New pharmacological treatments for obesity: incretin analogues, their mechanism of action, efficacy and safety

Incretin analogues are very effective weight loss medications resulting in weight loss approaching that achieved with bariatric surgery. They also have cardiovascular benefits. However, these treatments are costly and their availability has been limited because of overdemand.
- Obesity increases the risk of disease, including serious cardiometabolic disease.
- Incretin analogue treatment results in weight loss with a substantial reduction in cardiovascular risk factors. The newer agents have been associated with weight loss of 15 to 20% of bodyweight, which approaches that seen with bariatric surgery.
- For the first time, a pharmacological obesity treatment, semaglutide (at a weekly dose of 2.4 mg), has been shown to reduce major adverse cardiovascular events in people with obesity and established cardiovascular disease.
- Incretin analogues also show beneficial effects in heart failure, peripheral vascular disease, renal disease, obstructive sleep apnoea and metabolic dysfunction-associated steatohepatitis and have potential neuroprotective properties.
- Incretin analogues are an increasingly important class of drugs that provide substantial health benefits for people with obesity, with adverse effects limited primarily to gastrointestinal disturbances. Equity of access is a problem because these medications are costly and there have been global shortages because of overdemand.
Obesity and its associated medical complications are a major problem in Australia and worldwide. In 2022 to 2023, 26.4% of children and adolescents aged 2 to 17 years and 65.8% of adults (aged 18 years and above) in Australia were classified as having overweight or obesity.1 Obesity increases the risk of disease, including serious cardiometabolic disease, and prevention is key, as sustained long-term weight loss is rare once obesity is established.
Bariatric surgery was previously the most effective and durable weight-loss intervention for obesity and has been associated with excellent health outcomes. Incretin-based pharmacotherapy now offers a medical alternative, resulting in a 15 to 20% body weight loss – close to that achieved with bariatric surgery – and providing considerable cardiovascular benefit. Weight loss is sustained while patients remain on incretin treatment. However, these treatments are costly and their popularity has resulted in shortages, creating stress and uncertainty for people with obesity.
What are incretins and how do they result in weight loss?
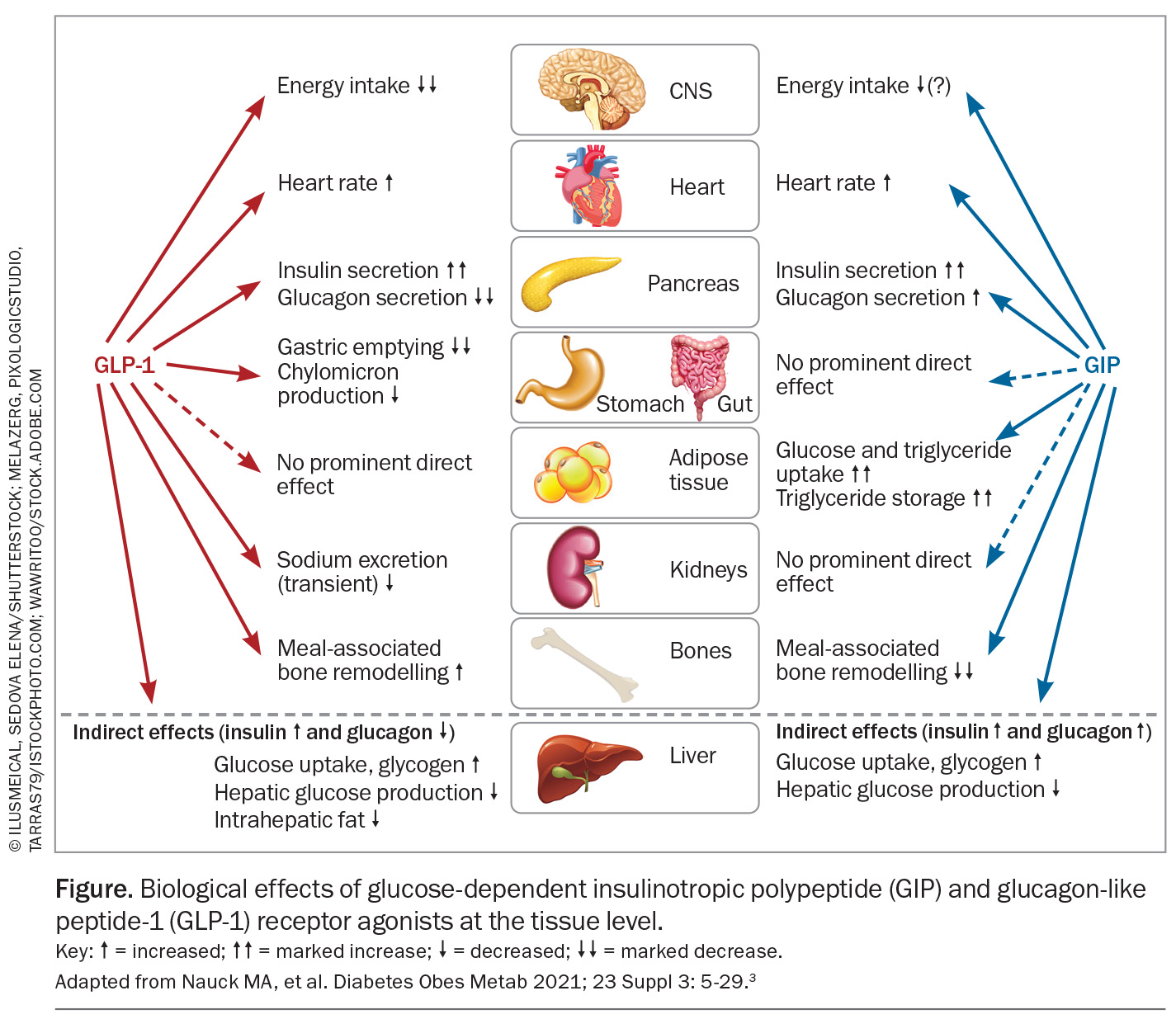
Incretins are hormones produced in the gastrointestinal tract that are rapidly secreted in response to a meal. They communicate nutrient intake to systems that regulate postprandial homeostasis, including the stimulation of pancreatic insulin release in a glucose-dependent manner. This is known as the incretin effect, and it occurs after oral but not intravenous administration of glucose. In addition, incretins regulate gastric motility, nutrient absorption, blood flow and food intake.2 The two dominant incretins are glucagon-like peptide-1 (GLP-1), secreted by enteroendocrine L cells (located in the ileum and colon), and glucose-dependent insulinotropic peptide (GIP), secreted by K cells (located mainly in the duodenum). These hormones are rapidly degraded by dipeptidyl peptidase-4 and neutral endopeptidase 24.11, with renal clearance resulting in a short half-life of two minutes.3 GLP-1 is also produced in cerebral preproglucagon neurons, where it is released locally and acts as a neurotransmitter. Peripheral and central GLP-1 systems appear to be separate and act independently.4
Of the two dominant incretins, GIP has been shown to have a greater effect on insulin release following oral glucose ingestion in healthy humans. At euglycaemia or hyperglycaemia, GLP-1 suppresses glucagon secretion. When blood glucose levels are 5 mmol/L or less, GLP-1 has no effect, and may even increase glucagon secretion. In contrast, GIP stimulates glucagon secretion, with enhanced activity at lower glycaemic levels.
