Modern therapies in vitiligo management

Vitiligo is a chronic autoimmune disorder that causes a loss of skin pigment. Although traditional treatments focus on halting disease progression and restoring colour, relapse is common. New therapies such as Janus kinase inhibitors, as well as advances in biologics and immunomodulatory agents, offer renewed hope for patients with this challenging condition.
- Vitiligo results from autoimmune destruction of melanocytes, leading to depigmented macules.
- Traditional treatment options include topical corticosteroids, calcineurin inhibitors, topical vitamin D analogues, phototherapy and surgery.
- Topical corticosteroids remain the cornerstone of vitiligo treatment, particularly for localised disease and extrafacial lesions.
- Narrowband ultraviolet B remains the preferred treatment modality for rapidly progressive or extensive disease, or when whole-body phototherapy is indicated.
- Janus kinase inhibitors, such as ruxolitinib, are emerging as game changers in repigmentation therapy.
- Ongoing research into biologics and immunomodulatory agents continues to expand the therapeutic options for vitiligo.
Vitiligo is a progressive autoimmune skin depigmentation disorder that affects about 1% of the global population.1,2 It is the most common cause of depigmentation.3 Clinical characteristics include white, nonscaly macules that are caused by the destruction of melanocytes in the skin or hair.1 The proposed pathogenesis involves a combination of genetics and environmental factors such as sunburn or stress, along with metabolic alterations that trigger disease onset and progression.3,4
Vitiligo can significantly affect a patient’s quality of life and may lead to psychological distress due to stigmatisation.5 The burden is greater in patients with more than 5% affected body surface area, darker skin, or facial or hand involvement.6 Patients often report impacts on daily activities, employment and mental health. Many are conscious of clothing choices and prioritise concealing visible lesions, contributing to psychosocial strain. Mental health conditions such as anxiety and depression are, therefore, prevalent in this population.6
Vitiligo has traditionally been difficult to manage because there are few definitive treatments. Management varies by type, severity and extent of disease. In recent years, new therapeutic options have emerged, with several licensed for autoimmune-mediated dermatoses, and ruxolitinib is now approved by the US Food and Drug Administration (FDA) for vitiligo. Other therapeutic agents remain under investigation in early- and late-phase clinical trials. This article summarises the management options used currently and emerging therapies for vitiligo.
Pathogenesis
The pathogenesis of vitiligo is complex, involving multiple mechanisms leading to a loss of functional melanocytes.4 Proposed mechanisms are both genetic and environmental. Disease may begin in sun-exposed areas during warmer months and can be triggered by pregnancy, trauma, severe sunburn or emotional stress.7 Lesions tend to develop in areas of friction, reflecting Koebnerisation, and occur on the face, dorsal surface of the hands, nipples, axillae, umbilicus, sacrum and surrounding orifices.7,8
Vitiligo is associated with other autoimmune disorders in affected patients and their relatives, supporting a genetic component to the disease.1 These associated diseases include autoimmune thyroid disease, rheumatoid arthritis, psoriasis, type 1 diabetes and pernicious anaemia.1 Over 50 susceptibility loci have been identified, including mutations affecting immune system regulation, cellular apoptosis and melanocyte-specific genes such as TYR and MC1R.4,9
