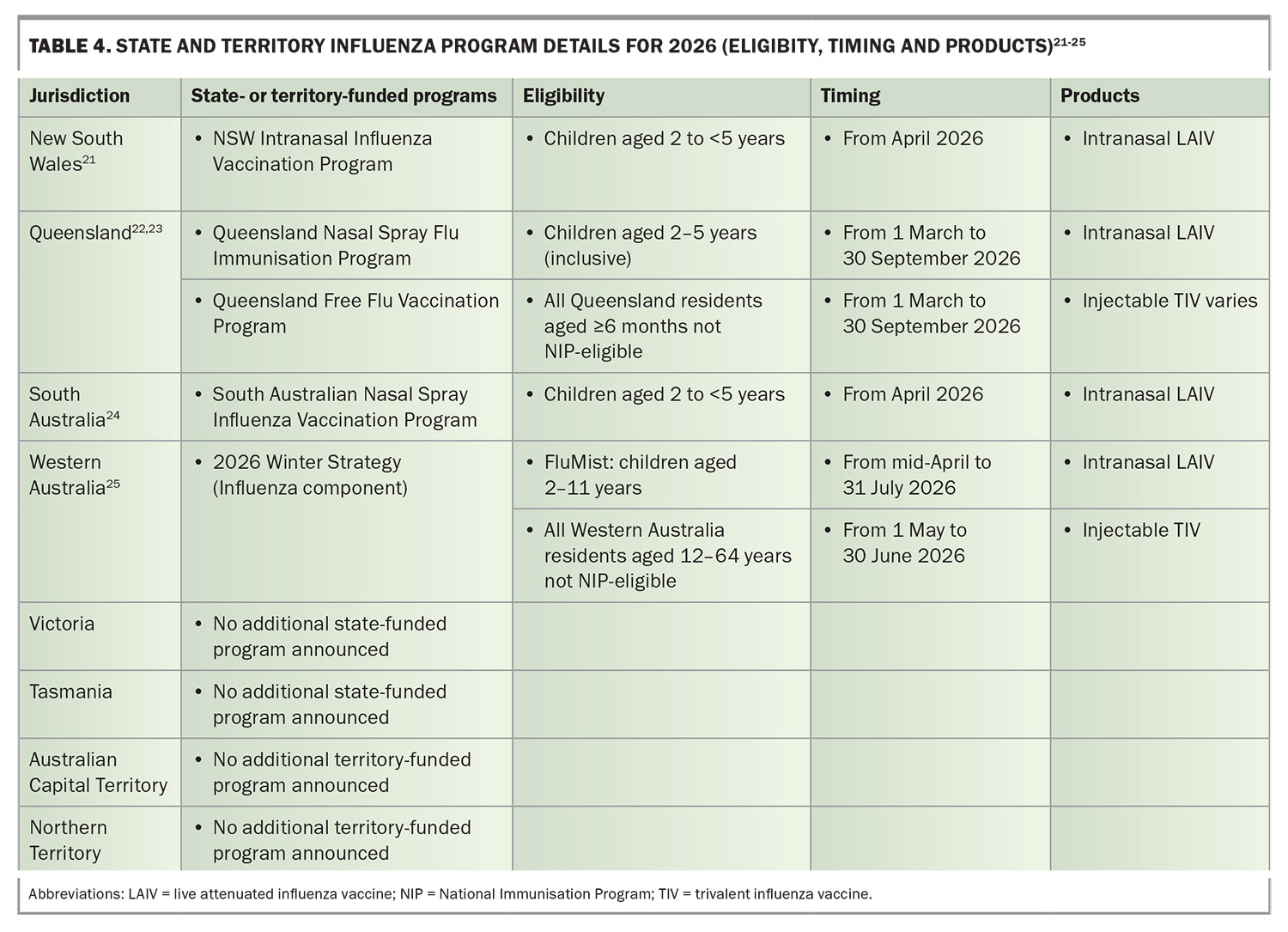
The major 2026 program change is the arrival of an intranasal LAIV in Australia for the first time. It is available nationally via private prescription (about $50 to $70) for children and adolescents aged 2 to younger than 18 years, with additional state-funded programs in New South Wales, Queensland, South Australia and Western Australia. Importantly, for some families, a needle-free option may meaningfully reduce barriers to uptake.
