Targeting the weight within: obesity pharmacotherapy in cardiovascular disease
Obesity is a major contributor to cardiovascular disease and mortality. New pharmacological treatments, including incretin therapies, have shown significant weight loss and cardiovascular benefits. Large trials have demonstrated that these agents reduce cardiovascular events in patients with cardiovascular disease and improve outcomes in heart failure, kidney disease and metabolic dysfunction-associated steatohepatitis. This article reviews the cardiovascular impact of weight loss and the role of therapies such as semaglutide and tirzepatide in reducing cardiovascular disease risk.
- Obesity independently increases cardiovascular mortality, and weight loss of 5 to 10% yields meaningful reductions in cardiovascular disease risk factors; weight loss of 10% and above is associated with improved cardiovascular outcomes.
- Lifestyle interventions remain foundational, but maintaining weight loss is challenging, with most patients regaining 80% of lost weight within five years.
- Glucagon-like peptide-1 receptor agonists have demonstrated significant weight loss and cardiovascular benefits in both patients with and without diabetes, including a reduction in major adverse cardiovascular events.
- Next-generation therapies are emerging, showing promise for greater weight loss and potential cardiovascular improvements, with ongoing trials evaluating long-term outcomes.
Obesity is a major public health problem that contributes both directly and indirectly to cardiovascular disease (CVD) and mortality. In 2015, obesity was estimated to have accounted for four million deaths globally, two-thirds of which were caused by CVD.1 The management of obesity is rapidly evolving with the introduction of new pharmacological treatments that achieve weight loss comparable to that seen with bariatric surgery. Modern pharmacological agents such as glucagon-like peptide-1 (GLP-1) receptor agonists and combination glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 receptor agonists have also been shown to improve cardiovascular outcomes in patients with obesity.
Why is treatment of obesity important?
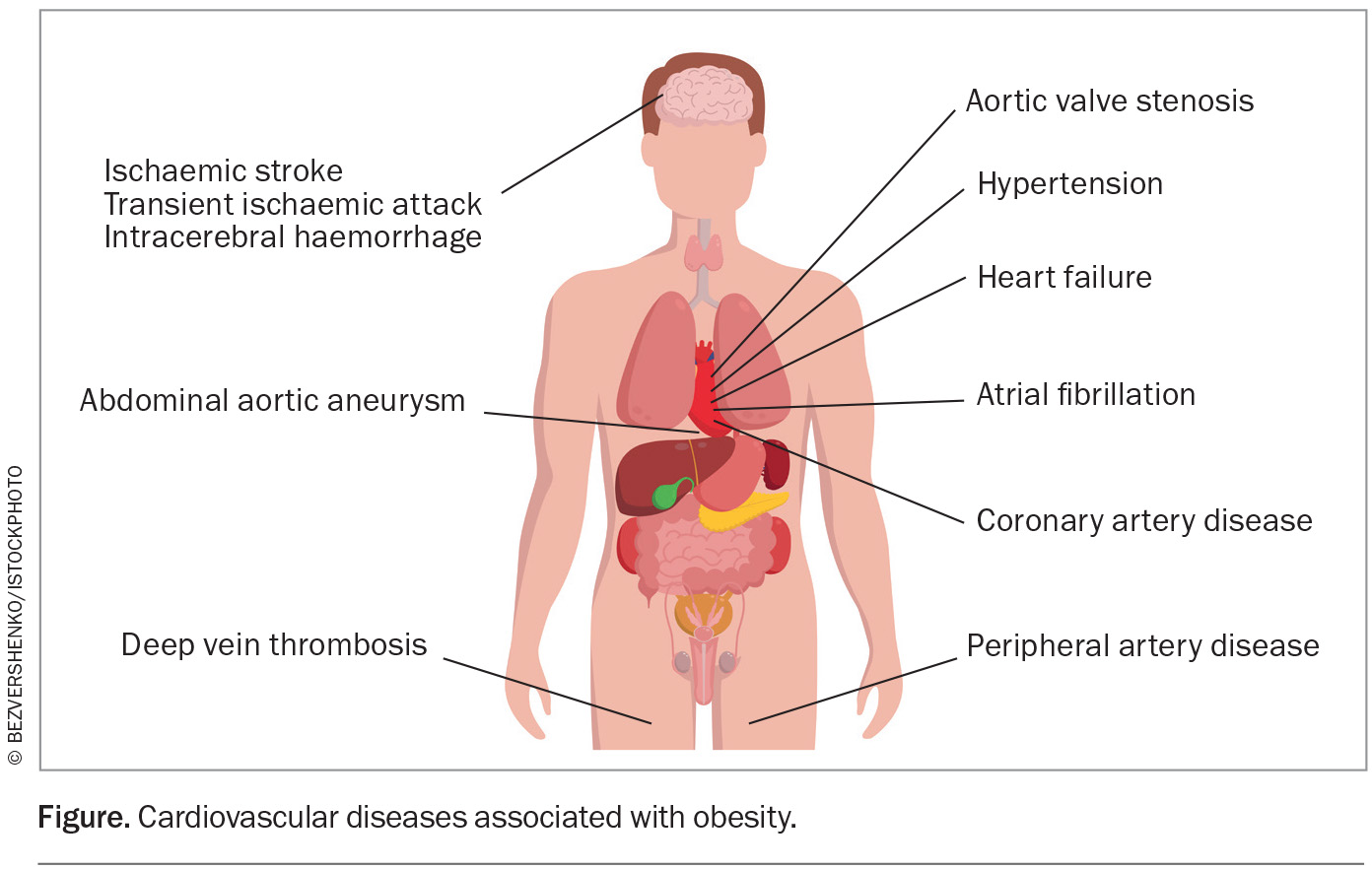
Obesity is a multifaceted disease that contributes directly and indirectly to atherosclerotic CVD, heart failure, atrial fibrillation and multiple CVD risk factors, including dyslipidaemia, type 2 diabetes, hypertension and sleep disorders (Figure). It is increasingly recognised that obesity leads to increased CVD mortality independent of cardiovascular risk factors.2-11 Modest reductions in weight of 5 to 10% can produce clinically significant improvements in CVD risk factors. Moreover, the degree of benefit in reducing CVD risk factors and improving clinical outcomes increases with higher percentages of weight loss.9,12-15 The most widely accepted body mass index (BMI) classifications of obesity are shown in Box 1.
