Metabolic dysfunction-associated fatty liver disease: assessment in primary care
Metabolic dysfunction-associated fatty liver disease is increasingly common in Australia and screening is recommended for at-risk individuals. Noninvasive testing for liver fibrosis and assessment for comorbidities guide management and appropriate specialty referrals for these patients.
- Metabolic dysfunction-associated fatty liver disease (MAFLD) is the most common liver condition in Australia and is predicted to overtake alcohol-associated liver disease as the major cause of cirrhosis and requirement for liver transplantation.
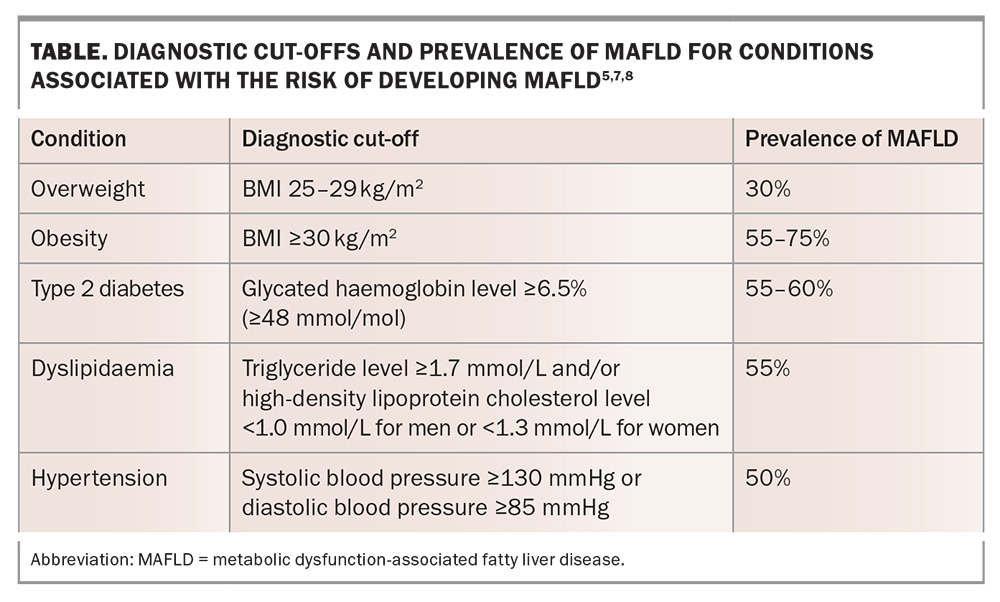
- MAFLD is defined by the presence of hepatic steatosis, plus obesity or overweight, type 2 diabetes or two or more metabolic risk factors.
- Abdominal ultrasound is the recommended first-line investigation to help diagnose hepatic steatosis.
- Patients with MAFLD are recommended to undergo noninvasive liver fibrosis testing based on the Fibrosis-4 (FIB-4) index. Patients with FIB-4 scores greater than 2.7 are at risk of advanced liver fibrosis and should be referred to a specialist.
- Indeterminate FIB-4 scores (i.e. between 1.3 and 2.7) should prompt further investigation to rule out fibrosis with liver elastography or a serum biomarker test (Hepascore or Enhanced Liver Fibrosis test).
- Low-risk patients with MAFLD (FIB-4 score <1.3) should be screened for underlying fibrosis in the community at least every three years.
The prevalence of metabolic dysfunction-associated fatty liver disease (MAFLD), formerly known as nonalcoholic fatty liver disease, is estimated to be 30% in Australia. Liver-associated deaths secondary to MAFLD are estimated to increase by 85% in Australia over the next decade.1-4 MAFLD is frequently encountered in primary care, prompting the publication of guidelines for GPs by the Gastroenterological Society of Australia in 2024. These guidelines aim to increase the identification and assessment of patients with MAFLD.5 This article summarises these recommendations with a practical focus for GPs in Australia.
Screening and diagnosis
MAFLD is defined by the presence of hepatic steatosis on imaging, plus obesity or overweight, type 2 diabetes and at least two of the following metabolic risk factors:
- central obesity
- hypertension
- hypertriglyceridaemia
- low level of high-density lipoprotein cholesterol
- prediabetes.1
The prevalence of MAFLD is 50 to 75% among adults who fulfil these criteria, leading to the recommendation that these patients should be assessed for MAFLD.5,6 The diagnostic cut-offs of these criteria are outlined in the Table.5,7,8 Abdominal ultrasound is the first-line test to help diagnose hepatic steatosis in people at risk of MAFLD, as it is relatively inexpensive, noninvasive and readily available. The sensitivity of ultrasound is lower in patients with obesity or a low degree of steatosis. Alternative measures of assessing hepatic steatosis include controlled attenuation parameter using FibroScan and MRI; however, they are not readily accessible in the community and do not attract Medicare rebates.