Jurisdictional overlay matters. In New South Wales, the Intranasal Influenza Vaccination Program is funded for children aged 2 to younger than 5 years, and was introduced in response to low coverage in this age group.21 NSW Health reports 24.4% coverage in children aged 6 months to younger than 5 years in 2025, with children aged 0 to 4 years accounting for 13% of influenza notifications to 31 October 2025. In Queensland, all residents aged 6 months and older can access free influenza vaccination under the 2026 Free Flu Vaccination Program, and a separately funded Nasal Spray Flu Immunisation Program for children aged 2 to 5 years inclusive.22,23 In South Australia, a free Nasal Spray Influenza Vaccination Program is available for children aged 2 to younger than 5 years.24 Furthermore, state materials from Western Australia indicate funded intranasal LAIV for children aged 2 to younger than 12 years as part of the broader 2026 Winter Strategy.25 Published operational details are summarised by state and territory in Table 4.21-25
Clinicians should explicitly consider three questions with each patient.
- Are they NIP-eligible?
- Does their state or territory provide additional funded access?
- Which product is the most appropriate for their age and clinical circumstances?
Diagnosis in a season with drift risk
Accurate and timely diagnosis of influenza is essential both for effective individual patient management and for the collection of epidemiological data that inform broader public health responses. Influenza is typically characterised by a constellation of features, including sudden-onset fever, chills, cough, sore throat, malaise, headache and myalgia; however, these manifestations substantially overlap with those of other acute respiratory illnesses, including the common cold, respiratory syncytial virus infection and early bacterial pneumonia.26 Relying on clinical features alone therefore poses a genuine diagnostic challenge and can lead to misdiagnosis, inappropriate management and missed opportunities to administer targeted antiviral therapy.27
One important consequence of diagnostic inaccuracy is the inappropriate use of antibacterial agents for a viral illness, contributing to antimicrobial resistance and delaying the use of effective antivirals in high-risk patients.28,29 Antiviral agents are most efficacious when administered within the first 48 hours of symptom onset, meaning that delays in accurate diagnosis can forfeit the critical therapeutic window and prolong the course of illness.30,31
Clinical prediction models that incorporate a range of symptoms have demonstrated variable diagnostic accuracy, with reported sensitivities of 36 to 80% and specificities of 78 to 98%.32,33 During the peak influenza season when community prevalence is high, the positive predictive value of a clinical diagnosis is considerably enhanced, and a presumptive diagnosis of influenza-like illness may reasonably guide initial management without awaiting laboratory confirmation, particularly when rapid testing is unavailable.34 Outside of peak season, or when the clinical picture is atypical, laboratory confirmation becomes more important.
Nucleic acid amplification testing (e.g. polymerase chain reaction [PCR]) remains the preferred modality for laboratory confirmation given its high sensitivity and specificity (about 94% and 99%, respectively, for influenza A).35 In Australia, PCR-based testing is available through both public hospital laboratories and a national network of private pathology services, which offer bulk-billed or low-cost testing with a valid referral. Multiplex PCR assays that detect a wide range of respiratory pathogens from a single specimen have expanded significantly in scope, particularly since the COVID-19 pandemic. Rapid antigen tests, available in point-of-care and self-test formats from pharmacies and supermarkets, offer results within 15 to 30 minutes and are useful if immediate management decisions are required, although they are less sensitive than PCR. The most commonly used specimens are nasopharyngeal or nose-and-throat swabs, whereas bronchoalveolar lavage, aspirate and sputum samples may also be used in appropriate clinical settings.
In 2026, the possibility of A(H3N2) subclade K antigenic drift provides additional rationale for laboratory testing in high-risk patients.2 Confirming the causative pathogen enables prompt, targeted antiviral therapy, supports infection prevention decisions and contributes to the ongoing national surveillance of circulating strains. In primary care, PCR testing should be considered for:
- patients at a higher risk of complications
- those requiring hospital assessment
- residents or staff in residential care or other congregate settings
- situations where influenza confirmation can guide antiviral use or isolation advice.12,16
In hospital and emergency department settings, rapid testing pathways support both patient flow and infection prevention measures.