Oxidative stress plays a key role in the pathogenesis of vitiligo by causing intrinsic defects in melanocytes.4 Under oxidative stress, melanocytes release reactive oxygen species that damage DNA and compromise protein and lipid function through oxidation and peroxidation.3,4 The innate immune system bridges the oxidative stress pathways and the adaptive immune response.10
Innate immune response activation occurs when damage-associated molecular patterns bind to and activate pattern recognition receptors.11 These damage-associated molecular patterns chaperone proteins and enhance their presentation to dendritic cells, thereby initiating cytotoxic T-cell responses of the adaptive immune system.3,11
Activated cytotoxic CD8+ T-cells then target melanocytes in the epidermis and dermis, and express perforin and granzyme B to induce cell apoptosis.2,3 Activated CD8+ T-cells also release cytokines such as interferon gamma and tumour necrosis factor, which promote further T-cell recruitment and melanocyte destruction.2,3,11 In addition, interferon gamma signalling recruits Janus kinase (JAK)1 and JAK2.12 Chronic inflammation and ongoing melanocyte damage are perpetuated by sustained JAK signalling and the continuous production of inflammatory cytokines.4,12
Clinical presentation
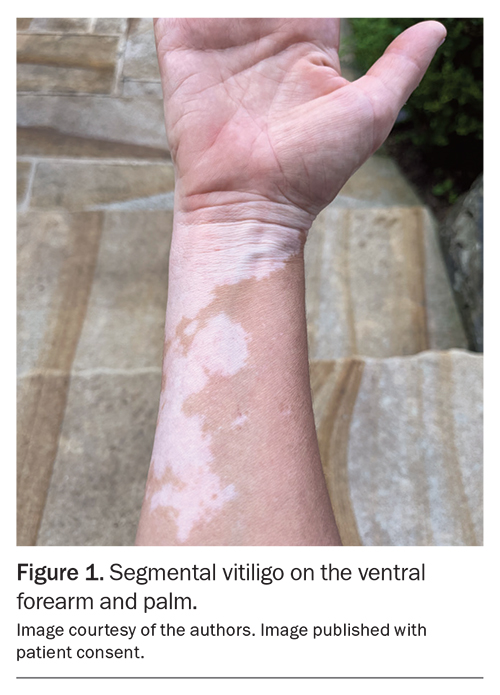
Vitiligo equally affects adults and children of all sexes and ethnicities, and can be differentiated into segmental and nonsegmental types.1 Segmental vitiligo is characterised by asymmetric, unilateral patches (Figure 1).7 The onset of disease typically occurs before the age of 30 years, usually in childhood, and follows a limited course with rapid stabilisation.1,7 Further disease extension after 24 months is rare.13 Segmental vitiligo can be further classified as unisegmental, bisegmental or plurisegmental.1
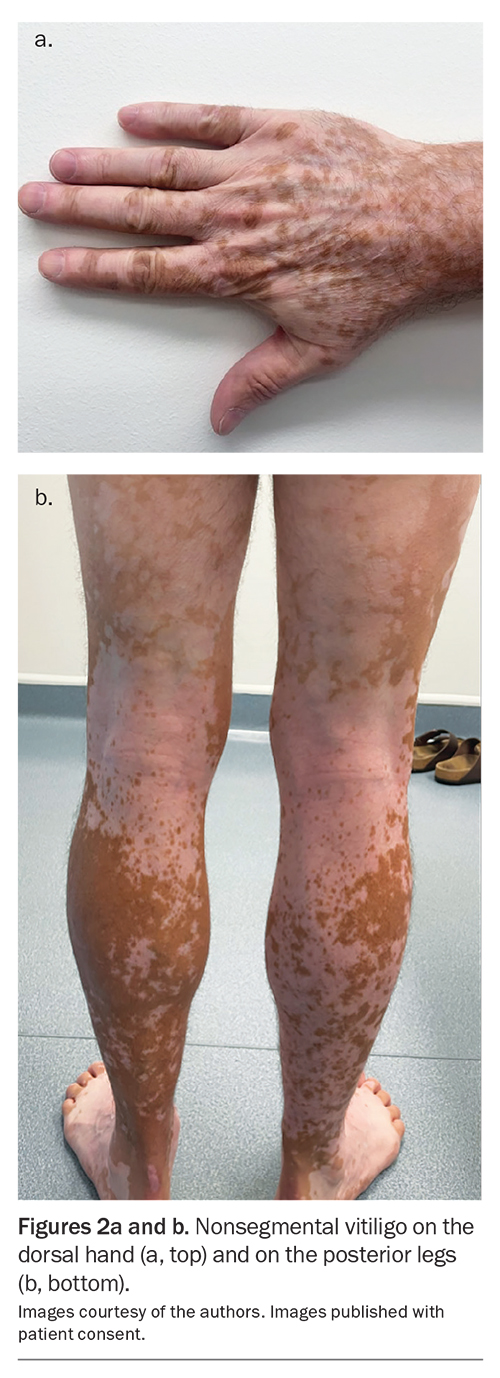
The more common nonsegmental vitiligo tends to present bilaterally, often affecting the acrofacial region or extensor surfaces (Figures 2a and b).1,13 Subtypes of nonsegmental vitiligo include acrofacial, mucosal, generalised, universal, mixed (when associated with segmental vitiligo) and rare variants.13 Nonsegmental forms typically evolve over time and may progress to a generalised distribution.1
Diagnosis
Vitiligo is diagnosed clinically, based on its characteristic appearance of white patches or macules surrounded by normal skin.8,14 Examination may include the use of a Wood’s lamp, which helps differentiate hypopigmented from depigmented skin when the diagnosis is uncertain.14 Other tools aiding diagnosis include dermoscopy and skin biopsy. Differential diagnoses for depigmentation to consider include, but are not limited to, pityriasis alba, hypopigmented mycosis fungoides, naevus depigmentosus, idiopathic guttate hypomelanosis, tinea versicolour and occupational or iatrogenic exposures such as to phenols.7,14
Management
Managing vitiligo remains a clinical challenge, as there is currently no definitive cure. A range of therapeutic options is available, with treatment tailored to individual factors such as disease type, extent and activity. The primary goals of vitiligo treatment are to halt disease progression, induce repigmentation and maintain pigmentation while preventing relapse.15 In active disease, the focus is on controlling inflammation and halting spread, whereas in stable disease, efforts shift towards repigmentation and long-term maintenance. Treatment typically requires a prolonged course, often six to 24 months, to achieve optimal outcomes.15
Traditional treatments
Traditional management strategies for vitiligo include topical and systemic therapies, phototherapy and more advanced options such as depigmentation and surgical intervention. These are summarised in Box 1.
