Obesity and obstructive sleep apnoea: exploring the role of tirzepatide

Obstructive sleep apnoea is a common, multifactorial condition with obesity as its most important modifiable driver. Weight loss can markedly reduce disease severity, and the recent TGA approval of tirzepatide for obstructive sleep apnoea in adults with obesity offers a novel adjunct to continuous positive airway pressure, broadening treatment strategies and improving long-term health outcomes.
Note
This is an online update of the original version of this article that was published in the September 2025 issue of Medicine Today. This update was prepared for World Obesity Day 2026.
- Obstructive sleep apnoea (OSA) is highly prevalent, affecting 936 million people worldwide. It is characterised by repetitive upper airway obstruction during sleep, leading to partial or total cessation of breathing, oxygen desaturation and sleep disruption.
- OSA is strongly associated with obesity, although other factors also contribute, including craniofacial features, genetic and racial predisposition, functional endotypes and age.
- Weight gain of 10% can increase the risk of developing moderate-to-severe OSA by up to sixfold, whereas weight loss can significantly reduce disease severity.
- Continuous positive airway pressure (CPAP) remains the gold-standard treatment for OSA, but adherence challenges limit its long-term effectiveness.
- Tirzepatide, which is TGA approved for the management of moderate-to-severe OSA in adults with obesity, helps achieve substantial weight loss and reduce OSA severity, although it should be used as an adjunct rather than a replacement for CPAP.
- Ongoing resistance-based exercise and adequate protein intake to maintain muscle mass are integral when using tirzepatide for chronic weight management.
Obstructive sleep apnoea (OSA) is a common and increasingly prevalent disorder characterised by repetitive episodes of upper airway obstruction during sleep, leading to intermittent hypoxia, fragmented sleep and a cascade of physiological disturbances.
Among the myriad of risk factors, obesity stands as the most significant and modifiable driver of OSA. In recent years, the development of novel pharmacotherapeutic agents such as tirzepatide has offered hope for managing select patients with OSA, particularly those whose condition is closely linked to excess adiposity.1 This article explores the association between obesity and OSA and examines the emerging role of tirzepatide in the management of this challenging disorder.
Assessment for obstructive sleep apnoea
OSA is a condition characterised by repetitive obstruction of the upper airway during sleep, resulting in partial cessation (hypopnoea) or total cessation (apnoea) of breathing, reduced blood oxygen saturation and sleep disturbance. Globally, an estimated 936 million people live with the condition.2 The classic symptoms of snoring, excessive daytime sleepiness and witnessed apnoeas are more frequently seen in men, whereas women may present with less specific symptoms including poor sleep quality, fatigue and mood disturbance.3 Presentation may also occur without overt symptoms, such as when there is reported concern of loud and excessive snoring or pauses in breathing observed by the patient’s bed partner. Occasionally, presentation follows a motor vehicle accident in which there was suspicion of sleep onset while driving; however, sleep restriction and circadian rhythm disorders are also potential causes. Furthermore, with the use of consumer health devices becoming more common, patients may present to discuss reports generated by these devices and the implications on their overall wellbeing. Untreated OSA has been linked to adverse cardiometabolic health, including an increased risk of atrial fibrillation.
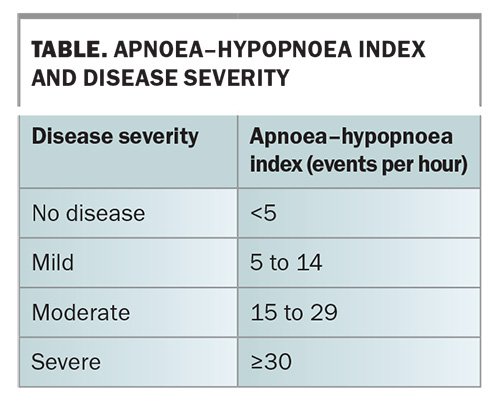
The severity of OSA is commonly classified using the apnoea–hypopnoea index (AHI), which measures the number of respiratory disturbances (apnoeas and hypopnoeas) per hour of sleep (Table).4 This value is derived from a diagnostic sleep study, either performed at home or in a sleep laboratory.