Treatment options
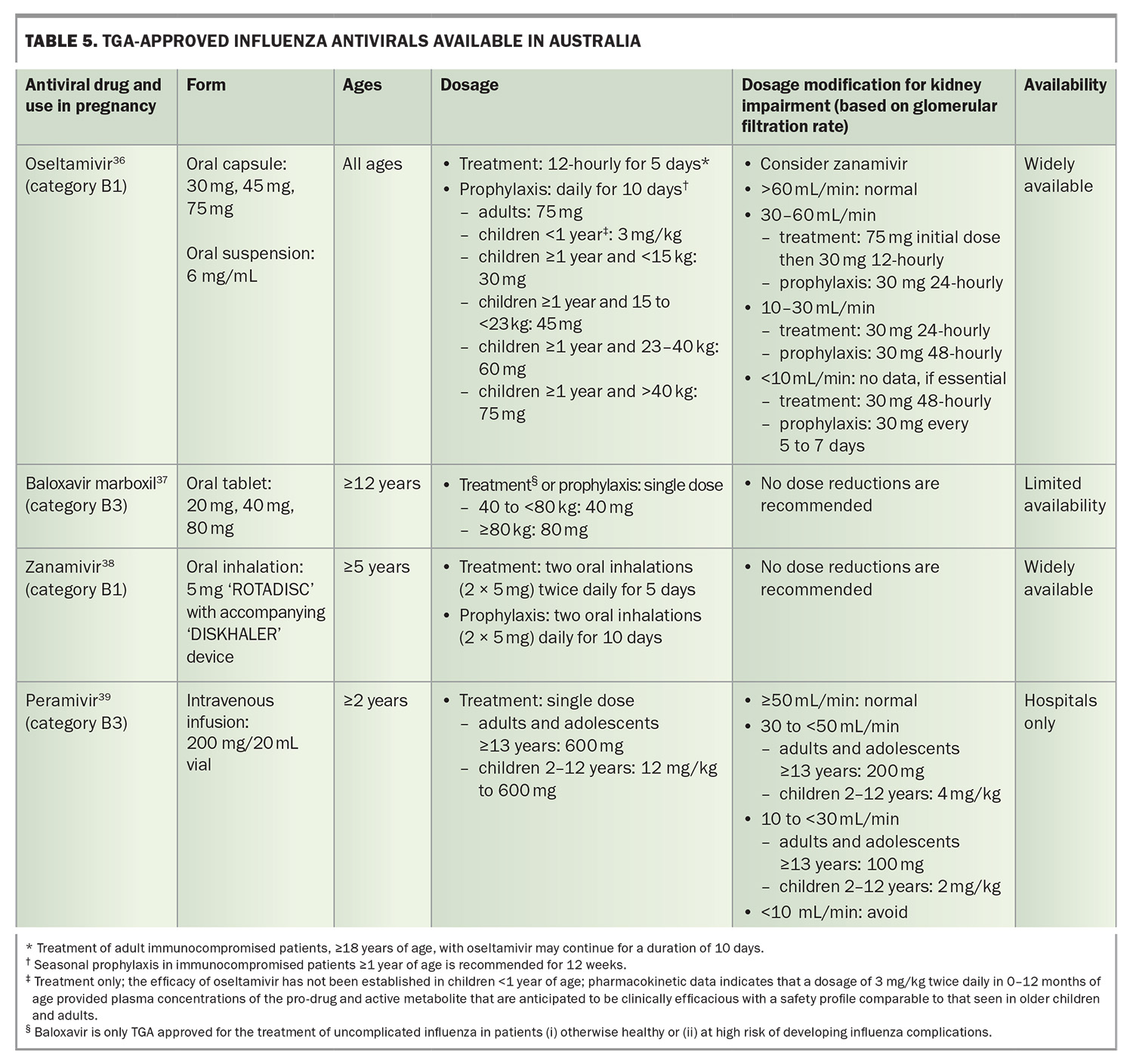
Antiviral medications play a critical role in the management of influenza, particularly in individuals at higher risk of poor outcomes (Table 5).36-39 Neuraminidase inhibitors remain the most widely used class in Australia; oseltamivir (oral capsule or suspension) and zanamivir (inhaled powder) are commonly prescribed, with peramivir available for severe cases requiring intravenous administration.12 These are all the most effective when initiated within 48 hours of symptom onset, with dosing adjusted for age, weight and renal function. Beyond reducing symptom duration, neuraminidase inhibitors can decrease the risk of complications and hospitalisation in high-risk groups.40
Baloxavir marboxil represents a distinct mechanistic class: a cap-dependent endonuclease inhibitor that disrupts viral replication by targeting the polymerase acidic protein, a component of the viral RNA polymerase complex essential for viral gene transcription.41 Like neuraminidase inhibitors, baloxavir is the most effective when administered within 48 hours of symptom onset, but offers two practical advantages as a single-dose regimen that simplifies adherence and leads to a rapid reduction in viral load, often apparent within one day of administration.41 Baloxavir is TGA approved, although its availability has historically been limited; clinicians should verify current supply with local pharmacies or hospital formularies.37,42
Concerns have previously been raised regarding the emergence of resistance to baloxavir, particularly PA-I38X substitutions in the polymerase acidic protein observed in clinical trials and real-world surveillance.43 In the context of 2026, however, the resistance picture is reassuring. Testing of 71 Australian A(H3N2) subclade K isolates demonstrated full susceptibility to all neuraminidase inhibitors, and sequencing of 240 viruses detected no PA-I38X or related substitutions conferring reduced baloxavir susceptibility.2 WHO global surveillance data are consistent with this finding, with no A(H3N2) viruses showing reduced susceptibility to either drug class identified in the period preceding the 2026 season.13,44 Ongoing surveillance for resistance patterns nevertheless remains essential, given the rapid evolutionary pace of A(H3N2) viruses.
