From soil to sepsis: melioidosis, an emerging tropical infectious disease

Melioidosis is an infectious disease caused by the environmental bacterium Burkholderia pseudomallei, and it is endemic to tropical regions of Australia. Even with optimal supportive care, about 10% of Australians with melioidosis will die from their infection.
- Melioidosis, a disease caused by the environmental bacterium Burkholderia pseudomallei, has a case fatality rate in Australia of about 10%.
- Melioidosis primarily affects individuals with comorbidities that include diabetes, hazardous alcohol consumption, chronic lung disease, chronic kidney disease, immunosuppression and malignancy.
- The disease is endemic in tropical Australia and Southeast Asia; however, cases are increasingly being reported in the Pacific, South Asia, Africa and the Americas.
- Melioidosis most commonly presents as pneumonia, but can also affect the skin, musculoskeletal system, abdominal viscera and the CNS.
- Diagnosis is confirmed by culturing B. pseudomallei from clinical specimens. Routine imaging of the chest, abdomen and pelvis is used to assess disease extent.
- Treatment involves an intensive intravenous antibiotic phase followed by a prolonged oral eradication phase. Side effects of this therapy can make adherence to treatment challenging.
- Prevention relies on minimising environmental exposure and optimising the management of predisposing comorbidities, as there is no approved vaccine and chemoprophylaxis has limited utility.
The organism that causes melioidosis, Burkholderia pseudomallei, is an environmental bacterium that lives in soil and surface water in tropical and subtropical regions. Most cases in endemic tropical regions occur during the monsoonal wet season when moist soil provides optimal conditions for the organism’s growth.1
Exposure to B. pseudomallei occurs via percutaneous inoculation, inhalation or ingestion, and many individuals with melioidosis have a history of occupational or recreational exposure to soil or surface water. However, most people who encounter the organism do not develop melioidosis.2 Indeed, about 90% of individuals who are diagnosed with melioidosis have a comorbidity that predisposes them to developing the disease.3 This is most commonly diabetes mellitus, but melioidosis also occurs more frequently in individuals with hazardous alcohol consumption, chronic lung disease, chronic kidney disease, immunosuppression and malignancy.3-5 Many of these comorbidities are seen more often in individuals who are socioeconomically disadvantaged, which is one of the reasons why the incidence of melioidosis is higher in Australia’s Aboriginal and Torres Strait Islander people.6