A systematic review and meta-analysis of randomised controlled trials evaluating weight reduction diets, with or without exercise, reported an 18% reduction in all-cause mortality over a median follow up of two years in adults with obesity.7 Over 55% of the weighting in this meta-analysis was contributed by the LOOK AHEAD trial, which assessed whether intensive lifestyle interventions for weight loss would reduce cardiovascular morbidity and mortality in individuals with overweight or obesity and type 2 diabetes (T2DM). The intensive lifestyle intervention group achieved greater weight loss (6.0% vs 3.5%) and reductions in glycated haemoglobin (HbA1c) as well as in all traditional CVD risk factors, except for LDL-cholesterol. However, despite the favourable effects on CVD factors, the trial did not demonstrate a significant reduction in cardiovascular outcomes. It was therefore discontinued at a median follow up of 9.6 years following a futility analysis.11,12
These findings suggest that although lifestyle-based weight loss interventions confer short-term benefit, it is difficult for most patients to maintain sufficient weight loss to achieve long-term reductions in cardiovascular events. Furthermore, maintaining weight loss remains a significant challenge, with up to 80% of lost weight expected to be regained over the subsequent five years.16
Recent developments support the growing recognition that larger degrees of weight loss (10% to 20% of body weight) may result in meaningful reductions in cardiovascular outcomes.17,18 A post hoc analysis of LOOK AHEAD found that participants who achieved at least a 10% body weight loss in the first year had a 21% lower rate of major adverse cardiac events.17 Similar findings have been reported in analyses of subjects who underwent metabolic surgery compared with those who did not.11,19-23 Although these findings are derived from nonrandomised cohort studies, metabolic surgery resulting in 20 to 35% total body weight loss was associated with lower rates of all-cause mortality, cardiovascular mortality, incident heart failure, myocardial infarction and stroke (p <0.001 for all comparisons).23
Accordingly, metabolic surgery should be considered an important adjunct to reduce CVD risk in patients with a BMI of 40 kg/m2 and above, or 35 kg/m2 and above in the presence of obesity-associated comorbidities, particularly when lifestyle and pharmacological therapy alone are insufficient.7,23
Evidence for new pharmacologic therapy
Although lifestyle interventions remain the cornerstone of weight management, sustaining long-term weight loss is challenging. The management of obesity is evolving with the advent of pharmacological agents that produce substantial weight loss approaching that achieved with metabolic surgery.
Glucagon-like peptide-1 receptor agonists
GLP-1 is an endogenous incretin hormone produced in the intestines following food intake. It enhances insulin secretion and suppresses glucagon release. Cardiovascular outcome studies have evaluated the safety and efficacy of GLP-1 receptor agonists in patients with T2DM, with meta-analyses reporting significant reductions in major adverse cardiovascular events (hazard ratio [HR] 0.86, 95% confidence interval [CI] 0.80–0.93) without an increased risk of severe hypoglycaemia.24 Although these studies did not specifically enrol patients with overweight or obesity, the average BMI at baseline exceeded 30 kg/m2. This included the LEADER and SUSTAIN-6 trials, which both demonstrated that injectable GLP-1 receptor agonists (liraglutide up to 1.8 mg once daily or semaglutide up to 1.0 mg once weekly, respectively) significantly reduced the risk of major adverse cardiovascular events in patients with T2DM who were at high cardiovascular risk.25,26
The STEP series of clinical trials evaluated semaglutide up to 2.4 mg once weekly in people with obesity. The STEP 1 trial showed that from baseline to week 68, the mean change in body weight in subjects without diabetes was –14.9% in the semaglutide group compared with –2.4% with placebo (p <0.001).27 The STEP 5 trial reported sustained weight loss with semaglutide out to 104 weeks.28
The SELECT trial was the first randomised, placebo-controlled cardiovascular outcome trial evaluating semaglutide in patients without diabetes who had overweight or obesity.29 A total of 17,604 patients aged 45 years or older with pre-existing CVD and a BMI of 27 kg/m2 and above (but without diabetes) were randomised to subcutaneous semaglutide once weekly (titrated up to 2.4 mg) or placebo. At a mean follow up of 39.8 months, semaglutide significantly reduced the incidence of death from cardiovascular causes, nonfatal myocardial infarction or nonfatal stroke (6.5% vs 8.0%, HR 0.80, 95% CI 0.72–0.90). Patients receiving semaglutide also experienced greater improvements in a variety of secondary endpoints, including reductions in body weight, HbA1c level, systolic and diastolic blood pressure and high-sensitivity C-reactive protein and lipid levels.29
Although the reduction in cardiovascular events may be mediated through weight loss and improvements in traditional CVD risk factors (e.g. lipid levels, glycaemia, blood pressure), the early separation of event curves between the semaglutide and placebo groups even before substantial weight loss occurred suggests additional direct cardioprotective effects. This may involve modulation of inflammatory and prothrombotic pathways or other pleiotropic mechanisms.
