Alopecia and crusted scalp in a young boy
A kerion, a severe inflammatory variant of tinea capitis, can result in irreversible follicular destruction and scarring alopecia. Careful attention to clinical findings, including lesion morphology and trichoscopic and systemic features, are helpful in achieving a correct diagnosis in a child presenting with hair loss.
- A kerion is a severe inflammatory variant of tinea capitis characterised by boggy plaques with overlying crust.
- Early recognition and prompt treatment of a kerion are essential to reduce the risk of permanent alopecia, which can have significant psychosocial implications later in life.
- In the time of rising antimicrobial resistance, appropriate identification of the causative organism is crucial. Empirical antifungal therapy, along with adjunctive therapies, should be initiated promptly and not delayed, while targeted antifungal therapy should commence as soon as the specific pathogen is identified.
- Repeating a fungal culture at the end of treatment is recommended to ensure complete clearance and reduce the risk of recurrence.
- Environmental decontamination measures are helpful to reduce fungal reservoirs and risk of recurrence.
- Oral antibiotic therapy should be used if there is evidence of a secondary bacterial infection.
Case scenario
A healthy 5-year-old boy presented with a one-month history of progressively enlarging patches of scalp alopecia accompanied by erythema, scaling and pustules. The condition had failed to respond to an initial course of oral antibiotics. There was no history of exposure to household pets or other animals. His family members did not report similar symptoms.
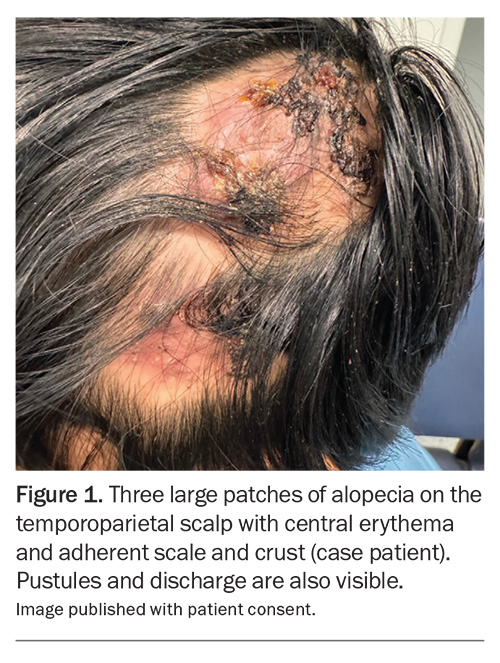
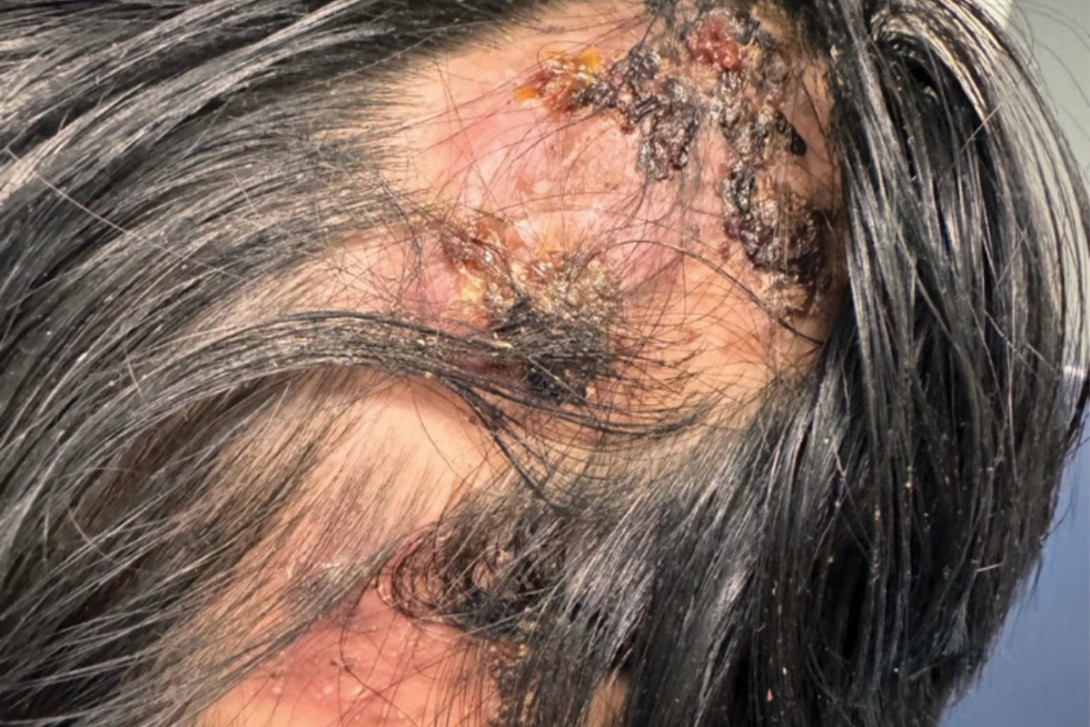
On examination, three large patches of alopecia were observed on the patient’s left temporoparietal scalp (Figure 1). These were noted to be tender, erythematous plaques with overlying adherent scale and crust. Scattered folliculocentric pustules and areas of boggy, purulent discharge were also present. Short, broken hair shafts and several corkscrew hairs were visible on trichoscopy within the affected areas. There was no palpable lymphadenopathy, and the child was systemically well.