Global distribution
B. pseudomallei is endemic in tropical Australia and Southeast Asia, but the area of endemicity is expanding, with cases being reported increasingly in the Pacific, South Asia, Africa and the Americas, and in individuals who have travelled to these regions.7,8 The incidence of melioidosis is increasing in Australia and is highest in the Top End of the Northern Territory and tropical North Queensland. Almost 300 cases were reported in Australia in the first five months of 2025. The increase in cases in Australia has been linked to urban expansion, and clustering after severe weather events has also been documented.9-12 The recent appearance of cases in Southeast Queensland has been linked to two climate change-associated La Niña events.13 However, despite the recent increase in disease incidence, most individuals at risk of melioidosis – even those living in endemic regions – have never heard of this life-threatening condition.14
Clinical presentation
About 85% of cases of melioidosis will present acutely and these patients will usually have had symptoms for less than a week, but more subacute presentations (with symptoms present for several months) are also seen. Reactivation from latent infection occurs, and may be precipitated by immunosuppression or concurrent illness, but it is far less frequent than previously believed and represents only about 3% of all cases.15
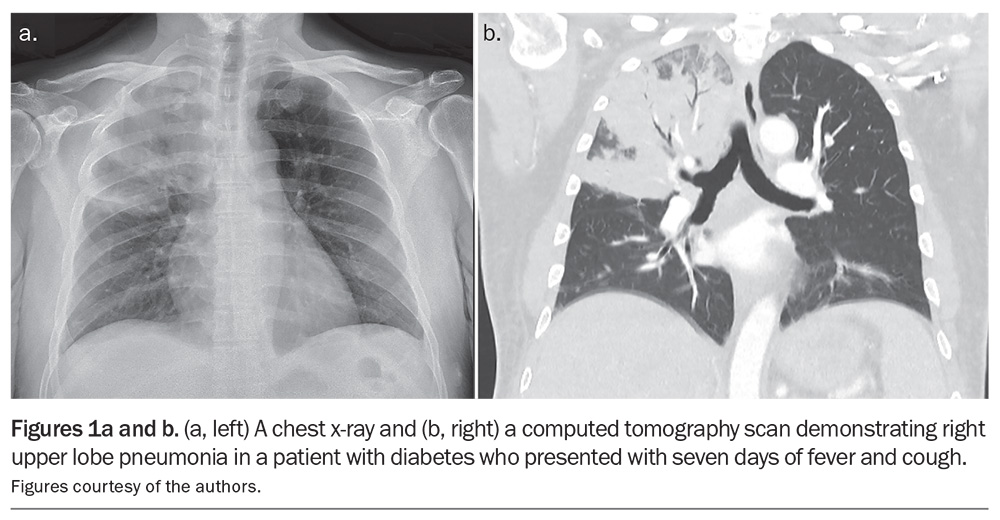
The clinical manifestations of melioidosis are protean and multiple organs are often involved concurrently, but pneumonia is present in most cases (Figure 1 and Figure 2).4,16 Patients with pneumonia usually present with a short history of fever, cough and dyspnoea. However, they may also present with weeks of respiratory symptoms that have not responded to the usual empirical therapies. Bacteraemia is present in up to 70% of cases of melioidosis and about 25% of Australian patients present with septic shock.17,18
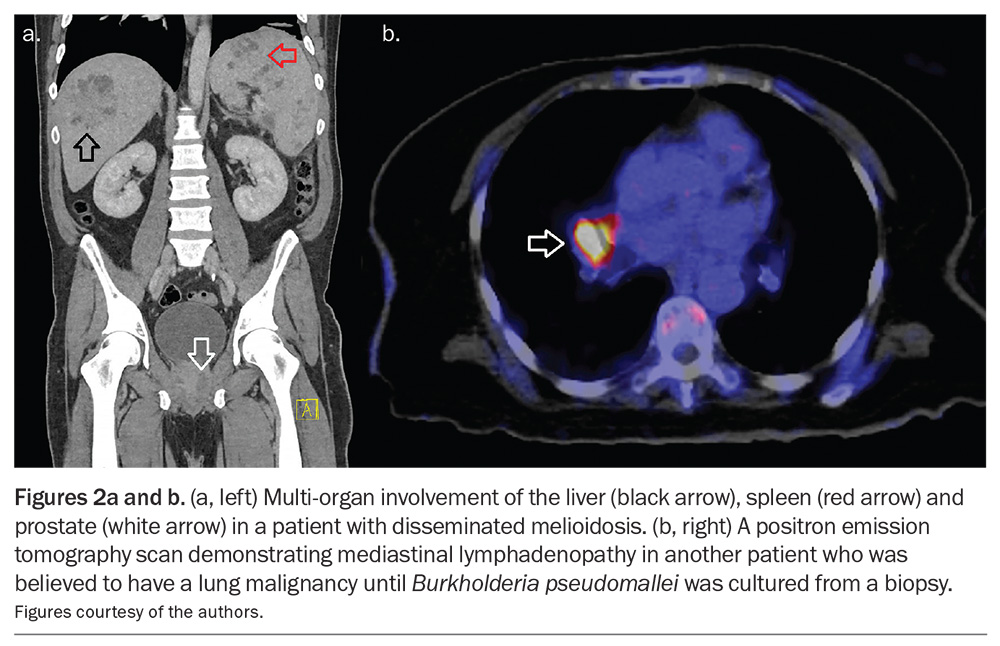
Other typical presentations of melioidosis include skin and soft tissue infections (~15% of cases), infections of the bones and joints (~10% of cases) and abscesses of the liver, spleen or both (~5% of cases). Prostatic involvement, which occurs in about 20% of affected men, is a characteristic manifestation and all men with melioidosis should be screened for this complication (Figure 2).19 CNS involvement occurs in only 4% of cases, but it is one of the most feared manifestations and may present as encephalomyelitis, brain abscess, meningitis or as an extradural collection.20 B. pseudomallei may also cause lymphadenopathy, nodules or masses that may be mistaken for cancer, emphasising the importance of sending biopsies for culture in the appropriate clinical context (Figure 2).21
