The physical health of patients with mental illness – complex interactions require complex interventions
The physical health of people with schizophrenia can be compromised due to a dynamic interplay between numerous additive risk factors, which result in considerable morbidity and mortality. Importantly, many of these risks are readily modifiable in a general practice setting through the provision of patient health education, thorough and regular screening, early intervention and directed treatment.
- Schizophrenia significantly increases the risk of comorbid physical health conditions, particularly cardiovascular disease and diabetes, leading to significantly elevated risks of morbidity and early mortality.
- Iatrogenic factors, such as the metabolic side effects of antipsychotic medications, further complicate the physical health of individuals with schizophrenia, increasing the risk of metabolic syndrome and related diseases.
- GPs play an important role in the assessment and management of both the mental and physical health of people with schizophrenia through early intervention, patient education and appropriate on-referral.
- Barriers to physical health care for people with schizophrenia, including social isolation, stigma and difficulty accessing health care, contribute to delayed diagnoses and care disruption.
- Collaboration between GPs, psychiatrists and other healthcare providers, along with the use of shared care models, can improve the overall health and quality of life for people with schizophrenia.
Schizophrenia can be a complex condition to manage in general practice. A practitioner could be forgiven for assuming this complexity results specifically from the psychiatric symptomology of the illness: paranoia, delusional misinterpretation or hallucinations as drivers of unpredictability or hostility. However, in actuality, the complexity is often derived from less visible factors: the physical rather than the mental health concerns. It also finds seat in numerous system factors with which both the patient and practitioner must contend. This article outlines the common physical health issues that a person with schizophrenia might face, and provides an up-to-date synopsis of current approaches to the care of the physical health of people with schizophrenia.
Up to 90% of people with schizophrenia have a comorbid chronic physical illness.1 Life expectancy is reduced, on average, by 18.7 years in men and 16.3 years in women.2 Although the risk of death by suicide, accident and misadventure is elevated in this group, the majority of the disparity in longevity is accounted for by physical health conditions, in particular cardiovascular and oncological diseases.3
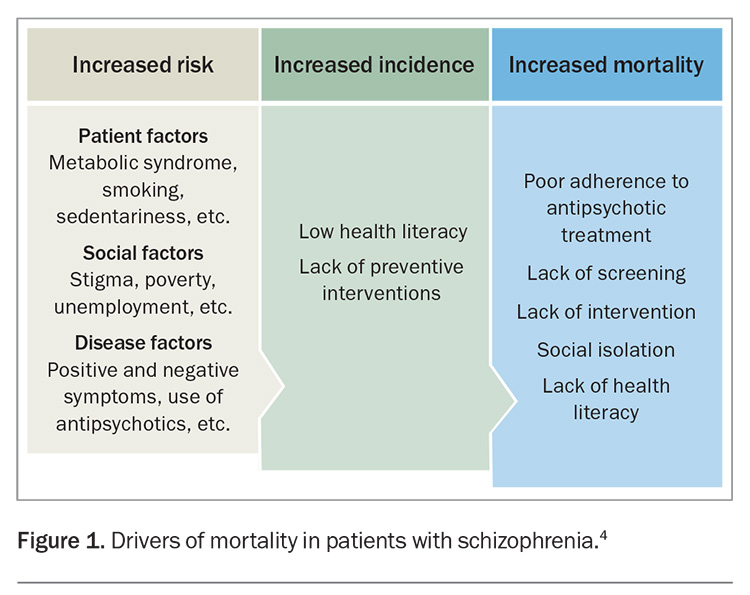
The dynamics that drive compromised physical health and mortality in people with schizophrenia are outlined in Figure 1.4 Factors associated with the core symptoms of the disorder result directly and indirectly in numerous additive risk factors including reduced activity, addiction, dietary imbalances and social isolation. They are also further compounded by a reduced propensity to seek care, difficulty communicating concerns, decreased ability to follow instructions and breakdown of natural supports.
