Typical reflux disease: recognition and management of resistance to therapy
Diagnosis of gastro-oesophageal reflux disease depends on recognising the cardinal symptoms of heartburn and regurgitation, with faulty diagnosis contributing to apparent failures of medical management. Management of persistent reflux symptoms, including heartburn, regurgitation and chest pain, that are not well controlled by medical therapy may require insightful physiological management. Laparoscopic surgery can offer excellent and durable outcomes in this patient group.
- Proton pump inhibitors (PPIs) are the mainstay of treatment for most patients with gastro-oesophageal reflux disease, but 20 to 30% of patients remain symptomatic despite therapy.
- If medical treatment does not adequately control symptoms, diagnosis requires an oesophageal physiology study (manometry and 24-hour pH impedance testing) while the patient is not taking PPIs.
- Endoscopy is only 30% sensitive for the diagnosis of gastro-oesophageal reflux disease.
- Double-dose PPIs are only marginally effective for symptom control in the medium term.
- Systematic reviews have found that minimally invasive laparoscopic antireflux surgery is superior to medical management in treating refractory reflux disease.
Gastro-oesophageal reflux disease (GORD) is a chronic disease caused by effortless reflux of gastric fluid into the oesophagus or extraoesophageal space, causing troublesome symptoms.1 The prevalence of GP-diagnosed GORD in Australia has been estimated at 11.3%.2 Prevalence has been reported in the USA as 18.1 to 27.8%, and in Europe as 8.8 to 25.9%.3 Medically refractory GORD refers to reflux symptoms that continue despite lifestyle changes and the use of proton pump inhibitors (PPIs). This resistance to therapy can lead to significantly reduced health-related quality of life for patients.4
This article aims to guide the selection of patients with typical reflux who have poorly controlled disease and may be suitable for minimally invasive surgery to further reduce symptoms and improve their quality of life. The relative merits of managing severe reflux disease with continued medication, increased medication and ancillary therapies are discussed, along with the efficacy of surgery.
Mechanisms of abnormal reflux
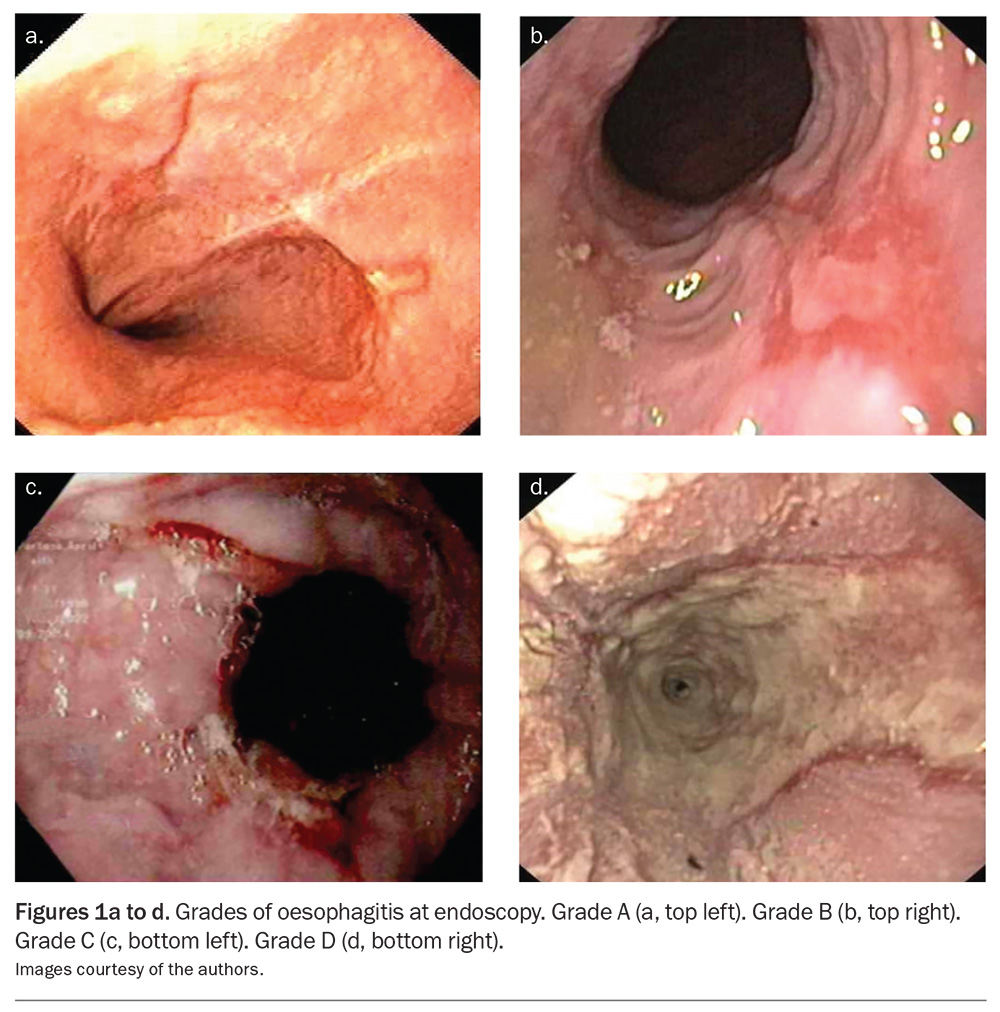