GLP-1 and GIP receptors are widespread in humans with abundant levels in pancreatic beta and, to a lesser degree, alpha cells. They have also been found in subcutaneous and visceral adipose tissue, as well as the heart, lungs, kidneys, blood vessels, bone and the gastrointestinal tract. In rodents and nonhuman primates, receptors have also been found in brain tissue involved in appetite regulation, satiety, energy intake and expenditure (i.e. hypothalamus and brain stem nuclei), as well as regions involved in synaptic plasticity, memory, reward functions and emotional responses (Figure).3
Effects of incretins on the gastrointestinal tract
Gut-released GLP-1 acts as a hormone and binds primarily to receptors in the gastrointestinal system. It also acts on vagal afferent neurons that signal the brain on gut nutrient status. Physiological and pharmacological doses of GLP-1 slow gastric emptying, but GIP has no such effect. Although tachyphylaxis to this effect may occur with long-acting GLP-1 receptor agonists, GLP-1-associated slowing of gastric emptying does not appear to be responsible for the nausea and vomiting often reported with the use of GLP-1 receptor agonist treatment. High doses of GLP-1 receptor agonist resulting in complete cessation of gastric emptying are not usually associated with gastrointestinal symptoms, which appear to be centrally mediated.3
In slowing gastric emptying, gastric acid and pancreatic exocrine secretion are also reduced by GLP-1. GLP-1 and GLP-1 receptor agonists also reduce intestinal motility and this may affect nutrient absorption. These effects have been referred to as the ‘ileal brake’, which signals the body to stop eating and cease digestion and nutrient absorption. This may contribute to the diarrhoea sometimes reported with GLP-1 receptor agonist treatment.
Energy intake and expenditure
Physiological plasma levels of GLP-1 and GIP do not affect appetite. However, at pharmacological concentrations, GLP-1 and possibly GIP reduce appetite and prospective food intake, and increase satiety.5 GLP-1 receptor agonists can stimulate GLP-1 receptors in the area postrema, a circumventricular organ in the lower brainstem that has fenestrated capillaries and can sense hormonal signals in the general circulation, and appetite centres in the hypothalamus, which have fenestrated capillaries in the median eminence. The area postrema is responsible for emesis and nausea and is likely the main contributor to these GLP-1 receptor agonist-associated side effects. Peripherally administered liraglutide is taken up by the hypothalamus (arcuate and paraventricular nuclei) where it stimulates pro-opiomelanocortin and amphetamine-regulated transcript to reduce appetite and induce weight loss.6 However, GLP-1 receptor agonists and gut-derived GLP-1 do not cross the blood–brain barrier and cannot access GLP-1 receptors in the brain parenchyma, which can only be accessed by brain (pre-proglucagon neuron)-derived GLP-1.4 Paradoxically, GIP-receptor knockout mice studies showed that, when fed a high-fat diet, the mice were resistant to weight gain.4
Based on animal studies, it has been suggested that antagonism of the GIP receptor may enhance GLP-1 receptor activity, whereas chronic GIP-receptor agonism may desensitise GIP-receptor activity mimicking antagonism.2 However, other animal studies suggest central GIP-receptor stimulation reduces food intake and body weight.7 At physiological or pharmacological doses, GLP-1 has not been shown to affect energy expenditure.3 However, at pharmacological doses, GIP agonism appears to increase fat oxidation by promoting futile calcium cycling, thereby diminishing the metabolic adaptation often observed with weight loss.8,9
Incretin therapies currently available in Australia
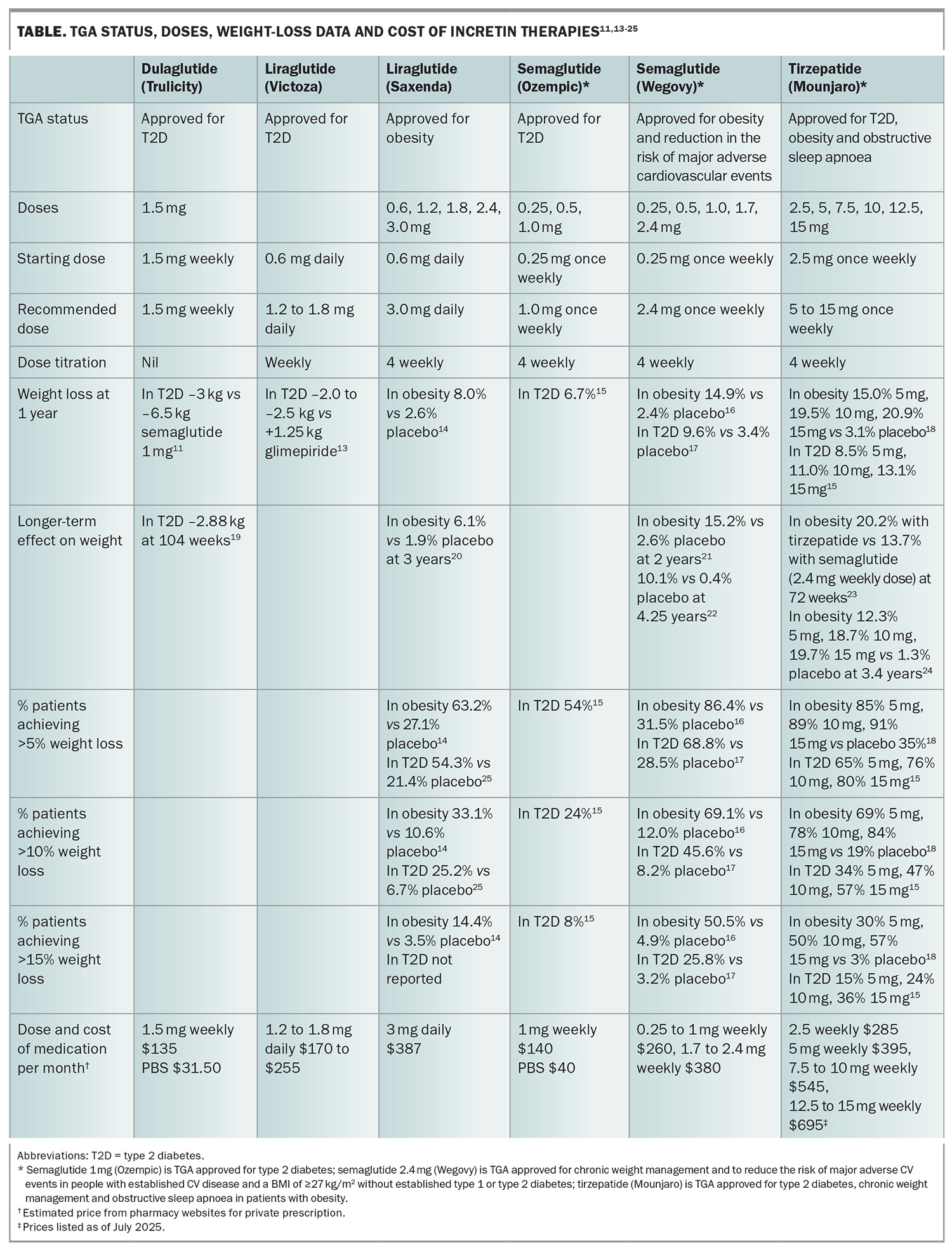
Dulaglutide, liraglutide and semaglutide – synthetic versions of human GLP-1 – and tirzepatide, a single-molecule dual GLP-1 and GIP receptor coagonist, are available in Australia. Liraglutide, semaglutide (2.4 mg weekly dose) and tirzepatide have been trialled for the management of obesity. Although dulaglutide has not been studied as an obesity treatment, it results in modest weight loss in people with type 2 diabetes, with head-to-head trials showing it to be less effective than liraglutide 1.8 mg, semaglutide 1.0 mg and tirzepatide.10-12