In practice, clinicians should not await laboratory confirmation before initiating antiviral therapy in high-risk patients, especially during periods of established community transmission. Priority groups for early empirical treatment include:
- those with severe or progressive disease
- pregnant women
- significantly immunocompromised individuals
- those with chronic cardiorespiratory disease
- very young children.12,16
Evidence and guidelines also support treatment initiation beyond 48 hours in these groups when illness is severe or complicated, as clinical benefit remains plausible even outside the optimal window. Clinicians in institutional settings such as residential aged care should additionally be familiar with local public health guidance on outbreak management, where combined treatment and postexposure prophylaxis strategies are warranted.
Several specific clinical scenarios warrant particular mention. First, a negative influenza PCR result does not always exclude influenza; empirical antiviral therapy should be continued if clinical suspicion remains high, for example, during an institutional outbreak where the pre-test probability is elevated and sampling conditions may have affected test sensitivity. Second, for patients with influenza and significant immunocompromise, extending the duration of oseltamivir or zanamivir treatment to 10 days should be considered, noting that prolonged neuraminidase therapy can be associated with the emergence of antiviral resistance (most commonly to oseltamivir). Third, for patients who fail to improve as expected, clinicians should investigate for alternative or additional diagnoses – particularly secondary bacterial pneumonia caused by organisms such as Streptococcus pneumoniae or Staphylococcus aureus – or refer for specialist evaluation, including consideration of whether the infecting strain may carry antiviral resistance mutations.45
One distinctly Australian irritation remains unchanged; influenza antivirals are not PBS listed, so the cost can still be a barrier.