Multiple physiological abnormalities may lead to abnormal frequency or duration of reflux or distribution of reflux events, causing symptoms. Disease may be confined within the oesophagus or become extraoesophageal, causing different symptoms depending on the sites affected. Symptoms and oesophagitis (Figure 1) are caused by an imbalance between the duration of acid exposure of the oesophagus and mucosal defences; this can also lead to ‘flooding’ of extraoesophageal sites.
Reflux events may be initiated by failure of the antireflux barrier, due to an abnormal crural diaphragm (CD), diminished lower oesophageal sphincter (LOS) function or delayed oesophagogastric clearance. Symptoms and mucosal damage are influenced by the gastric acid pocket located just below the LOS, the aggressiveness of constituents of reflux fluid, oesophageal clearance and the balance of mucosal resistance.5,6 The antireflux barrier between the stomach and the oesophagus comprises the LOS, the CD and the gastric sling filaments. The anatomically normal region may allow abnormal reflux through increased transient LOS relaxation, low LOS basal pressure, swallow-induced relaxations and straining. The severity of acid exposure corresponds to the level of physiological abnormality and is more extreme once anatomical changes, namely hiatal herniation, occur. A hiatal herniation dislocates the otherwise coinciding LOS and CD, rendering the antireflux mechanism less effective and allowing more frequent reflux events.7
Refluxate may consist of acid, especially from the acid pocket below the LOS, and is not well titrated by feeding. Acid reflux is associated with heartburn, regurgitation and chest pain when disease is located within the oesophagus. Extraoesophageal proximal manifestations include throat symptoms, reflux cough, choking and inhalation. These do not reflect atypical reflux disease but are symptoms of volume reflux occurring in a patient with index symptoms of heartburn or regurgitation. Volume reflux is common in patients with treatment-resistant disease. Reflux events may occur during treatment with PPIs and may be associated with the sensation of regurgitation – a breakthrough symptom that patients find disturbing.8,9
Biliary pancreatic fluid may be present in refluxate in a third of patients and is especially prominent in patients with Barrett’s oesophagus.10 The noxious effect of bile is not influenced by acid suppression and is a possible cause of refractory symptoms.
