Not just gas: rethinking abdominal distension in clinical practice

Abdominal bloating and distension are common yet often misunderstood symptoms that cause major patient distress. Recognition of abdominophrenic dyssynergia as a key driver is shifting management away from reducing intestinal gas production or enhancing intestinal transit, and towards targeted biofeedback and breathing therapies.
- Abdominophrenic dyssynergia is a newly recognised underlying functional cause of abdominal bloating and distension, shifting the focus away from excess gas production as the primary cause.
- Visible distension results from maladaptive muscular responses, particularly paradoxical diaphragmatic contraction and abdominal wall relaxation, rather than increased intra-abdominal gas.
- Electromyography-guided biofeedback therapy demonstrates a 70 to 80% success rate in correcting abdominophrenic dyssynergia, representing a major therapeutic breakthrough.
- Simplified ‘chest up, abdomen in’ breathing techniques provide accessible alternatives to complex biofeedback, reinforcing that treatment should target the underlying dysfunction rather than focusing solely on gas reduction or transit enhancement.
- Future priorities include broader validation and accessibility, development of simplified protocols, integration with digital tools and multimodal strategies to personalise treatment and expand access beyond specialist centres.
Abdominal bloating and distension are among the most common and troublesome gastrointestinal symptoms encountered in clinical practice. Although these symptoms have traditionally been attributed to excess intestinal gas production, emerging research reveals a more complex pathophysiology centred on abnormal viscerosomatic responses. Here, the current and evolving understanding of abdominophrenic dyssynergia, a novel mechanistic explanation for functional causes of bloating, is discussed. The promising biofeedback and breathing techniques that are changing treatment approaches are also examined.
Burden and emerging mechanisms
Abdominal bloating affects nearly one in five individuals in the general population yet remains one of the most poorly understood gastrointestinal symptoms.1 The sensation of bloating, characterised by feelings of abdominal pressure, fullness and trapped gas, is frequently but not always accompanied by visible abdominal distension. The distinction between abdominal bloating and distension is summarised as:
- bloating – subjective sensation of increased abdominal pressure or fullness
- distension – objective, visible increase in abdominal girth.
These symptoms can significantly impact quality of life, with more than 75% of patients describing their symptoms as moderate to severe.1,2
For decades, the medical community assumed that bloating and distension result from increased intra-abdominal gas volumes. However, groundbreaking research from Barcelona has fundamentally challenged this paradigm, showing that patients experiencing severe bloating episodes often have no significant increase in intestinal gas content compared with healthy controls.3,4 Instead, a complex interplay between the gut–brain axis and somatic musculature appears to drive these debilitating symptoms.
Traditional understanding: the gas-centric model
The conventional approach to bloating has long focused on identifying and treating the presumed causes of excessive intestinal gas production and retention.
Microbiome-associated bacterial fermentation has been increasingly recognised as a primary pathogenic mechanism, with conditions such as small intestinal bacterial overgrowth and the microbiome alterations characteristic of irritable bowel syndrome representing specific manifestations of this process. Fermentation of unabsorbed carbohydrates yields hydrogen, methane and carbon dioxide, which may contribute to symptom generation. In parallel, carbohydrate malabsorption (including lactose intolerance, fructose malabsorption and sensitivity to fermentable oligosaccharides, disaccharides, monosaccharides and polyols) is proposed to exacerbate symptoms through both osmotic effects and colonic fermentation.5,6
Delayed intestinal transit, whether idiopathic or medication-induced, was previously believed to result in gas accumulation and subsequent distension.7 The logic was that slower transit allows more time for bacterial fermentation and reduces the ability to expel gas.
The observation that some patients develop severe symptoms despite normal intestinal gas volumes led to the concept of heightened visceral perception and central sensitisation to physiologic gas loads.7 This visceral hypersensitivity model suggested that the problem lay not in gas quantity but in the brain’s interpretation of normal intestinal sensations.
The limitations of traditional thinking
Although the mechanisms described above undoubtedly contribute to symptoms in some patients, several observations challenged the gas-centric paradigm:
- the patients with the most dramatic visible distension often showed no correlation between symptom severity and measured gas volumes4,8
- rapid onset of distension (within minutes of eating) occurred too quickly to be explained by bacterial fermentation
- anti-gas treatments (e.g. simethicone, activated charcoal) showed limited efficacy in controlled trials9
- dietary restrictions targeting gas production provided only partial relief for many patients.