Communication and system readiness
Vaccination is not only a biomedical intervention but also a communication process between clinicians and patients. The Australian Technical Advisory Group on Immunisation emphasises that a healthcare provider recommendation is the strongest predictor of influenza vaccination uptake.16 Addressing vaccine hesitancy therefore requires open, respectful and evidence-based conversations. Research suggests that clear explanations of personal risk, vaccine benefits and safety profiles are more effective than confrontational messaging. Studies examining clinician–patient vaccine discussions have demonstrated that strong recommendations combined with empathetic listening improves acceptance and trust.46-48
The introduction of an intranasal LAIV in 2026 provides an opportunity to address ‘needle fatigue’ in children and families.49 However, clinicians must communicate eligibility clearly. The LAIV is contraindicated in cases of moderate-to-severe immunocompromise and is not recommended in pregnancy, where injectable inactivated vaccines remain the preferred option (Box 2).12,50
Equity in vaccine access deserves explicit attention in 2026. Indigenous populations experience disproportionately higher rates of influenza-related hospitalisation and mortality, yet uptake remains below targets in this group.51-54 Culturally safe communication, community-controlled health service engagement and proactive outreach, rather than relying solely on opportunistic vaccination, are the strategies most likely to close this gap.55 Clinicians working with Aboriginal and Torres Strait Islander communities should be familiar with NIP eligibility (which extends to all ages ≥6 months) and with any local jurisdiction programs that may facilitate easier access. Key vaccination resources including the Australian Immunisation Handbook (https://immunisationhandbook.health.gov.au), the National Centre for Immunisation Research and Surveillance (https://ncirs.org.au) and Sharing Knowledge About Immunisation (https://skai.org.au) provide clinicians with specific information to help discuss vaccinations with their Aboriginal and Torres Strait Islander patients.
Finally, it is essential that all influenza vaccinations are reported to the AIR, including the LAIV.
Conclusion
The 2026 seasonal influenza season arrives with strong evolutionary and programmatic signals. The emergence of A(H3N2) subclade K demonstrates how quickly antigenic drift can reshape influenza epidemiology and why continuous surveillance and vaccine strain updates remain essential. The available data do not suggest a more intrinsically severe virus, but they do show a lineage capable of extending a season, reducing reactivity to the prior year’s vaccine strain and reminding us that influenza’s talent for surprise is not going away.
Clinicians should take a layered approach to prevention by applying the NIP eligibility consistently, overlay state and territory program details, select the most appropriate vaccine product for the patient in front of you and use timely testing and antivirals where they may change outcomes.
Ultimately, success in 2026 will be measured not only by virological trends but by whether we close known uptake gaps among children, pregnant women, Aboriginal and Torres Strait Islander communities and medically at-risk individuals. Achieving this will require clearer recommendations, easier access and fewer missed opportunities for vaccination across the healthcare system. That challenge is entirely familiar. The virus, as ever, is not obliged to be. MT
COMPETING INTERESTS: Professor Griffin has received speaker honoraria from Seqirus. Dr Armstrong and Dr Spence: None.
References
1. Australian Centre for Disease Control. National Notifiable Disease Surveillance System (NNDSS) dashboard. Canberra: Australian Government Department of Health, Disability and Ageing; 2026. Available online at: https://nindss.health.gov.au/pbi-dashboard/ (accessed March 2026).
2. Dapat C, Peck H, Jelley L, et al. Extended influenza seasons in Australia and New Zealand in 2025 due to the emergence of influenza A(H3N2) subclade K viruses. Euro Surveill 2025; 30: 2500894.
3. Australian Bureau of Statistics (ABS). Deaths Due to Acute Respiratory Infection in Australia, January 2026. Canberra: ABS; 2026. Available online at: https://www.abs.gov.au/statistics/health/causes-death/deaths-due-acute-respiratory-infections-australia/jan-2026 (accessed March 2026).
4. Australian Centre for Disease Control. Australian Respiratory Surveillance Report – 1 to 28 December 2025. Canberra: Australian Government Department of Heath, Disability and Ageing; 2026. Available online at: https://www.cdc.gov.au/resources/publications/australian-respiratory-surveillance-report-1-28-december-2025 (accessed March 2026).
5. Australian Government Department of Health, Disability and Ageing. Influenza (Flu) Immunisation Data – 1 March to 5 October 2022–2025. Canberra: Australian Government; 2025. Available online at: https://www.health.gov.au/sites/default/files/2025-10/influenza-flu-immunisation-data-1-march-to-5-october-2022-2025 (accessed March 2026).
6. Petrova VN, Russell CA. The evolution of seasonal influenza viruses. Nat Rev Microbiol 2018; 16: 47-60.
7. Belongia EA, McLean HQ. Influenza vaccine effectiveness: defining the H3N2 problem. Clin Infect Dis 2019; 69: 1817-1823.
8. World Health Organization (WHO). Seasonal Influenza - Global Situation. Geneva: WHO; 2025. Available online at: https://www.who.int/emergencies/disease-outbreak-news/item/2025-DON586 (accessed March 2026).
