Obstructive sleep apnoea in adults and children – a primary care overview

Obstructive sleep apnoea should be considered in patients who present with snoring, pausing or gasping during sleep, excessive daytime sleepiness or resistant hypertension. GPs are ideally placed to screen, work up, initiate pre-phase treatments and co-ordinate multidisciplinary management for adult and paediatric patients with suspected obstructive sleep apnoea.
- Obstructive sleep apnoea (OSA) is a highly prevalent chronic disorder with major health consequences, and GPs plays an essential role in early identification, risk stratification and long-term management.
- Diagnosis in adults relies on validated screening tools and sleep studies, and management focuses on lifestyle optimisation, such as weight loss, as foundational steps.
- Positive airway pressure therapy remains the first-line treatment for moderate or severe OSA in adults, with adherence improved through tailored mask selection, sleep physician-supervised troubleshooting and nasal optimisation.
- Adjunctive therapies, including positional vibration devices and specialised pillows, offer benefit for selected adult patients.
- Sleep surgery and mandibular advancement splints are salvage options tailored to patient anatomy and OSA severity.
- Emerging options, such as hypoglossal nerve stimulation and weight loss medications, are under investigation in Australia, and may benefit patients with specific anatomical and physiological profiles.
- In children, GP management prioritises treating allergic rhinitis, correcting iron and vitamin D deficiency and referring for adenotonsillectomy when appropriate, as this is the most effective intervention.
Obstructive sleep apnoea (OSA) is a chronic, progressive condition characterised by recurrent upper airway obstruction during sleep, resulting in intermittent hypoxia, sleep fragmentation and heightened sympathetic nervous system activity. Prevalence studies estimate that moderate or severe OSA affects almost one billion people globally, with higher rates observed in older individuals, males and those with excess weight.1 The primary care setting plays a pivotal role in the early detection and ongoing management of OSA, particularly among patients presenting with comorbid conditions such as resistant hypertension, atrial fibrillation, obesity and depressive symptoms.2
OSA has a significant impact on the quality of life of both patients and their partners and family members. Typical symptoms include loud snoring, witnessed apnoeas and excessive daytime sleepiness. Moderate or severe disease is associated with additional health risks, such as cardiovascular disease and type 2 diabetes, as well as an increased risk of motor vehicle and workplace accidents caused by excessive daytime sleepiness.3
In the paediatric population, symptoms often present as snoring, gasping, breath-holding and bruxism during sleep. Daytime consequences in children include hyperactivity, behavioural issues, irritability and reduced attention in school.4 Management in children is different to that in adults.
Pathophysiology and risk factors
The incidence of OSA increases with age, likely due to increasing body weight as well as age-related hormonal and biological changes. The pathophysiology of OSA is heterogeneous, involving a combination of anatomical and physiological factors that contribute to upper airway obstruction.3
Soft tissue collapsibility from the palate to the larynx plays a role. Anatomically, narrow upper airways are more prone to collapse than broader airways. Surgical management, discussed below, aims to expand the upper airway through soft tissue reconstruction or bone remodelling procedures.2 Patients with OSA also have reduced pharyngeal dilator muscle tone during sleep, and clinical trials in Australia are investigating hypoglossal nerve stimulation to enhance nocturnal upper airway patency.5
Managing obstructive sleep apnoea in adults
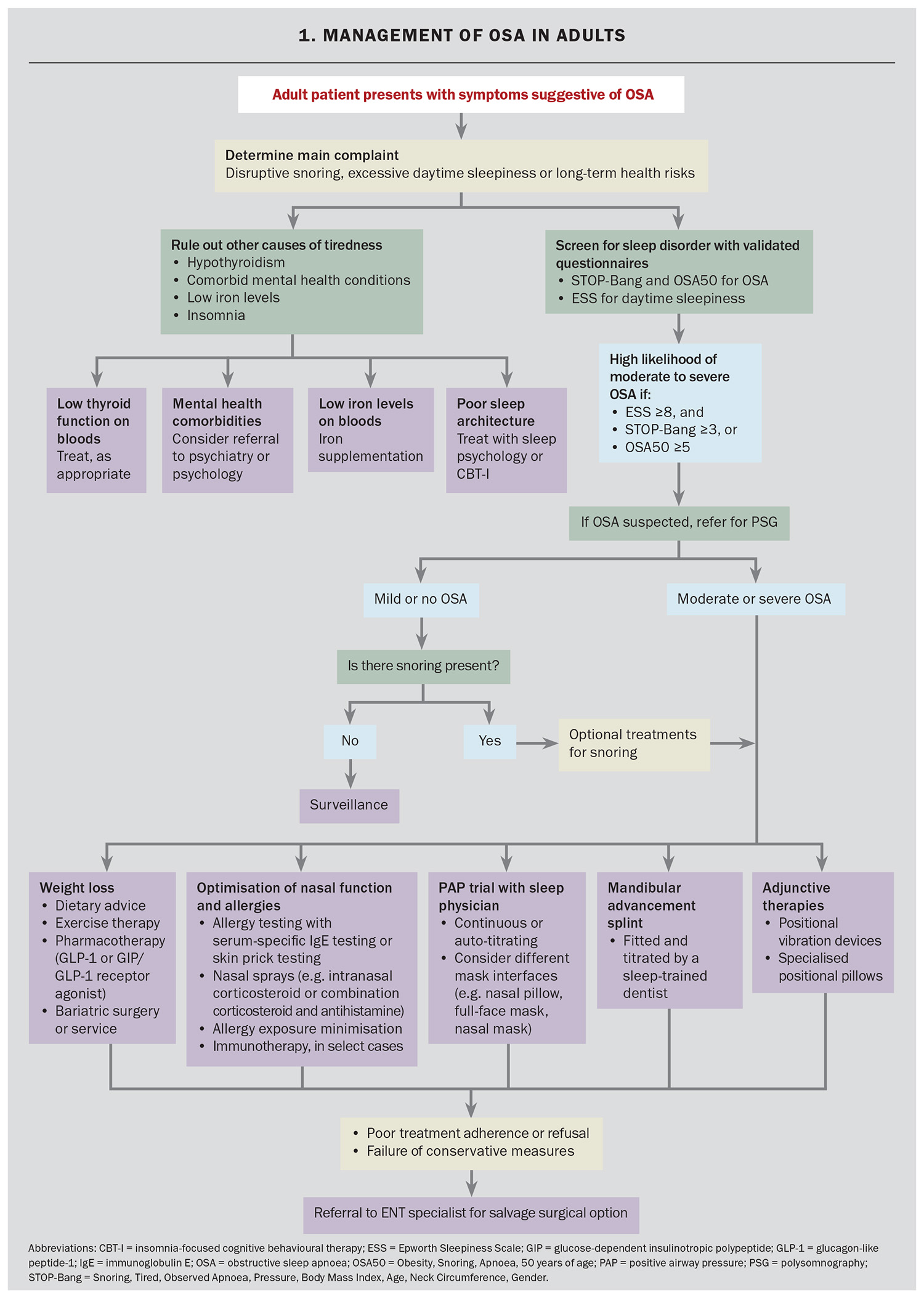
The management of OSA in adults is often complex and multifactorial, requiring a multidisciplinary approach (Flowchart 1). OSA is a chronic condition and managed such that the goal is not necessarily ‘cure’. The aim should be improvement in sleep symptoms and mitigation of long-term risks associated with OSA.2 GPs play a key role within the multidisciplinary care model for OSA. An Australasian Sleep Association position statement provides a comprehensive overview of the workup, assessment and management strategies for patients with OSA.2