Recent studies also report beneficial effects of semaglutide beyond cardiovascular risk reduction. At a target dose of 2.4 mg once weekly (which is TGA approved for chronic weight management), semaglutide led to significant weight loss, improved quality of life and increased six-minute walk distance in patients with obesity and heart failure with preserved or mildly reduced ejection fraction, with or without T2DM.30,31 Notably, weight reduction in these trials was associated with reductions in NT pro B-type natriuretic peptide, suggesting beneficial effects on myocardial remodelling.
Semaglutide (aiming for a target dose of 1.0 mg weekly), which is TGA approved for T2DM) was also found to reduce the risk of clinically important kidney outcomes in patients with T2DM and chronic kidney disease (defined by either estimated glomerular filtration rate [eGFR] 50 to 70 mL/min/1.73 m2 and urinary albumin-to-creatinine ratio >300 and <5000; or eGFR 25 to <50 mL/min/1.73 m2 and urinary albumin-to-creatinine ratio of >100 and <5000). The primary outcome – a composite of kidney failure (dialysis, transplantation or an eGFR of <15 mL/min/1.73 m2), at least 50% reduction in eGFR from baseline or death from kidney-associated or cardiovascular causes – was 24% lower in the semaglutide group (HR 0.76, 95% CI 0.66–0.88) at a median follow up of 3.4 years. Similar results were observed for a composite of the kidney-specific components of the primary outcome (HR 0.79, 95% CI 0.66–0.94) and for death from cardiovascular causes (HR 0.71, 95% CI 0.56–0.89). Major cardiovascular events were also reduced by 18% (HR 0.82, 95% CI 0.68–0.98).32