Comorbid conditions
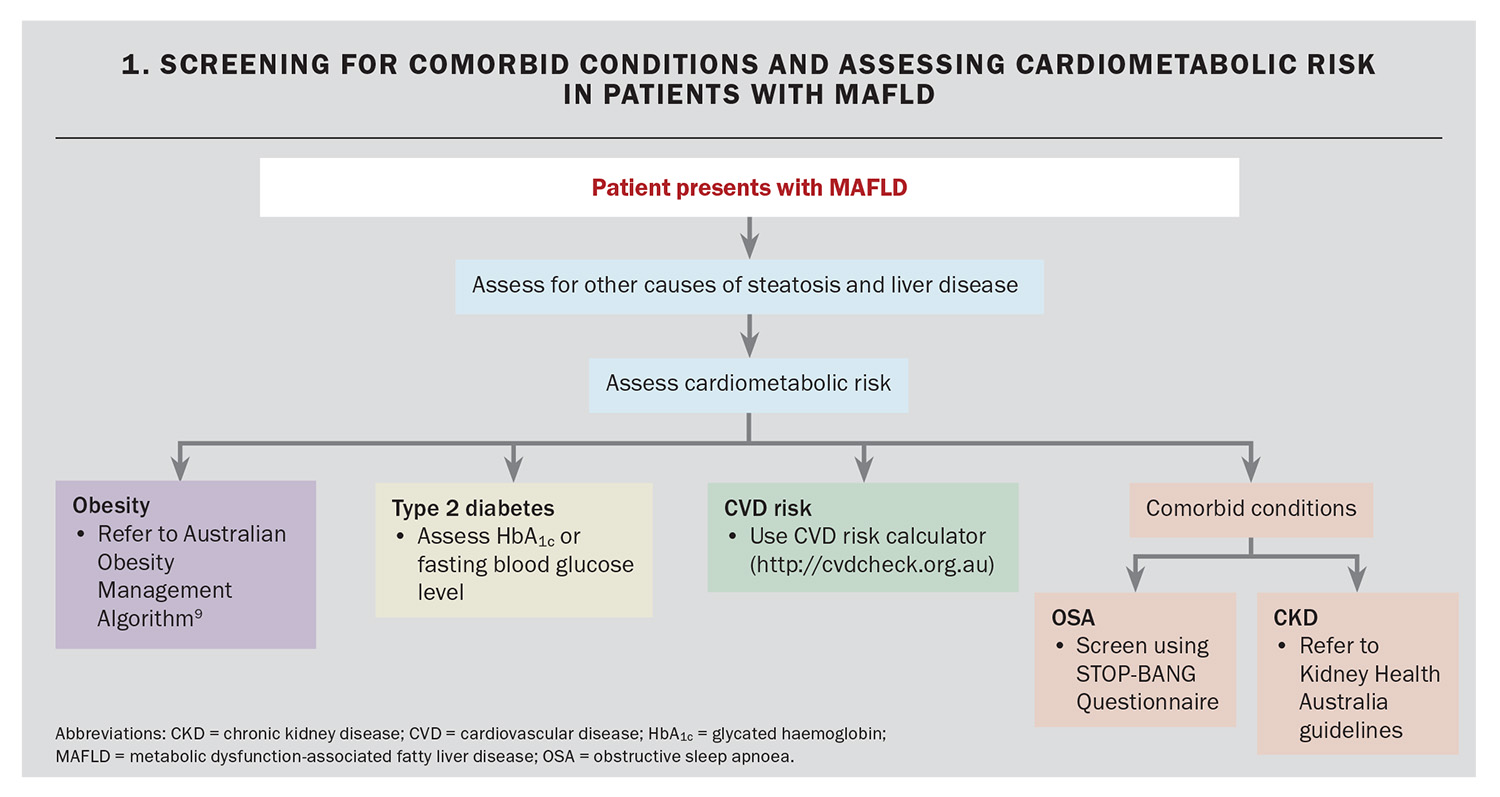
As part of the holistic care of patients with MAFLD, other conditions associated with metabolic dysfunction, such as obesity, should be assessed according to current Australian guidelines.9 Aboriginal and Torres Strait Islander populations are at particular risk as there is a greater prevalence of metabolic disease in these patients and the presence of metabolic disease is a major determinant of morbidity and mortality.10
MAFLD is also associated with a twofold increased risk of developing type 2 diabetes, the presence of which also increases the risk of developing cirrhosis in patients with MAFLD.11 Thus, patients with MAFLD should be assessed and monitored for type 2 diabetes using measurements of fasting blood glucose or glycated haemoglobin levels.