In addition to those discussed below, patients should be advised on the general measures of cosmetic camouflage (including make-up, dyes, self-tanning lotions and tattoos), asked to follow strict sun protection and offered psychological support.
Topical treatments
Topical corticosteroids
Topical corticosteroids remain the cornerstone of vitiligo treatment, reducing inflammation, particularly for localised disease and extrafacial lesions. Potent to very potent corticosteroids can be applied daily for up to three to six months. To minimise local adverse effects such as skin atrophy, striae and telangiectasia, intermittent regimens (e.g. two weeks on, two weeks off) are recommended, especially in sensitive areas. Caution is advised when using topical corticosteroids on the eyelids or large body surface areas because of the risk of systemic absorption. In children, topical corticosteroids are considered safe for short-term continuous use (two to four months), with intermittent treatment preferred for extended therapy.16
Topical calcineurin inhibitors
Topical calcineurin inhibitors, such as tacrolimus ointment (0.03% and 0.1% strengths) and pimecrolimus cream (1% strength), are widely used in the treatment of vitiligo, particularly in sensitive areas like the face, eyelids and intertriginous regions, where topical corticosteroids carry a higher risk of adverse effects. However, in Australia, they are used off label for vitiligo.17
The efficacies of topical calcineurin inhibitors are comparable to those of topical corticosteroids, with a better safety profile and a limited risk of skin atrophy.18,19 These agents inhibit T-cell activation and cytokine release, helping to suppress the autoimmune destruction of melanocytes. Treatment can be applied twice daily for six months or more.16 They are typically well tolerated, but localised adverse effects such as mild burning, stinging or erythema can occur. Topical calcineurin inhibitors are considered safe for long-term use and may be applied alone or in combination with phototherapy to enhance repigmentation.20
Topical vitamin D analogues
Vitamin D analogues, such as calcipotriol, are used off label in vitiligo management for their immunomodulatory and melanocyte-stimulating effects, either as monotherapy or as adjuncts to other treatments. These agents inhibit T-cell proliferation and promote melanocyte differentiation, contributing to repigmentation. Although its evidence remains limited compared with corticosteroids and calcineurin inhibitors, topical calcipotriol has been shown to enhance repigmentation, particularly when combined with phototherapy.21 Application is generally well tolerated, although mild irritation or erythema may occur.21 Further studies are needed to clarify the optimal role of vitamin D analogues in vitiligo treatment regimens.
Phototherapy and photochemotherapy
Narrowband ultraviolet B
Phototherapy plays a central role in the management of vitiligo, with treatment tailored according to lesion type, distribution and disease extent. It can be delivered as whole-body therapy or targeted to localised lesions using excimer lamps, excimer lasers or home devices.
Narrowband ultraviolet B (NB-UVB) is considered the first-line modality for rapidly progressive or extensive vitiligo and is the preferred option for whole-body phototherapy. Early initiation of NB-UVB is recommended, as it may both halt disease progression and induce repigmentation.22 Treatment is typically administered using a fixed dosing schedule that can be adjusted according to the clinical response.23 NB-UVB is safe for use in children as young as 3 years of age, provided treatment is supervised by caregivers, although practical challenges such as adherence and tolerance of enclosed treatment spaces may limit its use in younger patients.24 The main drawbacks of NB-UVB are the need for prolonged therapy and frequent clinic visits, which can be burdensome. Nevertheless, it is generally well tolerated, with erythema and xerosis being the most common side effects, and there is no convincing evidence of an increased risk of skin cancer.25 Anti-inflammatory topical creams are often used in conjunction with phototherapy to lower the risk of skin irritation.