Most puzzling was the observation that some patients could develop significant abdominal distension after consuming foods they believed would cause bloating, even when those foods were unlikely to produce excess gas.10 This suggested that psychological and neurological factors played a more prominent role than previously recognised.
The pathophysiology revolution: abdominophrenic dyssynergia
Redefining the mechanism
The classic model of bloating attributed symptoms primarily to excessive gas production resulting from bacterial fermentation of undigested carbohydrates.5 Although this mechanism likely contributes in a subset of patients, advanced CT imaging studies have revealed a more complex pathophysiology.4 Patients with disorders of gut–brain interaction which experience severe bloating exhibit a paradoxical viscerosomatic pattern, described as abdominophrenic dyssynergia.11
In contrast to these patients, healthy individuals respond to increased intra-abdominal content through a co-ordinated accommodation reflex: the diaphragm relaxes and moves upwards while the anterior abdominal muscles contract. This physiological adjustment permits vertical expansion of the abdominal cavity, minimising anterior protrusion of the abdominal wall.12
What is abdominophrenic dyssynergia?
Abdominophrenic dyssynergia represents a complete reversal of the normal accommodation pattern.11 When patients with functional gastrointestinal disorders experience intestinal stimuli, whether from normal food intake, gas or even the anticipation of symptoms, they exhibit:
- paradoxical diaphragmatic contraction and descent
- simultaneous relaxation of the internal oblique and anterior abdominal muscles
- abnormal caudoventral redistribution of abdominal contents.
This maladaptive response drives visible abdominal distension without any increase in actual intra-abdominal volume.8 The descent of the diaphragm pushes abdominal contents downwards and forwards, while the relaxed abdominal wall fails to provide the necessary muscular tone to contain this redistribution.
Figure 1 illustrates the normal accommodation response to intestinal stimuli and the paradoxical reaction seen in abdominophrenic dyssynergia.3
Beyond the abdomen: thoracic consequences
The implications of abdominophrenic dyssynergia extend beyond abdominal symptoms. The paradoxical diaphragmatic descent is accompanied by compensatory elevation of the costal wall, driven by intercostal muscle contraction.11 This response functionally mimics the chest wall mechanics seen in severe asthma, explaining why patients with significant abdominal distension often report associated dyspnoea, chest tightness and an inability to inhale deeply.
Association with disorders of gut–brain interaction
Disorders of gut–brain interaction, formerly known as functional gastrointestinal disorders, include conditions such as irritable bowel syndrome, functional dyspepsia and functional abdominal bloating or distension, and are so named because their defining feature is impaired bidirectional communication between the gut and brain via the nervous system.
Owing to a paucity of studies, the true prevalence of abdominophrenic dyssynergia remains unknown. However, given that disorders of gut–brain interaction affect around 40% of the global population, and that abdominophrenic dyssynergia has been documented in association with these conditions, it is likely to be both common and under-recognised.9 Importantly, abdominophrenic dyssynergia should not be regarded as a distinct functional disease entity, but rather as a mechanistic explanation for nonorganic causes of bloating.
Overlaps among disorders of gut–brain interaction are common, with many patients fulfilling diagnostic criteria for multiple such disorders, reflecting the shared pathophysiological mechanisms and multifactorial nature of these conditions.12 Although abdominal bloating is a common feature across multiple gastrointestinal disorders, its prominence and underlying mechanisms vary.
There is also potential overlap between disorders of gut–brain interaction and established organic gastrointestinal diseases, such as inflammatory bowel disease, illustrating that functional mechanisms can coexist with structural pathology. At present, clinicians are largely guided by the Rome IV criteria, which allow for symptom-based categorisation of disorders of gut–brain interaction. The Rome IV diagnostic criteria for functional abdominal bloating or distension (a disorder of gut–brain interaction where bloating is the predominant symptom), are shown in Table 1.13 Although useful for classification, these criteria do not elucidate the underlying mechanisms driving the individual patient’s bloating symptoms, highlighting the need for mechanistic understanding to complement symptom-based diagnosis.
Identifying abdominophrenic dyssynergia in practice
In clinical practice, abdominophrenic dyssynergia may be suggested by a pattern of features that help distinguish it from other mechanisms of bloating, including:
- postprandial symptom onset – distension often occurs within minutes of eating, regardless of meal composition
- visual prominence – the distension is typically dramatic and easily visible
- behavioural triggers – symptoms may be precipitated by anticipation or stress
- associated respiratory symptoms – chest tightness and shortness of breath during episodes; patients often describe an inability to take a deep breath
- diurnal variation – symptoms often worsen throughout the day and improve overnight.