Effective management of adult OSA begins with identifying the patient’s primary concerns, whether it be excessive daytime sleepiness, disruptive snoring affecting their partner or long-term health risks. Tailoring treatment goals to these concerns fosters engagement and improves patient outcomes. OSA management hinges on the foundational lifestyle strategies of sustained weight loss; avoidance of sedatives and alcohol; smoking cessation; and optimisation of nasal patency, sleep position and sleep architecture. It is important to assess for other comorbid sleep disorders, including insomnia, periodic limb movement disorder and parasomnias.
In Australia, the initial assessment of suspected OSA begins with validated screening tools such as the Snoring, Tired, Observed Apnoea, Pressure, Body Mass Index, Age, Neck Circumference, Gender (STOP-Bang) and Obesity, Snoring, Apnoea, 50 years of age (OSA50) questionnaires in adults at high risk of having OSA. The Epworth Sleepiness Scale (ESS) can be used to help quantify daytime sleepiness.6 The Snoring Severity Scale (SSS) is another useful tool for assessment in patients pre- and post-treatment.7 A diagnostic sleep study (polysomnography [PSG]) should be arranged based on clinical suspicion. Type 2 (home-based) studies are suitable for some patients with a high pre-test probability, whereas type 1 (in-laboratory) PSG remains the gold standard. Medicare rebates are available for type 1 and 2 sleep study referrals from GPs in patients who have a high risk of moderate to severe OSA, indicated by an ESS score of 8 or above and either a STOP-Bang score of 3 or above or OSA50 score of 5 or above. PSG is essential not only for diagnosing OSA but also for grading severity, stratifying risk and establishing a baseline to guide management.3
Moderate to severe OSA is associated with an increased risk of motor vehicle accidents and, thus, the fitness to drive of affected individuals must be considered for road safety.8 Any clinical concerns should be referred to a sleep physician for assessment, particularly for individuals with excessive sleepiness or those who hold a commercial driver’s licence, because commercial drivers are on the road for extended periods and accidents are more likely to involve members of the public (e.g. buses carrying passengers).
Weight loss
Weight loss is an important strategy in OSA management. The progressive nature of OSA is partly attributable to age-related weight gain, with excess adipose tissue accumulating around the neck, tongue and palate, narrowing the upper airway and increasing the risk of collapse during sleep. Multidisciplinary collaboration is essential for successful weight loss. This includes dietetics input for optimising dietary habits and exercise physiology input to help implement healthy exercise routines. Referral to GPs with expertise in obesity or to bariatric surgeons should be considered in appropriate cases.9
Recent advances in pharmacotherapy for weight loss have shown promise. The A Study of Tirzepatide in Participants With Obstructive Sleep Apnea (SURMOUNT-OSA) trial demonstrated that 12 months of treatment with tirzepatide, a dual glucose-dependent insulinotropic polypeptide/glucagon-like peptide-1 receptor agonist, led to an average 20% weight loss and a 50% reduction in the apnoea–hypopnoea index (AHI; the number of apnoeas and hypopnoeas per hour of sleep) among patients with moderate or severe OSA.10 Although these results are encouraging, longer-term studies are needed to establish safety, side effect profiles and durability of effect.
Tirzepatide has TGA approval for the treatment of moderate to severe OSA in adults with obesity.
Positive airway pressure therapy
The cornerstone of OSA management in adults is the use of positive airway pressure (PAP) therapy. It is typically commenced for those with moderate or severe OSA and functions by maintaining airway patency during sleep to prevent upper airway collapse and intermittent hypoxia.11 PAP therapy has been shown to improve sleep architecture, reduce daytime sleepiness and potentially mitigate long-term cardiovascular and neurocognitive sequelae associated with untreated OSA.12 Adherence to PAP is generally defined as device usage for more than five hours per night on at least five nights per week.13 Despite its in-laboratory efficacy, adherence remains a significant challenge in clinical practice.
