What’s the diagnosis?
A persistent skin eruption following chemotherapy
Case presentation
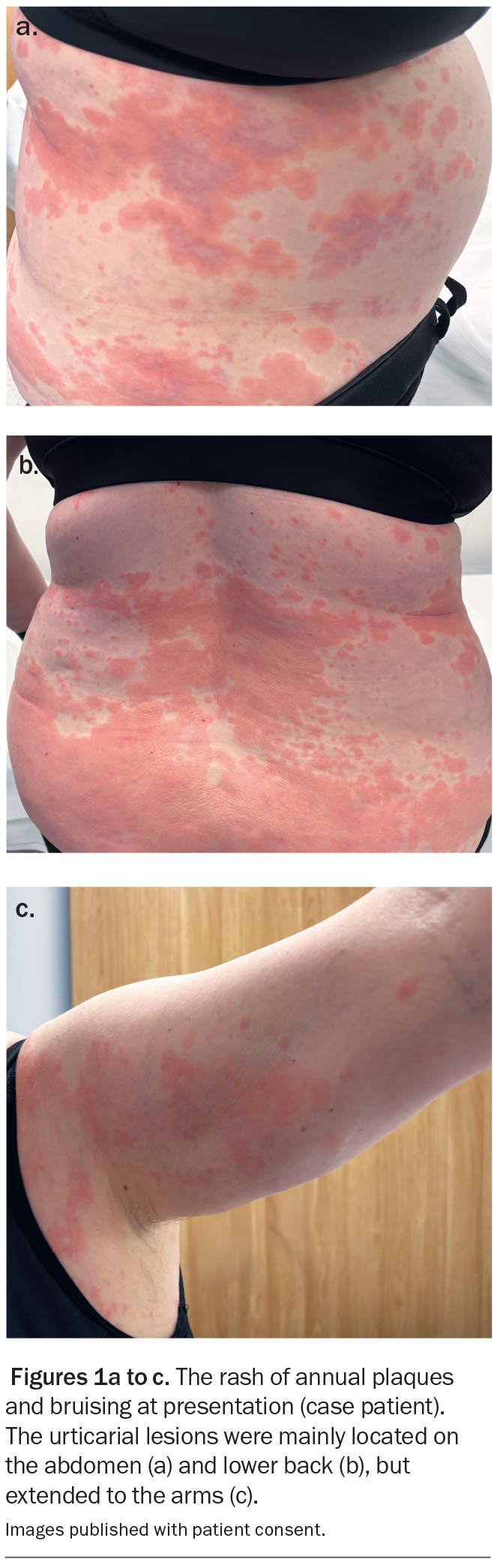
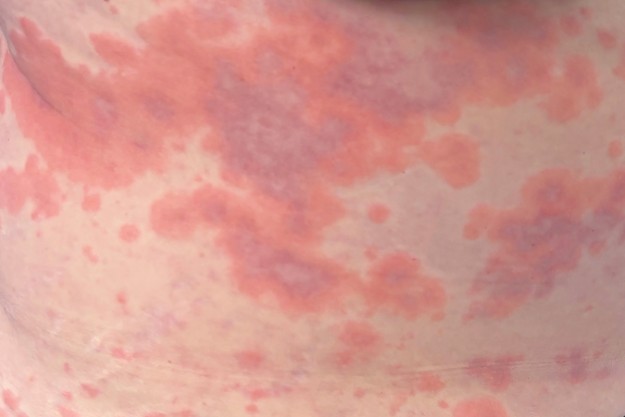
A 59-year-old woman presents with a widespread urticarial eruption and bruising (Figures 1a to c). She was recently diagnosed with breast cancer and has been receiving chemotherapy (anthracycline and taxane-based). About two weeks after the second round of chemotherapy, she developed the painful and pruritic skin eruption and was admitted to hospital, where she was treated with betamethasone dipropionate 0.05% ointment, loratadine and oral prednisone. Today, she has ongoing wheals despite treatment with oral prednisone (7.5 mg daily) and loratadine.