The role of expectation
Perhaps one of the most intriguing aspects of abdominophrenic dyssynergia is its susceptibility to psychological triggers. Studies have shown that patients can develop significant abdominal distension simply by consuming foods they believe will cause bloating, even when those foods are physiologically unlikely to produce gas.10 This finding underscores the significant influence of gut–brain axis interactions in driving the condition.
When to investigate further
The approach to investigation should be guided by clinical presentation, patient age and associated symptoms, with targeted testing preferred over extensive evaluation. A wide range of conditions may be associated with bloating and abdominal distension, as shown in Box 1 and Box 2.1 Patients should be assessed for red-flag features that may indicate organic disease, and these are listed in Box 3.
Although the exclusion of organic pathology remains an important step, it is equally important to recognise that disorders of gut–brain interaction may present alongside established organic disease, with the two frequently coexisting. Understanding the mechanisms underlying functional bloating and distension therefore requires consideration of both structural and functional contributors.
The treatment revolution: targeting the true mechanism
Targeted retraining of diaphragmatic and abdominal muscle co-ordination can restore physiological balance and relieve abdominal distension, as abdominophrenic dyssynergia arises when abnormal postural and muscular patterns override the normal reflex accommodation of the abdominal wall. In this way, the phenomenon is best understood not as a distinct disease entity but as a maladaptive, behaviour-conditioned response that can be corrected through re-establishing proper muscle dynamics.14
Electromyography-guided biofeedback: the gold standard
The most significant advancement in treating abdominophrenic dyssynergia has been the development of electromyography (EMG)-guided biofeedback therapy.15 This sophisticated technique enables patients to observe the real-time activity of their diaphragmatic and abdominal wall muscles, giving them immediate feedback on their viscerosomatic responses.
The biofeedback protocol involves:
- real-time EMG monitoring of diaphragmatic and intercostal muscle activity
- visual feedback allowing patients to observe their muscle activation patterns
- targeted training to reduce diaphragmatic and intercostal muscle activity during episodes
- co-ordination exercises to restore normal abdominophrenic synchrony.
Clinical trials have demonstrated notable success rates, with 70 to 80% of patients achieving significant symptom improvement.15,16 Patients learn to consciously override the dyssynergic response, leading to measurable reductions in abdominal girth and subjective symptom scores.
The ability to demonstrate objective improvements in both abdominal girth measurements and validated symptom scores offers concrete, quantifiable evidence supporting the efficacy of this intervention. Such measurable outcomes not only reinforce the clinical relevance of the treatment but also provide a robust framework for assessing therapeutic benefit, enabling comparison across studies and guiding patient management in a more evidence-based manner.
Although promising results have been reported from a single European centre,14 EMG-guided biofeedback is not yet widely available outside this setting and is currently not available in Australia.
Making treatment accessible with simplified breathing techniques
While EMG-guided biofeedback represents the therapeutic ideal, its complexity and limited availability have prompted the development of simplified breathing techniques that target the same underlying mechanisms.
The ‘chest up, abdomen in’ technique
The approach to take a breath in with the ‘chest up’ and ‘abdomen in’ teaches patients to counteract abdominophrenic dyssynergia through co-ordinated movement. Unlike normal diaphragmatic breathing, it is a somewhat counterintuitive retraining manoeuvre that consists of the following:
- an inspiration phase – gentle elevation of the chest wall (costal expansion) combined with conscious abdominal wall contraction
- an expiration phase – controlled chest wall relaxation combined with slow abdominal wall relaxation (while attempting to maintain abdominal tone).
Figure 2 illustrates this technique.14 Patients are advised to practise the technique for about 5 minutes before and after meals, as well as during symptomatic episodes.14
Diaphragmatic re-education
Although traditional diaphragmatic breathing exercises are beneficial for anxiety and certain respiratory conditions, they need to be modified for effective management of abdominophrenic dyssynergia. The adaptations include:16
- reverse pattern training – teaching patients to avoid excessive diaphragmatic descent
- abdominal wall strengthening – progressive exercises to improve anterior wall tone
- co-ordination drills – practising the opposition between diaphragmatic and abdominal wall activity.