Over the past two decades, technological advancements have sought to improve patient comfort and thereby improve adherence rates. Devices have progressed from fixed-pressure continuous PAP to auto-titrating PAP, which dynamically adjusts pressure in response to airway resistance, minimising the sensation of excessive pressure and improving tolerability. In parallel, there has been a diversification of mask interfaces, including full-face masks, nasal masks and nasal pillows, tailored to accommodate patient anatomy, facial hair, nasal obstruction and claustrophobia.12
Despite these innovations, long-term adherence to PAP therapy remains suboptimal, with studies reporting rates of only 40 to 50% at 12 months.13,14 Factors contributing to poor adherence include discomfort, noise, dry mucosa, nasal congestion, mask leak and a perceived lack of symptom improvement. Behavioural and educational interventions, including follow up with sleep physicians, motivational interviewing and the use of adherence-tracking apps may improve outcomes.12 Clinicians play a role in early troubleshooting and tailoring therapy to individual patient needs to maximise the sustained benefit.
Nasal management
Optimising nasal patency is a valuable step in enhancing upper airway function and overall quality of life. Although it may not significantly reduce OSA severity or snoring, improved nasal airflow can enhance a patient’s tolerance and adherence to PAP therapy by reducing the required pressure setting.15
Before specialist referral, medical optimisation of nasal patency through allergy identification and intranasal corticosteroid therapy is a valuable ‘pre-phase’ approach. Allergy testing for common aeroallergens, including dust mites, grass pollens, animal dander and mould, can help identify contributory triggers. Once identified, allergen avoidance strategies may reduce mucosal inflammation in patients with allergic rhinitis.16 The Australasian Society of Clinical Immunology and Allergy provides evidence- based algorithms for diagnosing and managing allergic and nonallergic nasal conditions.16
Initiating intranasal corticosteroids empirically in patients with turbinate hypertrophy and rhinorrhoea is worthwhile, as they can reduce turbinate hypertrophy and dry out excessive nasal mucus, improving nasal patency. Safe corticosteroids often used long term in intranasal therapy include mometasone furoate and fluticasone propionate. Studies have demonstrated these to be effective in the management of allergic rhinitis with minimal to no systemic adverse effects.17-20 Combined corticosteroid and antihistamine treatments such as fluticasone propionate/azelastine hydrochloride and mometasone furoate/olopatadine hydrochloride can be used to further manage nasal obstruction associated with allergic rhinitis.16 Referrals for allergy desensitisation therapy can be considered in selected cases; this may be in the form of sublingual or subcutaneous immunotherapy.16
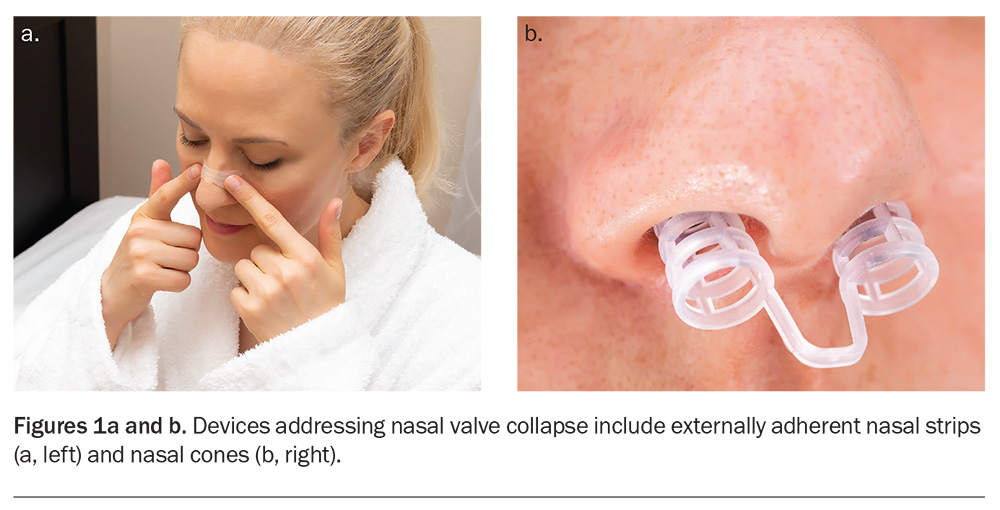
Nasal valve collapse is a common contributor to nasal obstruction. The internal nasal valve, formed by the junction of the nasal septum, upper lateral cartilage and the anterior head of the inferior turbinate, is the narrowest segment of the nasal airway and a defined site of resistance. Turbulent airflow can induce dynamic collapse at this site via the Bernoulli effect.21 Before considering surgical intervention, a range of conservative, over-the-counter options may be trialled. Externally adherent nasal strips, silicone nasal pillows and nasal cones mechanically support the internal and external nasal valves, helping to improve nasal airflow, and may guide ongoing use or surgical stabilisation procedures (Figures 1a and b).
Differential diagnoses and comorbidity assessment
Excessive daytime sleepiness, lethargy and fatigue have a broad differential diagnosis and are not always attributable to OSA. An important step in evaluation is to exclude common, reversible medical contributors such as iron-deficiency anaemia and hypothyroidism.
In patients with OSA, coexisting insomnia should also be considered.22 A detailed history is essential to identify sleep-onset insomnia, sleep-maintenance insomnia or both. Patients may report consistently taking longer than 30 minutes to fall asleep or prolonged difficulty returning to sleep after nocturnal awakenings, both of which suggest an insomnia component.22
In Australia, referral to a sleep psychologist for insomnia-focused cognitive behavioural therapy (CBT-I) is recommended as first-line treatment, with studies demonstrating this method to be effective.23,24 If multiple sessions are required, these can often be incorporated into a mental health care plan to enable Medicare rebates. Evidence supports CBT-I as more effective than pharmacotherapy, including melatonin, for long-term management of insomnia,25 with further clinical practice guidelines available via the Australasian Sleep Association.23 The CBT-I provider registry is available at http://sleepcentral.org.au/central/central/contents/clinicians-CBTi.aspx. Other alternatives to CBT-I are available online through university and government initiatives.