The patient’s medical history includes hypercholesterolaemia, hypertension, rosacea and irritable bowel syndrome. Her regular medications are ezetimibe and atorvastatin, irbesartan, diltiazem, metformin, hydrochlorothiazide and pantoprazole.
On examination, multiple annular dermal plaques with central bruising are observed on the patient’s abdomen, chest and upper limbs. She denies systemic symptoms, such as fever, shortness of breath, diarrhoea, abdominal pain or arthralgia, and feels systemically well.
Differential diagnoses
Conditions to consider among the differential diagnoses include the following.
Acute spontaneous urticaria
Acute spontaneous urticaria is defined by the spontaneous occurrence of wheals (hives) or angioedema, or both, lasting less than six weeks without an identifiable trigger.1 Around one in five individuals experience an episode of urticaria in their lifetime; it is more common in females than males and in individuals aged 20 to 30 years.2,3 Most cases are idiopathic, but potential triggers include infections (particularly viral infections), medications and food allergens.4
Clinically, patients who have acute spontaneous urticaria present with transient, erythematous wheals that will typically last for less than 24 hours. The lesions are intensely pruritic and do not leave residual pigmentation. Angioedema may occur concurrently, especially on the lips, eyelids or extremities.1
The diagnosis of acute spontaneous urticaria is based on the history and examination findings. Investigations are rarely required unless systemic symptoms (such as abdominal pain, fever, arthralgia) or atypical features are present.
For the case patient, the persistence of individual lesions beyond 24 hours and the presence of residual pigmentation made acute spontaneous urticaria an unlikely diagnosis.
Erythema annulare centrifugum
Erythema annulare centrifugum (EAC) is a benign reactive dermatosis characterised by annular, erythematous plaques with a distinctive trailing scale and central clearing.5 This rare condition can occur at any age and affects both sexes equally. Lesions typically develop on the thighs, buttocks or trunk and expand slowly over time. They are usually nonpruritic and asymptomatic.6
EAC is frequently idiopathic but can be associated with infections (e.g. dermatophyte infections), malignancy, medications or autoimmune disease.6,7 The diagnosis is primarily clinical, but a skin biopsy may reveal a superficial or deep perivascular lymphocytic infiltrate.6,8 In many cases, lesions will resolve spontaneously, but recurrence is possible if the underlying trigger persists.
For the case patient, the lesions migrated more quickly than is typically seen in EAC. In addition, the lack of scale and absence of central clearing made this diagnosis unlikely.
Wells’ syndrome
Wells’ syndrome (eosinophilic cellulitis) is a rare inflammatory dermatosis characterised by oedematous, cellulitis-like plaques.9 It can occur at any age but shows a slight female predominance. The exact cause is unknown but associations have been noted with infections, medications, insect bites and haematological malignancies.9
Clinically, patients present with the acute onset of tender, erythematous plaques, which are generally located on the extremities and may resemble infectious cellulitis. The lesions are often painful or burning and typically resolve with postinflammatory hyperpigmentation.10
The diagnosis of Wells’ syndrome is based on clinical suspicion supported by histopathological examination, which reveals a dense eosinophilic infiltrate with characteristic flame figures.11
For the case patient, the annular urticarial wheals with associated bruising and their location were not consistent with the acute onset of cellulitis-like plaques that typically affect the extremities, which made Wells’ syndrome unlikely.
Subacute cutaneous lupus erythematosus
Subacute cutaneous lupus erythematosus (SCLE) is a subtype of cutaneous lupus erythematosus, characterised by photosensitive lesions on sun-exposed areas.12 It has a strong association with anti-Ro/SSA antibodies and may be idiopathic or autoimmune in origin; however, up to 70% of cases are induced by drugs, with terbinafine and TNF-alpha inhibitors being known triggers.12-14 SCLE most commonly affects women between the ages of 30 and 50 years and is strongly associated with the HLA-DR3 haplotype.14
Clinically, SCLE presents with erythematous annular or papulosquamous lesions that are nonscarring, predominantly on the upper chest, shoulders and forearms. Systemic symptoms are generally mild or absent, but SCLE can occasionally occur in association with systemic lupus erythematosus (SLE).12
The diagnosis of SCLE is supported by positive antinuclear antibodies (ANA) and anti-Ro/SSA serology, and histopathology typically shows interface dermatitis.12 SCLE tends to follow a relapsing-remitting course and, although the risk of progression to SLE is low (about 10 to 15%), patients should be monitored periodically for systemic involvement.
