Menopause after cancer: hormonal and nonhormonal management

Advances in cancer survival have led to more women living long enough to reach menopause or experiencing menopausal symptoms as a result of treatment. Management can be challenging in these cases, especially when hormone therapy is not appropriate. This article outlines the consequences of untreated menopause post-cancer, hormonal and nonhormonal treatment options, and when to refer to a specialist menopause service to support post-cancer patients in primary care.
- As cancer survival rates improve, more women are living long enough to reach menopause or enter early menopause induced by treatment, requiring management in the community setting.
- Cancer treatment-induced menopause often results in significant short- and long-term health impacts.
- Menopausal hormone therapy (MHT) remains the most effective treatment for menopause symptoms but is often contraindicated post-cancer, depending on hormone receptor status and cancer type.
- Several effective nonhormonal treatment options are available for women who are ineligible for, or prefer to avoid, MHT.
- GPs should refer women with complex or refractory menopause symptoms to a specialist menopause service such as the newly established Specialist Menopause Hubs in New South Wales.
As cancer survival rates improve in developed countries, survivorship issues related to menopausal symptoms, bone health and fertility implications are becoming more common.1 This pattern emerges from improved screening, earlier detection and treatment at younger ages.2 More cancer patients are also living long enough to reach menopause or enter early menopause as a side effect of their oncotherapy.3 Often, the symptoms of menopause are further exacerbated in some cancers by endocrine therapy that may continue for up to a decade. Such cases represent a particular challenge, as standard hormonal treatment regimens are often contraindicated.
Recently, an increased public awareness of the impacts of untreated menopause has led to a significant enhancement of government expenditure in this area. In New South Wales, four Specialist Menopause Hubs have been established as referral centres for complex and refractory menopause cases (https://aci.health.nsw.gov.au/networks/menopause/about).4 This project may see expansion to other states in the future.
These Specialist Menopause Hubs serve as referral and support services for GPs in the community, where most menopause care takes place. Women who are referred to a Specialist Menopause Hub will have their symptoms assessed and treatment initiated before returning to the community for ongoing care. GPs are therefore becoming increasingly involved in long-term menopause care, including in post-cancer cases. This article is a practical guide to caring for these women in the community.
Consequences of untreated iatrogenic menopause post-cancer
Short-term consequences
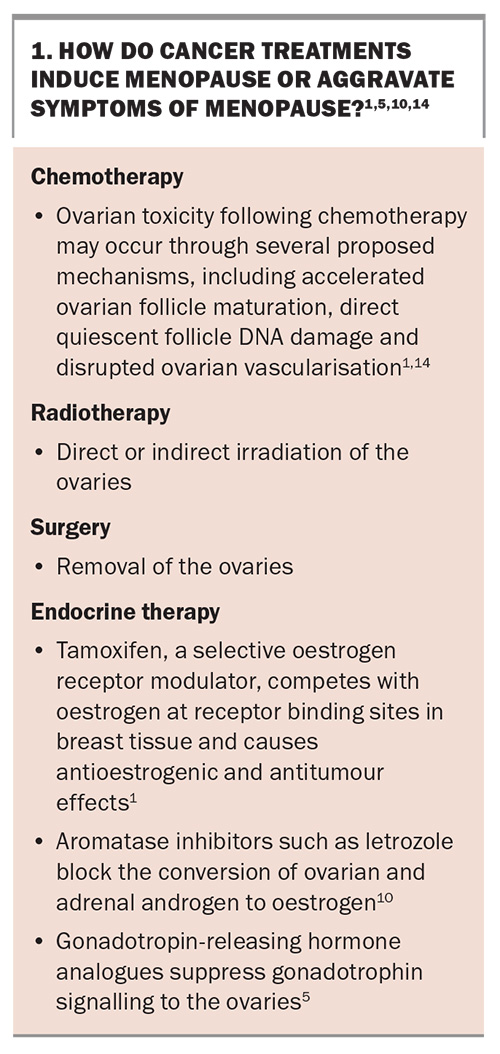
Cancer treatment can lead to an abrupt cessation of ovulatory function through surgical removal of the ovaries, chemotoxicity or radiotherapy.5-7 This type of acute iatrogenic menopause may be associated with more severe symptoms than those seen with physiological onset.6
Iatrogenic menopause may arouse sudden and severe vasomotor symptoms, sleep disturbance, fatigue, brain fog and vaginal dryness. These effects may be further compounded by ongoing chemotherapy, or endocrine therapy such as tamoxifen or aromatase inhibitors.5 Women often feel unprepared for and disturbed by these symptoms, as they expect to exit their arduous cancer treatment and return to ‘normal’ life.8 It is therefore important that they have access to effective information and treatment.