Excimer lamps and lasers provide an effective alternative to whole-body NB-UVB, offering shorter treatment durations and strong efficacy for localised disease, sometimes superior to whole-body NB-UVB.26 However, these modalities are impractical for large surface areas because of time and cost constraints. In addition, the risk of adverse effects such as erythema and blistering can depend on the operator’s technique.27 Home-based phototherapy units and handheld devices are additional options that may improve adherence, particularly for localised lesions, although their use is limited by device cost, maintenance requirements and technical considerations.28
Psoralen and ultraviolet A
Ultraviolet A (UVA) photochemotherapy, also known as psoralen and UVA, is no longer used in Australia, although it may be used elsewhere. It involves combining a photosensitising drug such as psoralen with long-wave UVA exposure. Psoralens can be administered orally, topically or via bath immersion.29 However, oral psoralen is no longer recommended because of the associated risk of systemic complications such as stomach pain and nausea.16
Photochemotherapy has demonstrated efficacy in the management of vitiligo, particularly for widespread disease, similar to NB-UVB. Side effects associated with psoralen and UVA include erythema and the risk of skin phototoxicity.25 Given the simplicity, comparable efficacy and lower risk profile, NB-UVB is preferred over psoralen and UVA.
Systemic treatments
Systemic oral therapies show variable efficacy in vitiligo, but in cases of active, rapidly progressive disease, oral corticosteroids can be used to arrest progression.30 For active disease, oral mini-pulse therapy is recommended, using moderate doses of corticosteroids. The suggested dosing schedule is twice weekly on two consecutive days for three to six months.31 Treatment should be reassessed at three months, and therapy should not exceed six months due to the risk of adverse events.16 Given the significant side effect profile of systemic corticosteroids, careful consideration of the risks and benefits is essential before initiation.
Other immunosuppressants, including methotrexate, cyclosporin and azathioprine, have been trialled in vitiligo, but evidence supporting their safety or efficacy in this setting is limited.32-34 These treatments are commonly used in Australia as part of ongoing management, but they are rarely employed as first-line therapy. At present, available biologic agents are not effective for the management of vitiligo.35
Depigmentation
Depigmentation therapy is an option for patients with widespread, treatment-resistant vitiligo. It is considered when more than 50% of the body surface area is affected and repigmentation strategies have failed or are undesired.36 The goal is to achieve a uniform skin tone by removing residual pigment from unaffected areas, most often using monobenzyl ether of hydroquinone. This agent is applied topically, often at a 20% concentration, and may induce generalised pigment loss over several months. However, treatment can lead to irritation, paradoxical repigmentation or pigment loss at distant, untreated sites.37
Depigmentation may also be achieved using physical methods such as cryotherapy or Q-switched lasers, particularly for localised areas, although these require experienced operators to minimise scarring and other adverse effects.38 Given the irreversible and highly visible nature of depigmentation, thorough counselling, psychosocial assessment and shared decision-making are essential before initiating therapy. Additional considerations should also be made in patients with darker skin tones due to sociocultural factors associated with treatment.16
Surgery
Surgical treatments for vitiligo are typically reserved for patients with stable, nonprogressive disease who have failed to respond to medical or light-based therapies. Key prerequisites for surgical intervention include disease stability for at least six to 12 months and the absence of the Koebner phenomenon. The risk of relapse should also be clearly communicated to patients considering this intervention.
Surgery aims to restore pigmentation by transplanting melanocytes into depigmented areas. Several techniques are available, including punch grafting, suction blister epidermal grafting, split-thickness grafting and cultured or noncultured melanocyte–keratinocyte transplantation. Although generally safe, each method carries specific risks and benefits. General complications include colour mismatch, scarring, graft displacement and postoperative hyper- or hypopigmentation.16,39
Emerging therapies
Several novel therapies are emerging in the modern dermatological management of vitiligo, reflecting advances in the understanding of vitiligo pathogenesis and immune modulation.40 These agents are summarised in Box 2. However, only a few of these newer treatments, including oral and topical JAK inhibitors, are approved by the TGA for prescription in Australia for conditions such as atopic dermatitis. None of them are currently TGA approved for vitiligo.
JAK inhibitors
The JAK–STAT (signal transducer and activator of transcription) signalling pathway is implicated in many autoimmune dermatological conditions. JAK inhibitors modulate the immune response by interfering with cytokine signalling through this pathway, offering therapeutic benefits in several inflammatory and autoimmune diseases.40 JAK inhibitors have rapidly evolved to play a key role in the management of conditions such as atopic dermatitis and psoriasis.41
Topical ruxolitinib
The JAK1/2 inhibitor ruxolitinib, in a topical formulation, is the first JAK inhibitor to be approved by the US FDA for vitiligo. In Australia, ruxolitinib is approved for use by the TGA for other conditions, although not for vitiligo. Phase 2 and 3 studies have demonstrated the clinical efficacy of topical ruxolitinib for both facial and body lesions.42 Ruxolitinib is also relatively safe, with no significant adverse events reported; the most frequently reported are acne and pruritus.42
Bimatoprost
The prostaglandin F2alpha analogue bimatoprost, commonly used for glaucoma and eyelash hypotrichosis, has shown promise as an off-label topical therapy for vitiligo in Australia. Small studies have demonstrated its capacity to induce perifollicular repigmentation, particularly in facial lesions, and it appears more effective when combined with phototherapy.43,44 Bimatoprost is generally well tolerated, with minor adverse effects such as local irritation, hypertrichosis and periocular pigmentation.44 Larger clinical trials are needed to better define its role in routine vitiligo management.