Cardiovascular disease is the most common cause of death among patients with MAFLD.12 Patients with MAFLD should be assessed and monitored for the presence and risk of cardiovascular disease in accordance with current Australian guidelines. Of note, statins should not be avoided and are safe for patients with MAFLD, including those with compensated cirrhosis.13
Baseline assessment for potential coexisting conditions of chronic kidney disease and obstructive sleep apnoea should be considered for people with MAFLD, given the shared pathogenesis between these conditions and MAFLD.14-17 An approach to screening for comorbid conditions and assessing cardiometabolic risk in patients with MAFLD is presented in Flowchart 1.9
Assessment
MAFLD and underlying liver disease
Alternative causes of hepatic steatosis include excessive alcohol intake, medications (including corticosteroids, methotrexate, antipsychotics, amiodarone, valproate and tamoxifen) and hepatitis C.5,18 People with MAFLD should be assessed for these causes of fatty liver and liver disease by taking a thorough medical history and using targeted serological testing, if appropriate. Current Australian recommendations for alcohol intake state that healthy men and women should drink no more than 10 standard drinks a week and no more than four standard drinks on any one day, while recognising that the risk of harm is lower when less alcohol is consumed.19 Alcohol consumption should be assessed in every patient with MAFLD, and those with advanced fibrosis or cirrhosis should be advised to be abstinent.
Additionally, people with MAFLD and elevated serum aminotransferase levels should undergo evaluation for hepatitis B and C, as well as iron overload. Furthermore, those with MAFLD who have additional risk factors based on history (e.g. intravenous drug use, high-risk sexual behaviour or a