Risk factors for obstructive sleep apnoea
Although obesity (body mass index [BMI] ≥30 kg/m2) stands as a leading contributor to OSA, it is far from the sole risk factor. The development and severity of OSA are influenced by a constellation of additional variables, both anatomical and nonanatomical. Genetic and racial predisposition, craniofacial features such as retrognathia or high-arched palate, and age all play a role. OSA has a higher prevalence in men, whereas its prevalence doubles for women after menopause.3,5
Functional predispositions, known as OSA endotypes, also contribute to the development of the disorder. These include reduced upper airway dilator muscle activity, reduced arousal threshold and increased loop gain for control of ventilation.6 Identifying a patient’s specific endotype may enable more personalised treatment approaches for OSA in the future.6
Together, these risk factors underscore the complex and multifactorial nature of OSA pathogenesis, shaping individualised approaches to prevention and treatment.
Obesity as a causative factor in obstructive sleep apnoea
The association between obesity and OSA is well established and multifaceted. According to the most recent data from the Australian Institute of Health and Welfare, about 31% of Australian adults aged 18 years and over are classified as obese, based on a BMI of 30 kg/m2 and over.7 Combining both overweight and obesity categories, around two-thirds (67%) of Australian adults fall into these classifications. Multiple epidemiological studies have shown that the prevalence of OSA increases in parallel with rising obesity rates.8 In adults, about 40% of individuals with OSA live with obesity.8
Longitudinal studies have confirmed that weight gain is a strong predictor of incident OSA, whereas weight loss is associated with significant improvements in OSA severity. For example, a 10% weight gain in adults can increase the risk of developing moderate-to-severe OSA by up to sixfold.9 Conversely, weight reduction through lifestyle modification, bariatric surgery or pharmacotherapy has been shown to improve the AHI, oxygen saturation and overall sleep quality.10
Multiple mechanistic pathways have been identified to explain how excess weight predisposes individuals to OSA. These are outlined in Box 1.11-15
Treatment of obstructive sleep apnoea
Weight management