The recent phase 3 ESSENCE trial showed that semaglutide 2.4 mg once weekly improved liver histology in patients with metabolic dysfunction-associated steatohepatitis with moderate or advanced liver fibrosis. Resolution of steatohepatitis and fibrosis reduction occurred in 32.7% of semaglutide-treated patients versus 16.1% in the placebo group (estimated difference 16.5 percentage points, 95% CI 10.2–22.8).33 Results from the STRIDE trial also found that semaglutide 1.0 mg once weekly is associated with an increase in walking distance in individuals with symptomatic peripheral artery disease and T2DM.34
Given the promising results of semaglutide 2.4 mg, the recent STEP-UP trial compared intensified semaglutide 7.2 mg with semaglutide 2.4 mg in adult patients with a BMI of 30 kg/m2 and above, without diabetes. Preliminary data presented at the recent 85th Scientific Sessions of the American Diabetes Association showed that semaglutide 7.2 mg achieved a mean weight loss of 21%, outperforming the 2.4 mg dose. More than 30% of participants receiving semaglutide 7.2 mg achieved a weight loss of 25% initial body weight, or greater.35
Oral GLP-1 receptor agonists are also under development. A phase 3 trial found that high-dose oral semaglutide (50 mg) produced a 12.7% placebo-adjusted weight loss at week 68, comparable to subcutaneous semaglutide in patients without diabetes.36 The recent SOUL trial evaluated the cardiovascular efficacy of oral semaglutide (up to 14 mg) in patients with T2DM (HbA1c 6.5–10.0%) and established atherosclerotic CVD, chronic kidney disease (eGFR <60 mL/min/1.73 m2), or both. The study demonstrated that oral semaglutide was associated with a lower risk of major adverse cardiovascular events (a composite of death from cardiovascular causes, nonfatal myocardial infarction or nonfatal stroke; HR 0.86, 95% CI 0.77–0.96), particularly nonfatal myocardial infarction (HR 0.74, 95% CI 0.61–0.89). However, a significant effect on major kidney outcomes was not observed (HR 0.91, 95% CI 0.80–1.05). Serious adverse event rates were comparable with placebo, although gastrointestinal events were slightly more frequent in the semaglutide group versus placebo (5.0% vs 4.4%).37
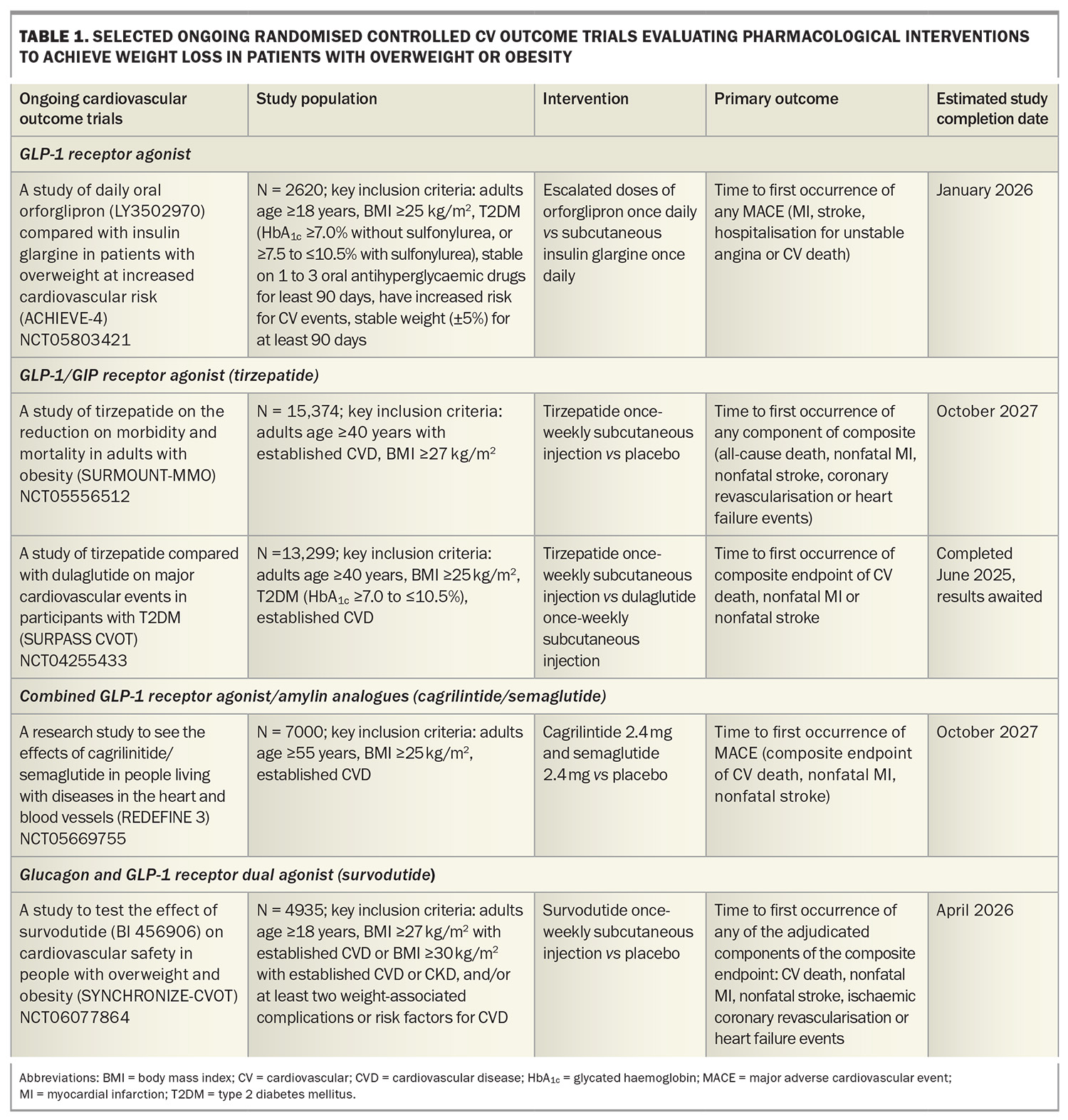
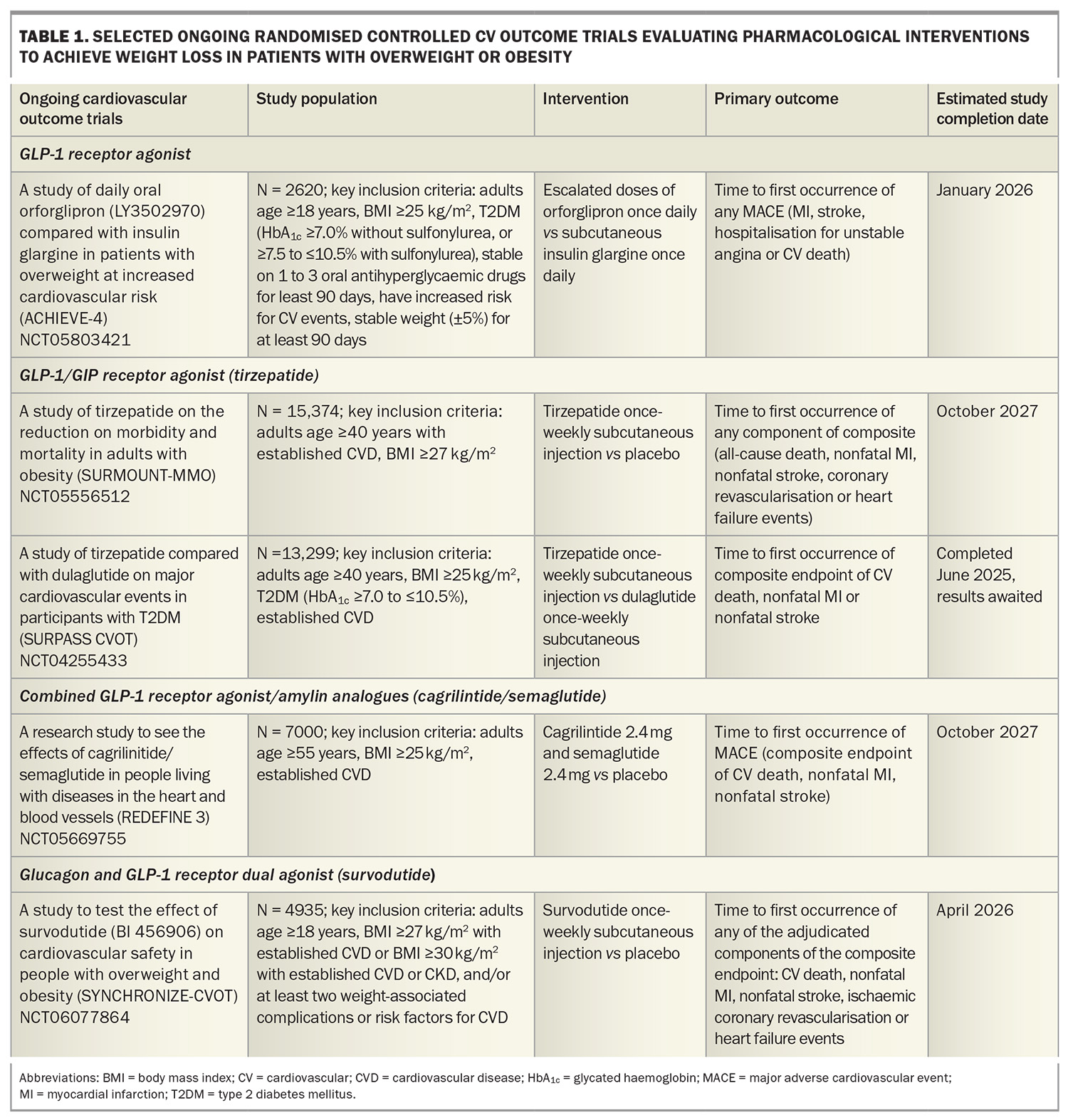
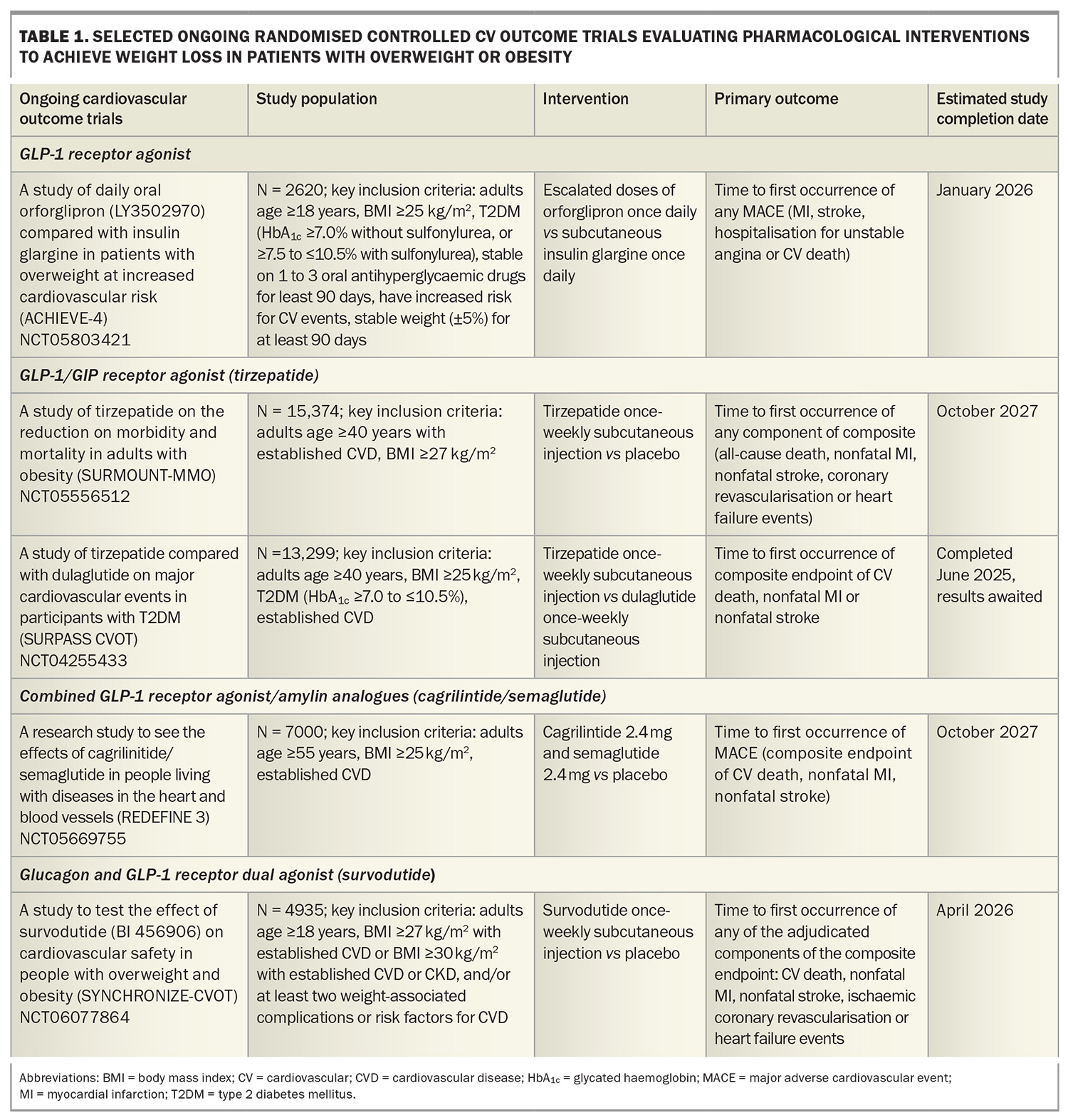
Orforglipron, an oral small-molecule, nonpeptide GLP-1 receptor agonist, has also demonstrated weight loss comparable to injectable GLP-1 receptor agonists.38 A phase 3 trial including adults with T2DM receiving orforglipron daily (3 mg, 12 mg or 36 mg) showed significant placebo-adjusted reductions in HbA1c (–1.07%) and weight loss (–5.9%) at the highest dose.39 There are ongoing studies evaluating the efficacy of orforglipron on weight loss and cardiovascular outcomes (clinical trial number NCT05803421; Table 1).
