Microscopic haematuria: a practical guide for GPs
Microscopic haematuria is a subtle finding that can reflect causes ranging from benign transient conditions to serious renal or urological disease. A structured, risk-based approach helps clinicians identify pathology without over-investigating low-risk patients.
- Microscopic haematuria requires systematic, risk-stratified evaluation, as it may indicate urological or glomerular disease despite often being asymptomatic and benign.
- Formal urine microscopy is essential to confirm haematuria and assess for glomerular features such as dysmorphic red blood cells, casts and proteinuria.
- Risk assessment (based on age, smoking history, sex, history of analgesic misuse and symptoms) guides the need for imaging, cystoscopy and specialist referral.
- Imaging choice should match risk, with ultrasound appropriate for low- or intermediate-risk patients and CT intravenous pyelogram preferred for high-risk patients or those with concerning features.
- Referral to a urologist is indicated for intermediate- or high-risk patients, whereas referral to a nephrologist is required when glomerular features are present or renal disease is suspected.
Microscopic haematuria is the presence of blood in the urine that is visible only when viewed under a microscope. Most GPs will see this regularly, often as an unexpected finding on routine testing. It can affect 2 to 5% of the Australian adult population and often presents a diagnostic challenge.1 The clinical significance of microscopic haematuria lies in its potential association with serious underlying pathology. Although it may be benign, observational studies have demonstrated that about 20% of patients with macroscopic haematuria have an underlying urinary tract malignancy, compared with 2 to 3% of those with microscopic haematuria.2 This relatively low but clinically significant cancer detection rate necessitates systematic evaluation protocols.
Careful assessment and evaluation are crucial to ensure prompt diagnosis and an appropriate referral pathway, given that the presentation may reflect a primary renal or urological disorder. This comes at a time when there are several ongoing randomised trials focused on screening for microscopic haematuria to increase the early diagnosis of bladder cancer.2-4 Recent updates from the 2025 American Urological Association guidelines provide improved risk stratification tools to help guide investigations.5
This article provides practical guidance on the investigation and management of microscopic haematuria, evidence-based approaches to risk assessment and appropriate referral pathways that balance diagnosis with avoiding over-investigation. A case vignette is presented in Box 1.
Definition and diagnosis
Microscopic haematuria is defined as the presence of three or more red blood cells (RBCs) per high-power field on microscopic examination of a properly collected midstream urine specimen. However, in Australian practice, automated laboratory analysers report results in RBCs per litre, and Australian guidelines accordingly use this unit, with a commonly used threshold of greater than 10 × 10⁶ RBCs/L.6 Urinary RBCs are not adjusted for concentration and hence can be under- or overestimated, emphasising the importance of repeat testing.
Importantly, urine dipstick testing alone is insufficient for diagnosis. A urine dipstick result that is positive for trace blood or greater reflects the detection of haemoglobin peroxidase activity. False positives can occur in the presence of myoglobinuria, concentrated urine, traces of povidone-iodine or menstrual contamination. Therefore, formal microscopic examination is necessary to confirm true haematuria.
Dysmorphic red blood cells and acanthocytes
In microscopic haematuria, the presence of dysmorphic RBCs (especially acanthocytes, a characteristic subtype) under phase contrast microscopy can suggest a glomerular source of bleeding. Dysmorphic RBCs are abnormally shaped RBCs resulting from mechanical trauma during passage through disrupted glomerular basement membranes and through osmotic changes during tubular passage.
Historically, a cut-off of greater than 80% dysmorphic RBCs was used to indicate glomerular disease, whereas less than 20% suggested a nonglomerular cause. However, there is considerable heterogeneity in the sensitivity and specificity of these cut-offs depending on the definitions, study populations and methods. In one cohort, one-third of patients with at least 40% dysmorphic RBCs had urological lesions that required treatment.7 Thus, urinary dysmorphic RBCs should be interpreted in the broader clinical context rather than used in isolation to guide referrals.