What diagnoses should be considered?
In paediatric scalp alopecia, careful attention to clinical features, including lesion morphology, trichoscopic findings and systemic features may help refine the diagnosis. In this case, the clinical picture was most consistent with a kerion, a severe inflammatory variant of tinea capitis characterised by boggy plaques with overlying crust. This inflammation is driven by a hypersensitivity reaction to dermatophyte antigens mediated by T-cell lymphocytes. A kerion predominantly affects young children and is most commonly seen in patients aged 3 to 7 years. Systemic features, such as fever and enlarged cervical lymph nodes, may accompany its presentation.1 The presence of corkscrew hairs supports the diagnosis of an endothrix dermatophyte infection, where fungal invasion of the hair shaft disrupts internal keratin, causing hairs to be weakened, twisted and easily broken.1
Noninflammatory tinea capitis, which typically presents as diffuse scale, grey patches with fine scaling, or black dot alopecia, is a diagnosis that should be considered. However, the marked inflammatory response and purulent discharge made it less likely in this case.
Bacterial folliculitis, while potentially mimicking a kerion with overlapping features (e.g. pustular lesions, erythema and crusting), generally lacks the hallmark boggy, indurated, alopecic plaques. Alopecia areata should be considered in patients with multifocal hair loss; however, its classic presentation involves smooth, well-circumscribed patches of alopecia with the absence of scale or pustules, which was inconsistent with the observed clinical findings in this case.
What investigations are appropriate for suspected tinea capitis?
The primary diagnostic methods for patients with suspected tinea capitis, whether inflammatory or not, involve hair pluckings, scalp scrapings and crust material obtained for direct microscopy and fungal culture. Microscopy offers prompt initial detection of fungal elements, such as hyphae or spores, but negative results are not definitive because of potential interobserver variability, inadequate sampling or low fungal loads within specimens.2,3 Fungal culture, which typically requires two to four weeks (and up to six weeks) for adequate growth, allows for accurate species identification. Clinicians should actively follow up on fungal culture results to determine the specific dermatophyte because this may inform the need to modify antifungal therapy. Importantly, if clinical features strongly suggest inflammatory tinea capitis then treatment must not be delayed while awaiting culture confirmation – empirical systemic antifungal therapy should be initiated promptly.
A fungal PCR test offers a significant advantage because of its high sensitivity as well as rapid turnaround time (24 to 48 hours) and is particularly useful in situations where a patient has received prior or partial antifungal treatment that would hinder culture growth or where the clinical presentation is atypical.4 However, depending on the assay used, certain uncommon fungal species may not be included in the PCR panel, and, therefore, it should always be performed alongside microscopy and culture to ensure diagnostic accuracy.
A Wood’s lamp examination may support clinical suspicion by revealing a characteristic green-yellow fluorescence associated with Microsporum species. However, this should not be used in isolation because its sensitivity is markedly reduced in patients with a kerion as significant inflammation can obscure the presence of the organism.5 When fluorescence is observed, Microsporum canis is the most likely causative species and should prompt targeted evaluation for zoonotic exposure, such as household pets (including the pets of family and friends), other animal contact and recent veterinary encounters.6
It is also important to consider the need for a bacterial swab to evaluate for possible secondary infection, which is commonly encountered.7 Coinfection may intensify the inflammatory response and necessitate adjunctive antibiotic therapy.