Weight management continues to be a priority in the current OSA management guidelines, aiming to reduce disease severity and improve long-term health outcomes.16-18 Sustained weight loss requires intentional lifestyle modifications, particularly caloric restriction and increased physical activity. Bariatric surgery has been shown to induce significant weight reduction, leading to improvements in OSA severity and associated cardiometabolic sequelae.19,20 However, the inherent risks of surgery may mean that bariatric surgery is not appropriate for some patients. Furthermore, the improvement in AHI following bariatric surgery may not necessarily be superior to that following weight loss achieved by conventional weight loss programs.21
Continuous positive airway pressure
The use of continuous positive airway pressure (CPAP) has been regarded as the gold standard for normalising respiration as treatment for OSA.22 It normalises respiration by reducing the number of apnoeas and hypopnoeas during sleep, resulting in improvements in both the AHI and hypoxic burden. From the patient’s perspective, the use of CPAP has been correlated with improvement of symptoms such as daytime sleepiness and snoring. CPAP as treatment for OSA has also been linked to improvements in blood pressure control and cardiovascular and possible all-cause mortality, with these benefits contingent on ongoing nightly adherence.17,23-27 Yet, the nature of the mask and machine used in CPAP may be perceived as unnatural by patients, with up to about 50% ceasing CPAP after three years.28
For patients who continue with CPAP, there may be incomplete resolution of their apnoea for various reasons, such as suboptimal equipment upkeep. Concerns have been raised regarding defective positive airway pressure devices and the presence of magnets in masks, which may pose risks to other medical devices such as implanted pacemakers and defibrillators.29,30 Another consideration is that CPAP use alone does not necessarily result in weight loss or sufficient improvement in cardiometabolic risk factors.31,32
Incretins
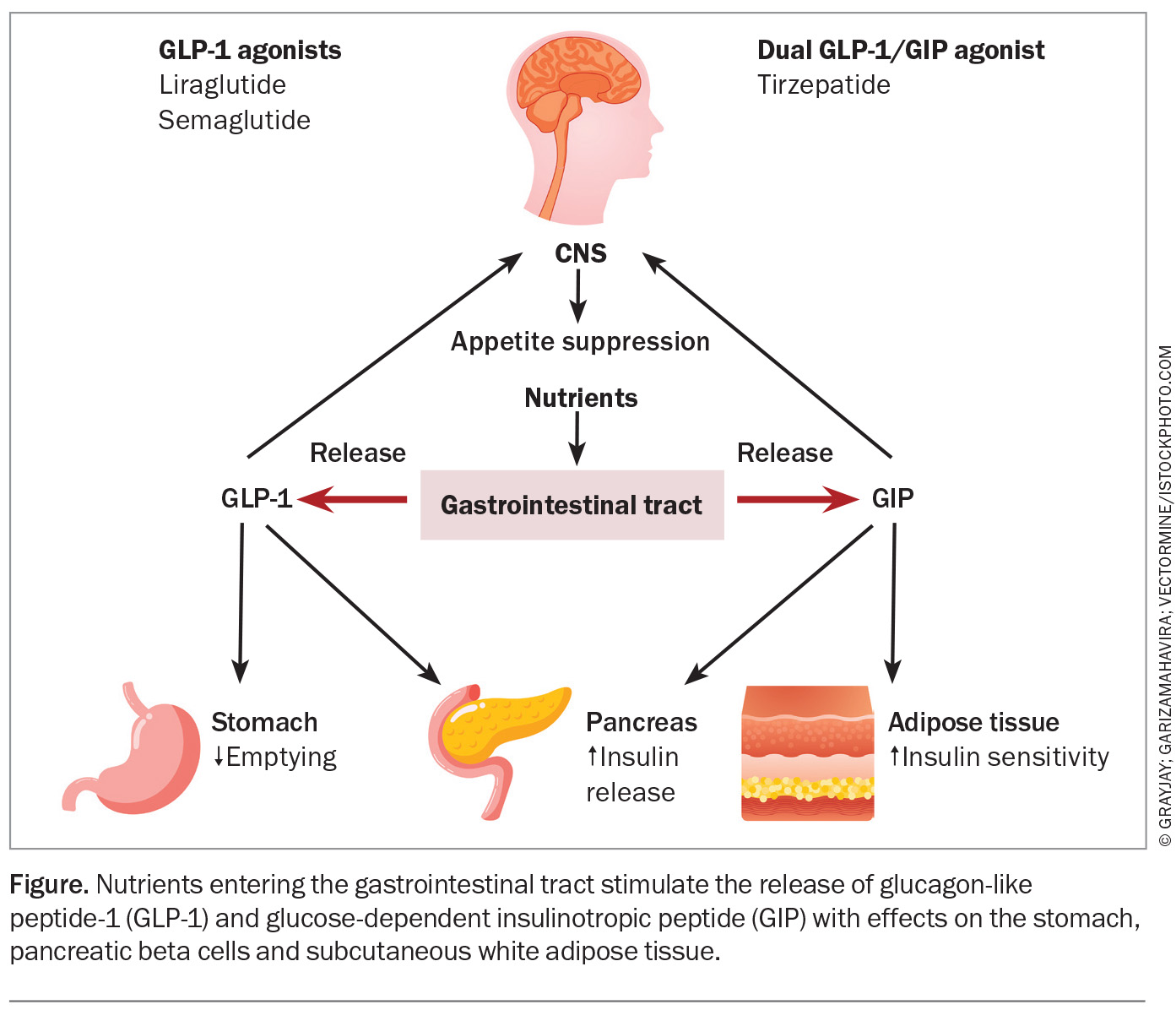
Numerous pharmacological agents have been developed for the management of obesity over the years. Hormones released from the gastrointestinal tract upon presentation of nutrients are known collectively as incretins. The two main incretins are glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP). The GLP-1 and GIP receptor agonists were originally developed for the management of type 2 diabetes through the potentiation of insulin secretion and other metabolic effects. However, they also had broader effects including delayed gastric emptying and suppression of appetite, contributing to the outcome of improved glycaemic control and sustained weight loss (Figure).