Oral JAK inhibitors
In a similar context, oral JAK inhibitors have an increasing body of evidence supporting their clinical efficacy and safety in vitiligo management. Several such agents have shown promise across case reports, small studies and late-phase clinical trials.
- Ritlecitinib (a JAK3 and tyrosine kinase inhibitor): produced meaningful improvements in a phase 2b trial, with a 21.2% mean reduction in the Facial Vitiligo Area Scoring Index at 24 weeks, and is currently undergoing phase 3 evaluation (clinical trial number: NCT05583526)45
- Tofacitinib (a JAK1 and JAK3 inhibitor): demonstrated partial to complete repigmentation in case studies and small studies, although durable results often require combination with phototherapy, and relapse may occur after discontinuation46,47
- Upadacitinib (a JAK1 inhibitor): showed about a 50% improvement in pigmentation in early studies and improved quality of life measures in a phase 2 trial, with phase 3 studies ongoing (clinical trial number: NCT06118411)48
- Baricitinib (a JAK1 and JAK2 inhibitor): produced significant repigmentation in small clinical series, and is being tested in phase 2 trials alongside phototherapy (clinical trial number: NCT04822584)49
- Povorcitinib (a JAK1 inhibitor): demonstrated favourable outcomes in a phase 2 trial involving 171 patients, showing improvements in the Vitiligo Area Scoring Index, and a phase 3 study is ongoing (clinical trial number: NCT06113445).50
Other agents, including ifidancitinib (a JAK1/3 inhibitor, clinical trial number: NCT03468855), delgocitinib (a pan-JAK inhibitor) and brepocitinib (a JAK1 inhibitor and tyrosine kinase 2 inhibitor, clinical trial number: NCT03715829) are at earlier stages of development.51
Although the evidence remains limited, concurrent ultraviolet B phototherapy has been shown to enhance the efficacy of JAK inhibitors in vitiligo.52 Safety concerns, particularly with systemic use, continue to be monitored, but JAK inhibitors represent a transformative frontier in vitiligo management, especially when combined with phototherapy to maximise repigmentation and potentially sustain disease control.
Other upcoming treatments
The novel treatment landscape for vitiligo is promising, with several potential therapies undergoing clinical trials. Cytokines are known to play a key role in the pathogenesis of vitiligo; however, established biologic agents show controversial evidence for efficacy and are not currently recommended as treatment.35
Newer biologics targeting more specific immune pathways are now under investigation. Anifrolumab, a fully human monoclonal antibody targeting the type I interferon receptor subunit 1, has been shown to modulate the heat shock protein 70–plasmacytoid dendritic cell axis.53 This pathway potentiates interferon alpha production as well as CXCL9 and CXCL10 expression, and has been implicated in the pathogenesis of vitiligo.50 A clinical trial is currently underway evaluating the efficacy of phototherapy alone versus combination treatment with anifrolumab and phototherapy (clinical trial number: NCT05917561). Interleukin-15 has also been identified as a key cytokine involved in disease persistence and relapse.54 AMG 714, a fully human immunoglobulin G, subclass 1, kappa light chain monoclonal antibody that inhibits interleukin-15 signalling, is being investigated in an ongoing phase 2 trial for both active and stable nonsegmental vitiligo (clinical trial number: NCT04338581).
Other emerging therapies include agents targeting immune checkpoints, cellular metabolism and melanocyte regeneration pathways. Abatacept, an immunoglobulin G1 fusion protein that binds cytotoxic T-lymphocyte-associated antigen 4, may attenuate vitiligo progression by modulating T-cell activation,55 with a phase 1 clinical trial currently underway (clinical trial number: NCT02281058). Phosphodiesterase-4 inhibitors, such as apremilast and crisaborole, may provide antioxidative, anti-inflammatory and promelanogenic benefits; however, clinical outcomes remain mixed, particularly when combined with phototherapy.56,57 Afamelanotide, a synthetic analogue of alpha-melanocyte-stimulating hormone, has shown promise in improving repigmentation alongside phototherapy,58 although its efficacy may be limited by the inability to regenerate melanocytes from stem cell reservoirs, and ongoing studies aim to further evaluate its therapeutic potential (clinical trial number: NCT05210582). Additionally, targeting T-cell metabolism with agents such as rapamycin and metformin is under investigation due to their roles in promoting autophagy, reducing oxidative stress and enhancing regulatory T-cell function (clinical trial numbers: NCT05342519 and NCT05607316).