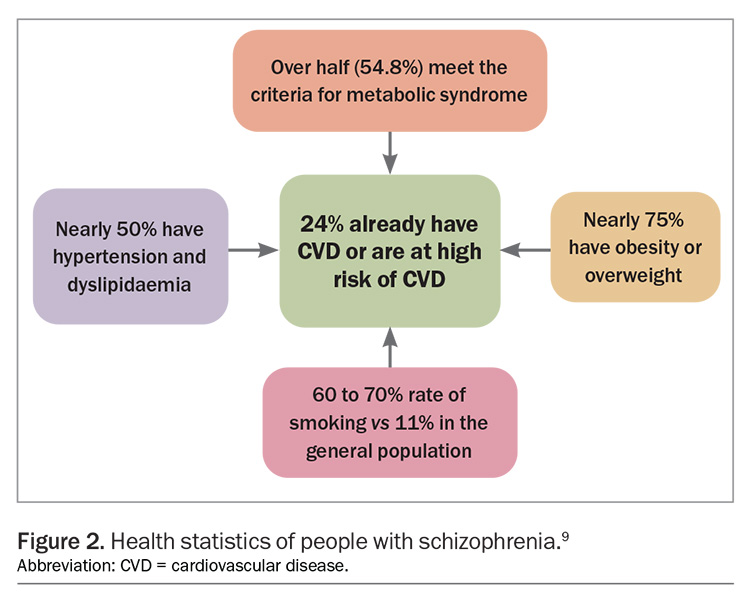
Alone, these risk factors represent adequate cause for concern and increased vigilance. However, aetiological models of schizophrenia reveal that there is substantial overlap with physical health issues, including abnormal fatty acid metabolism, generalised inflammation and mitochondrial dysfunction, and genetic predispositions to diabetes.5 Indeed, high insulin concentrations and low leptin concentrations are often present in the early stages of psychosis, even before antipsychotic treatment has started.6 As such, people with schizophrenia are two to five times more likely to develop type 2 diabetes.7 This being said, numerous modifiable risk factors represent useful targets for intervention, as outlined in Figure 2.8,9
It is also important to recognise iatrogenic drivers. Antipsychotics, mood stabilisers and some antidepressant medications can have metabolic implications. Second-generation antipsychotics (particularly olanzapine, clozapine and quetiapine) predispose an individual to metabolic syndrome, potentially linked to insulin resistance and dysregulated glucose metabolism.10 Sedating medications and substances of abuse further elevate the underlying risk of obstructive sleep apnoea.11
Concerningly, the overall cancer mortality rates are 40% higher in men and 20% higher in women with a mental illness, with the highest rates being for lung cancer in men and breast cancer in women.12,13 The causes for this are complex, but can largely be explained by the higher prevalence of smoking, substance abuse and chronic hepatitis B or C infections.14,15 In schizophrenia, this is likely exacerbated by low rates of help-seeking and personal advocacy, and by clinicians being more focused on mental state than physical health. Barriers to accessing health care (including discrimination, lack of integration of mental health and oncological services) and patient factors (including positive and negative symptomatology) can result in care disruption and, ultimately, delayed diagnosis and treatment.4 Further potential physical health concerns are outlined in Box 1.16-21
The impact of medications
Beyond the metabolic side effects outlined above, medications used to treat schizophrenia impart other adverse effects that affect quality of life and health.
Hyperprolactinaemia
Hyperprolactinaemia occurs secondary to dopaminergic blockade in the posterior pituitary, and its most significant elevations are seen with risperidone, paliperidone and amisulpride. Up to 70% of individuals on antipsychotic treatment have elevated prolactin concentrations, depending on the agent, with females more adversely affected than males.22 This has implications for sexual function and can result in gynaecomastia and galactorrhoea, as well as impacting bone health adversely.
Cardiac health
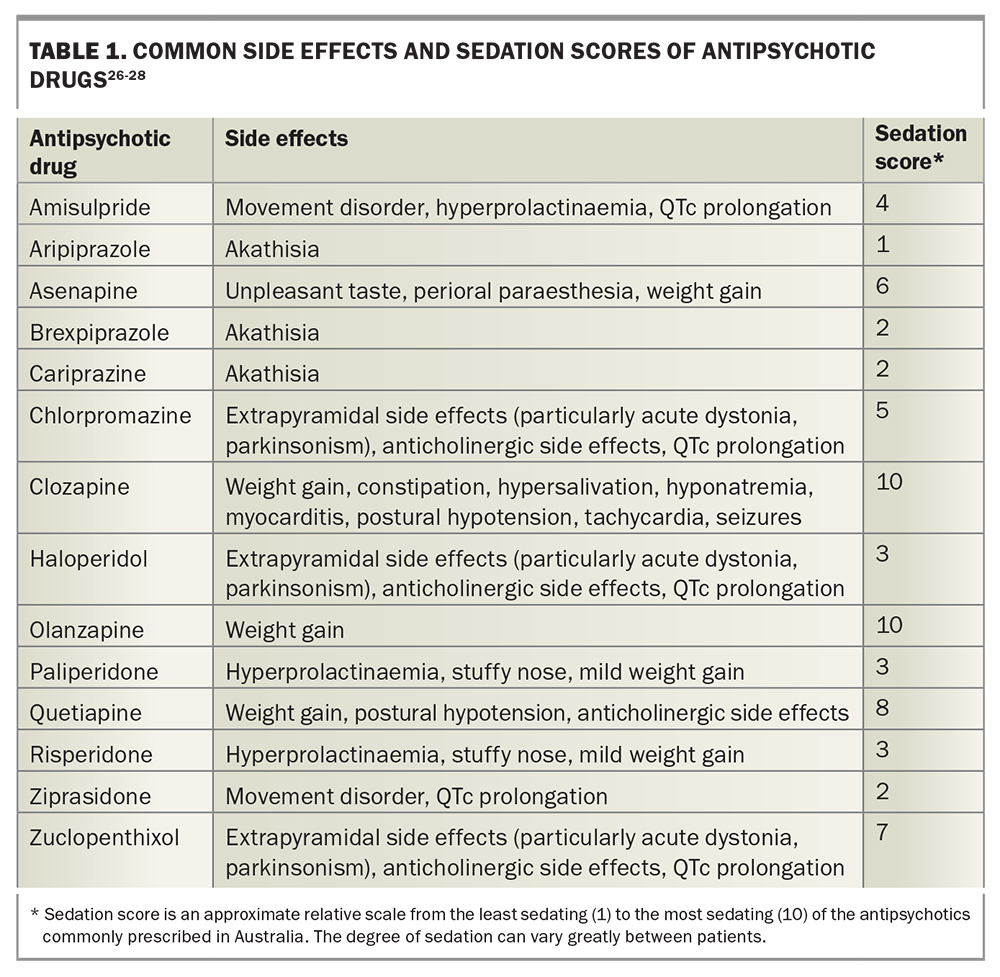
Myocarditis and cardiomyopathy have been shown to be associated with some antipsychotic medications, most commonly with clozapine.23,24 QTc prolongation is not usually a major concern with antipsychotics, apart from if high doses are used or if the patient has a baseline long QTc. Ziprasidone and amisulpride are the agents most strongly associated with QT prolongation, with a three- to sixfold increased odds ratio.25 Torsade de pointes can eventuate, and baseline and follow-up ECGs should be performed for any high-risk individuals, and especially for those in whom high doses of typical antipsychotics, ziprasidone or amisulpride are prescribed. A summary of other medication-specific issues are summarised in Table 1.26-28
The role of the GP
Assessment
Despite many barriers to care, roughly 80% of people in Australia with a psychotic disorder visit their GP in any 12-month period.29 General practice thus represents a vital setting for the provision of physical health care to individuals with schizophrenia. Given the short appointment times and limited availability, practitioners may, understandably, prioritise more apparent psychiatric issues over underlying physical sequelae. Assessment of an individual with schizophrenia should not differ fundamentally from the assessment of any other person. Although it is important to review current psychiatric treatments, medications and relevant side effects, the focus should remain on considering a person’s overall health.