How is a kerion managed?
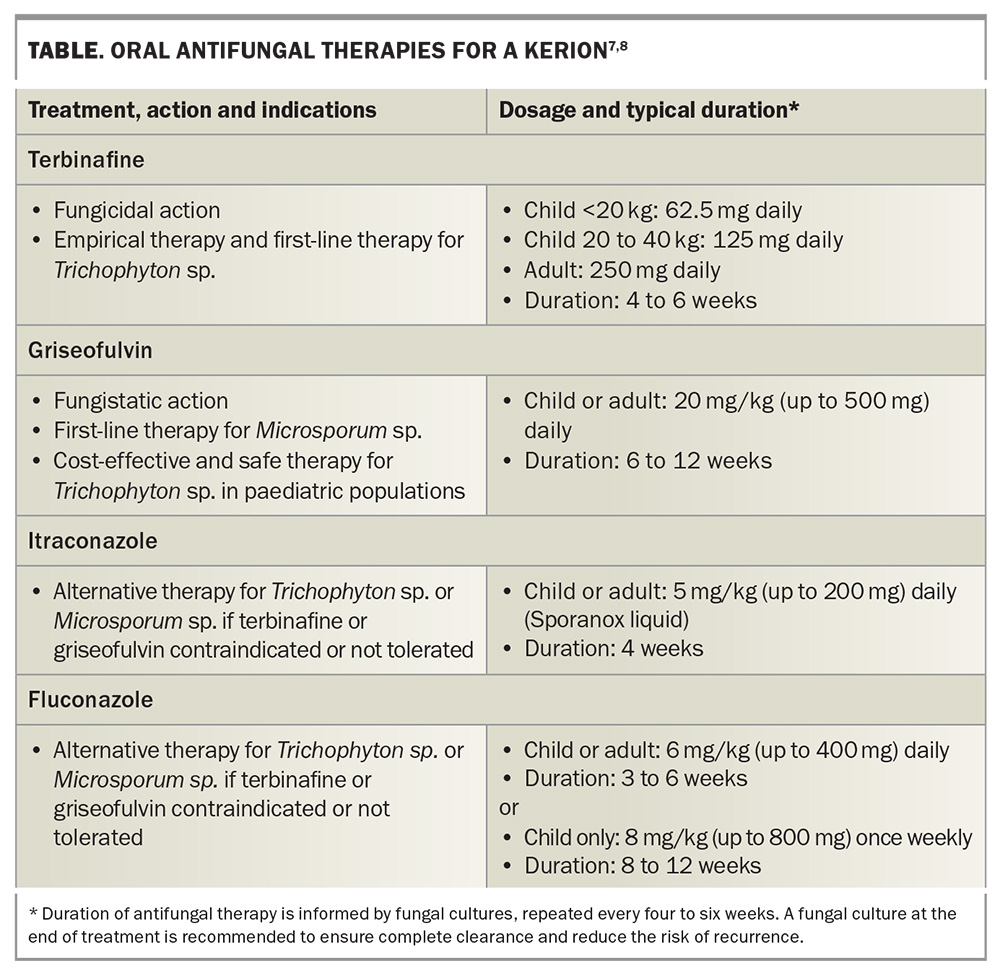
The management of kerion requires a holistic approach (Box). Oral antifungal therapy is the cornerstone. Terbinafine is used as empirical therapy and when Trichophyton species is identified, whereas griseofulvin is used for kerion caused by Microsporum species. Itraconazole and fluconazole are used when terbinafine or griseofulvin is contraindicated or not tolerated. Fungal cultures should be repeated every four to six weeks for patients receiving oral antifungal therapy; the duration should be extended if culture results remain positive and may be discontinued when they return negative and hair regrowth is evident. The use of oral antifungal therapy for treating a kerion is described in the Table.7,8 Topical antifungal therapy alone is not appropriate for tinea capitis because it cannot penetrate the hair follicle – systemic treatment is essential, with topical agents serving only as adjuncts.9
Adjunctive therapies include medicated shampoos that have a sporicidal activity (ketoconazole 2%, selenium sulfide 2.5%), applied three times weekly with a contact time of two to three minutes prior to rinsing, to help to prevent spread and reduce risk of reinfection. Saline soaks are used to remove surface crust, which promotes healing by alleviating itch and reducing risk of secondary infection and fungal spore burden.