Conclusion
Vitiligo is a complex autoimmune condition with a multifactorial pathogenesis involving genetic, oxidative and immune- mediated mechanisms. Although it has traditionally been difficult to treat, recent advances have expanded the therapeutic landscape, offering new hope for patients through targeted therapies. Current management relies on a combination of topical agents, phototherapy, systemic immunosuppressants and, in select cases, surgical or depigmentation approaches.
The development of JAK inhibitors and other immunomodulatory agents represents a major breakthrough, with several agents now TGA approved for other conditions, or in late-stage clinical trials. As understanding of the disease continues to evolve, ongoing research into emerging biologics and immune pathway targets is expected to transform vitiligo into a more controllable condition with individualised treatment approaches, improving both clinical outcomes and quality of life for affected individuals. MT
COMPETING INTERESTS: Dr Mostafa and Dr Chee are sub-investigators in a clinical trial for vitiligo sponsored by Pfizer. Associate Professor Shumack: None.
References
1. Ezzedine K, Eleftheriadou V, Whitton M, Van Geel N. Vitiligo. Lancet 2015; 386: 74-84.
2. Frisoli ML, Essien K, Harris JE. Vitiligo: mechanisms of pathogenesis and treatment. Annu Rev Immunol 2020; 38: 621-648.
3. Bergqvist C, Ezzedine K. Vitiligo: a review. Dermatology 2020; 236: 571-592.
4. Picardo M, Dell’Anna ML, Ezzedine K, et al. Vitiligo. Nat Rev Dis Primers 2015; 1: 1-16.
5. Parsad D, Dogra S, Kanwar AJ. Quality of life in patients with vitiligo. Health Qual Life Outcomes 2003; 1: 58.
6. Bibeau K, Ezzedine K, Harris JE, et al. Mental health and psychosocial quality-of-life burden among patients with vitiligo: findings from the global VALIANT study. JAMA Dermatol 2023; 159: 1124-1128.
7. Kovacs SO. Vitiligo. J Am Acad Dermatol 1998; 38: 647-668.
8. Alikhan A, Felsten LM, Daly M, Petronic-Rosic V. Vitiligo: a comprehensive overview: part I. Introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J Am Acad Dermatol 2011; 65: 473-491.
9. Upadhya S, Andrade MJ, Shukla V, Rao R, Satyamoorthy K. Genetic and immune dysregulation in vitiligo: insights into autoimmune mechanisms and disease pathogenesis. Autoimmun Rev 2025; 24: 103841.
10. Bergqvist C, Ezzedine K. Vitiligo: a focus on pathogenesis and its therapeutic implications. J Dermatol 2021; 48: 252-270.
11. Wang Y, Li S, Li C. Perspectives of new advances in the pathogenesis of vitiligo: from oxidative stress to autoimmunity. Med Sci Monit 2019; 25: 1017-1023.
12. Liu K, Zhou L, Shi M, et al. JAK inhibitors in immune regulation and treatment of vitiligo. Cytokine Growth Factor Rev 2024; 80: 87-96.
13. Ezzedine K, Lim HW, Suzuki T, et al. Revised classification/nomenclature of vitiligo and related issues: the Vitiligo Global Issues Consensus Conference. Pigment Cell Melanoma Res 2012; 25: E1.
14. van Geel N, Speeckaert R, Taïeb A, et al. Worldwide expert recommendations for the diagnosis and management of vitiligo: position statement from the International Vitiligo Task Force part 1: towards a new management algorithm. J Eur Acad Dermatol Venereol 2023; 37: 2173-2184.
15. Seneschal J, Boniface K. Vitiligo: current therapies and future treatments. Dermatol Pract Concept 2023; 13: e2023313S.
16. Seneschal J, Speeckaert R, Taïeb A, et al. Worldwide expert recommendations for the diagnosis and management of vitiligo: position statement from the international Vitiligo Task Force—part 2: specific treatment recommendations. J Eur Acad Dermatol Venereol 2023; 37: 2185-2195.
17. Lepe V, Moncada B, Castanedo-Cazares JP, Torres-Alvarez MB, Ortiz CA, Torres-Rubalcava AB. A double-blind randomized trial of 0.1% tacrolimus vs 0.05% clobetasol for the treatment of childhood vitiligo. Arch Dermatol 2003; 139: 581-585.
18. Chang HC, Hsu YP, Huang YC. The effectiveness of topical calcineurin inhibitors compared with topical corticosteroids in the treatment of vitiligo: a systematic review and meta-analysis. J Am Acad Dermatol 2020; 82: 243-245.