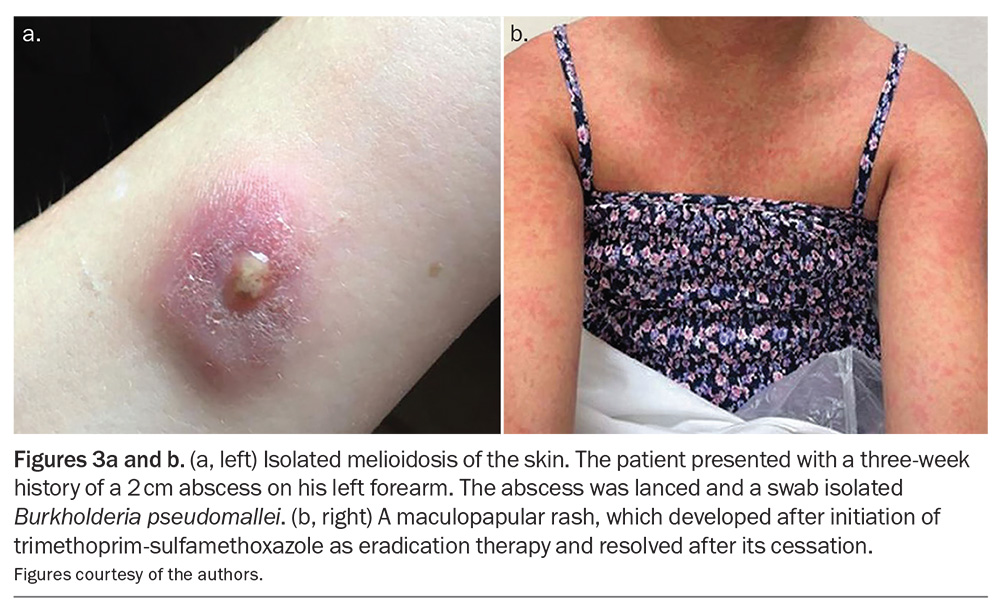
Less than 5% of cases of melioidosis occur in children, which may be explained by the fact that children are less likely to have predisposing comorbidities. Children with melioidosis usually have milder disease, with the most common manifestation being localised skin infection (Figure 3).22 However, disseminated disease can also be seen and fatalities have been reported in previously well children despite optimal supportive care.23 These fatal paediatric cases, and cases in adults without risk factors for melioidosis, may be due to the presence of virulence factors in the organism, a larger inoculum at the time of infection or undiagnosed immune dysfunction in the patient.20,24
Diagnosis
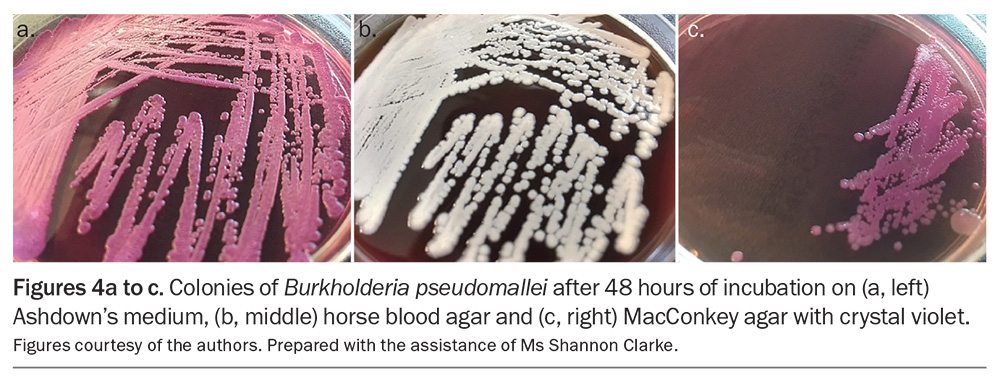
In endemic regions, the diagnosis of melioidosis is often suspected in patients with predisposing comorbidities who present with typical symptoms during the wet season. The diagnosis is confirmed by culturing B. pseudomallei, a Gram-negative rod, from blood, sputum, urine, pus or other clinical specimens; the organism grows well on traditional culture media (Figure 4).25 As multi-organ involvement is common and many patients are bacteraemic, the concurrent collection of multiple clinical specimens will facilitate diagnosis. True colonisation is extremely uncommon, therefore isolation of B. pseudomallei almost always warrants further investigation and treatment. In endemic regions, laboratories are adept at identifying B. pseudomallei; however, laboratories that are unfamiliar with the organism may disregard it inappropriately as an environmental contaminant.26
There is interest in the use of newer diagnostic strategies, including molecular testing, to expedite the diagnosis of melioidosis, but these assays are not yet widely available.27 Serology has limited utility in the diagnosis of the disease as it has poor sensitivity early in the disease course and may be positive in healthy individuals in endemic areas.27
Further diagnostic evaluation
Melioidosis can be rapidly fatal, but person-to-person transmission is extremely unusual, and no special precautions are required for health workers caring for patients with melioidosis. It is essential for clinicians who are unfamiliar with the infection to seek prompt specialist advice.