Mandibular advancement splints
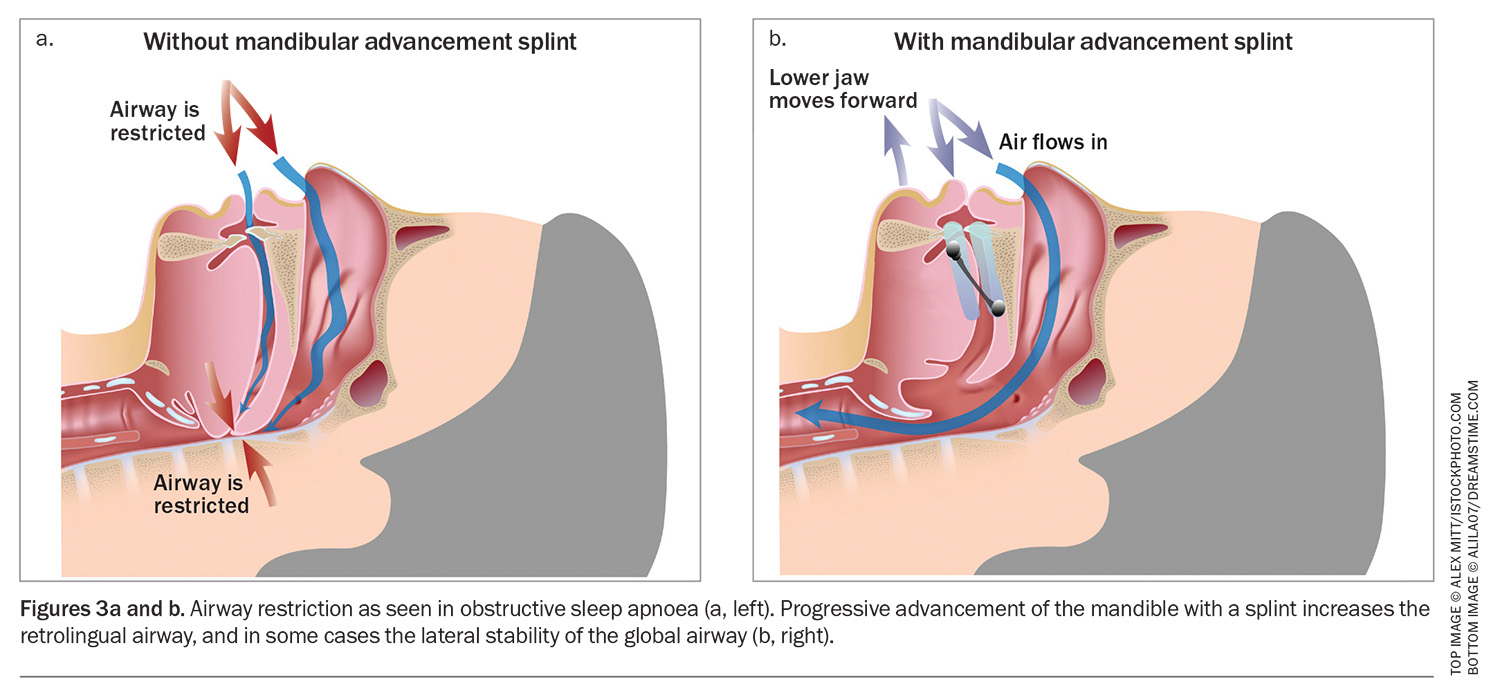
Mandibular advancement splints (Figure 2) can be used as a primary salvage treatment as standalone therapy or in combination therapy. Custom-made mandibular advancement splints are significantly more effective than over-the-counter ‘boil and bite’ devices.26 These appliances function by progressively advancing the mandible, thereby increasing the retrolingual airway space and, in some cases, improving lateral stability of the global airway (Figures 3a and b).
One limitation is that their effectiveness can only be assessed after formal fitting and a trial period, which may be costly. Mandibular advancement splints typically cost between $1000 to $2500, with private health insurance potentially rebating a portion depending on the level of cover. Additionally, some patients experience temporomandibular joint discomfort or changes in occlusion with prolonged use, potentially leading to reduced adherence.27
Treatment adjuncts
Various minimally invasive interventions may be trialled before specialist sleep surgeon or physician review, including sleep position trainer devices.
Positional therapy targets the reduction of supine sleep, which is associated with increased airway collapse in positional OSA. Some devices use sensors worn on the neck, chest or back to detect supine positioning, and gently prompt the user to adopt a side-sleeping posture through the delivery of gentle vibration signals. Additionally, specialised positional pillows that physically restrict supine sleep can be effective, although some patients report shoulder discomfort as a side effect.28
Mouth taping has gained popularity on social media as a potential remedy for snoring. This approach is not without risk. A recent systematic review suggests that mouth taping may be considered in very few carefully selected patients with primary snoring or mild OSA who have no underlying nasal obstruction. Appropriate patient selection is essential to minimise the risk of adverse events, including asphyxiation.29
Referral for surgical opinion
Surgical management is typically reserved for salvage cases in which PAP therapy is not tolerated by the patient or if conservative measures prove insufficient. The primary goal is to improve upper airway patency and reduce airway collapsibility.2 Studies have shown that surgical management of OSA can reduce long-term health consequences compared with no treatment or failed PAP therapy.30 Surgical interventions may be used alone or as part of a multimodal strategy alongside mandibular advancement splints, positional therapy or hypoglossal nerve stimulation.