The duration of exposure to refluxate may be extended by ineffective oesophageal and gastric clearance. Diminished efficacy of peristalsis contributes to delayed reflux bolus clearance. Chemical neutralisation by saliva reduces acid exposure time and is important for symptom control. Hiatal herniation may act as a reservoir for re-reflux and prolonged lower oesophageal exposure to refluxate.11
Diagnosis
Symptomatic evaluation
Typical reflux is largely diagnosed by identifying an abnormal frequency of heartburn and regurgitation symptoms before a patient has started taking medication. Other symptoms (e.g. belching, bloating, epigastric burn, gastric dyspepsia or throat burn) have a very low probability of being associated with reflux disease. Both the sensitivity and specificity of symptoms for diagnosing GORD are only about 65%.12 Diagnosis may be difficult because as many as two-thirds of patients with ‘refractory reflux’ do not have GORD.13
Differential diagnosis of reflux symptoms includes:
- abnormal oesophageal motility
- gastric emptying abnormality
- other oesophageal inflammatory conditions
- gallbladder disease
- peptic ulceration
- cardiorespiratory disease
- rumination syndrome
- bulimia
- irritable bowel syndrome
- neoplasia
- dyspepsia.
Reflux disease is the most common cause of noncardiac atypical chest pain.14 It is important to identify alarm symptoms that require urgent exclusion of other diseases, particularly malignancy. These symptoms include progressive new-onset symptoms, dysphagia, weight loss, vomiting, upper gastrointestinal bleeding, iron deficiency and new-onset dyspepsia.
Proton pump inhibitor test
Although it seems logical that symptoms that respond to a four-week trial of PPI therapy would be likely to confirm the diagnosis of GORD, this is poorly validated scientifically and has been considered neither sensitive nor specific. Making the diagnosis on the basis of symptomatic response to medical therapy is therefore not recommended. Nonetheless, if good symptom control is established, PPIs become effective treatment and further diagnostic assessment is generally not required.
In assessing the efficacy of PPI therapy, it is not enough to only ask the patient if the heartburn has resolved. The physiological cause of regurgitation events remains and reflux events will continue, although the patient will be less symptomatic when acid concentration is reduced. Regurgitation causing coughing, choking, disturbed sleep and postprandial chest pain may persist. The patient may have dysphagia secondary to continued oesophagitis or dysmotility. Heartburn must be retrosternal to have diagnostic efficacy for GORD, whereas gastric and throat burning are considered atypical symptoms that are not diagnostic of reflux disease and are not likely to respond to PPI therapy.
Endoscopy evaluation
Endoscopy has a sensitivity of only 30% for the diagnosis of GORD, diminishing its diagnostic value.15 It is largely used to determine complications of reflux disease and to exclude other conditions that may mimic it. Only about 10% of patients with reflux will show the presence of reflux oesophagitis on endoscopy. The only endoscopic criteria considered positive for diagnosis of reflux are grades C and D oesophagitis under the Los Angeles (LA) classification (Figure 1). LA grade A oesophagitis is poorly diagnostic of reflux disease. LA grade B oesophagitis, Barrett’s oesophagus and peptic stricture are highly suggestive of reflux disease.15 Presence of stricture may indicate a need for further treatment and exclusion of neoplasia. Identification of Barrett’s oesophagus is a likely indication for premalignant surveillance.