Almost all cases will require hospitalisation where patients will usually have CT imaging of the chest, abdomen and pelvis to identify the extent of disease.28 This will help define deep tissue collections that may require drainage and determine the duration of antibiotic therapy.29 In patients with CNS involvement or bone and joint involvement, CT imaging results may be normal and MRI is preferred. Computed tomography-positron emission tomography (CT-PET) may also help identify unrecognised foci of disease.30,31 For pregnant women and children, imaging strategies that minimise radiation exposure, including ultrasound, are a reasonable alternative, although these individuals require close follow-up.
Antibiotic treatment
There are two phases of antimicrobial therapy for melioidosis: an initial intravenous intensive phase and a subsequent oral eradication phase, which is prescribed to help prevent relapse. B. pseudomallei is intrinsically resistant to many commonly prescribed antibiotics, so during the initial intensive phase patients receive ceftazidime or meropenem. The duration of intensive therapy varies with the clinical phenotype, although a minimum of two weeks is recommended and source control is essential.29 After initial stabilisation and management in hospital, many patients are treated with outpatient parental antibiotic therapy. This therapy is given via a peripherally inserted central catheter with an elastomeric infuser device.32 This approach is safe, effective and is preferred by many patients.33
Relapse, which occurs in up to 4% of people, is more common in patients who are unable to adhere to prescribed antibiotic therapy and can be fatal.5 Adherence to currently recommended treatment regimens can be challenging: Australian treatment guidelines recommend a minimum of three months of high-dose trimethoprim-sulfamethoxazole (TMP-SMX) during the eradication phase, a phase of care that will frequently be co-managed by the GP.29 Over 50% of patients receiving high-dose TMP-SMX have side effects that necessitate cessation, dose reduction or a switch to alternative agents such as doxycycline or amoxycillin-clavulanate, which are less effective.34 The most common serious side effect from TMP-SMX is acute kidney injury, but fatal cutaneous reactions, including drug reaction with eosinophilia and systemic symptoms or toxic epidermal necrolysis, have also been seen.34
Outcomes and follow-up
Although the case fatality rate of melioidosis has declined significantly in Australia’s well-resourced health system over recent decades, it is still about 10%.4,5 Most individuals with melioidosis will have a comorbidity that has predisposed them to developing the disease. Their diagnosis of melioidosis is therefore an opportunity to identify this comorbidity and optimise its management to improve the individual’s long-term health outcomes.35
Prevention
Strategies to prevent melioidosis largely revolve around advising individuals with predisposing comorbidities to minimise exposure to soil and surface water during the wet season and to stay indoors during heavy rain. However, it is recognised that adherence to these recommendations is challenging, and they have not been shown to be effective in clinical trials.36
There are data to support chemoprophylaxis in selected populations, but the relatively low incidence of melioidosis in even high-risk populations – and the potential side effects of TMP-SMX – means that chemoprophylaxis has a very limited role in combating the disease.37-39 There is presently no approved vaccine for melioidosis, although public health strategies to reduce the incidence of predisposing risk factors – or at least optimise their management – might reduce the risk of melioidosis and the other complications of these comorbidities.16,35
Conclusions
The global incidence of melioidosis is rising but the disease remains under-recognised. This brief overview provides some insights to assist clinicians in identifying and managing patients with this life-threatening condition. The emergence of melioidosis is a further reminder of the complex interplay between host, pathogen and the environment that drives the incidence of many infectious diseases, and the resulting implications for the general population and the health systems that care for them. Some practice points for GPs are listed in the Box. MT
COMPETING INTERESTS: None.