For the case patient, the location and morphology of the painful wheals, which were mainly located on the abdomen and lower back, were not consistent with SCLE, which typically affects sun-exposed sites and presents with plaques that are not painful.
Urticarial vasculitis
This is the correct diagnosis. Urticarial vasculitis is a type of small vessel vasculitis characterised by wheal-like plaques that resemble urticarial hives.2,15 The lesions are histologically consistent with leukocytoclastic vasculitis involving small dermal blood vessels.15 Unlike for acute spontaneous urticaria, the lesions of urticarial vasculitis persist for more than 24 hours and are typically described as painful or burning rather than itchy, often resolving with a postinflammatory hyperpigmentation or purpura.16
Urticarial vasculitis is uncommon, with the exact prevalence unknown. In 2015, a Swedish study estimated the incidence at 0.7 cases per million, with a point prevalence of 9.5 per million.17 It can occur at any age but is more frequently reported in adults, with a female predominance.16 The condition exists on a spectrum of severity, ranging from limited cutaneous disease to systemic involvement.18 Most cases are idiopathic, but triggers include infections (e.g. hepatitis B or C, or other viral infections), medications (e.g. certain antibiotics, chemotherapy or NSAIDs), autoimmune disease (e.g. SLE) and malignancies.16
Urticarial vasculitis is considered a type III hypersensitivity reaction involving immune complexes deposition in small vessels and complement activation, leading to vascular inflammation.19 It is classified into two types on the basis of complement levels. Normocomplementaemic urticarial vasculitis (NUV) typically follows a milder course and is confined to the skin.15 Hypocomplementaemic urticarial vasculitis (HUV) tends to be more severe, with systemic features, such as fever, arthralgia, angioedema and involvement of the gastrointestinal or pulmonary systems.18 HUV is sometimes associated with connective tissue diseases, particularly SLE.15
Investigations
Urticarial vasculitis should be suspected in patients with urticarial lesions that persist for more than 24 hours with residual purpura or postinflammatory hyperpigmentation.2,16 The diagnosis requires a combination of compatible clinical features and biopsy confirmation of vasculitis.20 A skin biopsy of an early lesion is essential and will typically reveal leukocytoclastic vasculitis characterised by endothelial swelling, fibrin deposition, neutrophilic infiltration with leukocytoclasia and red blood cell extravasation.2,15 Direct immunofluorescence testing may show immunoglobulin and complement deposition around dermal blood vessels or along the dermal-epidermal junction.15 If these features are seen, further investigations may be appropriate to evaluate for systemic involvement and associated conditions – these include complement studies (C3, C4, C1q), erythrocyte sedimentation rate, urinalysis, renal function, ANA, anti-dsDNA, hepatitis serologies, and other autoimmune markers.15,20 Hypocomplementaemia is suggestive of more severe disease and systemic involvement.
Management
The management of urticarial vasculitis is guided by the severity and duration of disease. Treatment can be challenging and is not standardised.
Mild disease that is limited to the skin may remit spontaneously.21 For patients who have isolated cutaneous disease or moderate disease (persistent cutaneous symptoms with or without mild systemic features), first-line therapy focuses on symptomatic relief. Nonsedating H1 antihistamines and doxepin can be used to control pruritus and burning sensations. No single medicine has been shown to be superior to another and dosages may be increased above standard dosing to four times daily dosing.22 Examples include loratadine 10 mg and cetirizine 10 mg up to four times/day.22 However, antihistamines alone are often ineffective.20,21 NSAIDs are used to treat symptoms such as arthralgia and arthritis of urticarial vasculitis, noting that caution should be taken in patient with gastrointestinal irritation or kidney disease.22
The use of agents such as dapsone, colchicine or hydroxychloroquine to manage mild-to-moderate urticarial vasculitis, usually in combination with other treatment options, is supported by the literature.18,21-23 These agents have anti-inflammatory and immunomodulatory effects and are considered corticosteroid-sparing options for disease control.21
Systemic corticosteroids are the mainstay of treatment for urticarial vasculitis and often used to induce remission in moderate-to-severe flares.22,24 This may involve, for example, prednisone 0.5 to 1 mg/kg daily for about one week (tapering thereafter), but the exact dose of corticosteroids is based on patient factors and severity of disease. Improvement is usually noted within one to two days of commencing systemic therapy.