In Australia, liraglutide, semaglutide (2.4 mg weekly dose) and tirzepatide are TGA approved for weight management in adults. Saxenda, one brand of liraglutide, will be discontinued by the manufacturer at the end of 2025. Semaglutide (2.4 mg weekly dose) is also approved for use in adolescents aged 12 years and older with obesity. All these medications are administered by subcutaneous injection into the abdomen, upper arm or thigh. Their dosing, weight loss efficacy and cost are summarised in the Table.11,13-25 As with other weight loss treatments, people with type 2 diabetes tend to lose less weight than those without type 2 diabetes.15-18 These agents appear to be equally effective in patients who have undergone bariatric surgery.26
Body composition changes
Incretin analogues produce substantial weight loss approaching that achieved with bariatric surgery. Weight loss inevitably involves loss of both adipose tissue and lean tissue. However, treatment with these agents results in relatively greater fat loss, including visceral fat reduction, leading to an increased proportion of lean mass to total body mass.16,27
Efficacy in children and adolescents
Liraglutide 3 mg daily for one year was well tolerated in adolescents with obesity and resulted in a 4.6% reduction in body mass index (BMI) compared with placebo.28 In children aged six to 12 years with nonsyndromic obesity, liraglutide 3 mg daily for one year produced a 7.4% reduction in BMI compared with placebo.29 In adolescents with obesity, semaglutide 2.4 mg once weekly led to weight loss similar to that observed in adults (a 16.1% mean reduction in BMI from baseline vs 0.6% with placebo at 68 weeks), along with improvements in cardiometabolic risk factors such as waist circumference, glycated haemoglobin (HbA1c), LDL-cholesterol, triglyceride and alanine transaminase levels.30
What dose should be used?
For glycaemic control in people with type 2 diabetes, liraglutide doses above 1.8 mg once daily provide no substantial additional benefit. Similarly, semaglutide doses above 1 mg once weekly have a negligible effect on glycaemic control in most people with type 2 diabetes. However, tirzepatide demonstrates a dose-dependent effect on glycaemia: at 5 mg once weekly, 27% of people with type 2 diabetes achieved an HbA1c below 5.7%, compared with 46% at 15 mg once weekly.15 Although high doses generally result in greater weight loss, the incremental benefit diminishes beyond 10 mg once weekly.
The recommended dose of liraglutide for the management of type 2 diabetes is 1.2 to 1.8 mg daily, whereas the dose for weight loss is 3 mg daily. For semaglutide, the dose for type 2 diabetes is 1 mg once weekly, compared with 2.4 mg once weekly for weight loss. Tirzepatide’s maximum weekly dose is 15 mg for both type 2 diabetes and obesity. Clinically, many patients do not require the maximum recommended dose to achieve satisfactory weight loss. We recommend titrating the dose of an incretin analogue to the level required for adequate appetite control and weight loss, increasing only after the current dose is well tolerated. If a patient is experiencing side effects, dose escalation should be slower, and if severe, the dose should be reduced back to the dose that was tolerated. Patients should maintain that dose for about one month before attempting to increase the dose again.
How long should these medications be used?
Weight regain occurs when incretin analogues are discontinued. This was well demonstrated in a trial where participants taking semaglutide 2.4 mg weekly for weight loss regained two-thirds of their prior weight loss within a year of stopping the medication.31 This is unsurprising as, like nearly all medications, these agents are effective only while in use and lose efficacy once ceased. Whether weight loss can be maintained with lower doses after patients have sustained a stable reduced weight for several years remains to be determined.
