Relugolix, estradiol and norethisterone: a new therapeutic option for endometriosis

Endometriosis is a prevalent and debilitating condition, for which there are relatively few PBS-listed treatment options available. The combination of relugolix, estradiol and norethisterone acetate has recently been approved and PBS listed for use as a second-line option for endometriosis. Initial long-term data support its use for symptomatic relief, with a favourable side effect profile and convenient administration.
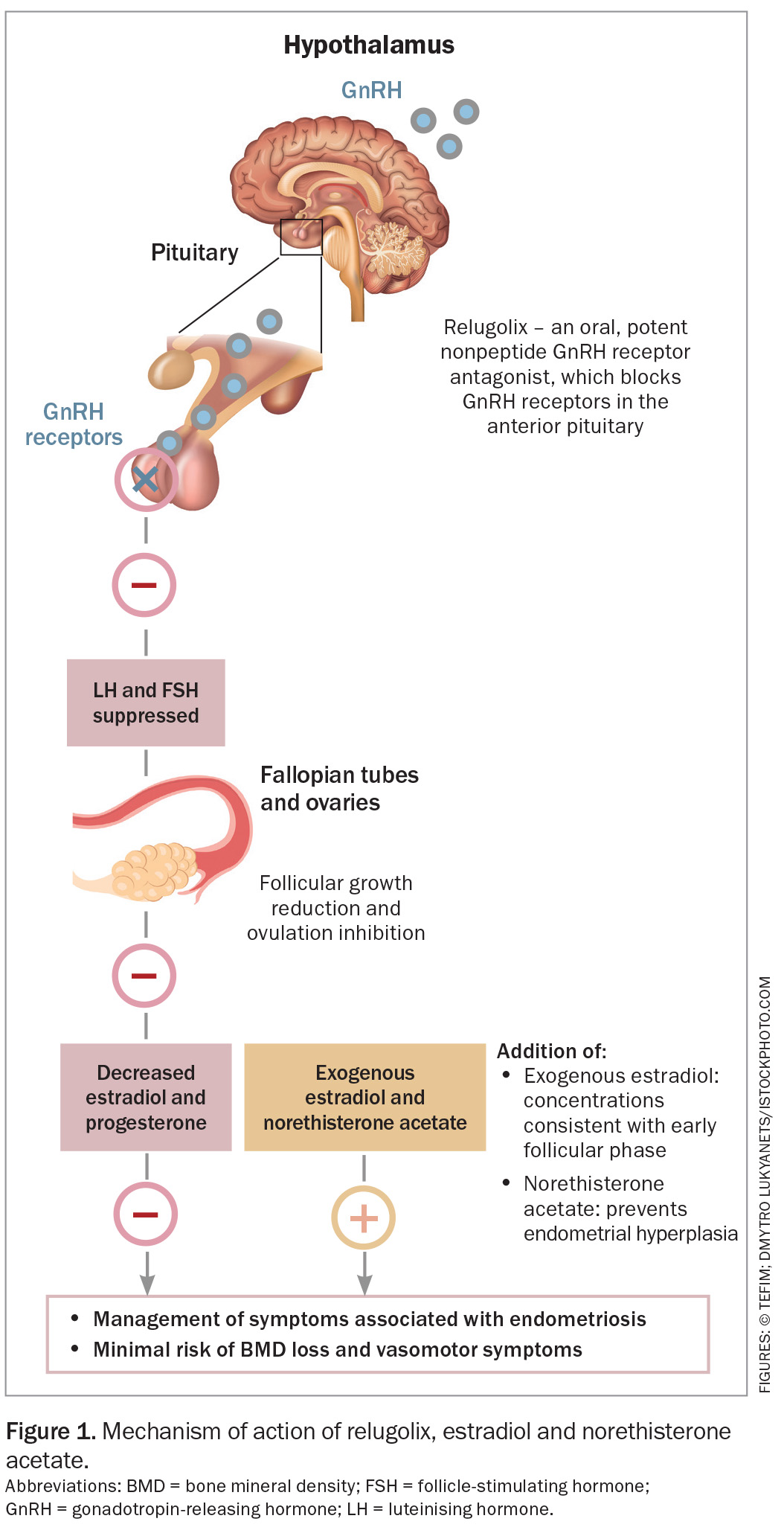
- The combination of relugolix, estradiol and norethisterone acetate contains a gonadotropin-releasing hormone antagonist to block endogenous hormone production and ovulation, with add-back oestrogen and progesterone to minimise menopausal symptoms and bone mineral density loss.