Oesophageal physiology
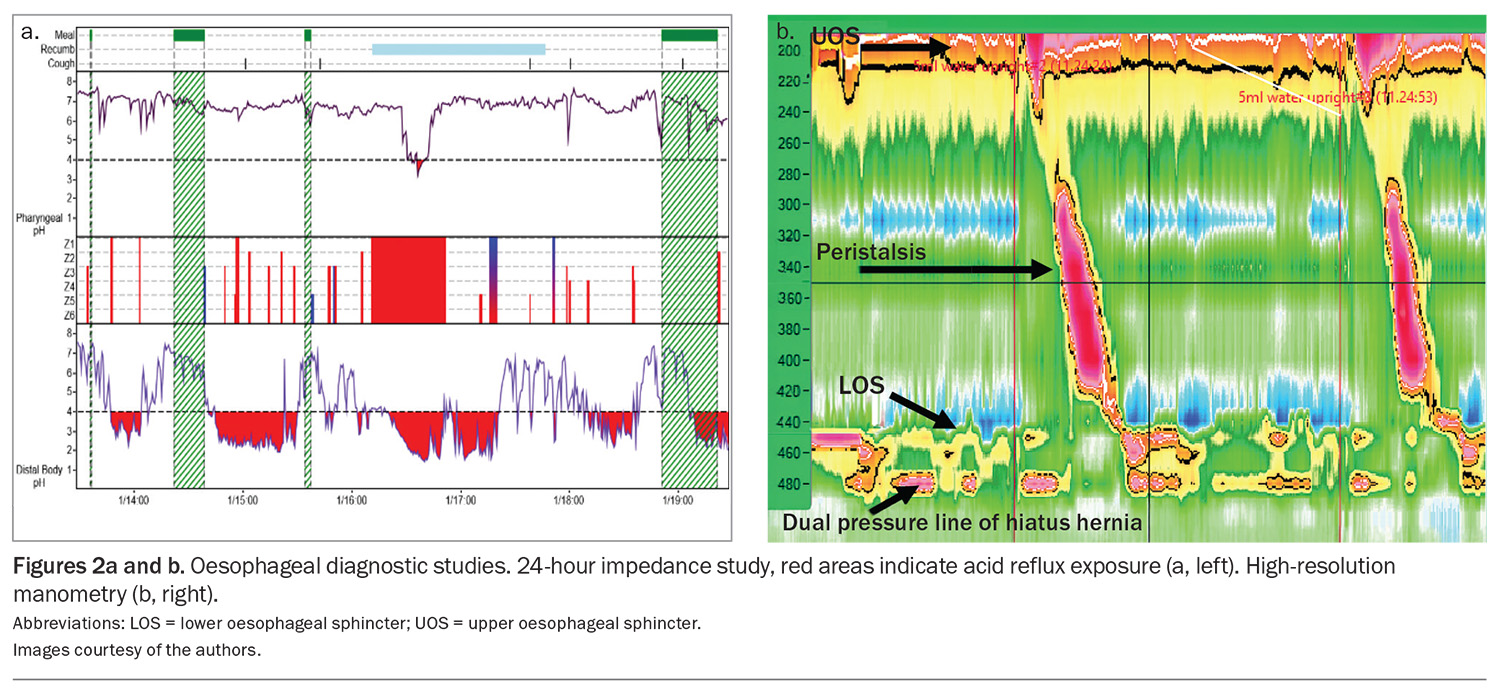
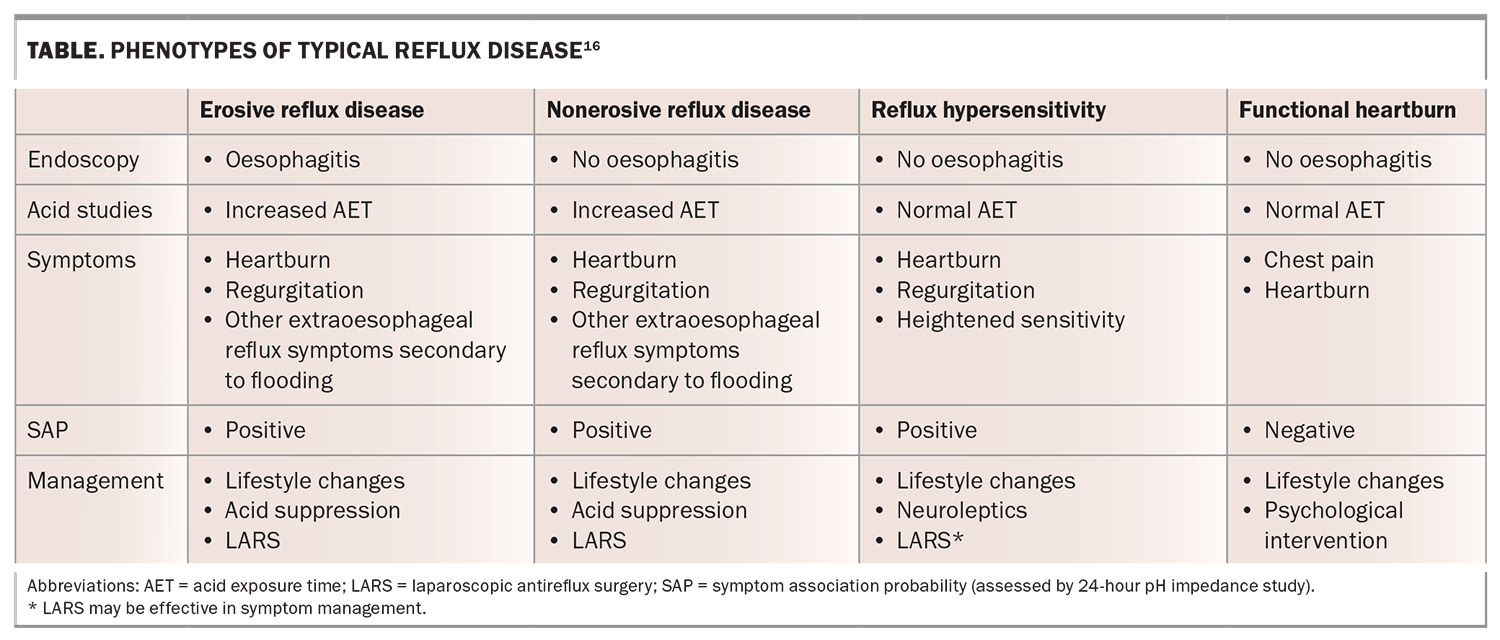
A 24-hour pH impedance study combined with oesophageal manometry (Figure 2) is generally the most accurate method of identifying typical reflux disease and the relationship between the reflux event and symptoms – known as symptom association probability (SAP). The SAP is vital in categorising patients by the various phenotypes of reflux disease, to enable appropriate treatment.16 Phenotypes include:
- GORD or erosive reflux disease, where oesophagitis occurs secondary to reflux disease
- nonerosive reflux disease, where reflux of acid or nonacid material is abnormally elevated but there is no oesophagitis
- reflux hypersensitivity, where there is normal acid or nonacid exposure and positive SAP
- functional heartburn, where there is no evidence of reflux disease and negative SAP, with normal reflux event frequency.17
Acid-related disease can be treated with acid suppression or surgery, whereas reflux hypersensitivity and functional heartburn require different therapy (Table).16
Oesophageal function testing can be used to aid diagnosis in patients with poor or incomplete symptomatic response to medication and with no or minimal oesophagitis seen on endoscopy. Multiple international guidelines recommend performing this test without acid suppression. There are no normative data supporting the use of reflux testing in patients when taking PPIs.
Therapy
Antisecretory therapy
The mainstay of treatment of persistent reflux symptoms is continuous PPI therapy. Although dose minimisation is preferable, involving use of antacids and intermittent PPIs or regular H2 antagonists, patients with severe symptoms require continuous treatment.18 When adequate symptom control is not achieved, a recent meta-analysis showed that doubling the dose of PPIs does not improve efficacy in the medium term.19
Conservative management with PPIs is beneficial in 70 to 75% of patients with typical reflux disease when adequately diagnosed. However, large PPI dosage evaluation studies have shown a lack of symptom control in the remaining 25 to 30% of patients, who may be candidates for surgical therapy to improve their quality of life.20-22