The GLP-1 receptor agonist liraglutide was approved in 2014 for the management of obesity in the USA and subsequently in Australia in 2015. More recently, the GLP-1 receptor agonist semaglutide 2.4 mg weekly dose was approved for the management of obesity in the USA in 2021 and in Australia in 2024.
In June 2025, the TGA broadened the indication for the GLP-1/GIP dual receptor agonist tirzepatide to include the treatment of moderate-to-severe OSA in adults with obesity. It previously had indications for the treatment of type 2 diabetes and chronic weight management. The additional indication was largely based on the findings of the SURMOUNT-OSA study published in 2024.1
For obesity-related OSA, weight reduction mediated by incretin-based treatments has been shown to improve the AHI with a mean change of 14.45 events per hour in a meta-analysis that included 1024 patients treated with liraglutide or tirzepatide for 12 weeks or more. Furthermore, the improvement for those receiving incretin-based treatment was greater compared with those receiving usual care by a mean of 11.6 events per hour.33
Exploring the role of tirzepatide
Tirzepatide is a novel, once-weekly injectable medication that acts as a dual agonist of the GLP-1 and GIP receptors. The SURMOUNT-OSA study used tirzepatide in two phase 3 double-blind randomised placebo-controlled trials in obese adults with moderate-to-severe OSA.1 Trial 1 consisted of participants who were not treated with CPAP (mean BMI 39.1 kg/m2, AHI 51.5 events per hour), whereas trial 2 consisted of participants who were treated with CPAP (mean BMI 38.7 kg/m2, AHI 49.5 events per hour). The study was not designed to compare tirzepatide against CPAP for the treatment of OSA, but did compare the effects of tirzepatide with placebo in both CPAP users and non-CPAP users with OSA and obesity. The primary endpoint for the study was the reduction in the AHI at 52 weeks.
In both trials, there were substantial improvements in the AHI and BMI for those allocated to tirzepatide dosed at 10 mg to 15 mg weekly compared with placebo after 52 weeks. Patients were commenced on tirzepatide 2.5 mg, and this was escalated every four weeks by 2.5 mg to a maximum tolerated dose of 10 mg or 15 mg weekly. The improvement in the AHI was 50.7% in trial 1 (no CPAP) and 58.7% in trial 2 (with CPAP), with 42.2% and 50.2% of participants in the respective trials reaching OSA remission (AHI <5 or AHI 5–14 with no sleepiness). The improvement in body weight for trial 1 and trial 2 was also substantial, with a 17.7% and 19.6% reduction, respectively. The measures of systolic blood pressure, systemic inflammation (as measured by high-sensitivity C-reactive protein levels), blood lipid profile, insulin resistance, AHI and event-specific hypoxic burden all improved for those treated with tirzepatide compared with placebo. Although improvements in the high-sensitivity C-reactive protein levels, insulin resistance and lipid profile were shown to be mediated by improvements in the AHI and hypoxic burden, the improvement in blood pressure control was not mediated by these factors. The implication of this finding is that treatment of both OSA and obesity in patients with a combination of both diseases is required for optimising cardiometabolic risk factors.34
Patient selection for tirzepatide therapy
Careful assessment of the patient’s entire clinical context is required when considering tirzepatide use in the context of OSA. The TGA indications for tirzepatide are presented in Box 2.
Arguably, the most pertinent factor in the consideration of tirzepatide use is the timing: how quickly does the desired outcome need to be achieved? The reduction in the BMI for those receiving tirzepatide and other incretin-based therapies is substantial but is not instantaneous. The reduction in the severity of OSA is also dependent on the amount of weight lost and the rate at which it is lost. It follows that the secondary benefits derived from substantial weight loss, such as improved blood pressure control, glycaemic control and cardiovascular morbidity, are not optimised until the excess adiposity is lost. Therefore, tirzepatide cannot be considered a replacement for CPAP or other core OSA therapies, but rather as an adjunct in cases where obesity is a contributing factor to OSA and where weight loss could meaningfully reduce disease severity.