Benefits beyond weight loss
Liraglutide, semaglutide (2.4 mg weekly dose) and tirzepatide lower blood pressure, improve lipid profiles, reduce blood glucose levels, reduce C-reactive protein levels and improve physical functioning.14,16,18 These effects appear associated with both weight loss and weight-independent effects of GLP-1 receptor agonists.32 Given these substantial beneficial effects on cardiometabolic risk factors, it is unsurprising that incretins have also been shown to reduce the risk of cardiometabolic diseases.
Prevention of type 2 diabetes
In adults with prediabetes and obesity, liraglutide 3 mg daily for three years reduced the risk of progression to type 2 diabetes by 79%, with a greater proportion reverting to normoglycaemia.20 Similarly, semaglutide 2.4 mg once weekly for one year in adults with prediabetes and obesity resulted in reversion to normoglycaemia in 81% compared with 14% on placebo.33 Even more impressively, three years of tirzepatide treatment reduced the risk of progression to type 2 diabetes by 93% among adults with prediabetes versus placebo (p <0.0001).24
Cardiovascular effects
Incretin analogues increase heart rate by about two to three beats per minute via indirect stimulation of sinoatrial nodal tissue through increased sympathetic nervous system activation. Although this could potentially have adverse cardiac effects, GLP-1 receptor agonists have been shown to protect cardiac myocytes against ischaemia in animal and human studies. Interestingly, GLP-1 receptor agonists oppose sympathetic effects on cardiac ventricular excitability, reducing ventricular arrhythmic risk through stimulation of cardiac parasympathetic neurons. This, along with their beneficial effect on lipid levels, inflammation and glucose uptake, likely contributes to their beneficial cardiac effects.32,34
After a median exposure of 3.8 years to liraglutide 1.8 mg daily in patients with type 2 diabetes, the primary composite outcome – death from cardiovascular causes, nonfatal myocardial infarction and nonfatal stroke – was reduced by 13%, with death from cardiovascular causes reduced by 22%.35
In people with type 2 diabetes and established cardiovascular disease, two years of low-dose semaglutide (0.5 or 1 mg) reduced the risk of a composite outcome of cardiovascular death, nonfatal myocardial infarction or nonfatal stroke by 26%, primarily driven by stroke reduction.36 In people without diabetes but with obesity and pre-existing cardiovascular disease, semaglutide 2.4 mg once weekly reduced the risk of death from cardiovascular causes, nonfatal myocardial infarction or nonfatal stroke by 20% over a median of 3.25 years.22 Semaglutide 2.4 mg once weekly has also been shown to result in greater improvements in exercise function and greater reduction in symptoms associated with heart failure in people with heart failure with preserved ejection fraction.37
In people with obesity and heart failure with preserved ejection fraction, one year of tirzepatide treatment lowered the risk of a composite of death from cardiovascular causes or worsening heart failure by 38%.38 A substudy of this trial showed tirzepatide reduced left ventricular mass and paracardiac adipose tissue.39 Cardiovascular outcome trials with tirzepatide are underway but real-world data suggest it may provide better cardiovascular outcomes in people aged over 40 with type 2 diabetes and established cardiovascular disease compared with GLP-1 receptor agonists.40
A recent trial showed that semaglutide 1 mg once weekly increased maximum walking distance by 13% in people with type 2 diabetes and peripheral vascular disease.41
Renal effects
In people with type 2 diabetes at high risk of cardiovascular disease, liraglutide 1.8 mg daily over a median follow up of 3.8 years resulted in a 22% reduction in renal outcomes compared with placebo. This result was driven mainly by a lower incidence of macroalbuminuria in the liraglutide group than in the placebo group.42 Similarly, semaglutide (0.5 or 1 mg once weekly) over two years in people with type 2 diabetes resulted in a 36% reduction in the progression of nephropathy compared with placebo.36 However, these studies were not primarily designed to assess renal outcomes and the benefits seen were mainly attributable to reductions in albuminuria. The Evaluate Renal Function with Semaglutide Once Weekly (FLOW) study was designed to assess the effect of semaglutide 1 mg once weekly on kidney outcomes in patients with chronic kidney disease and type 2 diabetes. The trial was stopped early based on a prespecified interim analysis, with a median follow up of 3.4 years. Semaglutide treatment resulted in a 24% reduction in major kidney disease, a composite of kidney failure, at least a 50% reduction in estimated glomerular filtration rate from baseline or death from kidney-associated or cardiovascular causes.43
Clinical trials of tirzepatide in people with renal disease are still ongoing. However, early evidence suggests potential renal benefits. In a post hoc analysis using pooled data from five trials (SURPASS-1 to -5) in people with type 2 diabetes and urinary albumin-to-creatinine ratio of 3.4 mg/mmol and above, tirzepatide was associated with a 19 to 26% reduction (depending on dose used) in urinary albumin-to-creatinine ratio compared with placebo or insulin. However, after 40 to 42 weeks, no significant difference in estimated glomerular filtration rate was observed.44 A retrospective cohort study of people with type 2 diabetes aged 18 years and older found that tirzepatide was associated with lower rates of all-cause mortality, cardiovascular events, acute kidney injury and adverse kidney events compared with GLP-1 receptor agonists.45
Liver disease
In a 72-week phase 2 study of patients (n = 320) with metabolic dysfunction-associated steatohepatitis (MASH), semaglutide (0.1, 0.2 or 0.4 mg daily) reduced steatosis but had no effect on fibrosis.46 In an open-label substudy of patients with type 2 diabetes and MASH (n = 502), tirzepatide resulted in a significant reduction in liver fat compared with insulin degludec after 52 weeks.47 In a smaller 48-week trial (n = 71) of semaglutide 2.4 mg once weekly in people with biopsy-confirmed MASH and compensated cirrhosis, there was no significant improvement in fibrosis or resolution of MASH compared with placebo.48 However, an interim analysis at 72 weeks of a phase 3 trial of semaglutide 2.4 mg once weekly for MASH showed histological improvements, including resolution of fibrosis, reduction of liver fibrosis and resolution of steatohepatitis in a significant number of patients.49