Lifestyle management
Although effective in patients with mild to moderate reflux disease, lifestyle management is less effective for those with more severe disease and often affects quality of life.23 Elevating the head of the bed may disturb sleep, and there is little evidence that this is effective. Equally, weight loss, cessation of smoking and avoiding late-evening gastric load, although practically useful and worth trialling for patients with mild disease, have very little supporting evidence and are less likely to be of benefit for patients with more severe disease.24
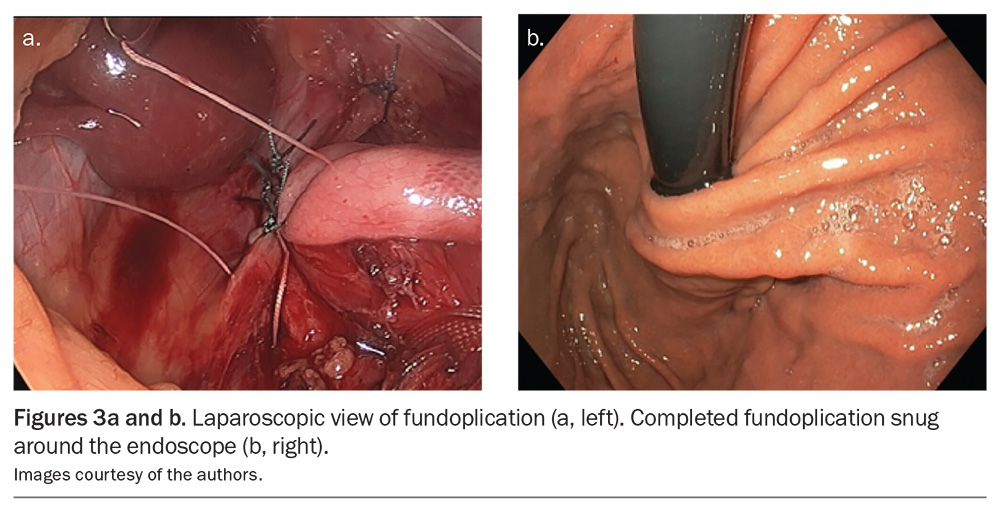
Minimally invasive laparoscopic antireflux surgery
Meta-analysis of randomised controlled trials (RCTs) of medical therapy with PPIs compared with laparoscopic antireflux surgery (LARS; Figure 3) has shown surgery to be more symptomatically effective than PPIs in the treatment of medically refractory GORD.25,26 There was a significant long-term reduction in heartburn in the LARS groups compared with the medical groups. Objective control of reflux disease, measured by 24-hour pH study, was substantially improved in the surgical groups.25 However, studies showed a relatively high level of bias and poor long-term evaluation, so further RCTs are suggested.26 No significant difference in adverse events was seen between surgery and PPI therapy. Importantly, there was no difference between the two groups in the symptom of dysphagia after recovery (a symptom frequently attributed to surgery).
It is important to note that most RCTs were conducted in an environment of full-time and experienced antireflux surgical practice, usually in a university hospital. Excellent results were obtained in 87 to 94% of patients, depending on responses to PPIs.27-29 High-volume surgeons have been reported to achieve superior early outcomes.30 The results, therefore, reflect expert management in a high-volume antireflux surgical service and may not be generalisable to broader surgical practice.
Morbidity and mortality from LARS are low. In a national study of 8947 patients, there was one surgery-related death, and the three-month reoperation rate was 0.4%.31 Morbidity is generally considered to be about 3 to 4%.32 Assessment of quality of life 20 years after total fundoplication in 131 patients found that 82% reported satisfaction.33