Long-term consequences
Although ovarian function may eventually return in some women, many will be rendered menopausal for the rest of their life.9 This early oestrogen deprivation is associated with a number of long-term health implications, including:
- accelerated bone loss and increased fracture risk10
- premature cardiovascular disease11
- severe and progressive genitourinary symptoms12
- persistent disturbing hot flushes and night sweats13
- infertility.
Therefore, it is important to discuss hormone replacement with eligible women and to consider nonhormonal alternatives for those who are ineligible for, or prefer to avoid, hormone therapy. The mechanisms by which cancer treatments induce menopause or aggravate symptoms are outlined in Box 1.1,5,10,14
Treatment selection
Hormone receptors
Although menopausal hormone therapy (MHT) is the most effective treatment for menopause symptoms, it is not suitable for all patients post-cancer. Several common cancers have receptors for oestrogen and progesterone, and hormone replacement can increase the likelihood of cancer recurrence.15 Cancers linked to oestrogen and progesterone receptors are presented in Box 2.3,16-18
The histopathology and hormone receptor status should therefore be confirmed in these cases, and cases of any rare malignancy, before commencing treatment. Most women with a history of oestrogen receptor-positive (ER+) or progesterone receptor-positive tumours require referral to a specialist menopause service, but nonhormonal treatment could be commenced in the meantime.
Hormonal treatment options
Systemic menopausal hormone therapy
In the absence of hormone receptors, systemic MHT may be considered.15 MHT is deemed the most effective approach for managing menopausal symptoms and offers potential benefits in preventing osteoporotic fractures, cognitive decline and cardiovascular issues, especially in those who are menopausal at under 45 years of age, while enhancing overall quality of life.12,19
Prescription of MHT in eligible women post-cancer follows the same principles as in physiological menopause cases, with some key additional considerations.
- Transdermal oestradiol is generally preferred because of the neutral effect on the risk of thromboembolism, which may be elevated during or after cancer treatment.20
- Younger women, or those with more abrupt cessations of ovulatory function, may require higher initial oestradiol doses.21
Ongoing follow-up and monitoring of hormone therapy should be carried out according to the usual prescribing principles. If menses return, the need for hormone replacement should be reviewed.
Vaginal hormonal therapies
For women with genitourinary symptoms of menopause, vaginal oestrogen is an effective treatment.22 Vaginal hormonal therapies act on the vaginal mucosa by increasing epithelial thickness, enhancing revascularisation and raising the number of superficial cells. These changes help restore the normal vaginal flora, including lactobacilli, and lower the vaginal pH, resulting in symptom improvement.23 Vaginal hormonal therapies may cause limited systemic absorption in some women.
An alternative local treatment is intravaginal prasterone, also known as dehydroepiandrosterone (DHEA).24,25 DHEA is converted locally into small amounts of active sex steroids, depending on the specific physiology of each cell and tissue.24 When used intravaginally, it has shown highly positive effects on the symptoms and signs of vulvovaginal atrophy, with minimal to no systemic oestrogen absorption and a theoretical improvement in libido.24 Vaginal prasterone is usually used nightly.
Typically, vaginal hormonal therapy is prescribed as an initial daily induction regimen for a minimum of two weeks before down-titration to at least twice a week on an ongoing basis.