At the time of this publication, both subcutaneous liraglutide and semaglutide (2.4 mg weekly dose) are TGA approved for chronic weight management in Australia. This includes adults with an initial BMI of 30 kg/m2 and above, or 27 to 29.9 kg/m2 with at least one weight-associated comorbidity (e.g hypertension, dyslipidaemia, obstructive sleep apnoea, cardiovascular disease, prediabetes or T2DM). Liraglutide and semaglutide (2.4 mg weekly dose) are not currently subsidised under the PBS and are only available via private prescription.
Combination incretin therapies
Next-generation pharmacological treatments for obesity include combinations of GLP-1 receptor agonists with other enteropancreatic hormones (such as GIP, glucagon and amylin). These combinations may enhance weight loss and cardiometabolic outcomes beyond those achieved with GLP-1 receptor agonist monotherapy.
Glucagon-like peptide-1/glucose-dependent insulinotropic polypeptide receptor agonists
Tirzepatide is a once-weekly, subcutaneous injectable peptide with dual agonist activity at both GLP-1 and GIP receptors. GIP activation appears to act synergistically with GLP-1 receptor activation, enabling greater weight reduction than GLP-1 receptor agonism alone.40
In patients with T2DM, the SURPASS-2 trial compared tirzepatide with semaglutide 1 mg (both as add-on therapies to metformin) and found tirzepatide was either noninferior or superior at all doses (5 mg, 10 mg 15 mg), for both HbA1c reduction and weight loss at 40 weeks. The 15 mg dose led to a mean HbA1c reduction of 2.3% and a mean weight loss of 11 kg.41
In patients with overweight or obesity without diabetes, the SURMOUNT-1 trial showed that tirzepatide 15 mg weekly led to a mean body weight reduction of 20.9% after 72 weeks of treatment, compared with 3.1% with placebo.42 In the recent SURMOUNT-5 trial in adult participants with obesity but without T2DM, tirzepatide (10 mg or 15 mg) achieved greater weight loss compared with semaglutide (1.7 mg or 2.4 mg weekly dose): –20.2% (95% CI –21.4 to –19.1) versus -13.7% (95% CI –14.9 to –12.6) at week 72. Tirzepatide was also associated with greater reductions in waist circumference and higher rates of achieving at least 25% weight loss.43
Cardiovascular outcomes data are awaited from the SURPASS CVOT trial (clinical trial number NCT04255433; Table 1) comparing tirzepatide and dulaglutide in patients with T2DM and cardiovascular disease. A recent press release reported topline results indicating that tirzepatide met the primary objective of noninferiority compared with dulaglutide for time to first occurrence of death from cardiovascular causes, nonfatal myocardial infarction or nonfatal stroke (HR 0.92, 95.3% CI 0.83–1.01), along with a reduction in all-cause mortality (HR 0.84, 95% CI 0.75–0.94).44 Formal presentation of these findings is anticipated later this year. Meanwhile, the ongoing SURMOUNT-MMO trial (clinical trial number NCT05556512; Table 1) evaluating tirzepatide in people at high cardiovascular risk who are overweight or obese (without diabetes) is expected to report in 2027.
In the SUMMIT trial, tirzepatide significantly reduced the risk of death from cardiovascular causes or worsening heart failure events in patients with heart failure with preserved ejection fraction (ejection fraction 50% and above) and obesity (BMI 30 kg/m2), compared with placebo (HR 0.62; 95% CI 0.41–0.95). Tirzepatide was also associated with an improved quality of life.45
The SURMOUNT-OSA trial reported that tirzepatide (10 mg or 15 mg) significantly reduced the apnoea–hypopnea index in people with moderate to severe obstructive sleep apnoea and obesity. Among participants using positive airway pressure, the apnoea–hypopnea index was reduced by up to 29.3 events per hour (95% CI –29.6 to –17.9), a 58.7% change from baseline, compared with a 5.3 events per hour (3.0%) reduction with placebo.46 Tirzepatide also improved sleep-associated patient-reported outcomes.46
Since September 2024, subcutaneous tirzepatide has been TGA approved for the treatment of overweight and obesity. Eligible adults include those with an initial BMI of 30 kg/m2 and above, or 27 to 29.9 kg/m2 in the presence of at least one weight-associated comorbidity. It is also approved for the treatment of obstructive sleep apnoea in adults living with obesity.
Novel ‘triple-G’ agonists
Retatrutide is a novel, once-weekly injectable agent that acts on GIP, GLP-1 and glucagon receptors, and has shown promise in clinical trials for significant weight loss. In a phase 2 trial involving people with obesity, the 12 mg weekly dose of retatrutide resulted in a placebo-adjusted mean weight reduction of 22.1% after 48 weeks of treatment.47 The phase 3 randomised controlled TRIUMPH-3 trial is currently underway to evaluate the effects of retatrutide in participants with obesity (BMI of 35 kg/m2 and above) and established CVD, focusing on percentage change in body weight (clinical trial number NCT05882045; Table 1).
Glucagon-like peptide-1 receptor agonist and amylin analogue combinations
Amylin is co-secreted with insulin from the pancreas and contributes to postprandial satiety regulation, delays gastric emptying and inhibits glucagon secretion. Cagrilintide is a long-acting amylin analogue that, when combined with the GLP-1 receptor agonist semaglutide, has demonstrated synergistic effects. In a phase 2 trial of adults with T2DM and a BMI of 27 kg/m2 and above, combination cagrilinitide/semaglutide achieved greater weight loss (–15.6%) at 32 weeks compared to semaglutide alone (–5.1%) or cagrilintide alone (–8.1%).48 The phase 3 REDEFINE-3 trial will further assess the impact of cagrilinitide/semaglutide on major adverse cardiovascular events in people with obesity (with or without T2DM) and established CVD (clinical trial number NCT05669755; Table 1).