history of blood transfusions) should also be screened for viral hepatitis, regardless of liver enzyme levels, and an individual risk-based assessment should be carried out in each patient. Elevated ferritin levels are present in up to one-third of patients with MAFLD; however, this rarely indicates significant iron loading in the absence of an elevated transferrin saturation (>45%).
Liver fibrosis
People with MAFLD may develop liver injury and inflammation (known as metabolic dysfunction-associated steatohepatitis), which may lead to liver fibrosis. Advanced fibrosis (i.e. stages 3 and 4) is present in about 5% of patients with MAFLD and predicts an increased risk of future liver decompensation, hepatocellular carcinoma (HCC) and liver-associated mortality.20-22 Standard liver function tests and imaging modalities, including ultrasound and CT, are inaccurate in detecting advanced liver fibrosis and may yield normal findings in patients with cirrhosis.
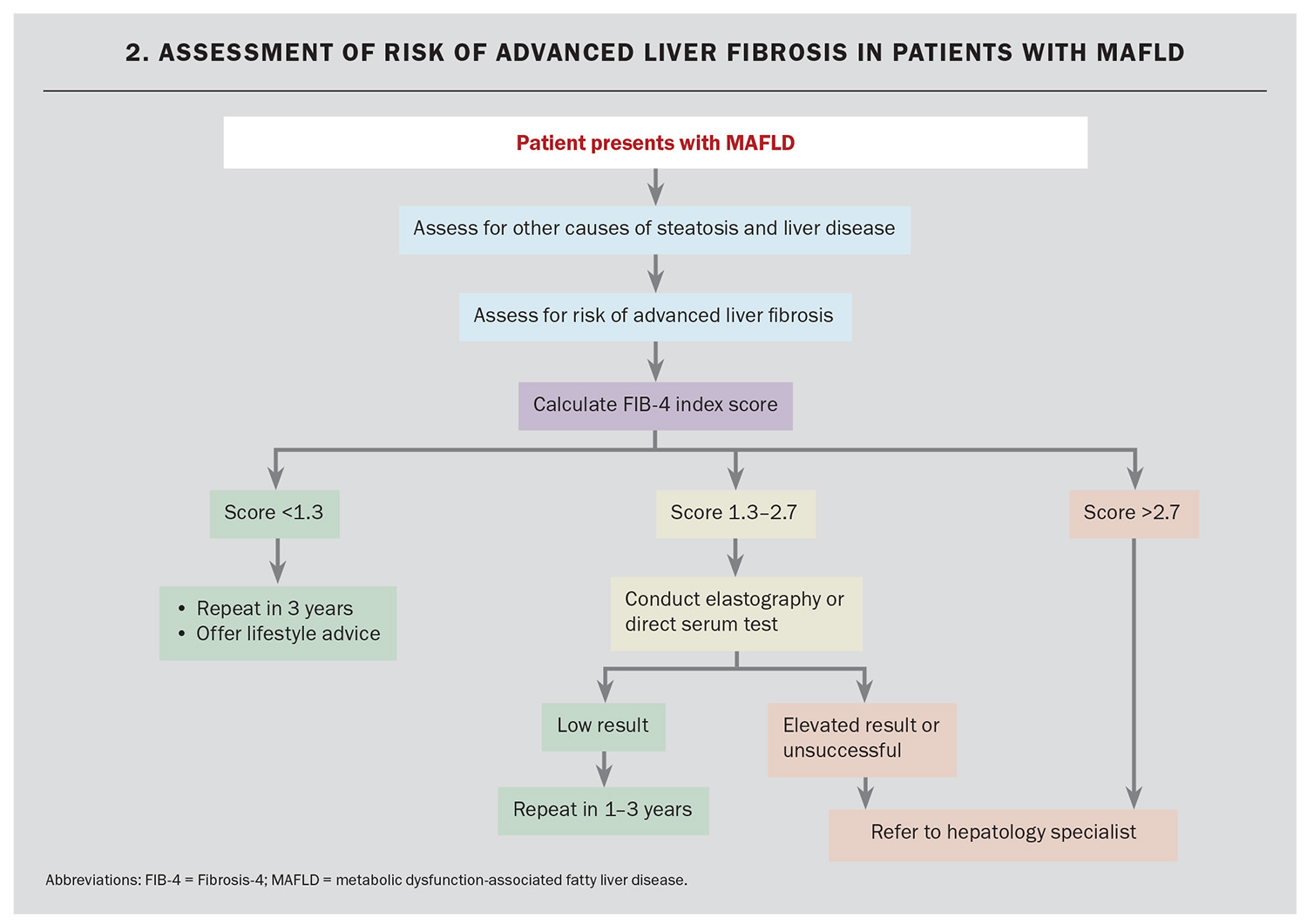
A noninvasive fibrosis test using commonly available parameters, such as the Fibrosis-4 (FIB-4) index, should be offered as an initial test to help rule out the risk of advanced liver fibrosis among patients with MAFLD (Flowchart 2). The FIB-4 score is calculated based on a patient’s age, aspartate aminotransferase level, alanine aminotransferase level and platelet count, and is readily available using online calculators (such as the one from the Liver Foundation, available online at https://liver.org.au/health-professionals/fib-4-calculator/). Age affects the interpretation of a FIB-4 score and, as such, the test should not be offered to those aged younger than 35 years.23 In this cohort, the prevalence of advanced liver fibrosis is very low. Second-line testing in the first instance, as detailed below, can be performed if there are clinical concerns.