Despite an evident need, people with schizophrenia experience lower rates of screening for risk factors of physical health problems, compared with the general population. This includes screening for tobacco use, despite its particularly high prevalence among people with serious mental illness.30 Further, compared with the general population, there is a 0.37-fold decrease in antihypertensive use and a 0.47-fold decrease in lipid-lowering drug use among people with schizophrenia.31
Examination and monitoring
History-taking should include a detailed account of substance use and sexual health, as well as any past history of hepatitis B and C immunisation and treatment. Often-forgotten elements include a detailed account of oral health, sleep hygiene and bowel habits, particularly in the context of long-term antipsychotic prescribing. A family and social history are also of particular importance, and involving next-of-kin in consultations can provide additional benefit.
Physical examination, including cardiovascular and respiratory assessments, is always relevant, particularly for people taking clozapine and other agents with a metabolic risk profile. Targeted neurological examination for those prescribed antipsychotics, particularly first-generation agents, can help identify extrapyramidal side effects that the patient may not be aware of or may not report. Regular observations of blood pressure, pulse and body weight are mandatory. ECGs should be considered for any individual on antipsychotics, particularly those that might prolong the QTc.
Investigations beyond routine metabolic screening should include liver function testing, both to monitor for fatty liver disease and any medication side effects. Nutritional status is also relevant, including vitamin B12 and D, as well as folate and iron studies. Targeted cancer screening should be considered based on history and examination findings; lung, prostate and bowel cancer screening should be prioritised, including the consideration of faecal occult blood testing. Breast cancer screening should be targeted particularly to those with elevated levels of prolactin, given the associated risk.32
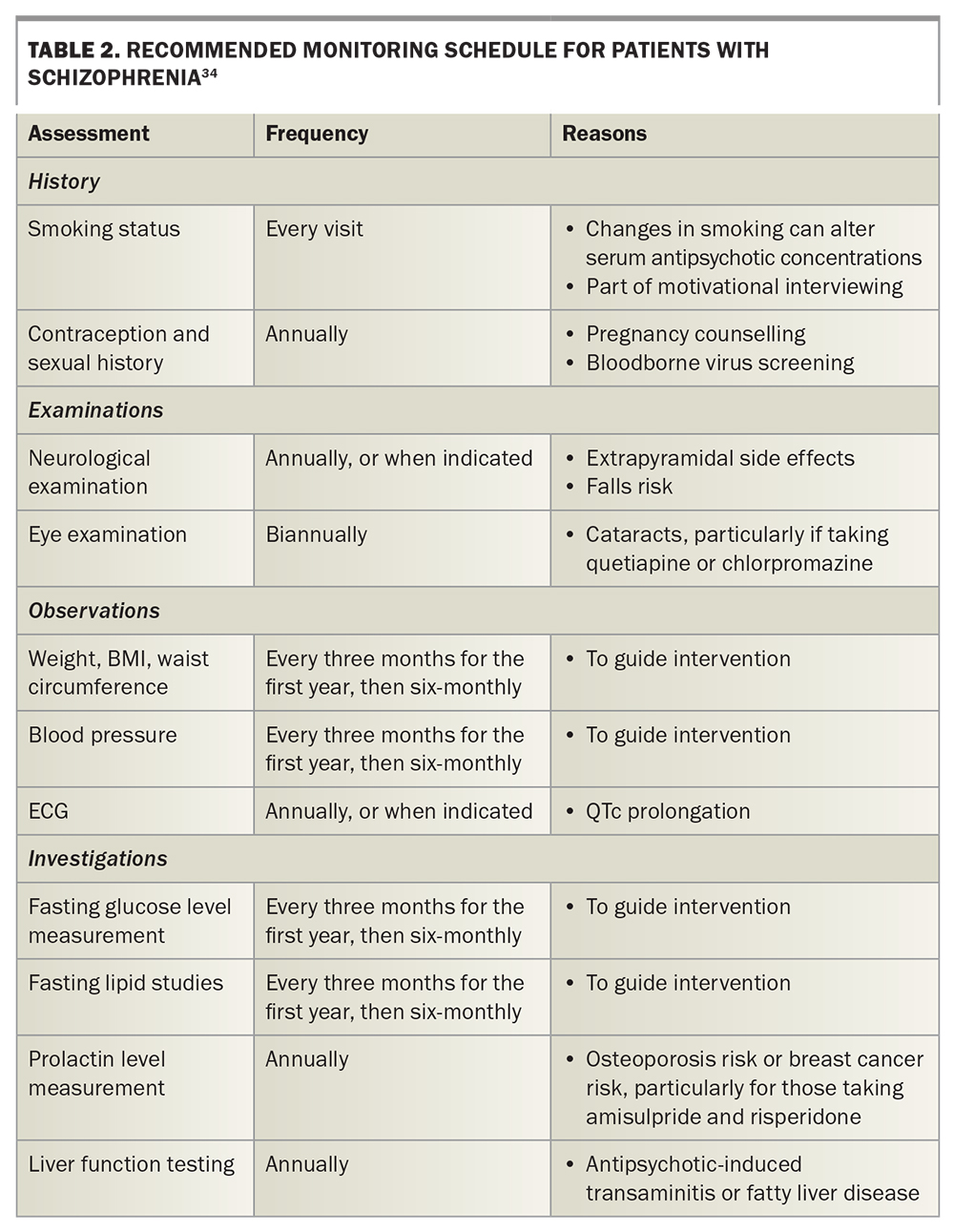
An appropriate schedule for monitoring physical health issues relevant to people with schizophrenia is outlined in Table 2.33 Where possible, this schedule should be adhered to in its entirety but may need to be targeted or modified based on the patient’s tolerance and frequency of engagement. For further detail, the reader is referred to the Royal Australian and New Zealand College of Psychiatrists expert consensus statement.34
Monitoring of tobacco smoking and nicotine intake, particularly sudden cessation, is important, as these can affect the serum concentration of various psychotropic agents, notably clozapine and olanzapine.35 Monitoring of drug levels is appropriate in this context.
Intervention
Nonpharmacological interventions
For metabolic health, the most effective interventions are consistently shown to be individual lifestyle counselling, exercise and dietary interventions, along with psychoeducation.36 These are all interventions that can be delivered effectively in the general practice setting. Aspects such as motivational interviewing and cognitive behavioural therapy techniques have also been shown to enhance the effectiveness of these general strategies.37 This is particularly pertinent, as general public health campaigns seem less effective in influencing the lifestyle choices of people living with a psychotic disorder.38