9. Neher RA, Huddleston J, Bedford T, et al. Nomenclature for tracking of genetic variation of seasonal influenza viruses. Influenza Other Respir Viruses 2026; 20: e70230.
10. Grimani A, Antonopoulou V, Meader N, et al. Motivational effectiveness of prosocial public health messaging to reduce respiratory infection risk: a systematic review and meta-analysis. Commun Med (Lond) 2025; 6: 42.
11. Scott J, Abers MS, Marwah HK, et al. Updated evidence for Covid-19, RSV, and influenza vaccines for 2025-2026. N Engl J Med 2025; 393: 2221-2242.
12. Australian Technical Advisory Group on Immunisation (ATAGI). Influenza (flu). Australian Immunisation Handbook. Canberra: Australian Government Department of Health, Disability and Ageing; 2026. Available online at: https://immunisationhandbook.health.gov.au/contents/vaccine-preventable-diseases/influenza-flu (accessed March 2026).
13. World Health Organization (WHO). Recommended Composition of Influenza Virus Vaccines for Use in the 2026 Southern Hemisphere Influenza Season. Geneva: WHO; 2025. Available online at: https://cdn.who.int/media/docs/default-source/influenza/who-influenza-recommendations/vcm-sh-2025/a.-26-september-2025-recommended-composition-of-influenza-virus-vaccines-for-use-in-the-2026-southern-hemisphere-influenza-season-full-report.pdf (accessed March 2026).
14. Monto AS, Zambon M, Weir JP. The end of B/Yamagata influenza transmission - transitioning from quadrivalent vaccines. N Engl J Med 2024; 390: 1256-1258.
15. Therapeutic Goods Administration. AIVC Recommendations for the Composition of Influenza Vaccines for Australia in 2026. Canberra: Australian Government Department of Health, Disability and Ageing; 2026. Available online at: https://www.tga.gov.au/resources/publication/meeting-statements/aivc-recommendations-composition-influenza-vaccines-australia-2026 (accessed March 2026).
16. Australian Technical Advisory Group on Immunisation (ATAGI). Statement on the Administration of Seasonal Influenza Vaccines in 2026. Canberra: Australian Government Department of Health, Disability and Ageing; 2026. Available online at: https://www.health.gov.au/resources/publications/atagi-statement-on-the-administration-of-seasonal-influenza-vaccines-in-2026 (accessed March 2026).
17. Australian Government Department of Health, Disability and Ageing. National Immunisation Program 2026 Influenza Vaccination: Program Advice for Health Professionals. Canberra: Australian Government; 2026. Available online at: https://www.health.gov.au/influenza-vaccination/resources/publications/2026-influenza-vaccination-program-advice-for-health-professionals (accessed March 2026).
18. Ashraf M, Stein AN, Youhanna J, et al. The impact of egg adaptation and immune imprinting on influenza vaccine effectiveness. Vaccine 2025; 62: 127393.
19. Subbarao K, Barr I. A tale of two mutations: beginning to understand the problems with egg-based influenza vaccines? Cell Host Microbe 2019; 25: 773-775.
20. Australian Government Department of Health, Disability and Ageing. National Immunisation Program Schedule. Canberra: Australian Government, 2026. Available online at: https://www.health.gov.au/sites/default/files/2026-01/national-immunisation-program-schedule.pdf (accessed March 2026).
21. NSW Health. 2026 NSW Intranasal Flu Vaccine Program – Information for Health Professionals. Sydney: NSW Health; 2025. Available online at: https://www.health.nsw.gov.au/immunisation/professionals/Pages/intranasal-flu-vaccine.aspx (accessed March 2026).
22. Queensland Health. 2026 Queensland Nasal Spray Flu Immunisation Program. Brisbane: Queensland Health; 2026. Available online at: https://www.health.qld.gov.au/clinical-practice/guidelines-procedures/diseases-infection/immunisation/schedule/2026-queensland-nasal-spray-flu-immunisation-program (accessed March 2026).