The decision to proceed with surgery should be individualised, considering the severity of OSA, patient anatomy, treatment goals and comorbidities.2 Contemporary modifications favour palate repositioning and reconstruction without soft tissue resection.31,32 An Australian randomised clinical trial on sleep apnoea multilevel surgery found improved AHI, ESS and quality of life scores up to 3.5 years following multilevel airway surgery.33,34 It is important to note that weight gain following surgery may lead to the recurrence of snoring and OSA and, thus, the maintenance of weight postoperatively is important.
Nasal surgery is considered in patients with OSA who have concurrent nasal obstruction with anatomical or pathological abnormalities. The primary goal of this ‘pre-phase’ treatment is to enhance nasal airflow and improve tolerance and adherence with PAP or other therapy. It is important to note that although nasal surgery may improve comfort and reduce PAP pressures, it does not independently resolve OSA or significantly reduce snoring.15
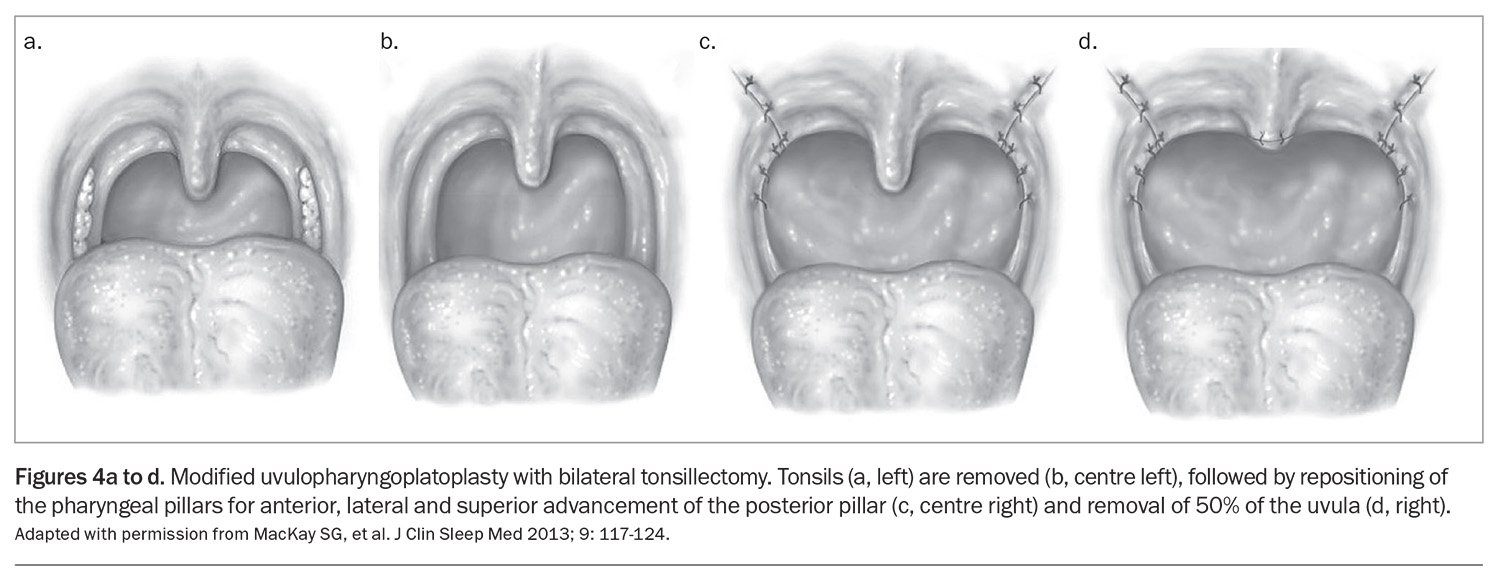
Surgical interventions for OSA are broadly categorised into bony and soft tissue procedures, both aiming to enlarge the upper airway and reduce collapsibility. The principles of surgery are based on careful dynamic anatomical assessment and aim to address multiple levels of the airway (e.g. tonsil, palate, tongue, epiglottis).2 The cornerstone of soft tissue pharyngeal surgery is modified uvulopalatopharyngoplasty, which involves tonsillectomy and anterior, lateral and superior repositioning of the soft palate to increase the retropalatal airway space (Figures 4a to d).35 This is often complemented by radiofrequency coblation of the tongue, a technique used to reduce oral tongue bulk and enhance oropharyngeal airflow.
Lingual tonsillectomy can be indicated in patients with hypertrophic lingual tonsils contributing to posterior displacement of the epiglottis and retrolingual obstruction. Reducing this lymphoid tissue can restore space at the tongue base and facilitate epiglottic anteversion, thereby improving laryngeal patency.36 Transpalatal advancement may be used in patients with vertically orientated, redundant soft palates. The procedure involves anterior mobilisation of a segment of the hard palate to reposition the soft palate into a more favourable oblique orientation.32 When combined with modified uvulopalatopharyngoplasty, this approach significantly enhances anteroposterior expansion of the retropalatal airway.