Glucagon and glucagon-like peptide-1 dual agonist
Survodutide is a dual GLP-1 and glucagon receptor agonist. In a phase 2 randomised controlled trial involving adults without diabetes and with a BMI of 27 kg/m2 and above, survodutide administered once weekly for 46 weeks led to dose-dependent weight loss. The 4.8 mg dose produced a placebo-adjusted weight reduction of 12.1%.49 Phase 3 trials – SYNCHRONISE-1 (in people without diabetes) and SYNCHRONISE-2 (in people with diabetes) – are ongoing to assess the efficacy and durability of weight loss with 3.6 and 6 mg doses over 76 weeks (clinical trial numbers NCT06066515 and NCT06066528). The SYNCHRONISE-CVOT trial will evaluate the cardiovascular safety of survodutide in participants with overweight or obesity and established CVD, chronic kidney disease or cardiovascular risk factors (clinical trial number NCT06077864; Table 1).
Conclusion
Obesity should be recognised as both an independent CVD risk factor and a chronic disease in its own right. An evolving body of evidence shows that weight reduction improves health outcomes beyond its effect on intermediary CVD risk factors. Lifestyle interventions remain the cornerstone of obesity management, and typically involve a multidisciplinary approach with GPs, cardiologists, endocrinologists and allied health professionals. Although addressing obesity can be challenging, all healthcare professionals should ask permission to discuss a patient’s weight in a sensitive, nonjudgemental manner (Box 2). Where lifestyle measures alone are insufficient, pharmacotherapy should be considered. Available options include GLP-1 receptor agonists such as semaglutide (2.4 mg weekly dose) and the dual GLP-1/GIP receptor agonist tirzepatide. Cardiovascular outcome trials evaluating novel agents that target multiple pathways – including combinations of GLP-1 agonism, GIP agonism, glucagon receptor agonism and amylin analogues – are ongoing and will help define the next generation of obesity treatments. MT
COMPETING INTERESTS: Dr Sun: None. Professor Atherton has received travel sponsorship and/or honoraria or consultancy payments (made to his employer) from AstraZeneca, Boehringer Ingelheim, Eli Lilly and Novo Nordisk.
References
1. Collaborators GBDO, Afshin A, Forouzanfar MH, et al. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med 2017; 377: 13-27.
2. Bogers RP, Bemelmans WJ, Hoogenveen RT, et al. Association of overweight with increased risk of coronary heart disease partly independent of blood pressure and cholesterol levels: a meta-analysis of 21 cohort studies including more than 300 000 persons. Arch Intern Med 2007; 167: 1720-1728.
3. Bianchettin RG, Lavie CJ, Lopez-Jimenez F. Challenges in cardiovascular evaluation and management of obese patients: JACC state-of-the-art review. J Am Coll Cardiol 2023; 81: 490-504.
4. Hritani R, Al Rifai M, Mehta A, German C. Obesity management for cardiovascular disease prevention. Obes Pillars 2023; 7: 100069.
5. Hubert HB, Feinleib M, McNamara PM, Castelli WP. Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham Heart Study. Circulation 1983; 67: 968-977.
6. Lavie CJ, Arena R, Alpert MA, Milani RV, Ventura HO. Management of cardiovascular diseases in patients with obesity. Nat Rev Cardiol 2018; 15: 45-56.
7. Ma C, Avenell A, Bolland M, et al. Effects of weight loss interventions for adults who are obese on mortality, cardiovascular disease, and cancer: systematic review and meta-analysis. BMJ 2017; 359: j4849.
8. Powell-Wiley TM, Poirier P, Burke LE, et al. Obesity and cardiovascular disease:a a scientific statement from the American Heart Association. Circulation 2021; 143: e984-e1010.
9. Ryan DH, Deanfield JE, Jacob S. Prioritizing obesity treatment: expanding the role of cardiologists to improve cardiovascular health and outcomes. Cardiovasc Endocrinol Metab 2023; 12: e0279.
10. Global Burden of Metabolic Risk Factors for Chronic Diseases C, Lu Y, Hajifathalian K, et al. Metabolic mediators of the effects of body-mass index, overweight, and obesity on coronary heart disease and stroke: a pooled analysis of 97 prospective cohorts with 1.8 million participants. Lancet 2014; 383(9921): 970-983.
11. Abdul Wahab R, le Roux CW. A review of the evidence on cardiovascular outcomes from obesity treatment. Obes Pillars 2023; 7: 100071.
12. Look ARG, Wing RR, Bolin P, et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med 2013; 369: 145-154.
13. Stevens VJ, Obarzanek E, Cook NR, et al. Long-term weight loss and changes in blood pressure: results of the Trials of Hypertension Prevention, phase II. Ann Intern Med 2001; 134: 1-11.
14. Morris E, Jebb SA, Oke J, et al. Effect of weight loss on cardiometabolic risk: observational analysis of two randomised controlled trials of community weight-loss programmes. Br J Gen Pract 2021; 71: e312-e319.
15. Tuomilehto J, Lindstrom J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001; 344: 1343-1350.
16. Anderson JW, Konz EC, Frederich RC, Wood CL. Long-term weight-loss maintenance: a meta-analysis of US studies. Am J Clin Nutr 2001; 74: 579-584.
17. Look ARG, Gregg EW, Jakicic JM, et al. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: a post-hoc analysis of the Look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol 2016; 4: 913-921.