23. Queensland Health. 2026 Free Flu Vaccination Program. Brisbane: Queensland Health; 2026. Available online at: https://www.health.qld.gov.au/clinical-practice/guidelines-procedures/diseases-infection/immunisation/schedule/2026-free-flu-vaccination-program (accessed March 2026).
24. Government of South Australia Health. Influenza Information for Health Professionals. Adelaide: Government of South Australia; 2026. Available online at: https://www.sahealth.sa.gov.au/wps/wcm/connect/public+content/sa+health+internet/clinical+resources/ clinical+programs+and+practice+guidelines/infectious+disease+control/influenza/influenza+information+for+health+ professionals (accessed March 2026).
25. Government of Western Australia Department of Health. 2026 Winter Strategy. Perth: Government of Western Australia; 2026. Available online at: https://www.health.wa.gov.au/~/media/Corp/Documents/Reports-and-publications/Winter-strategy/Winter-Strategy-2026.pdf (accessed March 2026).
26. Uyeki TM, Hui DS, Zambon M, Wentworth DE, Monto AS. Influenza. Lancet 2022; 400: 693-706.
27. Ebell MH, Rahmatullah I, Hulme C, et al. Accuracy of individual signs and symptoms and case definitions for the diagnosis of influenza in different age groups: a systematic review with meta-analysis. BMJ Open 2025; 15: e067574.
28. Uyeki TM, Bernstein HH, Bradley JS, et al. Clinical practice guidelines by the Infectious Diseases Society of America: 2018 update on diagnosis, treatment, chemoprophylaxis, and institutional outbreak management of seasonal influenza. Clin Infect Dis 2019; 68: 895-902.
29. Holmes EC, Hurt AC, Dobbie Z, Clinch B, Oxford JS, Piedra PA. Understanding the impact of resistance to influenza antivirals. Clin Microbiol Rev 2021; 34: e00224-20.
30. Fry AM, Goswami D, Nahar K, et al. Efficacy of oseltamivir treatment started within 5 days of symptom onset to reduce influenza illness duration and virus shedding in an urban setting in Bangladesh: a randomised placebo-controlled trial. Lancet Infect Dis 2014; 14: 109-118.
31. Liu JW, Lin SH, Wang LC, Chiu HY, Lee JA. Comparison of antiviral agents for seasonal influenza outcomes in healthy adults and children: a systematic review and network meta-analysis. JAMA Netw Open 2021; 4: e2119151.
32. Hung SK, Wu CC, Singh A, et al. Developing and validating clinical features-based machine learning algorithms to predict influenza infection in influenza-like illness patients. Biomed J 2023; 46: 100561.
33. Dugas AF, Valsamakis A, Atreya MR, et al. Clinical diagnosis of influenza in the ED. Am J Emerg Med 2015; 33: 770-775.
34. Communicable Diseases Network Australia (CDNA). Seasonal Influenza Infection: CDNA National Guidelines for Public Health Units. Canberra: Australian Government Department of Health; 2017. Available online at: https://www.cdc.gov.au/resources/publications/cdna-national-guidelines-flu (accessed March 2026).
35. Huang HS, Tsai CL, Chang J, Hsu TC, Lin S, Lee CC. Multiplex PCR system for the rapid diagnosis of respiratory virus infection: systematic review and meta-analysis. Clin Microbiol Infect 2018; 24: 1055-1063.
36. Therapeutic Goods Administration. Australian Product Information: Tamiflu ® (Oseltamivir Phosphate). Canberra: Australian Government Department of Health and Aged Care; 2023. Available online at: https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent=&id=CP-2012-PI-02051-3&d=20260329172310101 (accessed April 2026).
37. Therapeutic Goods Administration. Australian Product Information: Xofluza® (Baloxavir Marboxil) Tablets. Canberra: Australian Government Department of Health and Aged Care; 2023. Available online at: https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent=&id=CP-2020-PI-01260-1 (accessed April 2026).
38. Therapeutic Goods Administration. Australian Product Information: Relenza Rotadisk (Zanamivir). Canberra: Australian Government Department of Health, 2019. Available online at: https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent=&id=CP-2010-PI-04928-3 (accessed April 2026).
39. Therapeutic Goods Administration. Australian Product Information: Rapivab™ (Peramivir). Canberra: Australian Government Department of Health, 2021. Available online at: https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent=&id=CP-2021-PI-02235-1 (accessed April 2026).