These procedures may be performed concurrently or in a staged manner, depending on the patient’s anatomical features.36
Hypoglossal nerve stimulation
Hypoglossal nerve stimulation rose to prominence in the Stimulation Therapy for Apnea Reduction (STAR) trial.37 The trial targeted individuals with moderate to severe OSA (AHI score of 20 to 50), a body mass index less than 32 kg/m2 and no complete concentric palatal collapse on drug-induced sleep endoscopy. The results showed significant reductions in the AHI score and improvements in quality of life, with a three-year follow-up study demonstrating ongoing maintenance of these outcome measures.38 Despite technological advancements and the global uptake of hypoglossal nerve stimulation, it is only available through select clinical trials in Australia.5,39
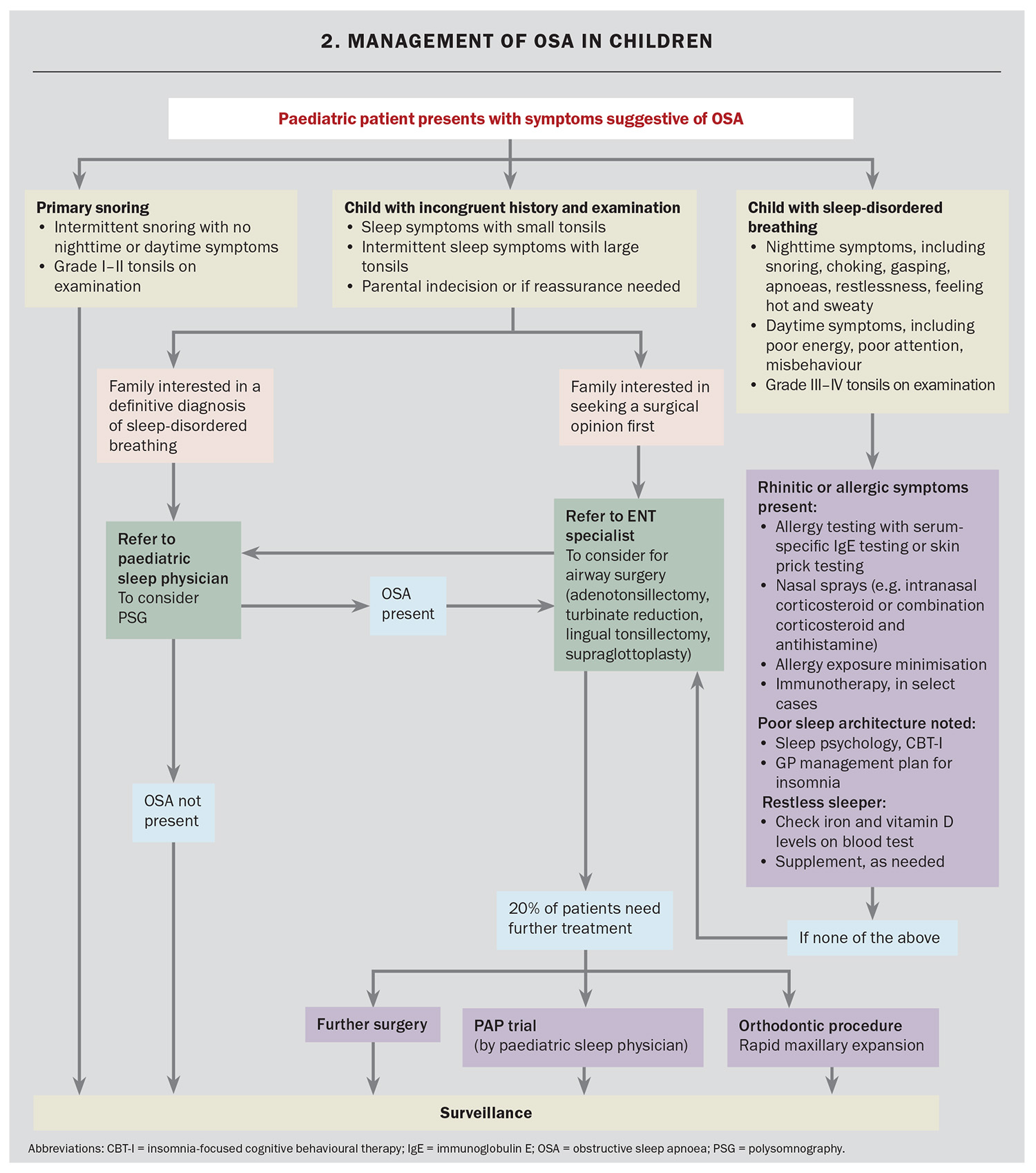
Managing obstructive sleep apnoea in children
Sleep-disordered breathing in children is a spectrum ranging from habitual primary snoring to repetitive episodes of airway obstruction during sleep.4 Primary snoring is not associated with pauses or disruption in breathing. Paediatric patients with isolated snoring, no other symptoms and small tonsils can be placed on surveillance. Surveillance is defined as six- to 12-monthly reviews by a GP, ENT specialist or paediatric sleep physician for progression or development of OSA symptoms.
If the history and examination are incongruent (e.g. snoring with small tonsils or large tonsils with intermittent snoring), a referral to an ENT surgeon or paediatric sleep physician for assessment with consideration of a sleep study is warranted.
Similar to OSA in adults, management of the condition in the paediatric population has many important considerations (Flowchart 2). OSA in children typically presents with snoring in conjunction with nighttime and daytime symptoms with grade III or IV tonsils on examination.40 Nighttime symptoms include gasping, feeling hot, restless sleep or bruxism. Daytime symptoms usually reflect the consequences of disrupted sleep, and include irritability, hyperactivity, poor concentration, academic underperformance and lethargy.4 If there is diagnostic uncertainty or parental indecision, a formal sleep study can be employed.
Prolonged use of pacifiers beyond the age of 4 years has been associated with altered maxillary development, potentially contributing to OSA later in life.41 Children with a high-arched palate should be referred to a specialist orthodontist for the consideration of rapid maxillary expansion, ideally once the first molars have erupted, usually between 6 and 8 years of age.42
In the primary healthcare setting, optimising nasal patency is an important step in managing OSA in children, particularly through the treatment of allergic rhinitis. Allergy testing, via either serum-specific IgE testing (previously known as a radioallergosorbent test) or skin prick testing by an immunologist, can help in identifying relevant triggers. Once identified, allergen avoidance is a key first-line strategy, especially when combined with intranasal corticosteroids.