Treatment of severe disease, which is most commonly seen in patients with HUV, involves systemic corticosteroids.22 Incorporating a corticosteroid-sparing agent is recommended. Use of dapsone, colchicine or hydroxychloroquine (if not already started) can help maintain disease control and allow corticosteroid tapering.
For patients with severe urticarial vasculitis, an immunosuppressant such as mycophenolate mofetil, cyclophosphamide, ciclosporin, azathioprine or methotrexate may be indicated to control disease and spare corticosteroids.18,21 Mycophenolate and ciclosporin have shown efficacy in case series for steroid-refractory urticarial vasculitis.25 Combination therapy (a corticosteroid plus one of these agents) is used in severe cases, with careful biochemical monitoring.
Biologic therapies are emerging as potential treatment options for refractory or relapsing urticarial vasculitis, with several case reports supporting their use.26 Rituximab, an anti-CD20 monoclonal antibody, has shown durable responses in patients with severe disease, particularly those with SLE overlap.27 These agents are currently not approved by the TGA for urticarial vasculitis (off-label use). Specialist consultation is advised when considering use of biologic therapies, particularly for patients with disease that is severe or refractory to treatment.
In extreme, life-threatening scenarios, such as HUV with severe immune-complex disease, intravenous immunoglobulin or plasma exchange have been used as adjunctive interventions.24 These therapies are reserved for refractory cases and managed in hospital settings.
Close monitoring is crucial in patients with severe urticarial vasculitis, including regular assessments of complement levels, organ function and drug-related toxicities. Dermatology referral is advised for skin biopsy and diagnostic confirmation, while rheumatology input is recommended for patients with systemic involvement or positive autoimmune serology. An allergist may be helpful when an immunological trigger is suspected or biologic therapy is being considered.
Outcome
For the case patient, a diagnosis of urticarial vasculitis was suspected on the basis of clinical features and confirmed after skin biopsy and histopathology demonstrating a leukocytoclastic vasculitis pattern. Direct immunofluorescence testing returned negative results and the results of blood tests indicated normal complement levels, consistent with NUV. The temporal relationship between the patient’s breast cancer treatment and symptom onset suggested that the condition was likely drug-induced, associated with the chemotherapy regimen.
The absence of systemic features made a future chemotherapy rechallenge reasonable, with close monitoring of symptoms. She underwent three weeks of radiation therapy and commenced letrozole. She was prescribed doxepin to assist with pruritus and sleep disturbance and will be reviewed regularly for ongoing dermatological care. MT
COMPETING INTERESTS: None.
References
1. Zuberbier T, Aberer W, Asero R, et al. The EAACI/GA(2) LEN/EDF/WAO guideline for the definition, classification, diagnosis, and management of urticaria: the 2013 revision and update. Allergy 2014; 69: 868-887.
2. Antia C, Baquerizo K, Korman A, Bernstein JA, Alikhan A. Urticaria: a comprehensive review. Epidemiology, diagnosis, and work-up. J Am Acad Dermatol 2018; 79: 599-614.
3. Saini SS. Chronic spontaneous urticaria: etiology and pathogenesis. Immunol Allergy Clin North Am 2014; 34: 33-52.
4. Kolkhir P, Church MK, Weller K, et al. Autoimmune chronic spontaneous urticaria: what we know and what we do not know. J Allergy Clin Immunol 2017; 139: 1772-1781.e1771.
5. McDaniel B, Cook C. Erythema annulare centrifugum. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025.
6. Kim DH, Lee JH, Lee JY, et al. Erythema annulare centrifugum: analysis of associated diseases and clinical outcomes according to histopathologic classification. Ann Dermatol 2016; 28: 257-259.