Practical implementation in clinical practice
A structured framework can guide the assessment of patients presenting with abdominal bloating and distension. Comprehensive symptom characterisation is the first step, with careful attention paid to triggers, timing and associated features. Exclusion of organic disease remains essential, using targeted investigations guided by the clinical presentation. Functional assessment should also be undertaken, with evaluation for associated disorders of gut–brain interaction, given the frequent overlap between these conditions. Finally, psychosocial screening is important to identify relevant comorbidities, such as anxiety, depression or a history of trauma, which may influence symptom burden and treatment response.
Treatment algorithm
Management of abdominophrenic dyssynergia is best approached through a stepwise algorithm.
- First-line interventions:
– patient education about abdominophrenic dyssynergia and the role of gut–brain mechanisms
– instruction in simple breathing techniques as outlined above
– basic dietary modifications, such as a trial of the low Fermentable Oligosaccharides, Disaccharides, Monosaccharides And Polyols diet. - Second-line therapies, if symptoms persist:
– structured breathing therapy with physiotherapy input
– targeted pharmacotherapy with tricyclic antidepressants or gabapentinoids
– psychological interventions such as cognitive behavioural therapy or gut-directed hypnotherapy. - Specialist referral may be warranted for certain patients:
– those with complex comorbid conditions
– those whose symptoms remain refractory despite first- and second-line measures.
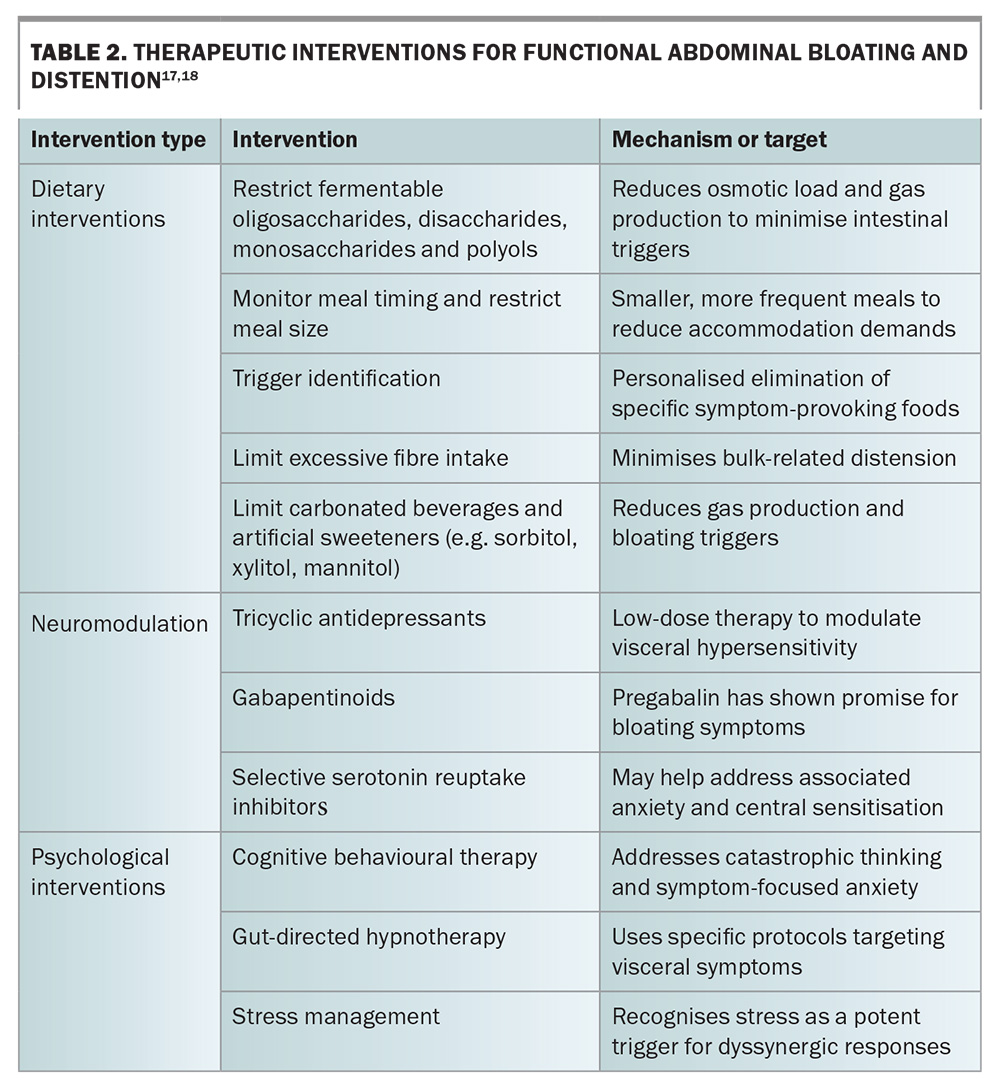
Successful treatment often requires a multimodal approach addressing multiple pathophysiological contributors, as summarised in Table 2.17,18
Future directions and research opportunities
The recognition of abdominophrenic dyssynergia as a central mechanism underlying functional bloating represents a significant conceptual advance in gastroenterology. However, progress in this field is hindered by the absence of standardised diagnostic criteria that can be applied reliably across diverse clinical settings. Current research protocols largely depend on advanced EMG and imaging methods; although these are highly informative, they remain restricted to specialist centres. This highlights the need for pragmatic diagnostic tools suitable for routine practice.