Aetiology and differential diagnosis
Transient causes
Numerous benign conditions can cause transient microscopic haematuria. Common causes include menstruation, vigorous physical exercise, sexual intercourse, urinary tract infections and recent urological procedures.
Glomerular causes
Glomerular haematuria encompasses a broad spectrum of kidney diseases. Immunoglobulin A nephropathy is the most common renal disorder, leading to chronic kidney disease and causing isolated microscopic haematuria. Many other less common glomerular causes exist but may include postinfectious glomerulonephritis, lupus nephritis, antineutrophil cytoplasmic antibody-associated vasculitis and genetic conditions such as the spectrum of Alport syndrome and thin basement membrane nephropathy. The likelihood of glomerular haematuria relies heavily on urine microscopy findings. In particular, the presence of dysmorphic RBCs, RBC or granular casts, or proteinuria warrants referral of the patient to a nephrologist.
Nonglomerular causes
Nonglomerular causes of haematuria include urological conditions affecting anywhere along the urinary tract and include renal parenchymal and systemic disorders that lead to bleeding into the urinary tract. Common causes include nephrolithiasis, urinary tract infections (cystitis, urethritis, pyelonephritis), benign prostatic hyperplasia and various cystic kidney diseases.
Malignancy, including cancers of the kidneys, ureters, bladder and prostate, is the most concerning among the nonglomerular causes of microscopic haematuria. Other contributors include coagulopathies, anticoagulant therapy, radiation cystitis and rare vascular anomalies such as nutcracker syndrome. Although anticoagulant therapy will increase the risk of microscopic haematuria, persistent haematuria still merits routine workup (as with rectal bleeding).
Clinical assessment
History taking
Key elements of history taking include reviewing:
- the duration and pattern of haematuria (e.g. through previous urine samples)
- relevant lifestyle changes at the time of testing (i.e. vigorous exercise)
- the medication history (particularly NSAIDs and anticoagulants)