Selection of patients for antireflux surgery
Patients who have an inadequate response to continuous acid suppression for control of symptoms, or who have continued complications of reflux disease despite properly administered medication, are potential candidates for LARS, following adequate investigation (Box). Patients with reflux hypersensitivity and functional heartburn are generally not considered for surgery.
Patients requiring a double dose of PPIs are likely to benefit, provided a substantive initial response to PPI therapy has been observed.19 The ICARUS guidelines (international consensus regarding preoperative examinations and clinical characteristics assessment to select adult patients for antireflux surgery) note that patients with a partial response to PPIs, hiatus hernia or oesophagitis of LA grade B or higher and Barrett’s oesophagus can expect a favourable symptomatic outcome from LARS.34
Minimally invasive antireflux surgery is underused, potentially reducing quality of life for a large group of patients.35 There are multiple consensus recommendations for performing endoscopy and manometry before antireflux surgery, and a 24-hour pH impedance reflux study is necessary in all patients without high-grade oesophagitis.34 Such patients can be evaluated by an experienced antireflux service and tested accordingly. Adequate patient selection is paramount, and specialised antireflux surgical groups are generally expert in reflux physiological testing. Various physiological anomalies exist, and personalised management is valuable in achieving optimal outcomes.36
Conclusion
There is a large group of patients with initial symptoms of heartburn and regurgitation (typical reflux) who have continued reflux symptoms that are incompletely managed by medication. These patients are potential candidates for minimally invasive antireflux surgery after adequate physiological investigation. RCTs of medical treatment compared with surgical management support this approach in reducing symptoms and improving quality of life for appropriately selected patients. MT
COMPETING INTERESTS: None.