For patients with confirmed allergies, combination therapies containing both a corticosteroid and an antihistamine, such as fluticasone propionate/azelastine hydrochloride and mometasone furoate/olopatadine hydrochloride, have demonstrated improved effectiveness due to their dual mechanism of action.16 Mometasone furoate and fluticasone propionate are safe for long-term use, as their systemic bioavailability is less than 1%. Their effects on the hypothalamic–pituitary axis and systemic absorption are exceedingly rare.18,20 These intranasal corticosteroids should be used for a minimum of six weeks to allow for adequate absorption into the nasal mucosa for peak effect.19
Children presenting with feeling hot, restless sleep without snoring or gasping should be assessed for iron and vitamin D deficiency, as correction through supplementation has been shown to improve sleep quality in patients with periodic limb movement disorders.43,44 Studies have suggested aiming for serum ferritin levels above 50 mcg/L and vitamin D levels above 75 nmol/L.45,46 Vitamin D supplementation is achieved through 1000 to 2000 units daily of cholecalciferol (vitamin D3). Oral iron supplementation is available in various forms with goals of 3 to 6 mg/kg per day. Iron absorption improves when taken with a source of vitamin C, such as orange juice. Vitamin D and iron levels can be rechecked at three months to assess for improvement or dose adjustment.
Once nasal anatomy and allergic contributors are optimised, the cornerstone treatment for paediatric OSA is adenotonsillectomy. Large-scale studies report significant clinical improvement in up to 80% of cases following this intervention.47 For the remaining 20% who do not experience adequate symptom resolution, further evaluation and tailored management by a paediatric sleep physician or ENT specialist through a multidisciplinary team is recommended. These patients are likely to be complex and would benefit from specialist input to address persistent contributing factors.
Conclusion
OSA is a common, chronic condition with far-reaching health and quality of life consequences. GPs play a crucial role in early identification, investigation and initiation of management, with timely referral to sleep or ENT specialists as appropriate. Management should be individualised, addressing both anatomical and behavioural contributors, and aligned with patient priorities.
Although PAP therapy remains the cornerstone of treatment in adults, a broad range of adjunctive therapies and surgical options are available for patients who are intolerant of or inadequately treated with conservative measures. For children, early intervention, particularly adenotonsillectomy in appropriately selected cases, can have significant developmental and cognitive benefits. A structured, multidisciplinary approach is essential to improve adherence, reduce morbidity and enhance long-term outcomes for both adult and paediatric patients. MT
COMPETING INTERESTS: Dr Kao, Dr Lathif and Associate Professor Jones: None. Professor Mackay has received consulting fees from CNXII Medical and as an advisor for an NIH-funded trial, payment or honoraria for a conference held by SomnoMed, travel for observing operations by Inspire, and support for his role as a speaker for the SISCO Sleep Surgery Workshop and the Prestige Sleep Dissection Workshop.
References
1. Benjafield AV, Ayas NT, Eastwood PR, et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med 2019; 7: 687-698.
2. MacKay SG, Lewis R, McEvoy D, Joosten S, Holt NR. Surgical management of obstructive sleep apnoea: a position statement of the Australasian Sleep Association. Respirology 2020; 25: 1292-1308.
3. Veasey SC, Rosen IM. Obstructive sleep apnea in adults. N Engl J Med 2019; 380: 1442-1449.
4. Evans HJ, Gibson NA, Bennett J, et al. British Thoracic Society guideline for diagnosing and monitoring paediatric sleep-disordered breathing. Thorax 2023; 78(Suppl 2): s1-s27.
5. Woodson BT, Kent DT, Huntley C, et al. Bilateral hypoglossal nerve stimulation for obstructive sleep apnea: a nonrandomized clinical trial. J Clin Sleep Med 2025; 21: 1883-1891.
6. Hamilton G, Li Chai-Coetzer C. Update on the assessment and investigation of adult obstructive sleep apnoea. Aust J Gen Pract 2019; 48: 176-181.
7. Sarkis LM, Jones AC, Ng A, Pantin C, Appleton SL, MacKay SG. Australasian Sleep Association position statement on consensus and evidence based treatment for primary snoring. Respirology 2023; 28: 110-119.
8. Austroads, National Transport Commission. Assessing fitness to drive for commercial and private vehicle drivers: medical standards for licensing and clinical management guidelines. Sydney: Austroads; 2022. Available online at: https://austroads.gov.au/drivers-and-vehicles/assessing-fitness-to-drive (accessed November 2025).
9. Hudgel DW, Patel SR, Ahasic AM, et al. The role of weight management in the treatment of adult obstructive sleep apnea. an official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med 2018; 198: e70-e87.
10. Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N Engl J Med 2024; 391: 1193-1205.
11. Ellender C, Vakulin A, Stocks N, Li Chai-Coetzer C. Management of obstructive sleep apnoea in primary care. Aust J Gen Pract 2024; 53: 363-369.
12. Bakker JP, Weaver TE, Parthasarathy S, Aloia MS. Adherence to CPAP: what should we be aiming for, and how can we get there? Chest 2019; 155: 1272-1287.
13. Weaver TE, Grunstein RR. Adherence to continuous positive airway pressure therapy: the challenge to effective treatment. Proc Am Thorac Soc 2008; 5: 173-178.
14. Rotenberg BW, Murariu D, Pang KP. Trends in CPAP adherence over twenty years of data collection: a flattened curve. J Otolaryngol Head Neck Surg 2016; 45: 43.
15. Camacho M, Riaz M, Capasso R, et al. The effect of nasal surgery on continuous positive airway pressure device use and therapeutic treatment pressures: a systematic review and meta-analysis. Sleep 2015; 38: 279-286.
16. Australasian Society of Clinical Immunology and Allergy (ASCIA). Allergic rhinitis clinical update. Sydney: ASCIA; 2024. Available online at: https://www.allergy.org.au/hp/papers/allergic-rhinitis-clinical-update (accessed November 2025).