Oral antibiotics are indicated if there is evidence of secondary bacterial infection and reduces contributory inflammation as well as risk of progression to scarring alopecia. Antibiotic selection should be guided by culture and sensitivity results; Staphylococcus aureus is the organism that is most commonly isolated.10 The course duration depends on infection severity.
Environmental decontamination measures are used to reduce fungal reservoirs. Items that are in contact with the hair (such as combs and hats) should not be shared with others, and clothing and bed linen should be laundered at high temperature (>60°C). Hair trimmers should be sterilised. If the kerion is suspected to have originated after a haircut in a community salon then informing the barber would be advisable. Family members should be examined and treated if there is evidence of infection. In addition, treating household pets when necessary is important to reduce the risk of reinfection and ongoing transmission in humans.
Educating the patient and caregivers about the condition and its management is important. Children can attend school while minimising the risk of infection by covering the lesions with a hat or dressings to prevent community spread. If a patient is experiencing significant emotional distress or anxiety related to hair loss, potential scarring or social stigma then referral to a counsellor or psychologist should be considered.
What are the risks of an untreated or mismanaged kerion?
An untreated or suboptimally managed kerion carries substantial risks to both the patient and public health. The exaggerated inflammatory response to dermatophyte antigens, often complicated by secondary bacterial infection, can result in irreversible follicular destruction and scarring alopecia, which is particularly concerning in paediatric patients because of the potential to significantly impact psychosocial wellbeing. Furthermore, the high transmissibility presents a public health risk, particularly in communal settings such as schools and households. Management failures, such as use of topical antifungal therapy instead of oral therapy or overlooking bacterial superinfection, may delay resolution and increase complication rates.
Dermatophyte resistance to standard antifungal treatments has been demonstrated. In cases of clinical nonresponse to treatment, species identification can guide the selection of alternative agents or combination therapy or indicate the necessity for further diagnostic testing.11 Of particular concern is Trichophyton indotineae, an emerging dermatophyte resistant to terbinafine. Alternative systemic treatments include oral itraconazole, super bioavailable itraconazole (SUBA-itraconazole) or oral voriconazole. To optimise clinical outcomes, combination therapy with topical agents such as ketoconazole, clotrimazole, or ciclopirox, may also be necessary.12 If encountered in clinical practice then management would be guided by an infectious disease specialist.
Case patient: clinical course and outcome
For the case patient, a clinical diagnosis of a kerion was made on the basis on the clinical presentation and characteristic trichoscopic findings. Scalp skin scrapings and hair pluckings were taken and returned negative results on microscopy; a bacterial swab demonstrated heavy growth of methicillin-sensitive S. aureus. While fungal culture results were pending, a multimodal management plan was implemented. The patient was treated with two courses of oral terbinafine (125 mg daily over 12 weeks), oral cephalexin (400 mg twice daily for two weeks) and adjunctive ketoconazole 2% shampoo. This regimen resulted in partial clinical improvement with a reduction in purulence, but erythema and crusting persisted.