Ultrasound-based assessment of diaphragmatic movement shows particular promise, offering a relatively accessible and cost-effective means of visualising paradoxical diaphragmatic activity in real time. In parallel, EMG-guided diaphragmatic re-education targeting the internal oblique muscle has emerged as an innovative therapeutic approach. Although early findings are encouraging, this strategy remains in its infancy and requires further clinical validation in larger, more diverse populations.
Optimising treatment pathways represents another important research priority. Simplified biofeedback protocols that preserve therapeutic efficacy and are feasible for delivery in home or community settings could greatly expand access to this highly effective intervention. Equally important is the systematic evaluation of breathing techniques alongside pharmacological and psychological therapies in rigorously designed combination trials. Such studies would provide the foundation for evidence-based multimodal treatment strategies, particularly for patients with complex or refractory symptoms. Identifying predictors of treatment response is also essential to move towards personalised management algorithms that maximise benefit while minimising treatment burden.
Technological innovation offers exciting opportunities to transform the clinical management of abdominophrenic dyssynergia and functional bloating. Smartphone applications capable of delivering real-time breathing guidance and symptom tracking could extend biofeedback principles directly into patients’ homes. Such tools may be especially valuable for patients living in rural regions. Wearable devices that monitor respiratory dynamics and abdominal girth during daily life could provide novel insights into triggers and treatment responses, whereas telemedicine platforms purpose-built for biofeedback training could help overcome barriers of geography and specialist availability. Together, these approaches can improve access to advanced therapies and embed abdominophrenic dyssynergia management into routine clinical practice.
Evidence to date, although limited, is encouraging. Three randomised controlled trials conducted at a single centre and enrolling about 100 patients in total, demonstrated that biofeedback guided by EMG or inductance plethysmography with adaptable belts improved both physiological co-ordination and patient-reported symptoms.14-16 Participants were able to modulate abdominothoracic muscular activity, and correction of postural tone restored abdominal girth to baseline levels. Notably, the benefits persisted beyond treatment sessions, with reduced distension observed during a subsequent 10-day evaluation period. An ancillary uncontrolled observation suggested continued improvement at six months, potentially reflecting sustained exercise practice.
Nevertheless, caution is warranted in interpreting these results. The trials were modest in size, conducted in a single research centre and excluded patients with abdominal distension in the absence of constipation, as well as those taking neuromodulators, limiting the generalisability of their findings. Additionally, some individuals with structural postural abnormalities, such as pronounced dorsal kyphosis or forward shoulder positioning, may require tailored interventions beyond standard training protocols.14 Addressing these limitations through larger multicentre studies with broader inclusion criteria will be essential to firmly establish the role of biofeedback and related interventions in clinical practice.
Implications for clinical practice
The recognition of abdominophrenic dyssynergia as a primary mechanism underlying functional bloating represents a paradigm shift with profound implications for patient care. Rather than focusing solely on reducing gas production or enhancing intestinal transit, clinicians can now offer targeted interventions that address the root cause of some bloating symptoms.
This mechanistic understanding also provides patients with a rational explanation for their experiences, potentially reducing the frustration and uncertainty that often accompanies disorders of gut–brain interaction. The ability to demonstrate objective improvements in abdominal girth measurements and symptom scores provides tangible evidence of treatment efficacy.
Conclusion
The recognition of abdominophrenic dyssynergia as a unifying explanation for functional bloating and distension represents one of the most significant advances in functional gastroenterology in recent years. By moving beyond simplistic gas-centric models, clinicians can now offer patients mechanistically driven treatments that target the true underlying pathophysiology.