7. Mu EW, Sanchez M, Mir A, Meehan SA, Pomeranz MK. Paraneoplastic erythema annulare centrifugum eruption (PEACE). Dermatol Online J 2015; 21: 13030/qt6053h29n.
8. Karen K. Erythema annulare centrifugum. DermNet; 2019. Available online at: https://dermnetnz.org/topics/erythema-annulare-centrifugum (accessed August 2025).
9. Heelan K, Ryan JF, Shear NH, Egan CA. Wells syndrome (eosinophilic cellulitis): proposed diagnostic criteria and a literature review of the drug-induced variant. J Dermatol Case Rep 2013; 7: 113-120.
10. Räßler F, Lukács J, Elsner P. Treatment of eosinophilic cellulitis (Wells syndrome) – a systematic review. J Eur Acad Dermatol Venereol 2016; 30: 1465-1479.
11. Sinno H, Lacroix JP, Lee J, et al. Diagnosis and management of eosinophilic cellulitis (Wells’ syndrome): a case series and literature review.
Can J Plast Surg 2012; 20: 91-97.
12. Sontheimer RD. Subacute cutaneous lupus erythematosus: 25-year evolution of a prototypic subset (subphenotype) of lupus erythematosus defined by characteristic cutaneous, pathological, immunological, and genetic findings. Autoimmun Rev 2005; 4: 253-263.
13. Borucki R, Werth VP. Cutaneous lupus erythematosus induced by drugs – novel insights. Expert Rev Clin Pharmacol 2020; 13: 35-42.
14. Vaglio A, Grayson PC, Fenaroli P, et al. Drug-induced lupus: traditional and new concepts. Autoimmun Rev 2018; 17: 912-918.
15. Marzano AV, Maronese CA, Genovese G, et al. Urticarial vasculitis: clinical and laboratory findings with a particular emphasis on differential diagnosis. J Allergy Clin Immunol 2022; 149: 1137-1149.
16. Gu SL, Jorizzo JL. Urticarial vasculitis. Int J Womens Dermatol 2021; 7: 290-297.
17. Sjöwall C, Mandl T, Skattum L, Olsson M, Mohammad AJ. Epidemiology of hypocomplementaemic urticarial vasculitis (anti-C1q vasculitis). Rheumatology (Oxford) 2018; 57: 1400-1407.
18. Jachiet M, Flageul B, Deroux A, et al. The clinical spectrum and therapeutic management of hypocomplementemic urticarial vasculitis: data from a French nationwide study of fifty-seven patients. Arthritis Rheumatol 2015; 67: 527-534.
19. Mehregan DR, Gibson LE. Pathophysiology of urticarial vasculitis. Arch Dermatol 1998; 134: 88-89.
20. Katelaris C. Treatment of urticaria. Aust Prescr 2001; 24: 124-126.
21. Kolkhir P, Grakhova M, Bonnekoh H, Krause K, Maurer M. Treatment of urticarial vasculitis: a systematic review. J Allergy Clin Immunol 2019; 143: 458-466.
22. Brewer J, Davis M. Urticarial vasculitis. UpToDate [website]; updated 2024.
23. Causes and assessment of urticaria. In: Therapeutic Guidelines: Dermatology. Melbourne: TG; 2022 [amended 2025]. Available online at: https://tg.org.au (accessed August 2025).
24. Greaves M. Chronic urticaria. J Allergy Clin Immunol 2000; 105: 664-672.
25. Grattan CE, O’Donnell BF, Francis DM, et al. Randomized double-blind study of cyclosporin in chronic ‘idiopathic’ urticaria. Br J Dermatol 2000; 143: 365-372.
26. Nikolai Dario Rothermel ND, Vera Ayala C, Gonçalo M, et al. Managing urticarial vasculitis: a clinical decision-making algorithm based on expert consensus. Am J Clin Dermatol 2024; 26: 61–75.
27. Alharbi S, Sanchez-Guerrero J. Successful treatment of urticarial vasculitis in a patient with systemic lupus erythematosus with rituximab. Clin Med Insights Arthritis Musculoskelet Disord 2020; 13: 1179544120967374.
Skin conditions
Cancer chemotherapy