- any associated urinary symptoms such as dysuria, nocturia, urinary frequency or flank pain.
Cancer risk assessment requires careful attention to the patient’s smoking history, use of Bex or Vincent’s powders, occupational exposures (particularly in industries using dyes, rubber, paints or chemicals), family history of urological malignancies and previous history of pelvic irradiation or chemotherapy. Age represents a key risk factor, with the malignancy risk increasing significantly in patients older than 35 to 40 years of age.
Physical examination
Physical examination should include blood pressure measurement, abdominal examination for masses or tenderness and assessment for signs of systemic disease. In male patients, digital rectal examination may be indicated to assess the prostate. In female patients, gynaecological and pelvic examinations may help exclude gynaecological sources of bleeding.
Investigation strategy
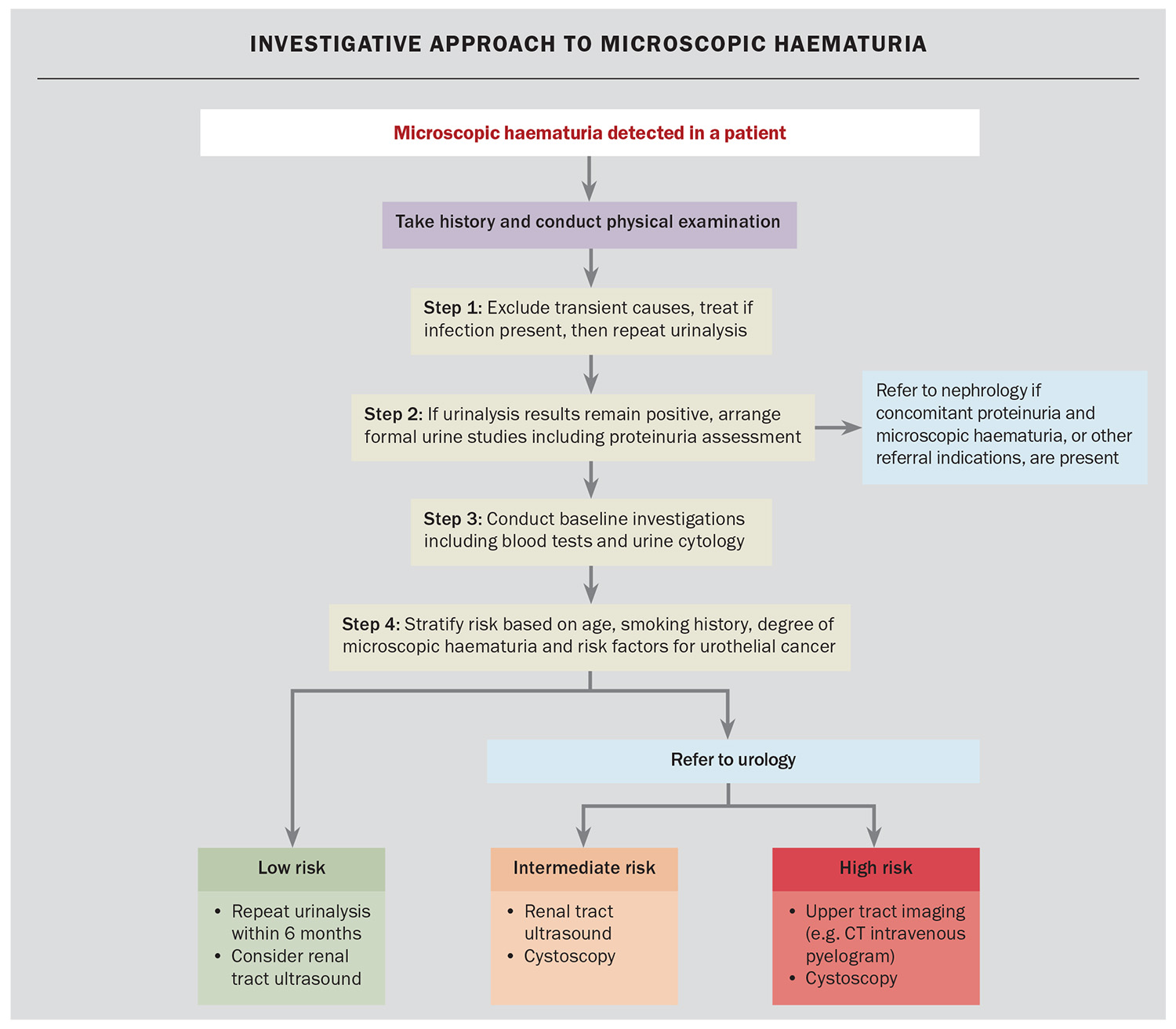
The diagnostic approach to microscopic haematuria is illustrated in the Flowchart.
Step 1: exclude transient causes and treat infection
- Repeat a formal urine microscopic assessment two weeks after menstruation, vigorous exercise or a confirmed urinary tract infection. Persistent microscopic haematuria warrants full evaluation. The importance of accurate urine testing for haematuria is outlined in Box 2.
Step 2: arrange formal urine studies
- Urine studies should include microscopy and culture, assessment of RBC morphology (for dysmorphic RBCs), evaluation for casts and measurement of the urine protein–creatinine ratio (uPCR).
- Concomitant proteinuria: uPCR ≥30 mg/mmol raises the likelihood of glomerular disease, and together with haematuria, doubles the risk of chronic kidney disease.8 These patients should be referred to a nephrologist.
Step 3: conduct baseline work-up
- Perform blood tests: full blood count; electrolytes, urea and creatinine; comprehensive metabolic panel; liver function tests; C-reactive protein level; and coagulation profile.
- Assess urine cytology on three consecutive days. These samples should always be a mid-morning, well-hydrated sample as overnight samples can contain degraded cells.