A FIB-4 score less than 1.3 excludes advanced fibrosis with a sensitivity of 74% (95% confidence interval [CI], 72-76%). As the specificity of the FIB-4 index reduces with age, a cut-off score of 2 is used for patients older than 65 years.23 Patients with low scores should have liver disease risk factors (e.g. diabetes, excess weight and alcohol consumption) managed and can be monitored in the community with repeat screening in three years. In the primary care setting, a FIB-4 score greater than 2.7 is 94% specific for underlying advanced fibrosis, and patients with this result should be referred to hepatology specialist services for confirmatory testing and management.24,25
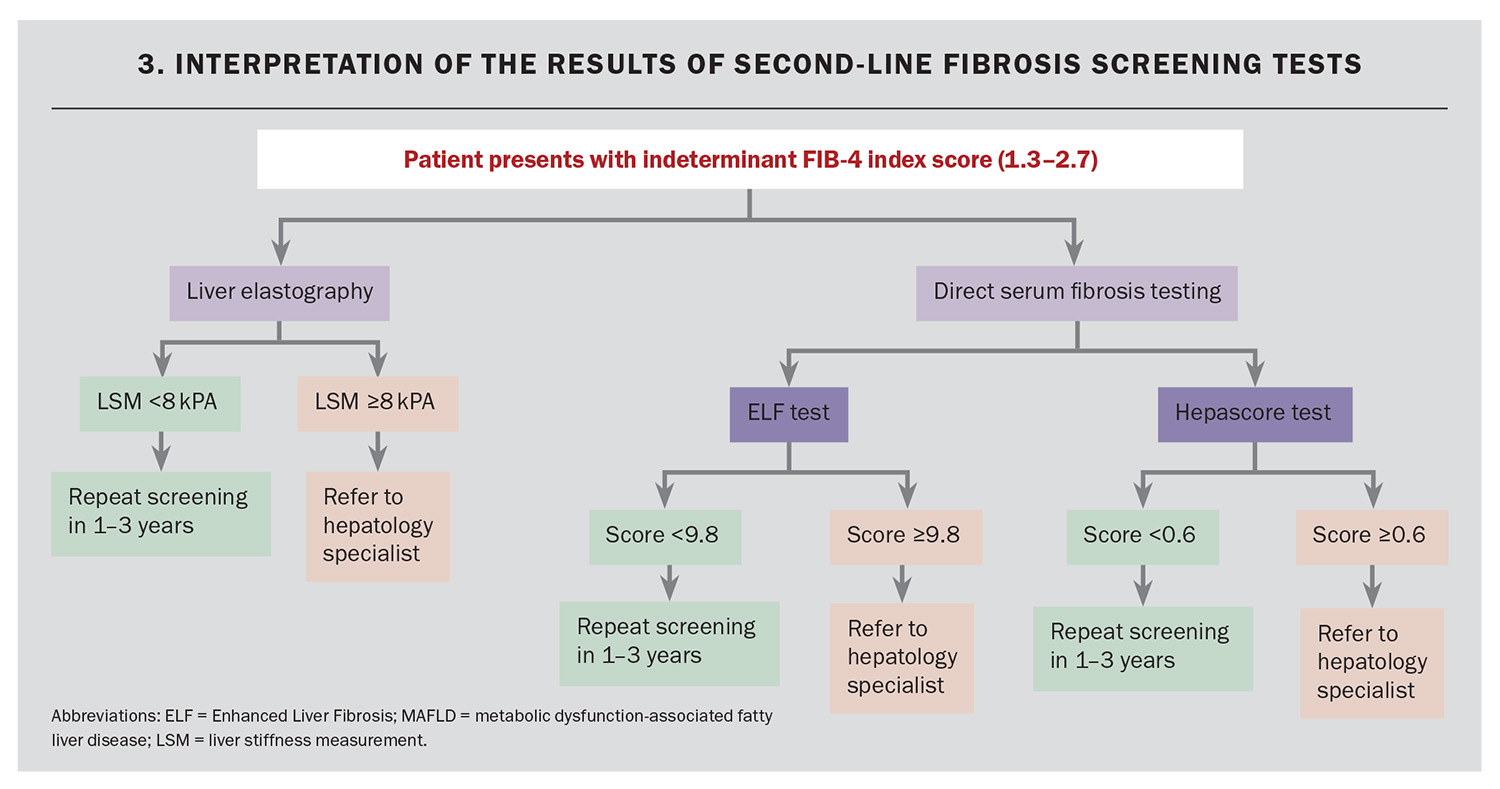
Results between these cut-offs (i.e. between 1.3 and 2.7) are indeterminate, and patients with these results should undergo second-line testing, where available (Flowchart 3).5 Second-line testing, including liver elastography (Fibroscan or shearwave elastography) or a direct serum fibrosis test (including Hepascore or Enhanced Liver Fibrosis [ELF] test), is recommended in this patient group owing to their higher accuracy.26 Liver elastography allows for liver stiffness measurement (LSM) and is quantified in kilopascals (kPa). The LSM correlates positively with liver fibrosis and predicts the likelihood of advanced liver fibrosis. Patients with an LSM less than 8 kPa are at low risk of liver fibrosis and liver-associated morbidity. It should be noted that the LSM may be falsely elevated in patients with conditions such as acute hepatitis, cholestasis (e.g. biliary obstruction), liver congestion (e.g. right heart failure) or focal liver lesions (e.g. tumours).27 Shearwave elastography has comparable accuracy with Fibroscan; however, shearwave requires greater technical expertise.