Focused education is of particular importance in the management of diabetes, particularly insulin-dependent disease. It has been shown that people with schizophrenia are more commonly unaware of their diabetes diagnosis, and some lack the skills and understanding to manage their blood glucose levels effectively.39
Opportunistic and regular exploration and education within GP consultations can be useful for a variety of other potential health issues. Managing cancer risk may be as simple as a brief exploration of sexual safety, sun safety, immunisation status and contraceptive usage. Recommending and providing immunisations, in particular for bloodborne viruses, and other preventive interventions can provide great benefit in this population. Discussing contraception with women of childbearing age is especially relevant for those on medications with known teratogenicity, such as lithium and sodium valproate. It may be relevant to explore the role of the combined oral contraceptive for some individuals, as exogenous oestrogen can enhance the mental health of some women with schizophrenia, albeit any such intervention requires a full risk–benefit analysis.40 If oestrogens are contraindicated, progesterone intrauterine devices or progesterone implants may be preferred.
Oral health is often compromised in people with schizophrenia. General advice includes regular brushing, using dental floss and using gum or alcohol-free mouthwash to reduce dry mouth.26
Pharmacological interventions
The use of pharmacological interventions for the treatment of physical illness in people with schizophrenia is not significantly different to those that pertain to the general population. However, certain patient factors, such as memory, thought and behavioural disorganisation, and alternative illness explanations, appear to affect patient adherence to treatment. Generally speaking, cut-offs for intervention for metabolic illnesses and risk factors are no different to those in the general population. These are summarised in Figure 3.41 Treatment also does not differ significantly and should be provided according to current guidelines for the general population.
That being said, more assertive intervention might be indicated in some cases, particularly if the metabolic impact of psychotropic medications is seen to be rapid. If reductions or alterations to treatment regimens are impossible or ineffective, the addition of topiramate or aripiprazole as a means to counteract these effects has good evidence of efficacy.36 Such additions should be made only with the input of the treating psychiatrist. There is an increasing movement towards the addition of antidiabetic medications as a first-line option in these cases. Although these are used off-label for this purpose, metformin has proven efficacy for modest weight loss and reducing insulin sensitivity, and the glucagon-like peptide-1 receptor agonists have been shown to be effective in people with schizophrenia.42 Polypharmacy can be an issue for people with schizophrenia, and involving the psychiatrist and pharmacist in rationalising these to limit drug interactions and reduce metabolic burden can be a useful intervention.