- Screen for sexually transmitted infections in appropriate populations.
Step 4: refer patient to a urologist based on risk stratification and baseline workup
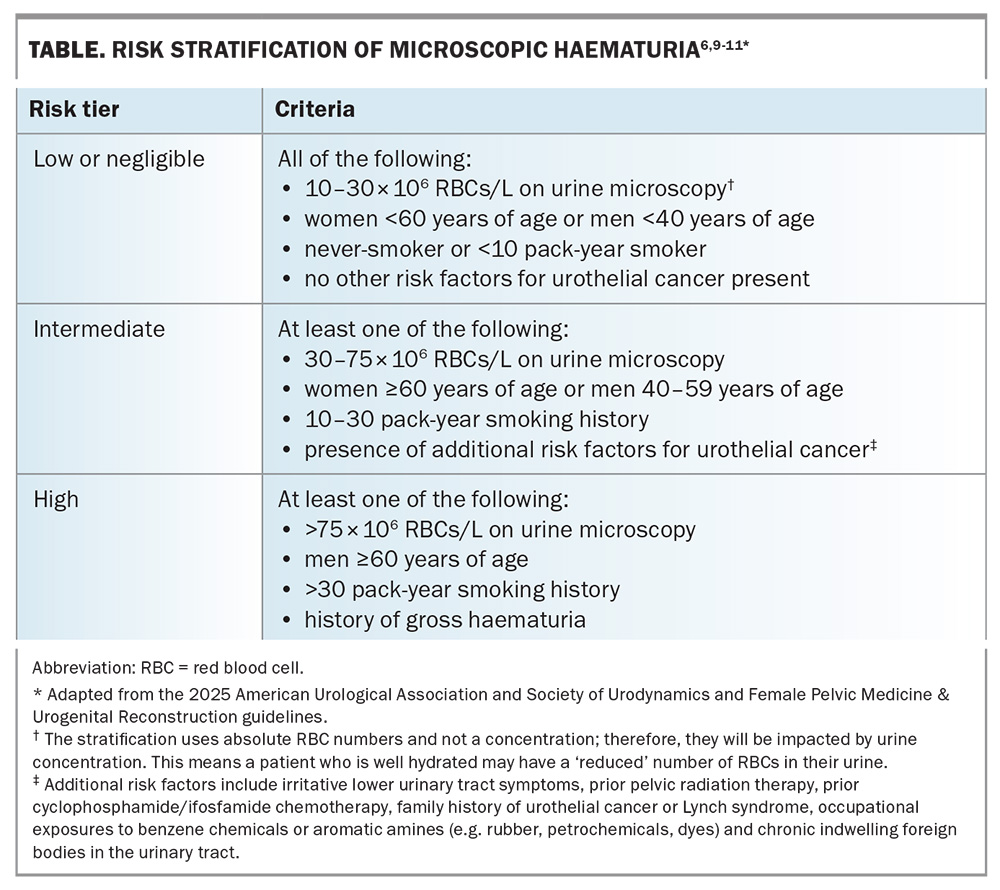
- The 2025 American Urological Association and Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction amendment divides patients into low- or negligible-, intermediate- and high-risk groups (Table).6,9-11 Age cut-offs differ by sex because women aged younger than 60 years carry a lower malignancy risk.
Step 5: consider imaging
- The choice of imaging modality depends on the clinical context and patient risk factors.
- Renal tract ultrasound is radiation-free, can detect most renal masses 2 cm and greater in size and is recommended for low- and intermediate-risk cohorts. The Detecting Bladder Cancer Using the UroMark Test (DETECT I) trial involving 3556 patients showed that ultrasound safely replaced CT intravenous pyelogram (CT IVP) in microscopic but not macroscopic haematuria.12 It is important to note that ultrasound has poor sensitivity for bladder lesions and hence is combined with cystoscopy in intermediate- or high-risk patients.