17. Sousa-Pinto B, Vieira RJ, Bognanni A, et al. Efficacy and safety of intranasal medications for allergic rhinitis: network meta-analysis. Allergy 2025; 80: 94-105.
18. Sastre J, Mosges R. Local and systemic safety of intranasal corticosteroids. J Investig Allergol Clin Immunol 2012; 22: 1.
19. McDonnell J, Weller K, Pien LC. Safety of intranasal steroids: an updated perspective. Curr Allergy Asthma Rep 2020; 20: 69.
20. Allen DB. Systemic effects of intranasal steroids: an endocrinologist’s perspective. J Allergy Clin Immunol 2000; 106: S179-S190.
21. Liu DH, Chen H, Wong BJ. Anatomy and physiology of the nasal valves. Otolaryngol Clin North Am 2025; 58: 189-203.
22. Sweetman A, Andronis C, Hancock K, Stocks N, Lack L, McEvoy R. General practitioner assessment and management of insomnia in adults. Aust J Gen Pract 2023; 52: 691-698.
23. Australasian Sleep Association Behavioural Management of Sleep Disorders education subcommittee. Sleep problems in psychological practice. InPsych 2022; 44.
24. Edinger JD, Arnedt JT, Bertisch SM, et al. Behavioral and psychological treatments for chronic insomnia disorder in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med 2021; 17: 255-262.
25. Morin CM, Buysse DJ. Management of insomnia. N Engl J Med 2024; 391: 247-258.
26. Love RL, Naughton MT, Cistulli P, McArdle N, MacKay SG. Complications and safe prescription of interventions for adult sleep disordered breathing in Australia. Aust J Otolaryngol 2019; 2: 9.
27. Mohammadieh AM, Sutherland K, Chan ASL, Cistulli PA. Mandibular advancement splint therapy. Adv Exp Med Biol 2022; 1384: 373-385.
28. Omobomi O, Quan SF. Positional therapy in the management of positional obstructive sleep apnea—a review of the current literature. Sleep Breath 2018; 22: 297-304.
29. Fangmeyer SK, Badger CD, Thakkar PG. Nocturnal mouth-taping and social media: a scoping review of the evidence. Am J Otolaryngol 2025; 46: 104545.
30. Every JD, Mackay SG, Sideris AW, Do TQ, Jones A, Weaver EM. Mean disease alleviation between surgery and continuous positive airway pressure in matched adults with obstructive sleep apnea. Sleep 2023; 46: zsad176.
31. MacKay SG, Love RL. Palatal surgery. In: Salman SO, ed. Modern management of obstructive sleep apnea. New York: Springer International Publishing; 2019. p. 49-57.
32. Tucker Woodson B, Kocdor P, Seyyedi M, Froymovich O. Transpalatal advancement pharyngoplasty and expansion sphincterplasty for obstructive sleep apnea. Oper Tech Otolaryngol Head Neck Surg 2015; 26: 95-99.
33. Pinczel AJ, Woods CM, Catcheside PG, et al. Sleep apnea multi-level surgery trial: long-term observational outcomes. Sleep 2024; 47: zsad218.
34. Crossnohere NL, Schuster ALR, Bruckel J, et al. Patient-reported outcome measures add value as clinical trial endpoints. Nat Med 2025; 31: 3601-3604.
35. MacKay SG, Carney AS, Woods C, et al. Modified uvulopalatopharyngoplasty and coblation channeling of the tongue for obstructive sleep apnea: a multi-centre Australian trial. J Clin Sleep Med 2013; 9: 117-124.
36. Lin H, Weaver E, Lin H, Friedman M. Multilevel obstructive sleep apnea surgery. Adv Otorhinolaryngol. 2017; 80: 109-115.
37. Strollo PJ Jr, Soose RJ, Maurer JT, et al. Upper-airway stimulation for obstructive sleep apnea. N Engl J Med 2014; 370: 139-149.
38. Alrubasy WA, Abuawwad MT, Taha MJJ, et al. Hypoglossal nerve stimulation for obstructive sleep apnea in adults: an updated systematic review and meta-analysis. Respir Med 2024; 234: 107826.
39. Kim DH, Kim SW, Han JS, Kim GJ, Basurrah MA, Hwang SH. Hypoglossal nerve stimulation effects on obstructive sleep apnea over time: a systematic review and meta-analysis. Otolaryngol Head Neck Surg 2024; 170: 736-746.
40. Haug KL, Clapp JT, Schwarze ML. Innovations in surgical communication- provide your opinion, don’t hide it. JAMA 2023; 158: 993-994.
41. Sexton S, Natale R. Risks and benefits of pacifiers. Am Fam Physician 2009; 79: 681-685.
42. Ngiam J, Cistulli PA. Dental treatment for paediatric obstructive sleep apnea. Paediatr Respir Rev 2015; 16: 174-181.
43. Ipsiroglu OS, Pandher PK, Hill O, et al. Iron deficiency and restless sleep/wake behaviors in neurodevelopmental disorders and mental health conditions. Nutrients 2024; 16: 3064.
44. Prono F, Bernardi K, Ferri R, Bruni O. The role of vitamin D in sleep disorders of children and adolescents: a systematic review. Int J Mol Sci 2022; 23: 1430.
45. Abboud M. Vitamin D supplementation and sleep: a systematic review and meta-analysis of intervention studies. Nutrients 2022; 14: 1076.
46. Leung W, Singh I, McWilliams S, Stockler S, Ipsiroglu OS. Iron deficiency and sleep–a scoping review. Sleep Med Rev 2020; 51: 101274.
47. Connolly HV, Tomaselli LT, McKenna Benoit MK. Adenotonsillectomy for pediatric obstructive sleep apnea: how to predict those at risk for postoperative complications. J Clin Sleep Med 2020; 16: 3-4.