References
1. Fairhead LJ, Smith S, Sim BZ, et al. The seasonality of infections in tropical Far North Queensland, Australia: a 21-year retrospective evaluation of the seasonal patterns of six endemic pathogens. PLOS Glob Public Health 2022; 2: e0000506.
2. Smith S, Marquardt T, Jennison AV, et al. Clinical manifestations and genomic evaluation of melioidosis outbreak among children after sporting event, Australia. Emerg Infect Dis 2023; 29: 2218-2228.
3. Prideaux L, Sandeman M, Stratton H, Kelleher AD, Smith S, Hanson J. Melioidosis in people living with diabetes; clinical presentation, clinical course and implications for patient management. Acta Trop 2025; 263: 107559.
4. Currie BJ, Mayo M, Ward LM, et al. The Darwin Prospective Melioidosis Study: a 30-year prospective, observational investigation. Lancet Infect Dis 2021; 21: 1737-1746.
5. Stewart JD, Smith S, Binotto E, McBride WJ, Currie BJ, Hanson J. The epidemiology and clinical features of melioidosis in Far North Queensland: implications for patient management. PLoS Negl Trop Dis 2017; 11: e0005411.
6. Hanson J, Smith S, Stewart J, Horne P, Ramsamy N. Melioidosis - a disease of socioeconomic disadvantage. PLoS Negl Trop Dis 2021; 15: e0009544.
7. Currie BJ. Melioidosis and Burkholderia pseudomallei: progress in epidemiology, diagnosis, treatment and vaccination. Curr Opin Infect Dis 2022; 35: 517-23.
8. Dan M. Melioidosis in travelers: review of the literature. J Travel Med 2015; 22: 410-414.
9. Kaestli M, Grist EP, Ward L, Hill A, Mayo M, Currie BJ. The association of melioidosis with climatic factors in Darwin, Australia: a 23-year time-series analysis. J Infect 2016; 72: 687-697.
10. Smith S, Horne P, Rubenach S, et al. Increased incidence of melioidosis in Far North Queensland, Queensland, Australia, 1998-2019. Emerg Infect Dis 2021; 27: 3119-3123.
11. Parameswaran U, Baird RW, Ward LM, Currie BJ. Melioidosis at Royal Darwin Hospital in the big 2009-2010 wet season: comparison with the preceding 20 years. Med J Aust 2012; 196: 345-348.
12. Ganeshalingam V, Kaestli M, Norton RE, Gassiep I. The effect of climate on melioidosis incidence in Townsville, Australia: a dry tropical region. Environ Health Prev Med 2023; 28: 33.
13. Gassiep I, Grey V, Thean LJ, et al. Expanding the geographic boundaries of melioidosis in Queensland, Australia. Am J Trop Med Hyg 2023; 108: 1215-1219.
14. Smith S, Buikstra E, Rubenach S, Preston-Thomas A, Hanson J. Limited awareness of melioidosis in high-risk populations despite an increasing incidence of the disease in Far North Queensland, Australia. Am J Trop Med Hyg 2022; 107: 1278-1280.
15. Howes M, Currie BJ. Melioidosis and activation from latency: the "time bomb" has not occurred. Am J Trop Med Hyg 2024; 111: 156-160.
16. Hanson J, Smith S. Melioidosis in northern Australia. Microbiol Aust 2022; 43: 120-124.
17. Salaveria K, Smith S, Liu YH, et al. The applicability of commonly used severity of illness scores to tropical infections in Australia. Am J Trop Med Hyg 2021; 106: 257-267.
18. Stephens DP, Thomas JH, Ward LM, Currie BJ. Melioidosis causing critical illness: a review of 24 years of experience from the Royal Darwin Hospital ICU. Crit Care Med 2016; 44: 1500-1505.
19. Kozlowska J, Smith S, Roberts J, Pridgeon S, Hanson J. Prostatic abscess due to Burkholderia pseudomallei: facilitating diagnosis to optimize management. Am J Trop Med Hyg 2018; 98: 227-230.
20. Gora H, Hasan T, Smith S, et al. Melioidosis of the central nervous system; impact of the bimABm allele on patient presentation and outcome. Clin Infect Dis 2022. Epub 2022.