- CT IVP remains the gold-standard investigation for upper renal tract malignancy and renal calculi in high-risk or symptomatic patients. Venous contrast has a low to negligible risk with all estimated glomerular filtration rates (eGFRs).13 We recommend that all intermediate- and high-risk patients who need contrast-enhanced imaging undergo CT IVP.
- Contemporary practice increasingly favours CT IVP for higher-risk patients while reserving ultrasound for lower-risk scenarios.
Step 6: perform cystoscopy
- Direct visualisation through cystoscopy with bilateral retrograde pyelography remains essential for comprehensive urinary tract assessment.
- Flexible cystoscopy visualises more than 90% of bladder tumours. It is mandatory for any episode of gross haematuria and persistent microscopic haematuria in intermediate- or high-risk patients.14
- It is important to note that urine cytology cannot replace cystoscopy with bilateral retrograde pyelography.14
- There is evolving use of urinary biomarkers to avoid the routine use of cystoscopy in some patients. Emerging evidence indicates that some urinary genomic tests have a negative predictive value of 99% for urothelial carcinoma and reduce the need for cystoscopy by 59%.15 However, these are not currently available in Australia.
Referral criteria and pathways
Urology referral
Referral to a urologist is indicated for all patients with visible macroscopic haematuria regardless of age, except in cases of clearly synpharyngitic haematuria (occurring simultaneously with an upper respiratory tract infection) or suspected glomerular basement membrane disease, where referral to a nephrologist is more appropriate. These patients will likely have deranged renal function, a high percentage of dysmorphic RBCs and a raised uPCR.
For microscopic haematuria, urological assessment is recommended for symptomatic patients of any age, patients who do not fit into the nephrology referral category and intermediate- or high-risk patients (as outlined in the Table).
Nephrology referral
Referral to a nephrologist is indicated for all patients in whom urological causes have been excluded or if there are findings to suggest a glomerular disease, such as a concurrent raised uPCR, dysmorphic RBCs, RBC casts or a family history of kidney disease. Some clear indications include:
- evidence of a declining eGFR (>10 mL/min/1.73 m² decrease over five years or >5 mL/min/1.73 m² decrease within one year)
- stage 4 or 5 chronic kidney disease (eGFR <30 mL/min/1.73 m²)
- significant proteinuria (uPCR >30 mg/mmol)
- isolated microscopic haematuria with hypertension in patients <40 years of age
- macroscopic haematuria coinciding with upper respiratory tract infection (synpharyngitic).
Nonreferral pathway
For low-risk patients, close monitoring without a referral may be appropriate. Many real-world studies have shown malignancy rates of less than 0.5% in these cohorts.16,17 Patients who fall into this category should undergo repeat urine microscopy within six months, followed by another assessment six months later if the first repeat test result is negative. If the results of both follow-up tests are negative, no further investigation is required. However, if haematuria persists at six months, renal tract imaging and referral to a urologist should be arranged. The case vignette is revisited in Box 3.