18. Neeland IJ, McGuire DK, Sattar N. Cardiovascular outcomes trials for weight loss interventions: another tool for cardiovascular prevention? Circulation 2021; 144: 1359-1361.
19. Aminian A, Zajichek A, Arterburn DE, et al. Association of metabolic surgery with major adverse cardiovascular outcomes in patients with type 2 diabetes and obesity. JAMA 2019; 322: 1271-1282.
20. Doumouras AG, Wong JA, Paterson JM, et al. Bariatric surgery and cardiovascular outcomes in patients with obesity and cardiovascular disease: a population-based retrospective cohort study. Circulation 2021; 143: 1468-1480.
21. Fisher DP, Johnson E, Haneuse S, et al. Association between bariatric surgery and macrovascular disease outcomes in patients with type 2 diabetes and severe obesity. JAMA 2018; 320: 1570-1582.
22. Sjostrom L, Peltonen M, Jacobson P, et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. JAMA 2014; 311: 2297-2304.
23. van Veldhuisen SL, Gorter TM, van Woerden G, et al. Bariatric surgery and cardiovascular disease: a systematic review and meta-analysis. Eur Heart J 2022; 43: 1955-1969.
24. Sattar N, Lee MMY, Kristensen SL, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol 2021; 9: 653-662.
25. Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375: 311-322.
26. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375: 1834-1844.
27. Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med 2021; 384: 989-1002.
28. Garvey WT, Batterham RL, Bhatta M, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med 2022; 28: 2083-2091.
29. Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med 2023; 389: 2221-2232.
30. Kosiborod MN, Abildstrom SZ, Borlaug BA, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med 2023; 389: 1069-1084.
31. Kosiborod MN, Petrie MC, Borlaug BA, et al. semaglutide in patients with obesity-related heart failure and type 2 diabetes. N Engl J Med 2024; 390: 1394-1407.
32. Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med 2024; 391: 109-121.
33. Sanyal AJ, Newsome PN, Kliers I, et al. Phase 3 trial of semaglutide in metabolic dysfunction-associated steatohepatitis. N Engl J Med 2025; 392: 2089-2099.
34. Bonaca MP, Catarig AM, Houlind K, et al. Semaglutide and walking capacity in people with symptomatic peripheral artery disease and type 2 diabetes (STRIDE): a phase 3b, double-blind, randomised, placebo-controlled trial. Lancet 2025; 405: 1580-1593.
35. Wharton S FP, Hjelmsaeth J, et al. Efficacy and Safety of Semaglutide 7.2mg in Obesity–STEP UP Trial. Chicago: 85th American Diabetes Association Scientific Sessions; 2025
36. Knop FK, Aroda VR, do Vale RD, et al. Oral semaglutide 50 mg taken once per day in adults with overweight or obesity (OASIS 1): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2023; 405: 705-719.
37. McGuire DK, Marx N, Mulvagh SL, et al. Oral semaglutide and cardiovascular outcomes in high-risk type 2 diabetes. N Engl J Med 2025; 392: 2001-2012.
38. Wharton S, Blevins T, Connery L, et al. Daily oral GLP-1 receptor agonist orforglipron for adults with obesity. N Engl J Med 2023; 389: 877-888.
39. Rosenstock J, Hsia S, Nevarez Ruiz L, et al. Orforglipron, an oral small-molecule GLP-1 receptor agonist, in early type 2 diabetes. N Engl J Med 2025. doi: 10.1056/NEJMoa2505669. Epub ahead of print.
40. Cho YK, La Lee Y, Jung CH. the cardiovascular effect of tirzepatide: a glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide dual agonist. J Lipid Atheroscler 2023; 12: 213-222.
41. Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med 2021; 385: 503-515.
42. Jastreboff AM, Aronne LJ, Ahmad NN, et al. tirzepatide once weekly for the treatment of obesity. N Engl J Med 2022; 387: 205-216.
43. Aronne LJ, Horn DB, le Roux CW, et al. Tirzepatide as compared with semaglutide for the treatment of obesity. N Engl J Med 2025; 393: 26-36.
44. Eli Lilly and Company. Lilly’s Mounjaro (Tirzepatide), a GIP/GLP‑1 Dual Agonist, Demonstrated Cardiovascular Protection in Landmark Head‑to‑Head Trial Reinforcing Its Benefit in Patients With Type 2 Diabetes and Heart Disease. Indianapolis: Eli Lilly and Company: 2025. Available online at: https://investor.lilly.com/news-releases/news-release-details/lillys-mounjaro-tirzepatide-gipglp-1-dual-agonist-demonstrated (accessed August 2025).
45. Packer M, Zile MR, Kramer CM, et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N Engl J Med 2025; 392: 427-437.
46. Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N Engl J Med 2024; 391: 1193-1205.
47. Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-hormone-receptor agonist retatrutide for obesity - a phase 2 trial. N Engl J Med 2023; 389: 514-526.
48. Frias JP, Deenadayalan S, Erichsen L, et al. Efficacy and safety of co-administered once-weekly cagrilintide 2.4 mg with once-weekly semaglutide 2.4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial. Lancet 2023; 402: 720-730.
49. le Roux CW, Steen O, Lucas KJ, Startseva E, Unseld A, Hennige AM. Glucagon and GLP-1 receptor dual agonist survodutide for obesity: a randomised, double-blind, placebo-controlled, dose-finding phase 2 trial. Lancet Diabetes Endocrinol 2024; 12: 162-173.