The pros and cons of CPAP have previously been discussed, but it is clear that in individuals with excessive sleepiness caused by OSA, CPAP leads to a significant reduction in sleepiness.22 This becomes even more important as a factor for consideration, as the use of CPAP in adults with OSA has been demonstrated to significantly lower the rate of motor vehicle accidents. Thus, CPAP remains the initial treatment of choice in many cases of OSA. The addition of tirzepatide to CPAP for OSA treatment leads to clinical benefit after some time, given the period required to achieve the necessary weight loss. Once a clinically significant amount of weight has been lost, then re-evaluation of the need for CPAP should occur.
The use of tirzepatide may be the only option for individuals with OSA who refuse or are unable to adhere to CPAP in potentially mitigating the poor health outcomes associated with untreated OSA.
Potential limitations and considerations of tirzepatide
Weight loss sustainability
There may be concern regarding the sustainability of the weight loss achieved with medications such as tirzepatide as the benefits may persist only while therapy is maintained. The SURMOUNT-4 study examined the maintenance of the weight reduction achieved with tirzepatide.35 In this study of over 600 patients, a mean weight loss of 20.9% was achieved in the initial 36 weeks of tirzepatide use. Participants were then randomised to receive either a continuation of tirzepatide or placebo for a further 52 weeks. The vast majority (about 90%) of those randomised to tirzepatide were able to maintain at least 80% of the weight lost during the lead-in period compared with 17% of those who received placebo. This suggests that a maintenance dose is required to retain the degree of weight loss seen with the use of tirzepatide. As obesity is recognised as a chronic disease, the relevance of and need for maintenance medications for weight management is apparent.
Side effects
In the SURMOUNT-OSA study, there were few side effects associated with tirzepatide use.1 Most of the adverse events were related to gastrointestinal symptoms such as nausea, constipation, diarrhoea and discomfort from gastro-oesophageal reflux disease. These were generally self-limiting and occurred during drug initiation and titration. There were few withdrawals from the study when doses were increased every four weeks; seven out of 224 patients withdrew from trial 1 and 12 out of 223 patients withdrew from trial 2 (4% of the total number of participants).
Special precautions and warnings for use
There are few contraindications for tirzepatide use. Acute pancreatitis has been reported with tirzepatide use,36 and there are no data on its safety in patients with a history of pancreatitis.37 The use of GLP-1 receptor agonists should be avoided in those with a family history or personal history of medullary thyroid cancer or multiple endocrine neoplasia type 2, with rodent studies showing an increased risk.38
An update to the Product Information for all GLP-1 or dual GLP-1/GIP receptor agonists available in Australia highlights the risk of aspiration during anaesthesia or deep sedation, owing to their effect of delayed gastric emptying.39 Anaesthetic guidelines are available for reference (see: https://www.anzca.edu.au/safety-and-advocacy/standards-of-practice/endorsed-guidelines-and-joint-statements).
Muscle loss
Weight loss occurring with the use of GLP-1 receptor agonists, GLP-1/GIP dual receptor agonists and bariatric surgery is invariably associated with some muscle loss, and thus potentially leading to unintended outcomes such as loss of strength or increased likelihood of falls. Patients should be advised on the need to maintain their muscle mass through resistance-based exercises and to prioritise adequate protein intake.
Cost, access and insurance coverage
The implementation of tirzepatide as an ongoing medication for OSA and weight management may also be significantly influenced by cost, access and insurance coverage.
Conclusion
Obesity and OSA are intricately linked, with excess body weight serving as a major driver of OSA pathogenesis and severity. Effective weight management remains a crucial component of OSA therapy. CPAP is the present gold-standard treatment for OSA as it effectively reduces AHI, although long-term adherence is variable. CPAP therapy alone does not necessarily lead to weight reduction or substantial improvements in cardiometabolic risk factors. The GLP-1 and dual GLP-1/GIP receptor agonists have emerged as adjuncts to the present gold-standard treatment with CPAP.