- The medication is PBS listed for women at least 18 years of age with visually confirmed endometriosis (via ultrasound, MRI or surgery) who have had an inadequate response to hormonal contraceptives or analgesics.
- The medication has a favourable side effect profile and is effective for dysmenorrhea and nonmenstrual pelvic pain. It is an adequate contraceptive when used for at least one month.
- Use for up to two years is associated with a loss in bone mineral density of less than 1%.
Endometriosis is a chronic, often debilitating condition marked by the presence of endometrial-like tissue outside the uterus. In Australia, endometriosis affects about one in seven women (including those born female) of reproductive age.1 The condition frequently presents with pelvic pain, dysmenorrhoea, dyspareunia, fatigue and infertility symptoms, which can be persistent or recurrent despite surgical or medical management.2 The diagnosis and management of endometriosis can be delayed resulting in an adverse impact on quality of life.
Endometriosis is a complex condition frequently associated with persistent pelvic pain, involving a multifaceted interplay between pelvic floor muscle sensitisation, bowel function and pain pathways, requiring a multidisciplinary team approach to care. These factors result in an estimated annual cost of $9.6 billion in terms of loss of productivity, loss of income, sick leave and missed opportunities in Australia.3 Laparoscopy was previously regarded as the gold standard for diagnosis and management, but the condition is now increasingly diagnosed with noninvasive techniques, such as ultrasound (specifically a deep infiltrating endometriosis scan) and MRI.4,5
In 2025, the Royal Australian and New Zealand College of Obstetricians (RANZCOG) published the Australian Living Evidence Guideline: Endometriosis.6 This guideline, commissioned by the Australian Government, aims to inform clinicians and the public about the emerging evidence around endometriosis relevant to the Australian context and to identify areas where further research is needed. As part of this process, RANZCOG is also tasked with developing a dissemination and implementation plan for both primary and secondary care.
Medical management remains the cornerstone of long-term treatment and prevention of recurrence. In this guideline (based on low certainty of evidence), the first-line hormonal treatments recommended for women with suspected or diagnosed endometriosis not trying to conceive are combined oral contraceptives and progestogens (oral formulations, injections, implants or intrauterine devices [IUDs]). These all yield similar rates of pain improvement. If first-line hormonal treatment does not improve the symptoms or there are side effects, consider referring the patient to gynaecology services for the consideration of second-line treatments, such as gonadotropin-releasing hormone (GnRH) agonists or antagonists. A strong recommendation when using these medications is to provide add-back hormonal therapy to prevent bone loss and menopausal symptoms.
The new combination of relugolix, estradiol and norethisterone acetate, a once-daily oral therapy containing a GnRH antagonist with add-back therapy in a single pill, has emerged as a promising option and has been listed on the PBS as of May 2025. Although the combination is also approved internationally for heavy menstrual bleeding due to uterine fibroids, it is not currently PBS listed for this indication in Australia, and this indication will not be addressed in this review.
What is relugolix, estradiol and norethisterone acetate?
The combination of relugolix, estradiol and norethisterone acetate is a fixed-dose therapy comprising:
- relugolix 40 mg: a nonpeptide GnRH receptor antagonist
- estradiol 1 mg: synthetic oestrogen
- norethisterone acetate 0.5 mg: synthetic progestogen.
Relugolix suppresses the pituitary release of luteinising hormone and follicle-stimulating hormone, reducing the ovarian production of oestrogen and progesterone, and preventing ovulation. The estradiol serves as add-back therapy, designed to counteract hypoestrogenic side effects such as vasomotor symptoms and bone mineral density (BMD) loss. Norethisterone acetate serves to prevent endometrial hyperplasia and endometrial cancer associated with continuous exogenous oestrogen therapy. With add-back therapy, systemic oestradiol concentrations are consistently maintained at a level equivalent to the early follicular phase of a natural menstrual cycle (Figure 1).7
PBS listing
As of 1 May 2025, the medication is listed on the PBS as a second-line treatment for endometriosis, following prior medical or surgical management. The cost has been reduced from $215 to $31 per month for eligible patients. Initiation is restricted to specialists with experience in the diagnosis and management of endometriosis (e.g. gynaecologists or women’s health GPs), and GPs may continue prescribing for maintenance therapy. Endometriosis must be visually proven via imaging (ultrasound or MRI) or surgery, with or without biopsy, before prescription of the drug, and the patient must be at least three months post-excisional surgery.