Direct serum fibrosis testing using Hepascore or the ELF test may be more readily available than liver elastography; however, these are not currently reimbursed by Medicare, limiting their use in the general population. Patients with a Hepascore less than 0.6 or an ELF test result less than 9.8 are at low risk of fibrosis and can be monitored in the community.
People with MAFLD and a high risk of advanced fibrosis should be referred for specialist review. Similarly, any patient with clinical, laboratory or imaging evidence of cirrhosis should be referred to a liver specialist.
Monitoring
People with MAFLD who have an initial noninvasive fibrosis test result indicating a low risk of advanced fibrosis (i.e. FIB-4 score <1.3) are recommended to undergo repeat noninvasive fibrosis testing in three years. People with MAFLD and a FIB-4 score between 1.3 and 2.7 who undergo elastography or a direct liver fibrosis serum test that indicates a low risk of advanced liver fibrosis should be offered repeat FIB-4 testing at least every three years. Patients with coexisting type 2 diabetes and poor glycaemic control or a rising aspartate aminotransferase level (by 10 IU/L) are at risk of more rapid fibrosis progression; repeat FIB-4 testing can be performed at a shorter interval (e.g. one to two yearly) in these individuals.
FIB-4 score calculation can be integrated into annual diabetes checks to identify those at risk of silent progression to cirrhosis. However, among people aged 75 years or older who have MAFLD and a low FIB-4 score (<1.3), the likelihood of developing cirrhosis or HCC over 10 years is <1%.28 Routine monitoring for fibrosis progression in this population should be performed on a case-by-case basis, depending on their coexisting conditions and life expectancy. Two cases involving different levels of risk of advanced fibrosis are presented in the Box.
Surveillance for HCC in people with cirrhosis increases the detection of small cancers, increases curative treatment options, increases survival and is cost effective.29 People with cirrhosis should undergo six-monthly surveillance for HCC using appropriate imaging, with or without serum alpha-fetoprotein testing.
Conclusion
MAFLD is an increasingly prevalent condition that requires risk assessment of underlying causes, coexisting conditions and complications (including liver fibrosis and cirrhosis). An ultrasound scan is the first-line investigation to assess for MAFLD and, once a diagnosis is made, screening should be performed for contributing conditions including type 2 diabetes, obesity, cardiovascular disease, chronic kidney disease and obstructive sleep apnoea. The first-line investigation for fibrosis includes calculation of the FIB-4 score, and individuals at risk of advanced fibrosis should be referred to a liver specialist. MT
COMPETING INTERESTS: Dr Lanigan: None. Professor Adams has received an institutional payment from Novo Nordisk; has received payments or honoraria from Novo Nordisk, CSL Behring and Dr Falk Pharma; has a patent associated with Hepascore; has been an Advisory Board Member with Novo Nordisk and CSL Behring; and is the Steering Committee Co-Chair for the Gastroenterology Society of Australia Consensus on assessment of MAFLD in primary care.
References
1. Eslam M, Newsome PN, Sarin SK, et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J Hepatol 2020; 73: 202-209.
2. Mahady SE, Adams LA. Burden of non-alcoholic fatty liver disease in Australia. J Gastroenterol Hepatol 2018; 33 Suppl 1: 1-11.
3. Adams LA, Roberts SK, Strasser SI, et al. Nonalcoholic fatty liver disease burden: Australia, 2019-2030. J Gastroenterol Hepatol 2020; 35: 1628-1635.
4. Younossi ZM, Golabi P, Paik JM, Henry A, Van Dongen C, Henry L. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology 2023; 77: 1335-1347.
5. MAFLD Consensus Statement Working Group. Recommendations for the assessment of metabolic dysfunction-associated fatty liver disease (MAFLD) in primary care: a consensus statement. Melbourne: Gastroenterological Society of Australia; 2024. Available online at: https://www.gesa.org.au/resources/clinical-practice-resources/metabolic-dysfunction-associated-fatty-liver-disease-mafld-consensus-statement/ (accessed June 2025).
6. Abeysekera KWM, Valenti L, Younossi Z, et al. Implementation of a liver health check in people with type 2 diabetes. Lancet Gastroenterol Hepatol 2024; 9: 83-91.
7. Vaz K, Kemp W, Majeed A, et al. Non-alcoholic fatty liver disease prevalence in Australia has risen over 15 years in conjunction with increased prevalence of obesity and reduction in healthy lifestyle. J Gastroenterol Hepatol 2023; 38: 1823-1831.