Conclusion
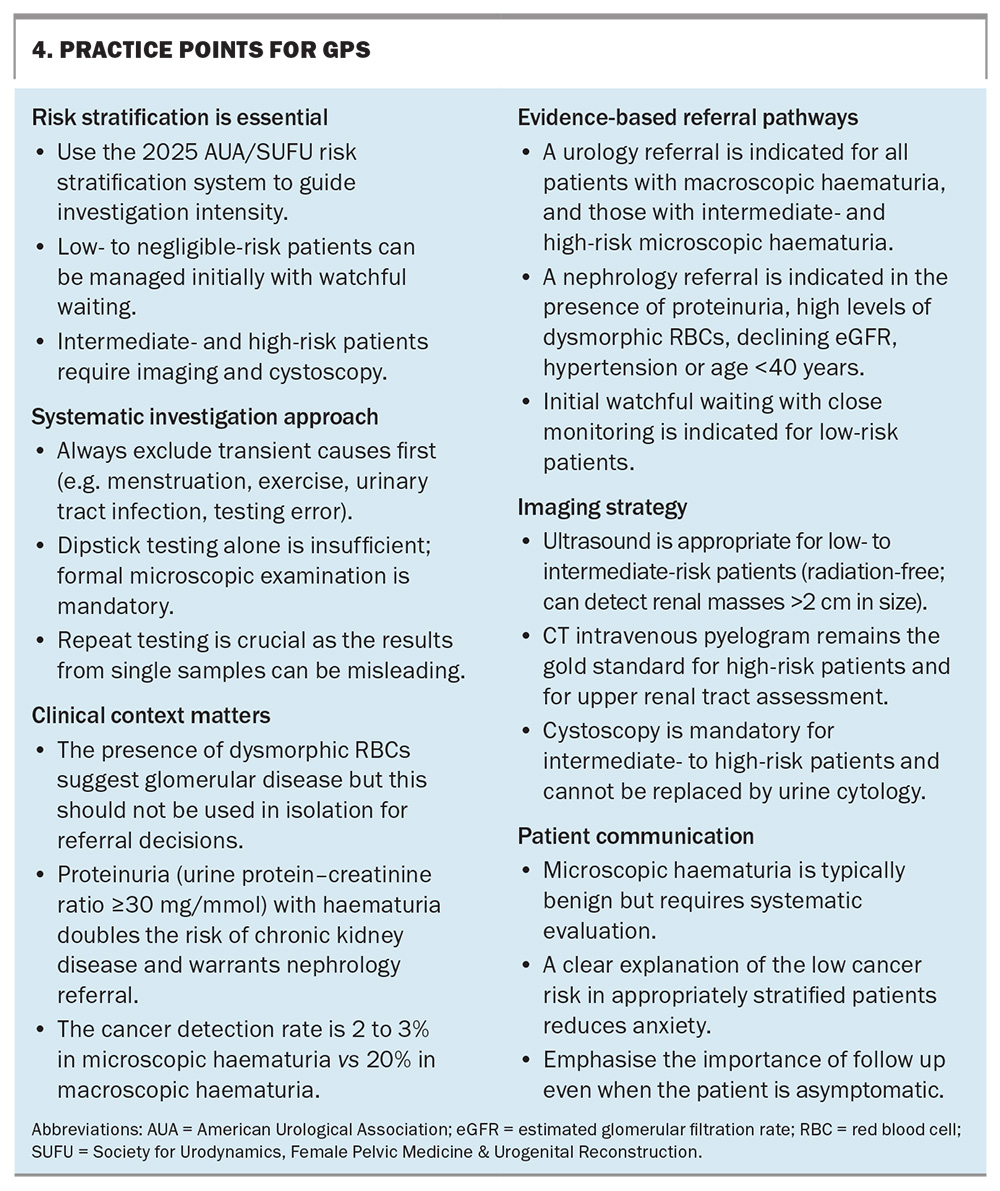
When microscopic haematuria is detected, evaluation should follow a systematic approach based on the patient’s risk of cancer or kidney disease. Initial management should focus on excluding transient causes and arranging appropriate baseline investigations, including formal urine microscopy. Risk assessment should incorporate factors such as age, smoking history, history of analgesic misuse, sex and associated symptoms.
In high-risk patients, prompt imaging and early referral to a urologist prevents delays in cancer diagnosis. Low-risk patients can be managed with careful monitoring and further investigation if haematuria persists. The presence of glomerular features (e.g. hypertension, existing kidney disease, proteinuria, dysmorphic RBCs or RBC casts on urine microscopy) warrants nephrology assessment, regardless of any concurrent urological evaluations.
Clear communication with patients is important; although microscopic haematuria is typically benign, appropriate investigation is still necessary. This reassurance assists with minimising anxiety while ensuring adherence to investigations and follow up, particularly as this patient cohort is often asymptomatic. Practice points for GPs are provided in Box 4.