How does the medication fit among treatment options for endometriosis in Australia?
The combination of relugolix, estradiol and norethisterone acetate should be considered second-line after failure of the combined oral contraceptive pill or IUD. Although other options for endometriosis treatment are available on the PBS, this combination is the only option that provides GnRH blockade and hormonal add-back therapy in a single tablet formulation, making it cheaper and more convenient than other options. Other PBS-listed second-line options include goserelin, nafarelin and dienogest.
Goserelin is a monthly injection that can be used for a maximum of six months on the PBS and is recommended to be administered with hormonal add-back therapy to prevent menopausal hot flushes, vulvovaginal dryness and BMD loss.8 A three-monthly injection is available; however, it is not PBS listed for endometriosis, and thus, patients need to visit their GP or specialist monthly for injections.
Dienogest is a daily progesterone-only tablet that has been shown to be noninferior to the combined oral contraceptive pill for endometriosis-associated pain and pain after laparoscopic excision of endometriosis.9,10 However, dienogest may reduce BMD by 2 to 4% and up to 55% of patients report abnormal uterine bleeding while taking the medication.11
Neither goserelin nor dienogest are reliable forms of contraception, and alternative nonhormonal contraceptive methods are required concurrently.
Efficacy
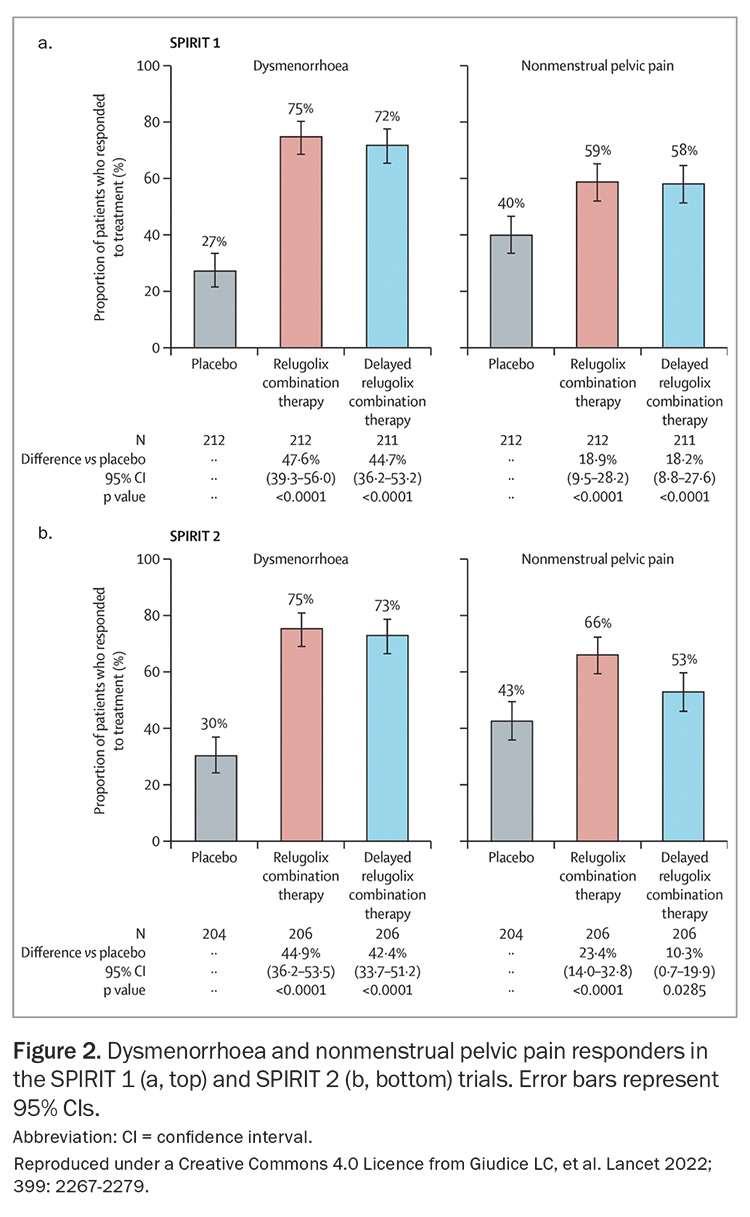
The pivotal SPIRIT 1 and SPIRIT 2 trials were double-blind, placebo-controlled trials that enrolled more than 1200 premenopausal women internationally with moderate-to-severe endometriosis-associated pain.12 Key findings for the combination of relugolix, estradiol and norethisterone acetate compared with placebo included:
- a significant reduction in dysmenorrhoea and nonmenstrual pelvic pain at 24 weeks (Figure 2):
– for dysmenorrhea, 75% of patients treated with the combination ‘responded’ (reduction in average 0–10 pain scores of >2.8) compared with 27 to 30% with placebo
– for nonmenstrual pelvic pain, 59 to 66% of patients treated with the combination ‘responded’ (reduction in average 0–10 pain scores of >2.1) compared with 40 to 43% with placebo - a reduction in analgesic use and improvement in patient-reported quality of life scores (e.g. Endometriosis Health Profile-30)
- sustained benefits documented over two years in extension studies.13
There are currently inadequate data to comment on the medication’s effect on endometriosis recurrence or head-to-head performance against other treatment options.