8. Alberti KG, Eckel RH, Grundy SM, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009; 120: 1640-1645.
9. Markovic TP, Proietto J, Dixon JB, et al. The Australian Obesity Management Algorithm: a simple tool to guide the management of obesity in primary care. Obes Res Clin Pract 2022; 16: 353-363.
10. Titmuss A, Davis EA, Brown A, Maple-Brown LJ. Emerging diabetes and metabolic conditions among Aboriginal and Torres Strait Islander young people. Med J Aust 2019; 210: 111-113.e1.
11. Adams LA, Anstee QM, Tilg H, Targher G. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 2017; 66: 1138-1153.
12. Kaya E, Yilmaz Y. Metabolic-associated fatty liver disease (MAFLD): a multi-systemic disease beyond the liver. J Clin Transl Hepatol 2022; 10: 329-338.
13. Zhou XD, Kim SU, Yip TC, et al. Long-term liver-related outcomes and liver stiffness progression of statin usage in steatotic liver disease. Gut 2024; 73: 1883-1892.
14. Byrne CD, Targher G. NAFLD: a multisystem disease. J Hepatol 2015; 62 Suppl 1: S47-S64.
15. Wang L, Liu H, Zhou L, et al. Association of obstructive sleep apnea with nonalcoholic fatty liver disease: evidence, mechanism, and treatment. Nat Sci Sleep 2024; 16: 917-933.
16. Jin S, Jiang S, Hu A. Association between obstructive sleep apnea and non-alcoholic fatty liver disease: a systematic review and meta-analysis. Sleep Breath 2018; 22: 841-851.
17. Chen S, Pang J, Huang R, Xue H, Chen X. Association of MAFLD with end-stage kidney disease: a prospective study of 337,783 UK Biobank participants. Hepatol Int 2023; 17: 595-605.
18. Liebe R, Esposito I, Bock HH, et al. Diagnosis and management of secondary causes of steatohepatitis. J Hepatol 2021; 74: 1455-1471.
19. Conigrave KM, Ali RL, Armstrong R, et al. Revision of the Australian guidelines to reduce health risks from drinking alcohol. Med J Aust 2021; 215: 518-524.
20. Ng CH, Lim WH, Hui Lim GE, et al. Mortality outcomes by fibrosis stage in nonalcoholic fatty liver disease: a systematic review and meta-analysis. Clin Gastroenterol Hepatol 2023; 21: 931-939.e5.
21. Ciardullo S, Perseghin G. Prevalence of NAFLD, MAFLD and associated advanced fibrosis in the contemporary United States population. Liver Int 2021; 41: 1290-1293.
22. Wong VW, Chu WC, Wong GL, et al. Prevalence of non-alcoholic fatty liver disease and advanced fibrosis in Hong Kong Chinese: a population study using proton-magnetic resonance spectroscopy and transient elastography. Gut 2012; 61: 409-415.
23. McPherson S, Hardy T, Dufour JF, et al. Age as a confounding factor for the accurate non-invasive diagnosis of advanced NAFLD fibrosis. Am J Gastroenterol 2017; 112: 740-751.
24. Kjaergaard M, Lindvig KP, Thorhauge KH, et al. Using the ELF test, FIB-4 and NAFLD fibrosis score to screen the population for liver disease. J Hepatol 2023; 79: 277-286.
25. Sung S, Al-Karaghouli M, Tam M, et al. Age-dependent differences in FIB-4 predictions of fibrosis in patients with MASLD referred from primary care. Hepatol Commun 2025; 9: e0609.
26. Boursier J, Guillaume M, Leroy V, et al. New sequential combinations of non-invasive fibrosis tests provide an accurate diagnosis of advanced fibrosis in NAFLD. J Hepatol 2019; 71: 389-396.
27. Castera L, Forns X, Alberti A. Non-invasive evaluation of liver fibrosis using transient elastography. J Hepatol 2008; 48: 835-847.
28. Hagstrom H, Talback M, Andreasson A, Walldius G, Hammar N. Ability of noninvasive scoring systems to identify individuals in the population at risk for severe liver disease. Gastroenterology 2020; 158: 200-214.
29. Lubel JS, Roberts SK, Strasser SI, et al. Australian recommendations for the management of hepatocellular carcinoma: a consensus statement. Med J Aust 2021; 214: 475-483.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.