For smoking cessation, nicotine replacement aids must be provided appropriately and at adequate doses, as smokers with schizophrenia are likely to be heavier smokers. Combining these aids with varenicline has a larger effect size for smoking cessation and has been shown to be effective and safe in people with schizophrenia. A small increased risk of potential adverse mental health impacts necessitates due caution and patient education, particularly regarding side effects such as sleep disturbance and mood change.43
Models of care delivery
Primary care remains the most appropriate setting in which interventions for physical health care should be delivered. Psychiatrists, alongside GPs, can work to reduce the marked discrepancy in health outcomes between this patient group and the general population, but their efforts can be hampered by the restriction of the current models of care. Short consultation times within general practice and psychiatry services often impede the effective care of individuals with complex problems.44 Longer consultation times, adjunct nurse-led appointments and shared clinics with mental health workers are important considerations in providing care for this population group. Furthermore, smartphone applications, activity monitoring devices and online programmes may be useful and might be integrated into care delivery models.45
Collaborative care is a paradigm to which primary care should aspire, particularly in the case of those with complex mental illnesses. Providing or engaging with a care co-ordinator or case manager is important, a role that may be held by a practice nurse. A shared care model that integrates case managers, pharmacists, psychiatrists and GPs to provide care promotes autonomy, convenience and care outside of the stigma of a mental health-specific service.46 Currently, this approach is most often used for patients on clozapine, but a similar model might be utilised for the care of other patient groups.
To improve co-operation between primary and tertiary services, service planners should consider investment in technologies, such as shared information platforms, to facilitate continuity. Currently, this might be achieved through use of My Health Record (https://www.digitalhealth.gov.au/
initiatives-and-programs/my-health-record), particularly for prescribing and vaccinations. In some cases, case conferencing or joint consultations with the patient may be useful, and GPs should consider being involved in multidisciplinary meetings where possible. Other techniques to engage better with people with schizophrenia are summarised in Box 2.
Referrals
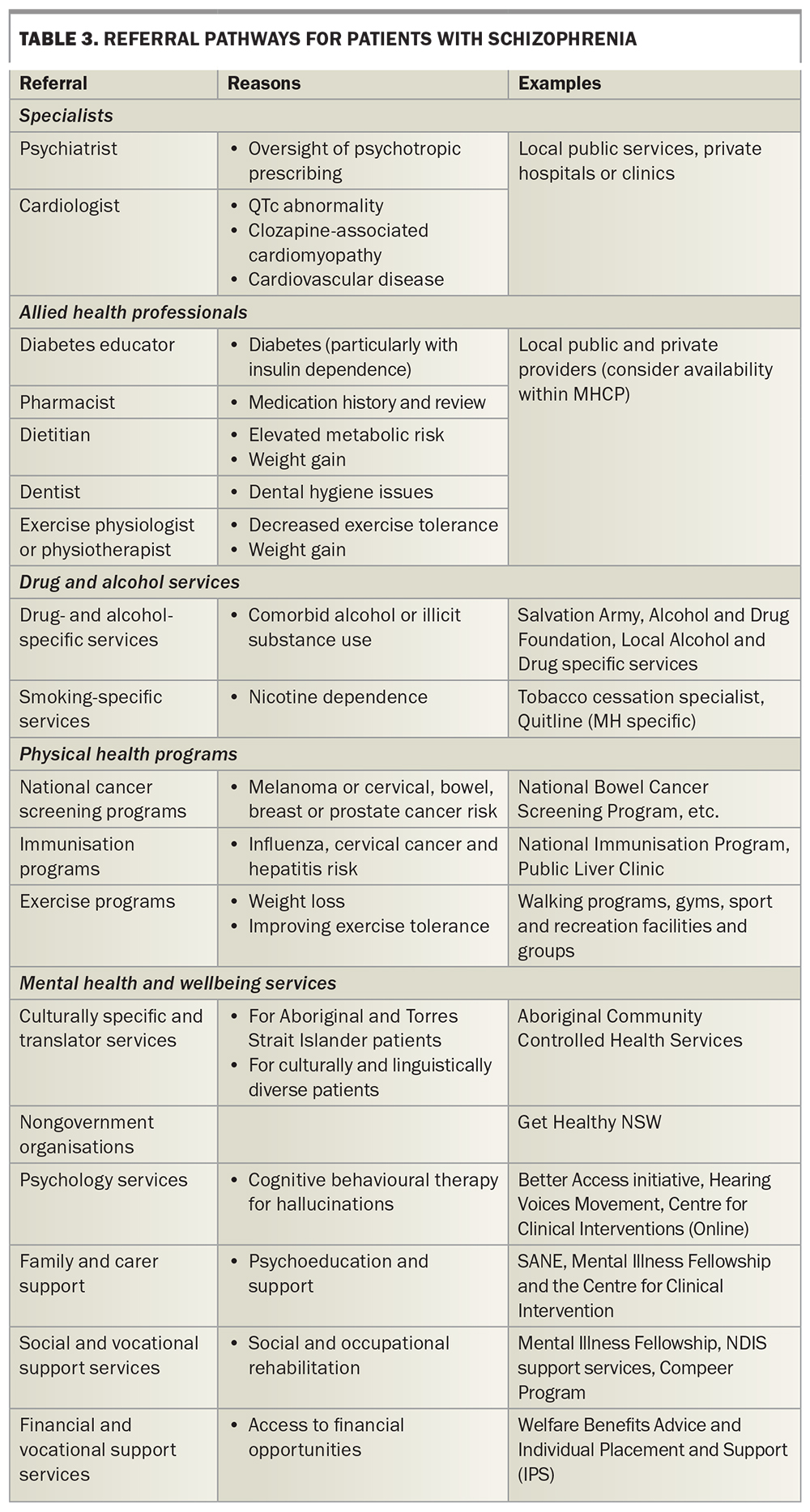
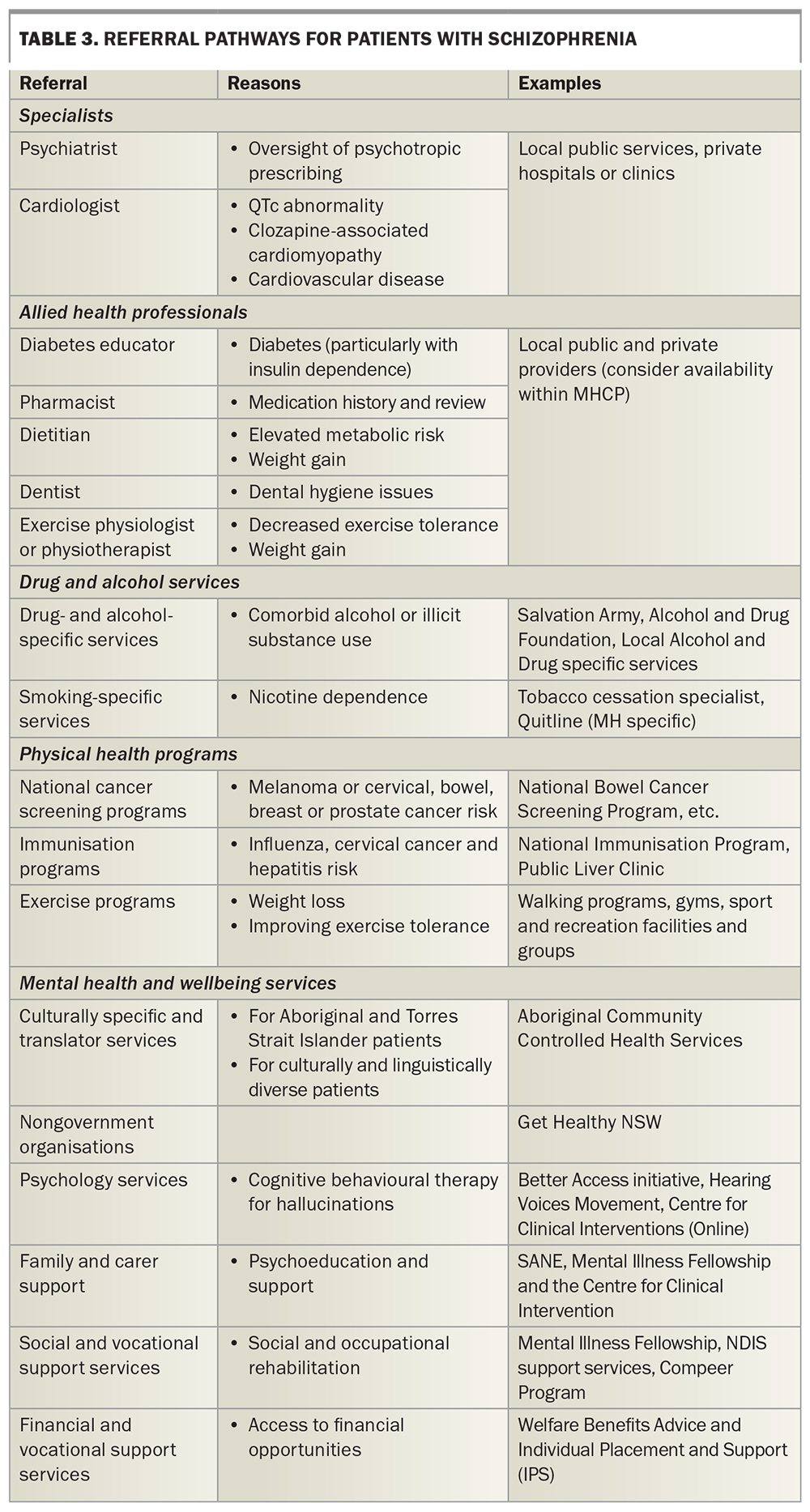
GPs, although well placed to co-ordinate care, should be adequately integrated into local therapeutic services and practices more widely. This integration provides for more expert and appropriate care of individuals and reduces the burden on GPs. Opportunities for collaboration and communication should be maximised, and being aware of local referral pathways and services is crucial. Relevant services and examples are listed in Table 3, although local resources may differ in availability depending on geographical and socioeconomic factors.