40. Tejada S, Jansson M, Sole-Lleonart C, Rello J. Neuraminidase inhibitors are effective and safe in reducing influenza complications: meta-analysis of randomized controlled trials. Eur J Intern Med 2021; 86: 54-65.
41. Hayden FG, Sugaya N, Hirotsu N, et al. Baloxavir marboxil for uncomplicated influenza in adults and adolescents. N Engl J Med 2018; 379: 913-923.
42. Therapeutic Goods Administration. Australian Public Assessment Report for Baloxavir Marboxil. Canberra: Australian Government Department of Health; 2020. Available online at: https://www.tga.gov.au/sites/default/files/auspar-baloxavir-marboxil-200604.pdf (accessed March 2026).
43. UK Health Security Agency. Guidance on Use of Antiviral Agents for the Treatment and Prophylaxis of Seasonal Influenza. London: UK Health Security Agency; 2026. Available online at: https://www.gov.uk/government/publications/influenza-treatment-and-prophylaxis-using-anti-viral-agents/guidance-on-use-of-antiviral-agents-for-the-treatment-and-prophylaxis-of-seasonal-influenza (accessed March 2026).
44. Hussain S, Meijer A, Govorkova EA, et al. Global update on the susceptibilities of influenza viruses to neuraminidase inhibitors and the cap-dependent endonuclease inhibitor baloxavir, 2020-2023. Antiviral Res 2025; 241: 106217.
46. Purcell N, Usman H, Woodruff N, et al. When clinicians and patients disagree on vaccination: what primary care clinicians can learn from COVID-19-vaccine-hesitant patients about communication, trust, and relationships in healthcare. BMC Prim Care 2024; 25: 412.
47. Fasce A, Mustata M, Deliu A, et al. A field test of empathetic refutational and motivational interviewing to address vaccine hesitancy among patients. NPJ Vaccines 2025; 10: 142.
48. Helps C, Leask J, Barclay L, Carter S. Understanding non-vaccinating parents’ views to inform and improve clinical encounters: a qualitative study in an Australian community. BMJ Open 2019; 9: e026299.
49. Jhaveri R, Skolnik N, Bandell A. Systematic review of evidence on feasibility of administering intranasal live attenuated influenza vaccine at home. NPJ Vaccines 2025; 10: 272.
50. Therapeutic Goods Administration. Australian Product Information: Flumist® (Influenza Virus Vaccine) Nasal Spray. Canberra: Australian Government Department of Health, Disability and Ageing; 2026. Available online at: https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent=&id=CP-2025-PI-02358-1&d=20260402172310101 (accessed April 2026).
51. Betts JM, Weinman AL, Oliver J, et al. Influenza-associated hospitalisation and mortality rates among global Indigenous populations; a systematic review and meta-analysis. PLOS Glob Public Health 2023; 3: e0001294.
52. Imai C, Jayasinghe S, McRae J, et al. ATAGI targeted review 2023: vaccination for preventing influenza in Australia. Commun Dis Intell (2018) 2024; 48.
53. Australian Institute of Health and Welfare, National Indigenous Australians Agency. 3.02 Immunisation - Aboriginal and Torres Strait Islander Health Performance Framework. Canberra: Australian Government; 2026. Available online at: https://www.indigenoushpf.gov.au/measures/3-02-immunisation (accessed March 2026).
54. Australian Institute of Health and Welfare. Aboriginal and Torres Strait Islander Specific Primary Health Care: Results from the OSR and nKPI. Canberra: AIHW; 2026. Available online at: https://www.aihw.gov.au/getmedia/e5c1cee6-e764-4b34-b19f-2d325d69a8f7/Aboriginal-and-Torres-Strait-Islander-specific-primary-health-care-results-from-the-OSR-and-nKPI.pdf?v=20260122092454&inline=true (accessed March 2026).
55. Harwood E, Taylor K, Clark K, et al. Yarning about vaccinations: empowering individuals to have supportive conversations with Aboriginal peoples about vaccinations, using a community-engaged approach. Aust N Z J Public Health 2025; 49: 100206.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.