Tirzepatide also shows promise. In a phase 2 trial of people with biopsy-confirmed MASH, tirzepatide treatment over 52 weeks led to resolution of MASH in 34 to 53% of participants (depending on the dose used) compared with placebo. There was also an indication of improved fibrosis without worsening of MASH with tirzepatide.50
Obstructive sleep apnoea
In a one-year study of adults with moderate to severe obstructive sleep apnoea and obesity – about half of whom were treated with positive airway pressure at baseline – tirzepatide 10 or 15 mg once weekly resulted in a clinically meaningful reduction in the apnoea–hypopnoea index compared with placebo. Tirzepatide also significantly reduced the sleep apnoea-specific hypoxic burden, a measure that comprises the frequency, duration and depth of oxygen desaturation associated with sleep events, and is considered a better predictor of cardiovascular risk and mortality in sleep apnoea. Treatment was also associated with reductions in body weight, high-sensitivity C-reactive protein level, systolic blood pressure and improvements in sleep-associated patient-reported outcomes.51
Neurodegenerative disease
Animal studies suggest that GLP-1 analogues exert a range of beneficial effects on cognitive function in models of Alzheimer’s disease. These include improvements in learning and memory, reductions in amyloid plaques and tau phosphorylation, attenuation of cortical and hippocampal neuronal loss, increased neurogenesis, improved synapsis number and plasticity, reduced brain oxidative stress, cerebral anti-inflammatory activity and improved brain insulin receptor localisation and signalling.52,53 GLP-1 receptor agonists have also shown benefit in animal models of Parkinson’s disease, with greater effects observed using GLP-1/GIP dual receptor agonists.52
Although dementia was not a primary outcome, a pooled analysis of 15,820 patients with type 2 diabetes randomised to GLP-1 receptor agonist treatment or placebo found a 50% reduction in dementia progression over a median of 3.6 years (15 cases in the placebo group vs 32 in the GLP-1 receptor agonist group).53 A nationwide cohort study of 120,054 patients with type 2 diabetes over a median follow up of 7.4 years found an 11% reduction in the rate of dementia with GLP-1 receptor agonists (95% of patients were on liraglutide) when used as a second-line treatment compared with other glucose-lowering therapies, with greater benefit seen with longer duration of use.53 However, these studies were observational or post hoc analyses, and dementia was not the primary outcome.52,53
Two recent trials further support a possible role for GLP-1 analogues in dementia prevention. An analysis of electronic health records from over one million people with type 2 diabetes and no prior diagnosis of dementia found that semaglutide use was associated with a 40 to 70% reduced risk developing dementia over three years, depending on the comparator medication, with the greatest benefit seen when compared to insulin.54 A systemic review and meta-analysis of randomised clinical trials evaluating cardioprotective glucose-lowering medications also found that GLP-1 receptor agonists were associated with a significant reduction in dementia risk.55 While promising, these findings are not definitive due to the lack of randomised clinical trials with dementia as the primary outcome, and further evidence is required. Two phase 3 trials are currently underway investigating once-daily oral semaglutide in early-stage Alzheimer’s disease (clinical trial numbers NCT04777396 and NCT04777409).
Small clinical trials of exenatide and liraglutide in people with Parkinson’s disease have shown improvements in motor symptoms, emotional wellbeing, activities of daily living and quality of life.52 However, in a 96-week phase 3 trial (n=194) of once-weekly exenatide 2 mg in people with moderately severe Parkinson’s disease (without diabetes), there was no significant improvement in Parkinson’s disease severity compared with placebo.56
Adverse effects
Gastrointestinal effects
The most common adverse events associated with GLP-1 receptor agonist treatment are gastrointestinal, particularly nausea, followed by diarrhoea, vomiting and constipation. These symptoms usually subside over time, and slow dose titration can minimise their occurrence. Nonetheless, gastrointestinal side effects led to treatment discontinuation in 4.5% of participants in semaglutide trials and 6.2% of those receiving the highest dose (15 mg weekly) of tirzepatide.16,18
Early concerns about pancreatic safety arose from reports of pancreatitis in animal studies with the use of sitagliptin, clinical reports of humans treated with exenatide and the detection of preneoplastic pancreatic ductal lesions in animals treated with GLP-1 receptor agonists. Use of these agents has been associated with a modest increase in lipase and amylase levels (by 15 and 37%, respectively, in one study46), but this is rarely of any clinical significance. A comprehensive review of preclinical toxicology in animals with and without diabetes by European and US regulatory authorities found no evidence of incretin-associated pancreatic toxicity. There has been no evidence of increased pancreatic disease in the large cardiovascular outcome trials studying the safety of GLP-1 receptor agonists.57
However, in a Canadian study using data from a large health claims database, comparing the use of a GLP-1 receptor agonist with naltrexone/bupropion to treat obesity, there was an increased risk of pancreatitis. Although the event rate was low (4.6/1000 person-years for semaglutide, 7.9 for liraglutide and 1.0 for bupropion/naltrexone), there was a ninefold increased risk with GLP-1 receptor agonists overall.58
Some studies have reported an increased risk of biliary disease, including cholelithiasis and acute cholecystitis, with the use of GLP-1 receptor agonists. However, animal studies do not support these agents having a direct effect on the biliary system. As weight loss increases the lithogenicity of bile, it remains unclear whether the increased risk of biliary disease seen with GLP-1 receptor agonists is due to the medication itself or to weight loss.57
Anaesthesia
Several studies, including a retrospective audit and a matched pair case-control study of 205 pairs of patients undergoing gastroscopy, have shown that patients taking GLP-1 receptor agonists have higher rates of retained solid gastric contents.59 There have also been case reports of pulmonary aspiration when under anaesthesia in patients taking GLP-1 receptor agonists.59 Given the long half-life of these agents, stopping them before surgery is of limited value, and is not feasible in emergency procedures.