The development of biofeedback and breathing techniques specifically aimed at abdominophrenic dyssynergia offers genuine hope for patients with these debilitating symptoms. For gastroenterologists, the challenge lies in translating this sophisticated mechanistic understanding into practical, accessible interventions that can be delivered across diverse healthcare settings. The potential to transform the lives of patients affected by these common yet poorly understood symptoms has never been greater. MT
COMPETING INTERESTS: None.
References
1. Jiang X, Locke GR, Choung RS, et al. Prevalence and risk factors for abdominal bloating and visible distension: a population-based study. Gut 2008; 57: 756-763.
2. Ringel Y, Williams RE, Kalilani L, et al. Prevalence, characteristics, and impact of bloating symptoms in patients with irritable bowel syndrome. Clin Gastroenterol Hepatol 2009; 7: 68-72.
3. Damianos JA, Tomar SK, Azpiroz F, Barba E. Abdominophrenic dyssynergia: a narrative review. Am J Gastroenterol 2023; 118: 41-45.
4. Accarino A, Perez F, Azpiroz F, et al. Abdominal distention results from caudo-ventral redistribution of contents. Gastroenterology 2009; 136: 1544-1551.
5. Staudacher HM, Whelan K, Irving PM, et al. Comparison of symptom response following advice for a diet low in fermentable carbohydrates (FODMAPs) versus standard dietary advice in patients with irritable bowel syndrome. J Hum Nutr Diet 2011; 24: 487-495.
6. Halmos EP, Power VA, Shepherd SJ, et al. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 2014; 146: 67-75.
7. Agrawal A, Whorwell PJ. Review article: abdominal bloating and distension in functional gastrointestinal disorders – epidemiology and exploration of possible mechanisms. Aliment Pharmacol Ther 2008; 27: 2-10.
8. Tremolaterra F, Villoria A, Azpiroz F, et al. Impaired viscerosomatic reflexes and abdominal-wall dystony associated with bloating. Gastroenterology 2006; 130: 1062-1068.
9. Lacy BE, Cangemi D, Vazquez-Roque M. Management of chronic abdominal distension and bloating. Clin Gastroenterol Hepatol 2021; 19: 219-231.
10. Barba E, Sanchez B, Burri E, et al. Abdominal distension after eating lettuce: the role of intestinal gas evaluated in vitro and by abdominal CT imaging. Neurogastroenterol Motil 2019; 31: e13703.
11. Villoria A, Azpiroz F, Burri E, et al. Abdomino-phrenic dyssynergia in patients with abdominal bloating and distension. Am J Gastroenterol 2011; 106: 815-819.
12. Burri E, Cisternas D, Villoria A, et al. Accommodation of the abdomen to its content: integrated abdomino-thoracic response. Neurogastroenterol Motil 2012; 24: 312-e162.
13. Rome Foundation. Rome IV diagnostic criteria for disorders of gut-brain interaction (DGBI). Raleigh: Rome Foundation; 2016. Available online at: https://theromefoundation.org/rome-iv/rome-iv-criteria/ (accessed September 2025).
14. Barba E, Livovsky DM, Accarino A, Azpiroz F. Thoracoabdominal wall motion-guided biofeedback treatment of abdominal distention: a randomized placebo-controlled trial. Gastroenterology 2024; 167: 538-546.e1.
15. Barba E, Burri E, Accarino A, et al. Abdominothoracic mechanisms of functional abdominal distension and correction by biofeedback. Gastroenterology 2015; 148: 732-739.
16. Barba E, Accarino A, Azpiroz F. Correction of abdominal distention by biofeedback-guided control of abdominothoracic muscular activity in a randomized, placebo-controlled trial. Clin Gastroenterol Hepatol 2017; 15: 1922-1929.
17. Ford AC, Moayyedi P, Chey WD, et al. American College of Gastroenterology monograph on management of IBS. Am J Gastroenterol 2018; 113: 1-18.
18. Fond G, Loundou A, Hamdani N, et al. Anxiety and depression comorbidities in irritable bowel syndrome (IBS): a systematic review and meta-analysis. Eur Arch Psychiatry Clin Neurosci 2014; 264: 651-660.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.

-Abraham-Not just gas.jpg)
-Abraham-Not just gas.jpg)



-Abraham-Not just gas.jpg)