Tirzepatide is now a viable therapeutic option for achieving sustained and significant weight loss that is sufficient to alter the course of OSA. Tirzepatide should not replace CPAP, but it is now another arrow in the quiver for use in OSA management. It raises the need for furthering education and collaboration among sleep medicine specialists, endocrinologists and primary care providers to facilitate a holistic approach to maximize patient outcomes. As our therapeutic arsenal expands, the hope is to provide more targeted, effective and sustainable solutions for those battling the dual burdens of obesity and OSA. MT
COMPETING INTERESTS: Dr Lee has a leadership or fiduciary role in the ASA Provision of CPAP Guideline Committee and the joint National Association of Testing Authorities and ASA Accreditation Advisory Committee for Sleep Disorders Services. Professor Yee has received speakers fees for Eli Lilly, TEVA and SomnoMed.
References
1. Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N Engl J Med 2024; 391: 1193-1205.
2. Benjafield AV, Ayas NT, Eastwood PR, et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med 2019; 7: 687-698.
3. Bonsignore MR, Saaresranta T, Riha RL. Sex differences in obstructive sleep apnoea. Eur Respir Rev 2019; 28: 190030.
4. Grigg-Damberger MM. The AASM Scoring Manual four years later. J Clin Sleep Med 2012; 8: 323-332.
5. Young T, Finn L, Austin D, Peterson A. Menopausal status and sleep-disordered breathing in the Wisconsin Sleep Cohort Study. Am J Respir Crit Care Med 2003; 167: 1181-1185.
6. Edwards BA, Landry SA, Thomson LDJ, Joosten SA. Sleep apnea endotypes and their implications for clinical practise. Sleep Med 2025; 126: 260-266.
7. Australian Institute of Health and Welfare (AIHW). Overweight and obesity. Canberra: Australian Government, AIHW; 2024. Available online at: https://www.aihw.gov.au/reports/overweight-obesity/overweight-and-obesity/contents/technical-notes (accessed September 2025).
8. Patel SR. The complex relationship between weight and sleep apnoea. Thorax 2015; 70: 205-206.
9. Peppard PE, Young T, Palta M, Dempsey J, Skatrud J. Longitudinal study of moderate weight change and sleep-disordered breathing. JAMA 2000; 284: 3015-3021.
10. Liu Y, Yang L, Stampfer MJ, Redline S, Tworoger SS, Huang T. Physical activity, sedentary behaviour and incidence of obstructive sleep apnoea in three prospective US cohorts. Eur Respir J 2022; 59: 2100606.
11. Davies R, Stradling. The relationship between neck circumference, radiographic pharyngeal anatomy, and the obstructive sleep apnoea syndrome. Eur Respir J 1990; 3: 509-514.
12. Schwartz AR, Gold AR, Schubert N, et al. Effect of weight loss on upper airway collapsibility in obstructive sleep apnea. Am Rev Respir Dis 1991; 144: 494-498.
13. Shah NM, Kaltsakas G. Respiratory complications of obesity: from early changes to respiratory failure. Breathe 2023; 19: 220263.
14. Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-α: direct role in obesity-linked insulin resistance. Science 1993; 259: 87-91.
15. Punjabi NM, Beamer BA. C-reactive protein is associated with sleep disordered breathing independent of adiposity. Sleep 2007; 30: 29-34.
16. Morgenthaler T, Kapen S, Lee-Chiong T, et al. Practice parameters for the medical therapy of obstructive sleep apnea. Sleep 2006; 29: 1031-1035.
17. Azarbarzin A, Sands SA, Stone KL, et al. The hypoxic burden of sleep apnoea predicts cardiovascular disease-related mortality: the Osteoporotic Fractures in Men Study and the Sleep Heart Health Study. Eur Heart J 2019; 40: 1149-1157.
18. Strobel RJ, Rosen RC. Obesity and weight loss in obstructive sleep apnea: a critical review. Sleep 1996; 19: 104-115.
19. Arterburn DE, Telem DA, Kushner RF, Courcoulas AP. Benefits and risks of bariatric surgery in adults: a review. JAMA 2020; 324: 879-887.
20. Al Oweidat K, Toubasi AA, Tawileh RBA, Tawileh HBA, Hasuneh MM. Bariatric surgery and obstructive sleep apnea: a systematic review and meta-analysis. Sleep Breath 2023; 27: 2283-2294.
21. Dixon JB, Schachter LM, O’Brien PE, et al. Surgical vs conventional therapy for weight loss treatment of obstructive sleep apnea: a randomized controlled trial. JAMA 2012; 308: 1142-1149.