19. Lee JH, Kwon HS, Jung H, et al. Treatment outcomes of topical calcineurin inhibitor therapy for patients with vitiligo: a systematic review and meta-analysis. JAMA Dermatol 2019; 155: 929-938.
20. Pharmaceutical Benefits Scheme (PBS). Pimecrolimus. Canberra: Australian Government Department of Health, Disability and Ageing; 2020. Available online at: https://www.pbs.gov.au/medicine/item/8802G (accessed October 2025).
21. Al-Smadi K, Ali M, Alavi SE, et al. Using a topical formulation of vitamin D for the treatment of vitiligo: a systematic review. Cells 2023; 12: 2387.
22. Esmat SM, El-Mofty M, Rasheed H, et al. Efficacy of narrow band UVB with or without OMP in stabilization of vitiligo activity in skin photo-types (III-V): a double-blind, randomized, placebo-controlled, prospective, multicenter study. Photodermatol Photoimmunol Photomed 2022; 38: 277-287.
23. Mohammad TF, Al-Jamal M, Hamzavi IH, et al. The Vitiligo Working Group recommendations for narrowband ultraviolet B light phototherapy treatment of vitiligo. J Am Acad Dermatol 2017; 76: 879-888.
24. Percivalle S, Piccinno R, Caccialanza M, Forti S. Narrowband ultraviolet B phototherapy in childhood vitiligo: evaluation of results in 28 patients. Pediatr Dermatol 2012; 29: 160-165.
25. Bae JM, Jung HM, Hong BY, et al. Phototherapy for vitiligo: a systematic review and meta-analysis. JAMA Dermatol 2017; 153: 666-674.
26. Le Duff F, Fontas E, Giacchero D, et al. 308-nm excimer lamp vs. 308-nm excimer laser for treating vitiligo: a randomized study. Br J Dermatol 2010; 163: 188-192.
27. Lopes C, Trevisani VFM, Melnik T. Efficacy and safety of 308-nm monochromatic excimer lamp versus other phototherapy devices for vitiligo: a systematic review with meta-analysis. Am J Clin Dermatol 2016; 17: 23-32.
28. Smith MP, Ly K, Thibodeaux Q, Bhutani T, Nakamura M. Home phototherapy for patients with vitiligo: challenges and solutions. Clin Cosmet Investig Dermatol 2019; 12: 451-459.
29. Shenoi SD, Prabhu S; Indian Association of Dermatologists, Venereologists and Leprologists. Photochemotherapy (PUVA) in psoriasis and vitiligo. Indian J Dermatol Venereol Leprol 2014; 80: 497-504.
30. Radakovic-Fijan S, Fürnsinn-Friedl AM, Hönigsmann H, Tanew A. Oral dexamethasone pulse treatment for vitiligo. J Am Acad Dermatol 2001; 44: 814-817.
31. Pasricha JS, Khaitan BK. Oral mini-pulse therapy with betamethasone in vitiligo patients having extensive or fast-spreading disease. Int J Dermatol 1993; 32: 753-757.
32. Singh H, Kumaran MS, Bains A, Parsad D. A randomized comparative study of oral corticosteroid minipulse and low-dose oral methotrexate in the treatment of unstable vitiligo. Dermatol Basel Switz 2015; 231: 286-290.
33. Patra S, Khaitan BK, Sharma VK, Khanna N. A randomized comparative study of the effect of betamethasone oral mini-pulse therapy versus oral azathioprine in progressive nonsegmental vitiligo. J Am Acad Dermatol 2021; 85: 728-729.
34. Mehta H, Kumar S, Parsad D, Bishnoi A, Vinay K, Kumaran MS. Oral cyclosporine is effective in stabilizing active vitiligo: results of a randomized controlled trial. Dermatol Ther 2021; 34: e15033.
35. Bae JM, Kim M, Lee HH, et al. Increased risk of vitiligo following anti-tumor necrosis factor therapy: a 10-year population-based cohort study. J Invest Dermatol 2018; 138: 768-774.
36. Grimes PE, Nashawati R. Depigmentation therapies for vitiligo. Dermatol Clin 2017; 35: 219-227.
37. Tan ES, Sarkany R. Topical monobenzyl ether of hydroquinone is an effective and safe treatment for depigmentation of extensive vitiligo in the medium term: a retrospective cohort study of 53 cases. Br J Dermatol 2015; 172: 1662-1664.
38. van Geel N, Depaepe L, Speeckaert R. Laser (755 nm) and cryotherapy as depigmentation treatments for vitiligo: a comparative study. J Eur Acad Dermatol Venereol 2015; 29: 1121-1127.