References
1. Vakil N, van Zanten SV, Kahrilas P, Dent J, Jones R. The Montreal definition and classification of gastroesophageal reflux disease: a global evidence-based consensus. Am J Gastroenterol 2006; 101: 1900-1920; quiz 1943.
2. Harrison C, Henderson JV, Miller G, Britt HC. The prevalence of diagnosed chronic conditions and multimorbidity in Australia: a method for estimating population prevalence from general practice patient encounter data. PLoS One 2017; 12: e0172935.
3. El-Serag HB, Sweet S, Winchester CC, Dent J. Update on the epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut 2014; 63: 871-880.
4. Revicki DA, Wood M, Maton PN, Sorensen S. The impact of gastroesophageal reflux disease on health-related quality of life. Am J Med 1998; 104: 252-258.
5. Boeckxstaens G. The relationship between the acid pocket and GERD. Gastroenterol Hepatol (N Y) 2013; 9: 595-596.
6. Fass R, Boeckxstaens GE, El-Serag H, Rosen R, Sifrim D, Vaezi MF. Gastro-oesophageal reflux disease. Nat Rev Dis Primers 2021; 7: 55.
7. Kahrilas PJ, Lin S, Chen J, Manka M. The effect of hiatus hernia on gastro-oesophageal junction pressure. Gut 1999; 44: 476-482.
8. Ribolsi M, Balestrieri P, Biasutto D, Emerenziani S, Cicala M. Role of mixed reflux and hypomotility with delayed reflux clearance in patients with non-cardiac chest pain. J Neurogastroenterol Motil 2016; 22: 606-612.
9. Naik RD, Meyers MH, Vaezi MF. Treatment of refractory gastroesophageal reflux disease. Gastroenterol Hepatol (N Y) 2020; 16: 196-205.
10. Richter JE. Importance of bile reflux in Barrett’s esophagus. Dig Dis 2000; 18: 208-216.
11. Antunes C, Aleem A, Curtis SA. Gastroesophageal reflux disease (archived). In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025.
12. Dent J, Vakil N, Jones R, et al. Accuracy of the diagnosis of GORD by questionnaire, physicians and a trial of proton pump inhibitor treatment: the Diamond Study. Gut 2010; 59: 714-721.
13. Herregods TV, Troelstra M, Weijenborg PW, Bredenoord AJ, Smout AJ. Patients with refractory reflux symptoms often do not have GERD. Neurogastroenterol Motil 2015; 27: 1267-1273.
14. Fass R, Achem SR. Noncardiac chest pain: epidemiology, natural course and pathogenesis. J Neurogastroenterol Motil 2011; 17: 110-123.
15. Gyawali CP, Fass R. Management of gastroesophageal reflux disease. Gastroenterology 2018; 154: 302-318.
16. Katzka DA, Pandolfino JE, Kahrilas PJ. Phenotypes of gastroesophageal reflux disease: where Rome, Lyon, and Montreal meet. Clin Gastroenterol Hepatol 2020; 18: 767-776.
17. Aziz Q, Fass R, Gyawali CP, Miwa H, Pandolfino JE, Zerbib F. Esophageal disorders. Gastroenterology 2016; 150: 1368-1379.
18. Keung C, Hebbard G. The management of gastro-oesophageal reflux disease. Aust Prescr 2016; 39: 6-10.
19. Zhang H, Yang Z, Ni Z, Shi Y. A meta-analysis and systematic review of the efficacy of twice daily PPIs versus once daily for treatment of gastroesophageal reflux disease. Gastroenterol Res Pract 2017; 2017: 9865963.