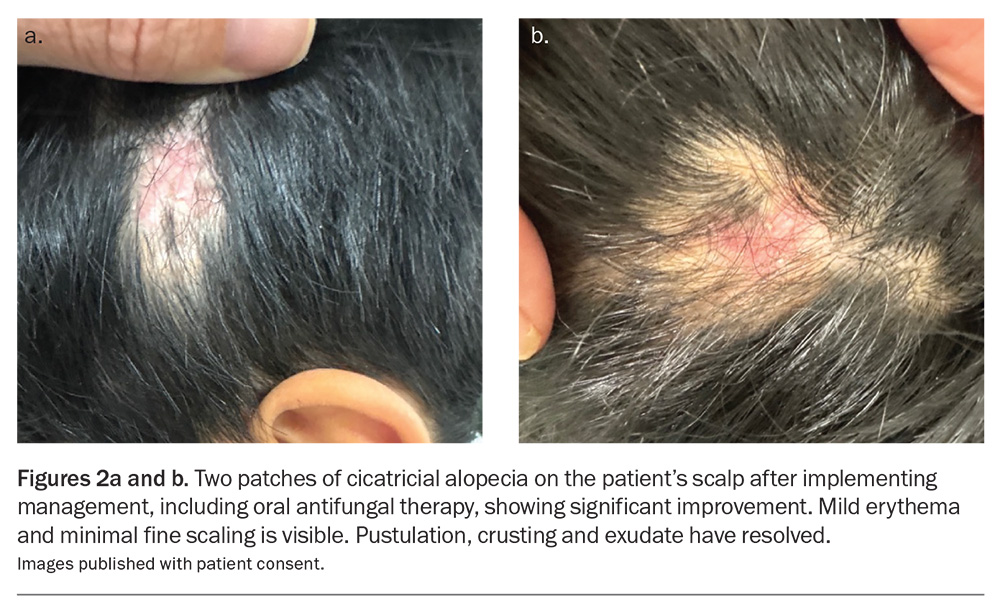
Fungal culture subsequently identified Trichophyton violaceum, an anthropophilic dermatophyte. In the context of an incomplete response to terbinafine, the systemic antifungal therapy was transitioned to oral griseofulvin (250 mg daily). Marked clinical improvement was observed after six weeks, with near-complete resolution of scale and residual erythema (Figures 2a and b). Substantial hair regrowth was evident across the previously affected scalp, but two small residual patches of alopecia remained. Trichoscopic examination demonstrated loss of follicular ostia consistent with focal scarring alopecia secondary to the preceding severe inflammatory kerion.
The patient was discharged from the dermatology service into the care of his GP with a plan to continue oral griseofulvin and ketoconazole shampoo (once weekly) until mycological clearance is confirmed on repeat fungal culture of skin scrapings. MT
COMPETING INTERESTS: None.
References
1. Leung AKC, Hon KL, Leong KF, Barankin B, Lam JM. Tinea capitis: an updated review. Recent Pat Inflamm Allergy Drug Discov 2020; 14: 58-68.
2. Ali AI, Aden AI, Mohamud AK. Dermoscopic findings in tinea capitis among under 18 children in dermatology polyclinic patients: a hospital-based cross-sectional study. Ann Med Surg (Lond) 2023; 86: 121-126.
3. Knoll MA, Steixner S, Lass-Flörl C. How to use direct microscopy for diagnosing fungal infections. Clin Microbiol Inf 2023; 29: 1031-1038.
4. Douglass Hanly Moir Pathology. Dermatophyte PCR [Internet]. Macquarie Park; 2025. Available online at: https://www.dhm.com.au/media/2uchuqnt/dhm-information-for-clinicians-dermatophyte-pcr-mar-2025.pdf (accessed February 2026).
5. Gnanasegaram M. Kerion. DermNet; 2012. Available online at: https://dermnetnz.org/topics/kerion (accessed February 2026).
6. Pasquetti M, Min A, Scacchetti S, Dogliero A, Peano A. Infection by Microsporum canis in paediatric patients: a veterinary perspective. Vet Sci 2017; 4: 46.
7. Therapeutic Guidelines (TG). Treatment of tinea. Melbourne: TG; 2025. Available from: https://www.tg.org.au/ (accessed February 2026).
8. Kelly BP. Superficial fungal infections. Pediatr Rev 2012; 33: e22-e37.
9. Kakourou T, Uksal U. Guidelines for the management of tinea capitis in children. Pediatr Dermatol 2010; 27: 226-228.
10. Friedland R, Sabbah F, Reiss-Huss S, Ben Amitai D. Epidemiologic features and risk of scarring in pediatric patients with kerion celsi. Pediatr Dermatol 2022; 39: 215-219.
11. Gupta AK, Venkataraman M. Antifungal resistance in superficial mycoses. J Dermatolog Treat 2022; 33: 1888-1895.
12. Sonego B, Corio A, Mazzoletti V, et al. Trichophyton indotineae, an emerging drug-resistant dermatophyte: a review of the treatment options. J Clin Med 2024; 13: 3558.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.