Safety and precautions
The combination of relugolix, estradiol and norethisterone acetate is generally well tolerated but requires thoughtful patient selection. Eight percent of study participants reportedly discontinued the medication due to the onset of adverse events, with the most common reason being headache. The risk of thromboembolic disorders, mood disorders and hypertension was not increased in trials up to two years, although these studies were not designed for this purpose.13 Any contraindications are generally associated with the administration of exogenous oestrogen and the potential for a small (<1%) reduction in BMD during use.
Contraindications
The medication is contraindicated in women with:
- a history of thromboembolism
- known thrombophilia
- migraine with aura
- severe liver disease
- hormone-sensitive malignancies
- galactose intolerance, total lactase deficiency or glucose–galactose malabsorption
- osteoporosis
- pregnancy or who are breastfeeding.
- Common side effects
- The following are some common side effects associated with the medication:
- headache
- nasopharyngitis
- hot flush or hyperhidrosis
- nausea
- mood changes
- changes in libido or vulvovaginal dryness
- abnormal uterine bleeding
- back pain or arthralgia.
Bone health
Although add-back therapy mitigates BMD loss, relugolix still poses a risk in vulnerable populations. Trial data indicate that patients experience a small reduction in BMD initially (<1%), which stabilises over two years of administration.13 Data beyond two years of use do not exist. There are inadequate data to determine whether this bone loss is reversible after cessation of the drug, and these data are unlikely to be available until the completion of phase 3B trials in 2030 (clinical trial number: NCT05862272). For this reason, DEXA scanning is recommended at baseline for all patients and annually thereafter during long-term treatment.
Drug interactions
Relugolix may interact with CYP3A4 and P-glycoprotein modulators, necessitating caution when coadministered with:
- P-glycoprotein inhibitors (potentially increased exposure to relugolix), such as:
– azithromycin
– clarithromycin
– tetracycline
– itraconazole (can be used with fluconazole)14 - CYP3A4 inducers (potentially reduced exposure to relugolix), such as:
– carbamazepine
– phenytoin
– HIV and hepatitis C virus protease inhibitors.
The full details on drug interactions are available in the official TGA Product Information.
Contraceptive properties
The combination of relugolix, estradiol and norethisterone acetate provides contraceptive efficacy after one month of use. Hormonal contraceptives must be ceased before initiation, and nonhormonal contraceptive methods used during the initial month. If doses are missed on two or more consecutive days, barrier contraception should be used for the next seven days of treatment. As the medication induces amenorrhoea in most patients, it may be difficult to recognise an inadvertent pregnancy.15 Patients should be informed of this risk and have a low threshold for completing a pregnancy test if unsure. Patients should expect to rapidly and predictably ovulate after cessation of the medication, with more than 95% of patients ovulating in the first month after cessation.7
Role of the GP and monitoring
GPs are vital in supporting the continuity of care through symptom monitoring, side effect management and patient education. Ongoing monitoring includes:
- symptom relief and adverse effects
- addressing modifiable risk factors for osteoporosis (e.g. smoking, sedentary lifestyle)
- arranging an annual dual x-ray absorptiometry bone scan
- ensuring that the medication is not continued beyond two years without consultation with a specialist.