20. Labenz J, Armstrong D, Lauritsen K, et al. A randomized comparative study of esomeprazole 40 mg versus pantoprazole 40 mg for healing erosive oesophagitis: the EXPO study. Aliment Pharmacol Ther 2005; 21: 739-746.
21. Castell DO, Kahrilas PJ, Richter JE, et al. Esomeprazole (40 mg) compared with lansoprazole (30 mg) in the treatment of erosive esophagitis. Am J Gastroenterol 2002; 97: 575-583.
22. Richter JE, Kahrilas PJ, Johanson J, et al. Efficacy and safety of esomeprazole compared with omeprazole in GERD patients with erosive esophagitis: a randomized controlled trial. Am J Gastroenterol 2001; 96: 656-665.
23. Ness-Jensen E, Hveem K, El-Serag H, Lagergren J. Lifestyle intervention in gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2016; 14: 175-182.e1-3.
24. Talley NJ, Zand Irani M. Optimal management of severe symptomatic gastroesophageal reflux disease. J Intern Med 2021; 289: 162-178.
25. Tristão LS, Tustumi F, Tavares G, Bernardo WM. Fundoplication versus oral proton pump inhibitors for gastroesophageal reflux disease: a systematic review and meta-analysis of randomized clinical trials. Esophagus 2021; 18: 173-180.
26. Garg SK, Gurusamy KS. Laparoscopic fundoplication surgery versus medical management for gastro‐oesophageal reflux disease (GORD) in adults. Cochrane Database Syst Rev 2015 (11): CD003243.
27. Mainie I, Tutuian R, Agrawal A, Adams D, Castell DO. Combined multichannel intraluminal impedance–pH monitoring to select patients with persistent gastro-oesophageal reflux for laparoscopic Nissen fundoplication. Br J Surg 2006; 93: 1483-1487.
28. Seo HS, Choi M, Son S-Y, Kim MG, Han D-S, Lee HH. Evidence-based practice guideline for surgical treatment of gastroesophageal reflux disease 2018. J Gastric Cancer 2018; 18: 313-327.
29. Lundell L, Bell M, Ruth M. Systematic review: laparoscopic fundoplication for gastroesophageal reflux disease in partial responders to proton pump inhibitors. World J Gastroenterol 2014; 20: 804-813.
30. Schlottmann F, Strassle PD, Patti MG. Antireflux surgery in the USA: influence of surgical volume on perioperative outcomes and costs-time for centralization? World J Surg 2018; 42: 2183-2189.
31. Maret-Ouda J, Yanes M, Konings P, Brusselaers N, Lagergren J. Mortality from laparoscopic antireflux surgery in a nationwide cohort of the working-age population. Br J Surg 2016; 103: 863-870.
32. Pessaux P, Arnaud JP, Ghavami B, et al. Morbidity of laparoscopic fundoplication for gastroesophageal reflux: a retrospective study about 1470 patients. Hepatogastroenterology 2002; 49: 447-450.
33. Kinsey-Trotman S, Bright T, Thompson S, Devitt PG, Watson D. Quality of life following laparoscopic Nissen fundoplication: outcome at 20 years [abstract]. ANZ J Surg 2017; 87 Suppl 1: 141.
34. Pauwels A, Boecxstaens V, Andrews CN, et al. How to select patients for antireflux surgery? The ICARUS guidelines (international consensus regarding preoperative examinations and clinical characteristics assessment to select adult patients for antireflux surgery). Gut 2019; 68: 1928-1941.
35. Thompson SK, Watson DI. We asked the experts: "When is a laparoscopic fundoplication warranted for gastroesophageal reflux disease?". World J Surg 2022; 46: 1711-1712.
36. Hayden J, Jamieson G. Optimization of outcome after laparoscopic antireflux surgery. ANZ J Surg 2006; 76: 258-263.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.