39. van Geel N, Ongenae K, Naeyaert JM. Surgical techniques for vitiligo: a review. Dermatol Basel Switz 2001; 202: 162-166.
40. Schwartz DM, Bonelli M, Gadina M, O’Shea JJ. Type I/II cytokines, JAKs, and new strategies for treating autoimmune diseases. Nat Rev Rheumatol 2016; 12: 25-36.
41. Solimani F, Meier K, Ghoreschi K. Emerging topical and systemic JAK inhibitors in dermatology. Front Immunol 2019; 10: 2847.
42.Rosmarin D, Passeron T, Pandya AG, et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N Engl J Med 2022; 387: 1445-1455.
43. Jha AK, Sarkar R, Udayan UK, Roy PK, Jha AK, Chaudhary RKP. Bimatoprost in Dermatology. Indian Dermatol Online J 2018; 9: 224-228.
44. Silpa-Archa N, Likittanasombat S, Apinuntham C, et al. The efficacy of bimatoprost ophthalmic solution combined with NB-UVB phototherapy in non-segmental and segmental vitiligo: a single-blind randomized controlled study. Sci Rep 2023; 13: 6438.
45. Ezzedine K, Peeva E, Yamaguchi Y, et al. Efficacy and safety of oral ritlecitinib for the treatment of active nonsegmental vitiligo: a randomized phase 2b clinical trial. J Am Acad Dermatol 2023; 88: 395-403.
46. Liu LY, Strassner JP, Refat MA, Harris JE, King BA. Repigmentation in vitiligo using the Janus kinase inhibitor tofacitinib may require concomitant light exposure. J Am Acad Dermatol 2017; 77: 675-682.e1.
47. Craiglow BG, King BA. Tofacitinib citrate for the treatment of vitiligo: a pathogenesis-directed therapy. JAMA Dermatol 2015; 151: 1110-1112.
48. Ezzedine K, Soliman AM, Rosmarin D, Pandya AG, Schlosser BJ, van Geel N. Patient-reported outcomes following 24 weeks of treatment with upadacitinib in adults with non-segmental vitiligo: results from a phase 2, randomized, double-blind, dose-ranging study. Br J Dermatol 2024; 190: ii64-ii65.
49. Dong J, Huang X, Ma LP, et al. Baricitinib is effective in treating progressing vitiligo in vivo and in vitro. Dose Response 2022; 20: 15593258221105370.
50. Pandya AG, Ezzedine K, Passeron T, et al. Efficacy and safety of the oral Janus kinase 1 inhibitor povorcitinib in patients with extensive vitiligo in a phase 2, randomized, double-blinded, dose-ranging, placebo-controlled study. J Am Acad Dermatol 2025; 93: 946-955.
51. Yagi K, Ishida Y, Otsuka A, Kabashima K. Two cases of vitiligo vulgaris treated with topical Janus kinase inhibitor delgocitinib. Australas J Dermatol 2021; 62: 433-434.
52. Phan K, Phan S, Shumack S, Gupta M. Repigmentation in vitiligo using janus kinase (JAK) inhibitors with phototherapy: systematic review and meta-analysis. J Dermatolog Treat 2022; 33: 173-177.
53. Jacquemin C, Rambert J, Guillet S, et al. Heat shock protein 70 potentiates interferon alpha production by plasmacytoid dendritic cells: relevance for cutaneous lupus and vitiligo pathogenesis. Br J Dermatol 2017; 177: 1367-1375.
54. Richmond JM, Strassner JP, Zapata L, et al. Antibody blockade of IL-15 signaling has the potential to durably reverse vitiligo. Sci Transl Med 2018; 10: eaam7710.
55. Dwivedi M, Laddha NC, Arora P, Marfatia YS, Begum R. Decreased regulatory T-cells and CD4(+) /CD8(+) ratio correlate with disease onset and progression in patients with generalized vitiligo. Pigment Cell Melanoma Res 2013; 26: 586-591.
56. Sun X, Sheng A, Xu AE. Successful treatment of vitiligo with crisaborole ointment: a report of two cases. Br J Dermatol 2023; 188: 436-437.
57. Khemis A, Fontas E, Moulin S, Montaudié H, Lacour JP, Passeron T. Apremilast in combination with narrowband UVB in the treatment of vitiligo: a 52-week monocentric prospective randomized placebo-controlled study. J Invest Dermatol 202; 140: 1533-1537.e2.
58. Lim HW, Grimes PE, Agbai O, et al. Afamelanotide and narrowband UV-B phototherapy for the treatment of vitiligo: a randomized multicenter trial. JAMA Dermatol 2015; 151: 42-50.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.