The American Society of Anesthesiologists recommends managing patients on GLP-1 receptor agonists as if they have a full stomach, including appropriate airway protection and rapid sequence induction.57 Similar recommendations have been made by Australian experts. Additionally, it has been recommended that patients having elective procedures consume only clear fluids for 24 hours before surgery. Australian guidelines also provide detailed advice on patient preparation before elective upper endoscopy.60
Medullary thyroid cancer
There is a theoretical increased risk of medullary thyroid cancer (MTC) as preclinical studies showed a link between GLP-1 receptor agonism to the development of C-cell hyperplasia and MTC in rodents. Although GLP-1 receptors are expressed in rodents and have a functional role in bone metabolism, the density of GLP-1 receptors in monkey and human thyroid cells is extremely low. Clinical trials have shown no evidence of increased calcitonin levels (which have been measured in all clinical trials of GLP-1 receptor agonists) or MTC rates with GLP-1 receptor agonist use. Nevertheless, as some human MTCs may express GLP-1 receptors, these agents are not recommended for individuals with a personal or family history of MTC or multiple endocrine neoplasia type 2.57
Ophthalmic complications
After two years of low-dose semaglutide 1 mg once weekly in people with type 2 diabetes, there was a 76% increased risk of worsening retinopathy compared with placebo, although the absolute event rate was low (3.0% with semaglutide vs 1.8% with placebo).36 Similarly, in a retrospective cohort study of 3435 people with type 2 diabetes exposed to tirzepatide for 180 days and over compared with a matched unexposed cohort, tirzepatide was associated with an increased risk of new-onset proliferative diabetic retinopathy – primarily in those with pre-existing nonproliferative diabetic retinopathy. There were very few cases in participants with no or very mild retinopathy at baseline, and no progression of mild nonproliferative diabetic retinopathy was observed.61 GLP-1 receptors are not present in ocular tissue, so it is likely that any worsening of retinopathy is associated with the rapid glucose lowering observed with these agents.3
Nonarteritic anterior ischaemic optic neuropathy (NAION), the second most common form of optic neuropathy and a leading cause of adult blindness, has also emerged as a potential concern. A retrospective matched cohort study found a significantly higher risk of NAION among people with diabetes and those with overweight or obesity taking semaglutide compared with those not on GLP-1 receptor agonist treatment.62 A case series linked the use of semaglutide or tirzepatide with an increased risk of NAION, sometimes after a single dose, including in one patient without diabetes and in another who experienced symptoms in both eyes following separate doses. Rapid correction of glycaemia with resultant optic nerve swelling has been hypothesised as a factor. Other reported complications included bilateral papillitis and paracentral acute middle maculopathy, both in people with type 2 diabetes. These findings suggest a higher risk of acute ocular complications in people with type 2 diabetes.63 However, several studies have found no association between synthetic incretins and NAION, and a recent review concluded that no definitive causal relationship has been established between these agents and NAION.64
Psychiatric disease
A bidirectional relationship exists between obesity and depression, with each increasing the risk of the other.65 Given this, it is not unexpected that case reports of suicide in people taking incretin treatment have been documented, and these are being monitored by the European Medicines Agency and the US Food and Drug Administration. However, a retrospective cohort study of electronic health records involving 240,618 patients with overweight or obesity prescribed semaglutide or non-GLP-1 receptor agonist antiobesity medications, and 1,589,855 patients with type 2 diabetes prescribed semaglutide or other hypoglycaemic agents, found a lower risk of suicidal ideation in patients prescribed semaglutide regardless of prior psychiatric history.66 Although people with significant psychiatric illness are typically excluded from clinical trials, no increase in psychiatric disorders has been observed in these studies. In contrast, improvements in quality of life measures have been reported with incretin therapy.
The future of incretins and new agents
Incretin analogues have proven to be very popular, with global shortages precipitated by high demand, which has been particularly problematic for patients. These medications must only be used by people with health problems and not by healthy individuals with minimal or no excess adiposity who are seeking weight loss for purely cosmetic reasons. However, as the production of approved medications increases, and with the arrival of new products currently undergoing phase 2 and 3 studies, availability is expected to improve. Products in more advanced studies include a combination of cagrilintide (an amylin analogue) and semaglutide; retatrutide, a triple compound of GIP, GLP-1 and glucagon receptor agonists; survodutide, a dual glucagon and GLP-1 receptor agonist; maridebart and cafraglutide, a dual GIP receptor antagonist and GLP-1 receptor agonist; and orforglipron, an oral nonpeptide GLP-1 receptor agonist.67
Conclusion
Incretin analogues are very effective weight loss medications with cardiovascular, renal, respiratory and hepatic benefits, with emerging evidence suggesting potential neuroprotective effects. Adverse effects are generally limited to gastrointestinal disturbances, which can be managed through dose reduction and caution around anaesthesia. Liraglutide, semaglutide and tirzepatide have been shown to reduce major cardiovascular events, and similar trials are ongoing for other investigational incretins. Currently, no medications for overweight and obesity are subsidised by the PBS, which is a significant concern given that obesity disproportionately affects people of lower socioeconomic status. The Pharmaceutical Benefits Advisory Committee will need to weigh the broad availability of incretin analogues for obesity management against the long-term health and economic costs of obesity to the Australian community. MT
COMPETING INTERESTS: Associate Professor Markovic is an investigator on pharmaceutical trials of semaglutide and CagriSema (NovoNordisk), tirzepatide and retatrutide (Lilly), BI 456906 (Boehringer Ingelheim) and maridebart/cafraglutide (Amgen); and is on the Advisory Board for Nestlé Health Science, VLCD. Associate Professor Hocking has received honoraria for lectures and manuscript writing from Lilly Australia, AstraZeneca, Amgen, Sanofi-Aventis, Nestlé Health Sciences, iNova and Servier; support for attending meetings from Lilly Australia, Novo Nordisk, Amgen and CSL Seqirus; participation on Advisory Boards from Lilly Australia, Novo Nordisk, Ethicon and AstraZeneca; is an investigator on pharmaceutical trials of semaglutide and CagriSema (NovoNordisk), tirzepatide and retatrutide (Lilly), BI 456906 (Boehringer Ingelheim) and maridebart/cafraglutide (Amgen); and is President of the National Association of Clinical Obesity Services.
References
1. Australian Institute of Health and Welfare (AIHW). Overweight and Obesity. Canberra: AIHW; 2024. Available online at: https://www.aihw.gov.au/reports/overweight-obesity/overweight-and-obesity (accessed July 2025).