22. Patil SP, Ayappa IA, Caples SM, Kimoff RJ, Patel SR, Harrod CG. Treatment of adult obstructive sleep apnea with positive airway pressure: an American Academy of Sleep Medicine systematic review, meta-analysis, and GRADE assessment. J Clin Sleep Med 2019; 15: 301-334.
23. Barbe F, Duran-Cantolla J, Capote F, et al. Long-term effect of continuous positive airway pressure in hypertensive patients with sleep apnea. Am J Respir Crit Care Med 2010; 181: 718-726.
24. Shafazand S, Patel SR. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension. J Clin Sleep Med 2014; 10: 341-343.
25. Drager LF, Malhotra A, Yan Y, et al. Adherence with positive airway pressure therapy for obstructive sleep apnea in developing vs. developed countries: a big data study. J Clin Sleep Med 2021; 17: 703-709.
26. Sanchez-de-la-Torre M, Gracia-Lavedan E, Benitez ID, et al. Adherence to CPAP treatment and the risk of recurrent cardiovascular events: a meta-analysis. JAMA 2023; 330: 1255-1265.
27. McEvoy RD, Antic NA, Heeley E, et al. CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med 2016; 375: 919-931.
28. Pepin JL, Bailly S, Rinder P, et al. CPAP therapy termination rates by OSA phenotype: a French nationwide database analysis. J Clin Med 2021; 10: 936.
29. Therapeutic Goods Administration (TGA). Philips recall action for CPAP, bi-level PAP devices and mechanical ventilators collection. Canberra: TGA; 2021.
Available online at: https://www.tga.gov.au/safety/information-about-specific-safety-alerts-and-recalls/philips-recall-action-cpap-bi-level-pap-devices-and-mechanical-ventilators-collection (accessed September 2025).
30. Therapeutic Goods Administration (TGA). CPAP and BiPAP face and nasal masks that contain magnets. Canberra: TGA; 2022. Available online at: https://www.tga.gov.au/news/safety-alerts/cpap-and-bipap-face-and-nasal-masks-contain-magnets (accessed September 2025).
31. Drager LF, Brunoni AR, Jenner R, Lorenzi-Filho G, Bensenor IM, Lotufo PA. Effects of CPAP on body weight in patients with obstructive sleep apnoea: a meta-analysis of randomised trials. Thorax 2015; 70: 258-264.
32. Chirinos JA, Gurubhagavatula I, Teff K, et al. CPAP, weight loss, or both for obstructive sleep apnea. N Engl J Med 2014; 370: 2265-2275.
33. Bardoczi A, Matics ZZ, Turan C, et al. Efficacy of incretin-based therapies in obesity-related obstructive sleep apnea: a systematic review and meta-analysis of randomized controlled trials. Sleep Med Rev 2025; 82: 102119.
34. Malhotra A, Grunstein R, Azarbarzin A, et al. Tirzepatide on obstructive sleep apnea-related cardiometabolic risk: secondary outcomes of the SURMOUNT-OSA randomized trial. Nat Med 2026; 32: 653-659.
35. Aronne LJ, Sattar N, Horn DB, et al. Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: the SURMOUNT-4 randomized clinical trial. JAMA 2024; 331: 38-48.
36. Mando N, Thomson E, Fowler M, Short L, Gillen N. Acute pancreatitis caused by tirzepatide. Cureus 2024; 16: e76007.
37. Chis BA, Fodor D. Acute pancreatitis during GLP-1 receptor agonist treatment. A case report. Clujul Med 2018; 91: 117-119.
38. Lisco G, De Tullio A, Disoteo O, et al. Glucagon-like peptide 1 receptor agonists and thyroid cancer: is it the time to be concerned? Endocr Connect 2023; 12: e230257.
39. Therapeutic Goods Administration (TGA). Medicines containing GLP-1 and dual GIP/GLP-1 receptor agonists. Canberra: TGA; 2025. Available online at: https://www.tga.gov.au/news/safety-updates/medicines-containing-glp-1-and-dual-gipglp-1-receptor-agonists (accessed September 2025).
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.