21. Baker K, Duncan T, Kung S, Smith S, Hanson J. Melioidosis masquerading as malignancy in tropical Australia; lessons for clinicians and implications for clinical management. Acta Trop 2024; 254: 107209.
22. McLeod C, Morris PS, Bauert PA, et al. Clinical presentation and medical management of melioidosis in children: a 24-year prospective study in the Northern Territory of Australia and review of the literature. Clin Infect Dis 2015; 60: 21-26.
23. Young A, Tacon C, Smith S, Reeves B, Wiseman G, Hanson J. Case report: fatal pediatric melioidosis despite optimal intensive care. Am J Trop Med Hyg 2017; 97: 1691-1694.
24. Sarovich DS, Price EP, Webb JR, et al. Variable virulence factors in Burkholderia pseudomallei (melioidosis) associated with human disease. PLoS One 2014; 9: e91682.
25. Fairley L, Smith S, Maisrikrod S, Henning L. Systematic review and meta-analysis of diagnostic tests for diagnosis of melioidosis. Acta Trop 2021; 214: 105784.
26. Meumann EM, Limmathurotsakul D, Dunachie SJ, Wiersinga WJ, Currie BJ. Burkholderia pseudomallei and melioidosis. Nat Rev Microbiol 2024; 22: 155-169.
27. Gassiep I, Ganeshalingam V, Chatfield MD, Harris PNA, Norton RE. Melioidosis: laboratory investigations and association with patient outcomes. Am J Trop Med Hyg 2021; 106: 54-59.
28. Smith S, Hanson J, Currie BJ. Melioidosis: an Australian perspective. Trop Med Infect Dis 2018; 3: 27.
29. Therapeutic Guidelines. Melioidosis. Melbourne: Therapeutic Guidelines Limited; 2025. Available online at: https://www.tg.org.au (accessed June 2025).
30. Bramwell J, Kovaleva N, Morigi JJ, Currie BJ. 18-fluorine-fluorodeoxyglucose positron emission computer tomography imaging in melioidosis: valuable but not essential. Trop Med Infect Dis 2025; 10: 69.
31. Boyle R, Withey G, Smith S, Hanson J. Mycotic aneurysms due to Burkholderia pseudomallei in Far North Queensland, tropical Australia: a case series and review of the literature. Acta Trop 2024; 260: 107480.
32. Sullivan RP, Marshall CS, Anstey NM, Ward L, Currie BJ. 2020 review and revision of the 2015 Darwin melioidosis treatment guideline; paradigm drift not shift. PLoS Negl Trop Dis 2020; 14: e0008659.
33. Dadwal P, Bonner B, Fraser D, et al. Bone and joint infections due to melioidosis; diagnostic and management strategies to optimise outcomes. PLoS Negl Trop Dis 2024; 18: e0012317.
34. Martin GE, Bramwell J, Gadil E, et al. Adverse reactions to trimethoprim/sulfamethoxazole for melioidosis eradication therapy: an evaluation of frequency and risk factors. Int J Infect Dis 2025; 150: 107283.
35. Hanson J, Smith S. High rates of premature and potentially preventable death among patients surviving melioidosis in tropical Australia. Am J Trop Med Hyg 2019; 101: 328-331.
36. Suntornsut P, Teparrukkul P, Wongsuvan G, et al. Effectiveness of a multifaceted prevention programme for melioidosis in diabetics (PREMEL): a stepped-wedge cluster-randomised controlled trial. PLoS Negl Trop Dis 2021; 15: e0009060.
37. Majoni SW, Hughes JT, Heron B, Currie BJ. Trimethoprim+sulfamethoxazole reduces rates of melioidosis in high-risk hemodialysis patients. Kidney Int Rep 2018; 3: 160-167.
38. Chau KWT, Smith S, Kang K, Dheda S, Hanson J. Antibiotic prophylaxis for melioidosis in patients receiving hemodialysis in the tropics? One size does not fit all. Am J Trop Med Hyg 2018; 99: 597-600.
39. Shukla T, Smith S, Johnstone K, Donald P, Hanson J. The characteristics and clinical course of patients with melioidosis and cancer. PLoS Negl Trop Dis 2024; 18: e0012631.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.