2. Campbell JE. Targeting the GIPR for obesity: to agonize or antagonize? Potential mechanisms. Mol Metab 2021; 46: 101139.
3. Nauck MA, Quast DR, Wefers J, Pfeiffer AFH. The evolving story of incretins (GIP and GLP-1) in metabolic and cardiovascular disease: a pathophysiological update. Diabetes Obes Metab 2021; 23(Suppl 3): 5-29.
4. Trapp S, Brierley DI. Brain GLP-1 and the regulation of food intake: GLP-1 action in the brain and its implications for GLP-1 receptor agonists in obesity treatment. Br J Pharmacol 2022; 179: 557-570.
5. Adriaenssens AE, Biggs EK, Darwish T, et al. Glucose-dependent insulinotropic polypeptide receptor-expressing cells in the hypothalamus regulate food intake. Cell Metab 2019; 30: 987-996.e6.
6. Baggio LL, Drucker DJ. Glucagon-like peptide-1 receptors in the brain: controlling food intake and body weight. J Clin Invest 2014; 124: 4223-4226.
7. Liskiewicz A, Khalil A, Liskiewicz D, et al. Glucose-dependent insulinotropic polypeptide regulates body weight and food intake via GABAergic neurons in mice. Nat Metab 2023; 5: 2075-2085.
8. Ravussin E, Sanchez-Delgado G, Martin CK, et al. Tirzepatide did not impact metabolic adaptation in people with obesity, but increased fat oxidation. Cell Metab 2025; 37: 1060-1074.e4.
9. Yu X, Chen S, Funcke JB, et al. The GIP receptor activates futile calcium cycling in white adipose tissue to increase energy expenditure and drive weight loss in mice. Cell Metab 2025; 37: 187-204.e7.
10. Dungan KM, Povedano ST, Forst T, et al. Once-weekly dulaglutide versus once-daily liraglutide in metformin-treated patients with type 2 diabetes (AWARD-6): a randomised, open-label, phase 3, non-inferiority trial. Lancet 2014; 384: 1349-1357.
11. Pratley RE, Aroda VR, Lingvay I, et al. Semaglutide versus dulaglutide once weekly in patients with type 2 diabetes (SUSTAIN 7): a randomised, open-label, phase 3b trial. Lancet Diabetes Endocrinol 2018; 6: 275-286.
12. Frias JP, Nauck MA, Van J, et al. Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet 2018; 392: 2180-2193.
13. Garber A, Henry R, Ratner R, et al. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 2009; 373: 473-481.
14. Pi-Sunyer X, Astrup A, Fujioka K, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med 2015; 373: 11-22.
15. Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med 2021; 385: 503-515.
16. Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med 2021; 384: 989-1002.
17. Davies M, Færch L, Jeppesen OK, et al. Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet 2021; 397: 971-984.
18. Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med 2022; 387: 1434-1435.
19. Weinstock RS, Guerci B, Umpierrez G, Nauck MA, Skrivanek Z, Milicevic Z. Safety and efficacy of once-weekly dulaglutide versus sitagliptin after 2 years in metformin-treated patients with type 2 diabetes (AWARD-5): a randomized, phase III study. Diabetes Obes Metab 2015; 17: 849-858.
20. le Roux CW, Astrup A, Fujioka K, et al. 3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes: a randomised, double-blind trial. Lancet 2017; 389: 1399-409.
21. Garvey WT, Batterham RL, Bhatta M, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med 2022; 28: 2083-2091.
22. Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med 2023; 389: 2221-2232.
23. Aronne LJ, Horn DB, le Roux CW, et al. Tirzepatide as compared with semaglutide for the treatment of obesity. N Engl J Med 2025; 393: 26-36.
24. Jastreboff AM, le Roux CW, Stefanski A, et al. Tirzepatide for obesity treatment and diabetes prevention. N Engl J Med 2025; 392: 958-971.
25. Davies MJ, Bergenstal R, Bode B, et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: the SCALE diabetes randomized clinical trial. JAMA 2015; 314: 687–699.
26. Suliman M, Buckley A, Al Tikriti A, et al. Routine clinical use of liraglutide 3 mg for the treatment of obesity: outcomes in non-surgical and bariatric surgery patients. Diabetes Obes Metab 2019; 21: 1498-501.
27. Volpe S, Lisco G, Racaniello D, et al. Once-weekly semaglutide induces an early improvement in body composition in patients with type 2 diabetes: a 26-week prospective real-life study. Nutrients 2022; 14: 2414.
28. Kelly AS, Auerbach P, Barrientos-Perez M, et al. A randomized, controlled trial of liraglutide for adolescents with obesity. N Engl J Med 2020; 382: 2117-2128.
29. Flox CK, Barrientos-Perez M, Bomberg, EM, et al. Liraglutide for children 6 to <12 years of age with obesity — a randomized trial. N Engl J Med 2024; 392: 555-565.
30. Weghuber D, Barrett T, Barrientos-Pérez M, et al. Once-weekly semaglutide in adolescents with obesity. N Engl J Med 2022; 387: 2245-2257.
31. Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab 2022; 24: 1553-1564.
32. Drucker DJ. The cardiovascular biology of glucagon-like peptide-1. Cell Metab 2016; 24: 15-30.
33. McGowan BM, Bruun JM, Capehorn M. Efficacy and safety of once-weekly semaglutide 2·4 mg versus placebo in people with obesity and prediabetes (STEP 10): a randomised, double-blind, placebo-controlled, multicentre phase 3 trial. Lancet Diabetes Endocrinol 2024; 12: 631-642.
34. Ang R, Mastitskaya S, Hosford PS, et al. Modulation of cardiac ventricular excitability by GLP-1 (glucagon-like peptide-1). Circ Arrhythm Electrophysiol 2018; 11: e006740.
35. Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375: 311-322.
36. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375: 1834-1844.
37. Kosiborod MN, Abildstrom SZ, Borlaug BA, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med 2023; 389: 1069-1084.
38. Packer M, Zile MR, Kramer CM, et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N Engl J Med 2025; 392: 427-437.
39. Kramer CM, Borlaug BA, Zile MR, et al. Tirzepatide reduces LV mass and paracardiac adipose tissue in obesity-related heart failure: SUMMIT CMR substudy. J Am Coll Cardiol 2025; 85: 699-706.