Conclusion
Although evidently complex, improving the physical health of people with schizophrenia can be a rewarding undertaking. Stigma, barriers to access and affordability combine with patient mental health factors to block people living with schizophrenia from achieving a quality and length of life that should be available to them. As GPs, there is a definite opportunity – through education, attention and proactive intervention – to improve the outcomes for this patient group greatly. With an approach tailored to the individual’s capacity and care needs, and with an integrated and evidence-based model of care, it is possible to limit the unfortunately common sequelae of the illness as a whole. This can have a wide-reaching positive impact on the individual’s mental, physical and emotional wellbeing, and further-reaching benefits to their families and the community at large. MT
COMPETING INTERESTS: Professor Castle has received grants for research on schizophrenia and antipsychotics from the National Health and Medical Research Council, Medical Research Future Fund, Barbara Dicker Foundation, Canadian Institute for Health Information, Brain Canada, Servier and Boehringer Ingelheim. He has also received travel support and honoraria for presentations and consultancy from Servier, Seqirus, Lundbeck, Mindcafe, Psychscene and Inside Practice. He was a coauthor of the Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for the management of schizophrenia and related disorders (2016), and an author on Being Equally Well (2021). He founded the Optimal Health Program and holds 50% of its intellectual property; and is a part owner (8%) of Clarity Healthcare. Dr Stubbs: None.
References
1. Lora A, Kohn R, Levav I, McBain R, Morris J, Saxena S. Service availability and utilization and treatment gap for schizophrenic disorders: a survey in 50 low- and middle-income countries. Bull World Health Organ 2012; 90: 47-54, 54A-54B.
2. Laursen TM. Life expectancy among persons with schizophrenia or bipolar affective disorder. Schizophr Res 2011; 131: 101-104.
3. Lawrence D, Hancock KJ, Kisely S. The gap in life expectancy from preventable physical illness in psychiatric patients in Western Australia: retrospective analysis of population based registers. BMJ 2013; 346: f2539.
4. Peritogiannis V, Ninou A, Samakouri M. Mortality in schizophrenia-spectrum disorders: recent advances in understanding and management. Healthc Basel Switz 2022; 10: 2366.
5. Castle DJ, Buckley PF, Gaughran FP. Physical Health and Schizophrenia. Oxford, UK: Oxford University Press; 2017.
6. Mizuki Y, Sakamoto S, Okahisa Y, et al. Mechanisms underlying the comorbidity of schizophrenia and type 2 diabetes mellitus. Int J Neuropsychopharmacol 2021; 24: 367-382.
7. Suvisaari J, Keinänen J, Eskelinen S, Mantere O. Diabetes and schizophrenia. Curr Diab Rep 2016; 16: 16.
8. Wildgust HJ, Beary M. Are there modifiable risk factors which will reduce the excess mortality in schizophrenia? J Psychopharmacol Oxf Engl 2010; 24: 37-50.
9. Galletly CA, Foley DL, Waterreus A, et al. Cardiometabolic risk factors in people with psychotic disorders: the second Australian national survey of psychosis. Aust N Z J Psychiatr 2012; 46: 753-761.
10. Suvisaari J, Keinänen J, Eskelinen S, Mantere O. Diabetes and schizophrenia. Curr Diab Rep 2016; 16: 16.
11. Stubbs B, Vancampfort D, Veronese N, et al. The prevalence and predictors of obstructive sleep apnea in major depressive disorder, bipolar disorder and schizophrenia: a systematic review and meta-analysis. J Affect Disord 2016; 197: 259-267.
12. Lawrence D, Holman CD, Jablensky AV, Threlfall TJ, Fuller SA. Excess cancer mortality in Western Australian psychiatric patients due to higher case fatality rates. Acta Psychiatr Scand 2000; 101: 382-388.
13. Tran E, Rouillon F, Loze JY, et al. Cancer mortality in patients with schizophrenia. Cancer 2009; 115: 3555-3562.
14. Musuuza JS, Sherman ME, Knudsen KJ, Sweeney HA, Tyler CV, Koroukian SM. Analyzing excess mortality from cancer among individuals with mental illness. Cancer 2013; 119: 2469-2476.
15. Ni L, Wu J, Long Y, et al. Mortality of site-specific cancer in patients with schizophrenia: a systematic review and meta-analysis. BMC Psychiatr 2019; 19: 323.
16. Sjögren R, Nordström G. Oral health status of psychiatric patients. J Clin Nurs 2000; 9: 632-638.
17. Schoepf D, Uppal H, Potluri R, Heun R. Physical comorbidity and its relevance on mortality in schizophrenia: a naturalistic 12-year follow-up in general hospital admissions. Eur Arch Psychiatry Clin Neurosci 2014; 264: 3-28.
18. Olfson M, Gerhard T, Huang C, Crystal S, Stroup TS. Premature mortality among adults with schizophrenia in the United States. JAMA Psychiatr 2015; 72: 1172-1181.
19. Rosenberg SD, Swanson JW, Wolford GL, et al. The five-site health and risk study of blood-borne infections among persons with severe mental illness. Psychiatr Serv Wash DC 2003; 54: 827-835.
20. Lacey C, Ellen S, Devlin H, Wright E, Mijch A. Hepatitis C in psychiatry inpatients: testing rates, prevalence and risk behaviours. Australas Psychiatry Bull R Aust N Z Coll Psychiatr 2007; 15: 315-319.
21. Sara G, Gould P, Curtis J, et al. Vaccine-preventable hospitalisations in adult mental health service users: a population study. Psychol Med 2023; 53: 7232-7241.
22. Montgomery J, Winterbottom E, Jessani M, et al. Prevalence of hyperprolactinemia in schizophrenia: association with typical and atypical antipsychotic treatment. J Clin Psychiatr 2004; 65: 1491-1498.
23. Hägg S, Spigset O, Bate A, Soderström TG. Myocarditis related to clozapine treatment. J Clin Psychopharmacol 2001; 21: 382-388.