COMPETING INTERESTS: None.
References
1. Chadban SJ, Briganti EM, Kerr PG, et al. Prevalence of kidney damage in Australian adults: the AusDiab kidney study. J Am Soc Nephrol 2003; 14(7 Suppl 2): S131-S138.
2. Takeuchi M, McDonald JS, Takahashi N, et al. Cancer prevalence and risk stratification in adults presenting with hematuria: a population-based cohort study. Mayo Clin Proc Innov Qual Outcomes 2021; 5: 308-319.
3. Joukar F, Hassanipour S, Atefi A, et al. Prevalence and associated factors for asymptomatic microscopic hematuria in adults in the PERSIAN Guilan cohort study (PGCS). Sci Rep 2024; 14: 3452.
4. Waisbrod S, Natsos A, Wettstein MS, et al. Assessment of diagnostic yield of cystoscopy and computed tomographic urography for urinary tract cancers in patients evaluated for microhematuria: a systematic review and meta-analysis. JAMA Netw Open 2021; 4: e218409-e218409.
5. Barocas DA, Lotan Y, Matulewicz RS, et al. Updates to microhematuria: AUA/SUFU guideline (2025). J Urol 2025; 213: 547-557.
6. Barocas DA, Boorjian SA, Alvarez RD, et al. Microhematuria: AUA/SUFU guideline. J Urol 2020; 204: 778-786.
7. Koo KC, Lee KS, Choi AR, Rha KH, Hong SJ, Chung BH. Diagnostic impact of dysmorphic red blood cells on evaluating microscopic hematuria: the urologist’s perspective. Int Urol Nephrol 2016; 48: 1021-1027.
8. Johnson DW, Jones GRD, Mathew TH, et al.; Australasian Proteinuria Consensus Working Group. Chronic kidney disease and measurement of albuminuria or proteinuria: a position statement. Med J Aust 2012; 197: 224-225.
9. Bromage SJ, Liew M, Moore K, Raju B, Shackley D. The evaluation of CT urography in the haematuria clinic. J Clin Urol 2013; 6: 153-157.
10. Eisenhardt A, Heinemann D, Rübben H, Heβ J. Haematuria work-up in general care-a German observational study. Int J Clin Pract 2017; 71: e12982.
11. Todenhöfer T, Hennenlotter J, Tews V, et al. Impact of different grades of microscopic hematuria on the performance of urine-based markers for the detection of urothelial carcinoma. Urol Oncol 2013; 31: 1148-1154.
12. Tan WS, Feber A, Sarpong R, et al.; DETECT I trial collaborators. Who should be investigated for haematuria? Results of a contemporary prospective observational study of 3556 patients. Eur Urol 2018; 74: 10-14.
13. McDonald JS, McDonald RJ, Carter RE, Katzberg RW, Kallmes DF, Williamson EE. Risk of intravenous contrast material-mediated acute kidney injury: a propensity score-matched study stratified by baseline-estimated glomerular filtration rate. Radiology 2014; 271: 65-73.
14. Heller MT, Tublin ME. In search of a consensus: evaluation of the patient with hematuria in an era of cost containment. AJR Am J Roentgenol 2014; 202: 1179-1186.
15. Lotan Y, Daneshmand S, Shore N. A multicenter prospective randomized controlled trial comparing Cxbladder Triage to cystoscopy in patients with microhematuria: the safe testing of risk for asymptomatic microhematuria trial. J Urol 2024; 212: 41-51.
16. Saxon GM, Patil D, Hammett J. Microhematuria in women: prevalence of malignancy and risk score evaluation. Urology 2022; 160: 34-39.
17. Sancı A, Oktar A, Gokce MI, et al. Comparison of microscopic hematuria guidelines as applied in 1018 patients with microscopic hematuria. Urology 2021; 154: 28-32.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.