40. Dani SS, Makwana B, Khadke S, et al. An observational study of cardiovascular outcomes of tirzepatide vs glucagon-like peptide-1 receptor agonists. JACC Adv 2025; 4: 101740.
41. Bonaca MP, Catarig AM, Houlind K, et al. Semaglutide and walking capacity in people with symptomatic peripheral artery disease and type 2 diabetes (STRIDE): a phase 3b, double-blind, randomised, placebo-controlled trial. Lancet 2025; 405: 1580-1593.
42. Mann JFE, Orsted DD, Brown-Frandsen K, et al. Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med 2017; 377: 839-848.
43. Perkovic V, Tuttle KR, Rossing P, et al. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med 2024; 391: 109-121.
44. Apperloo EM, Tuttle KR, Pavo I, et al. Tirzepatide associated with reduced albuminuria in participants with type 2 diabetes: pooled post hoc analysis from the randomized active- and placebo-controlled SURPASS-1-5 clinical trials. Diabetes Care 2025; 48: 430-436.
45. Chuang MH, Chen JY, Wang HY, Jiang ZH, Wu VC. Clinical outcomes of tirzepatide or GLP-1 receptor agonists in individuals with type 2 diabetes. JAMA Netw Open 2024; 7: e2427258.
46. Newsome PN, Buchholtz K, Cusi K, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med 2021; 384: 1113-1124.
47. Gastaldelli A, Cusi K, Fernández Landó L, Bray R, Brouwers B, Rodríguez Á. Effect of tirzepatide versus insulin degludec on liver fat content and abdominal adipose tissue in people with type 2 diabetes (SURPASS-3 MRI): a substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial. Lancet Diabetes Endocrinol 2022; 10: 393-406.
48. Loomba R, Abdelmalek MF, Armstrong MJ, et al. Semaglutide 2.4 mg once weekly in patients with non-alcoholic steatohepatitis-related cirrhosis: a randomised, placebo-controlled phase 2 trial. Lancet Gastroenterol Hepatol 2023; 8: 511-522.
49. Sanyal AJ, Newsome PN, Kliers I, et al. Phase 3 trial of semaglutide in metabolic dysfunction-associated steatohepatitis. N Engl J Med 2025; 392: 2089-2099.
50. Loomba R, Hartman ML, Lawitz EJ, et al. Tirzepatide for metabolic dysfunction-associated steatohepatitis with liver fibrosis. N Engl J Med 2024; 391: 299-310.
51. Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N Engl J Med 2024; 391: 1193-1205.
52. Ferrari F, Moretti A, Villa RF. Incretin-based drugs as potential therapy for neurodegenerative diseases: current status and perspectives. Pharmacol Ther 2022; 239: 108277.
53. Norgaard CH, Friedrich S, Hansen CT, et al. Treatment with glucagon-like peptide-1 receptor agonists and incidence of dementia: data from pooled double-blind randomized controlled trials and nationwide disease and prescription registers. Alzheimers Dement 2022; 8: e12268.
54. Wang W, Wang Q, Qi X, et al. Associations of semaglutide with first-time diagnosis of Alzheimer’s disease in patients with type 2 diabetes: target trial emulation using nationwide real-world data in the US. Alzheimers Dement 2024; 20: 8661-8672.
55. Seminer A, Mulihano A, O’Brien C, et al. Cardioprotective glucose-lowering agents and dementia risk: a systematic review and meta-analysis. JAMA Neurol 2025; 82: 450-460.
56. Vijiaratnam N, Girges C, Auld G, et al. Exenatide once a week versus placebo as a potential disease-modifying treatment for people with Parkinson’s disease in the UK: a phase 3, multicentre, double-blind, parallel-group, randomised, placebo-controlled trial. Lancet 2025; 405: 627-636.
57. Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab 2018; 27: 740-756.
58. Sodhi M, Rezaeianzadeh R, Kezouh A, Etminan M. Risk of gastrointestinal adverse events associated with glucagon-like peptide-1 receptor agonists for weight loss. JAMA 2023; 3330: 1795-1797.
59. Raven LM, Brown C, Greenfield JR. Considerations of delayed gastric emptying with peri-operative use of glucagon-like peptide-1 receptor agonists. Med J Aust 2023; 220: 14-16.
60. Australian and New Zealand College of Anaesthetists (ANZCA). Clinical Practice Recommendation on the Periprocedural Use of GLP-1/GIP Receptor Agonists. Melbourne: ANZCA; 2024. Available online at: https://www.anzca.edu.au/resources/professional-documents/endorsed-guidelines/periprocedural-glp-1-use-consensus-clinical-guide.pdf (accessed July 2025).
61. Buckley AJ, Tan GD, Gruszka-Goh M, Scanlon PH, Ansari I, Suliman SGI. Early worsening of diabetic retinopathy in individuals with type 2 diabetes treated with tirzepatide: a real-world cohort study. Diabetologia 2025. Online ahead of print. Doi: 10.1007/s00125-025-06466-8.
62. Hathaway JT, Shah MP, Hathaway DB, et al. Risk of nonarteritic anterior ischemic optic neuropathy in patients prescribed semaglutide. JAMA Ophthalmol 2024; 142: 732-739.
63. Katz BJ, Lee MS, Lincoff NS, et al. Ophthalmic complications associated with the antidiabetic drugs semaglutide and tirzepatide. JAMA Ophthalmol 2025; 143: 215-220.
64. Gregory V, Mollan SP. Are synthetic incretins associated with ischaemic optic neuropathy? Eye (Lond) 2025; 39: 808-809.
65. Luppino FS, de Wit LM, Bouvy PF, et al. Overweight, obesity, and depression: a systematic review and meta-analysis of longitudinal studies. Arch Gen Psychiatry 2010; 67: 220-229.
66. Wang W, Volkow ND, Berger NA, Davis PB, Kaelber DC, Xu R. Association of semaglutide with risk of suicidal ideation in a real-world cohort. Nat Med 2024; 30: 168-176.
67. Melson E, Ashraf U, Papamargaritis D, Davies MJ. What is the pipeline for future medications for obesity. Int J Obes 2025; 49: 433-451.