24. Coulter DM, Bate A, Meyboom RH, Lindquist M, Edwards IR. Antipsychotic drugs and heart muscle disorder in international pharmacovigilance: data mining study. BMJ 2001; 322: 1207-1209.
25. Bordet C, Garcia P, Salvo F, et al. Antipsychotics and risk of QT prolongation: a pharmacovigilance study. Psychopharmacology (Berl) 2023; 240: 199-202.
26. Hope J, Keks N. Chronic schizophrenia and the role of the general practitioner. Aust J Gen Pract 2015; 44: 802-808.
27. Eugene AR, Eugene B, Masiak M, Masiak JS. Head-to-head comparison of sedation and somnolence among 37 antipsychotics in schizophrenia, bipolar disorder, major depression, autism spectrum disorders, delirium, and repurposed in COVID-19, infectious diseases, and oncology from the FAERS, 2004-2020. Front Pharmacol 2021; 12: 621691.
28. Gao K, Mackle M, Cazorla P, Zhao J, Szegedi A. Comparison of somnolence associated with asenapine, olanzapine, risperidone, and haloperidol relative to placebo in patients with schizophrenia or bipolar disorder. Neuropsychiatr Dis Treat 2013; 9: 1145-1157.
29. Morgan VA, Waterreus A, Jablensky A, et al. People living with psychotic illness in 2010: the second Australian national survey of psychosis. Aust N Z J Psychiatr 2012; 46: 735-752.
30. Castillo-Sánchez M, Fàbregas-Escurriola M, Bergè-Baquero D, Fernández-SanMartín M, Goday-Arno A. Screening of cardiovascular risk factors in patients with schizophrenia and patients treated with antipsychotic drugs: are we equally exhaustive as with the general population? Clin Exp Hypertens N Y N 2017; 39: 441-447.
31. Lahti M, Tiihonen J, Wildgust H, et al. Cardiovascular morbidity, mortality and pharmacotherapy in patients with schizophrenia. Psychol Med 2012; 42: 2275-2285.
32. Rahman T, Sahrmann JM, Olsen MA, et al. Risk of breast cancer with prolactin elevating antipsychotic drugs: An observational study of U.S. women (ages 18–64). J Clin Psychopharmacol 2022; 42: 7-16.
33. Hope J, Keks N. Schizophrenia: the GP’s crucial management role. Medicine Today 2019; 20(2): 16-27.
34. Lambert TJ, Reavley NJ, Jorm AF, Oakley Browne MA. Royal Australian and New Zealand College of Psychiatrists expert consensus statement for the treatment, management and monitoring of the physical health of people with an enduring psychotic illness. Aust N Z J Psychiatr 2017; 51: 322-337.
35. Hilton T. Pharmacological issues in the management of people with mental illness and problems with alcohol and illicit drug misuse. Crim Behav Ment Health CBMH 2007; 17: 215-224.
36. Vancampfort D, Firth J, Correll CU, et al. The impact of pharmacological and non-pharmacological interventions to improve physical health outcomes in people with schizophrenia: a meta-review of meta-analyses of randomized controlled trials. World Psychiatry 2019; 18: 53-66.
37. Caemmerer J, Correll CU, Maayan L. Acute and maintenance effects of non-pharmacologic interventions for antipsychotic associated weight gain and metabolic abnormalities: a meta-analytic comparison of randomized controlled trials. Schizophr Res 2012; 140: 159-168.
38. Cook BL, Wayne GF, Kafali EN, Liu Z, Shu C, Flores M. Trends in smoking among adults with mental illness and association between mental health treatment and smoking cessation. JAMA 2014; 311: 172-182.
39. Galletly CA, Foley DL, Waterreus A, et al. Cardiometabolic risk factors in people with psychotic disorders: the second Australian national survey of psychosis. Aust N Z J Psychiatr 2012; 46: 753-761.
40. Kulkarni J, Hayes E, Gavrilidis E. Hormones and schizophrenia. Curr Opin Psychiatr 2012; 25: 89-95.
41. Mind(gardens) Neuroscience Network. Adult Positive Cardiometabolic Health Resource. Sydney: Mind(gardens) Neuroscience Network; 2023. Available online at: https://www.mindgardens.org.au/KBIMResources/ (accessed August 2025).
42. De R, Prasad F, Stogios N, et al. Promising translatable pharmacological interventions for body weight management in individuals with severe mental illness - a narrative review. Expert Opin Pharmacother 2023; 24: 1823-1832.
43. Mendelsohn CP, Kirby DP, Castle DJ. Smoking and mental illness. An update for psychiatrists. Australas Psychiatry Bull R Aust N Z Coll Psychiatr 2015; 23: 37-43.
44. Druss BG. Improving medical care for persons with serious mental illness: challenges and solutions. J Clin Psychiatr 2007; 68: 40-44.
45. Milne-Ives M, Lam C, De Cock C, Van Velthoven MH, Meinert E. Mobile apps for health behavior change in physical activity, diet, drug and alcohol use, and mental health: systematic review. JMIR MHealth UHealth 2020; 8: e17046.
46. Sharma A, Buckhalter S, Siskind DJ, Castle D. Clozapine shared care: mental health services and GPs working together for better outcomes for people with schizophrenia. Med J Aust 2022; 217: S36-S38.