- Patient education includes ensuring that the patient is aware of:
- expected disruptions to the menstrual cycle causing amenorrhea or irregular bleeding
- contraceptive coverage and timing.
Conclusion
The combination of relugolix, estradiol and norethisterone acetate expands the therapeutic landscape for clinicians managing endometriosis. Its convenience, inclusion of hormonal add-back therapy and PBS subsidy make it a welcome alternative for patients needing medium-term symptom relief. With appropriate monitoring and collaborative care between GPs and non-GP specialists, the medication has the potential to improve outcomes in a condition often underdiagnosed and undertreated. MT
COMPETING INTERESTS: None.
This article is for general information purposes only, and the full Product Information should be consulted before prescribing any of the mentioned medications.
References
1. Australian Institute of Health and Welfare (AIHW). Endometriosis. Canberra: AIHW; 2025. Available online at: https://www.aihw.gov.au/reports/chronic-disease/endometriosis/contents/how-common-is-endometriosis (accessed October 2025).
2. Horne AW, Missmer SA. Pathophysiology, diagnosis, and management of endometriosis. BMJ 2022; 379: e070750.
3. Armour M, Avery J, Leonardi M, et al. Lessons from implementing the Australian National Action Plan for Endometriosis. Reprod Fertil 2022; 3: C29-C39.
4. Avery JC, Deslandes A, Freger SM, et al. Noninvasive diagnostic imaging for endometriosis part 1: a systematic review of recent developments in ultrasound, combination imaging, and artificial intelligence. Fertil Steril 2024; 121: 164-188.
5. Avery JC, Knox S, Deslandes A, et al. Noninvasive diagnostic imaging for endometriosis part 2: a systematic review of recent developments in magnetic resonance imaging, nuclear medicine and computed tomography. Fertil Steril 2024; 121: 189-211.
6. The Royal Australian and New Zealand College of Obstetricians and Gynaecologists (RANZCOG). Australian Living Evidence Guideline: Endometriosis. Melbourne: RANZCOG; 2025. Available online at: https://ranzcog.edu.au/womens-health/endometriosis/ (accessed October 2025).
7. Duijkers IJM, Klipping C, Draeger C, et al. Inhibition of ovulation and pharmacologic mechanism of action of relugolix combination therapy. Fertil Steril 2025; 124: S0015-0282.
8. Alonso A, Gunther K, Maheux-Lacroix S, Abbott J. Medical management of endometriosis. Curr Opin Obstet Gynecol 2024; 36: 353-361.
9. El Taha L, Abu Musa A, Khalifeh D, Khalil A, Abbasi S, Nassif J. Efficacy of dienogest vs combined oral contraceptive on pain associated with endometriosis: randomized clinical trial. Eur J Obstet Gynecol Reprod Biol 2021; 267: 205-212.
10. Mehdizadeh Kashi A, Niakan G, Ebrahimpour M, et al. A randomized, double-blind, placebo-controlled pilot study of the comparative effects of dienogest and the combined oral contraceptive pill in women with endometriosis. Int J Gynaecol Obstet 2022; 156: 124-132.
11. Li RR, Xi Q, Tao L, Sheng W, Zhao CC, Wu YJ. A systematic review and Bayesian analysis of the adverse effects of dienogest. BMC Pharmacol Toxicol 2024; 25: 43.
12. Giudice LC, As-Sanie S, Arjona Ferreira JC, et al. Once daily oral relugolix combination therapy versus placebo in patients with endometriosis-associated pain: two replicate phase 3, randomised, double-blind, studies (SPIRIT 1 and 2). Lancet 2022; 399: 2267-2279.
13. Becker CM, Johnson NP, As-Sanie S, et al. Two-year efficacy and safety of relugolix combination therapy in women with endometriosis-associated pain: SPIRIT open-label extension study. Hum Reprod 2024; 39: 526-537.
14. Wang E-J, Lew K, Casciano CN, Clement RP, Johnson WW. Interaction of common azole antifungals with P glycoprotein. Antimicrob Agents Chemother 2002; 46: 160-165.
15. Al-Hendy A, Lukes AS, Poindexter AN, et al. Long-term relugolix combination therapy for symptomatic uterine leiomyomas. Obstet Gynecol 2022; 140: 920-930.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.