Nonhormonal treatment options
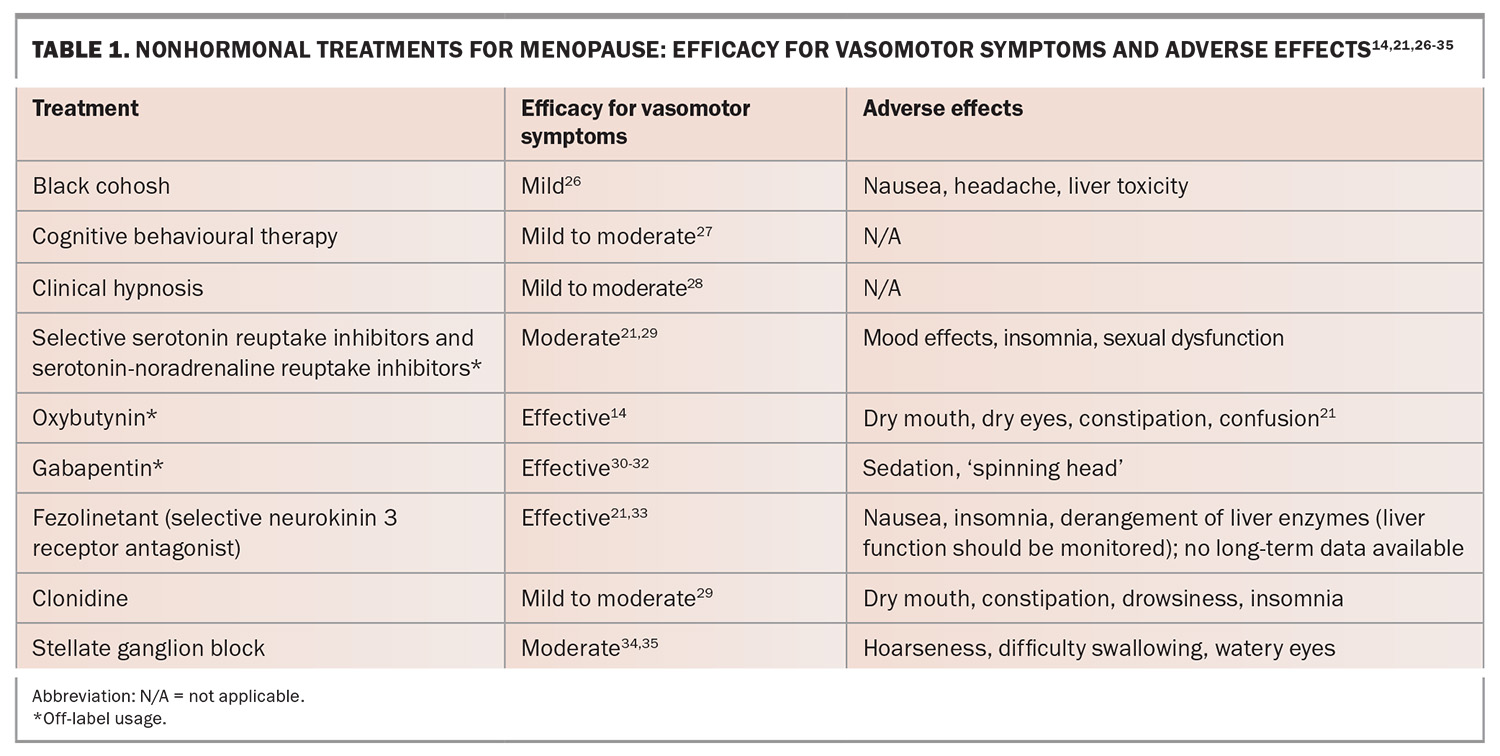
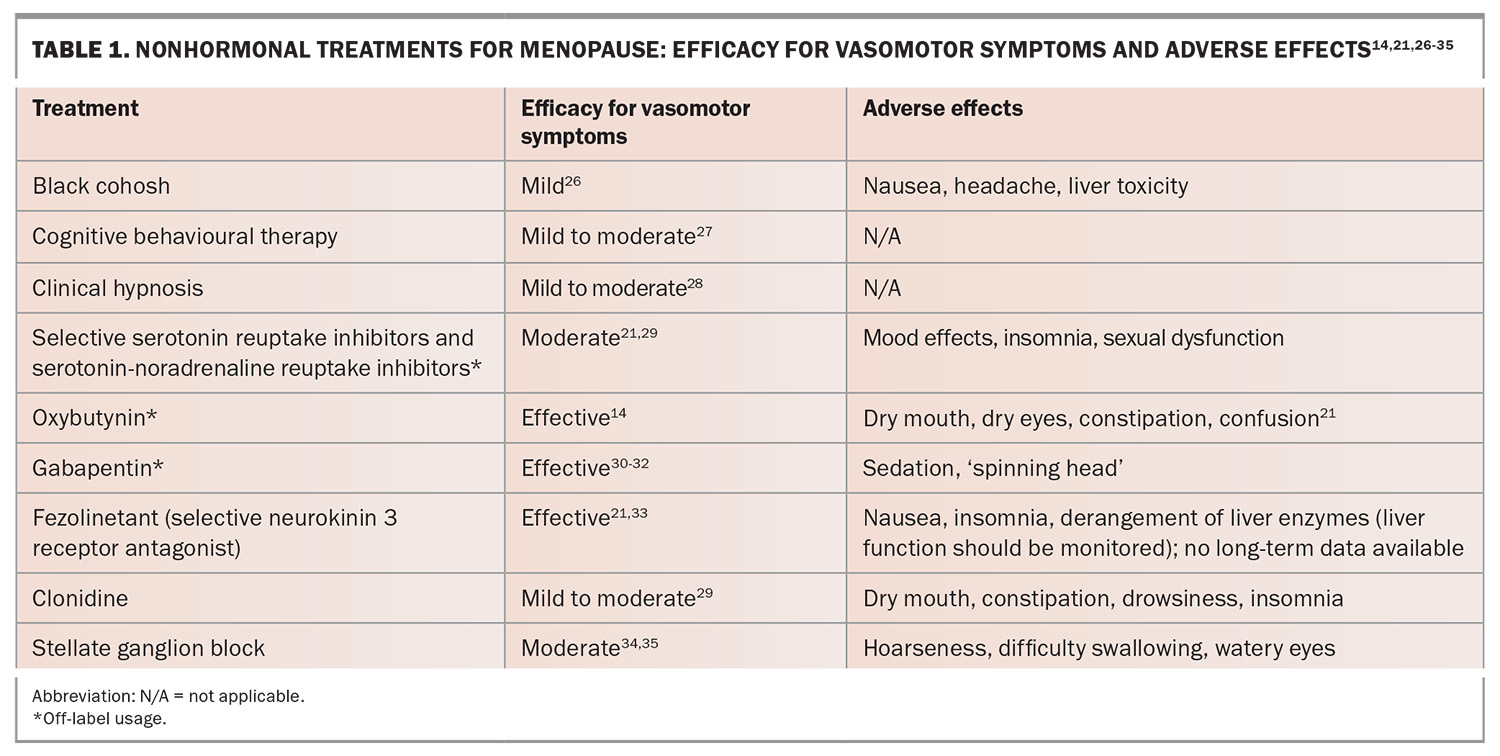
In lieu of hormonal options, extensive research has been conducted into nonhormonal treatments for menopausal symptoms. Nonhormonal options are typically less effective for treating menopausal symptoms; however, they remain the primary option for women with a history of ER+ cancers. A summary of the nonhormonal treatment options is presented in the Table.14,21,26-35 In general, lifestyle changes such as exercise, weight loss and smoking cessation may also help improve the tolerability of symptoms but do not directly modify vasomotor symptoms.
Treatment of menopause symptoms after breast cancer
Breast cancer is the most commonly diagnosed cancer worldwide and the most commonly diagnosed malignancy in women who are 40 years of age and younger.2 Women whose quality of life is significantly affected by menopausal symptoms may be more likely to discontinue breast cancer treatment, increasing the risk of recurrence and reducing survival.36 Therefore, proactive treatment of post-cancer menopause symptoms is crucial.
Hormone receptor-positive breast cancers
Women experiencing menopause symptoms following hormone receptor-positive breast cancers should be managed with nonhormonal treatment options, as outlined in the Table.14,21,26-35,37 Systemic hormone replacement in such cases has shown an increased risk of cancer recurrence.16,19 This risk also extends to the use of the synthetic steroid tibolone, which has similarly been linked to higher rates of breast cancer recurrence.19,38 Many women will tolerate daytime hot flushes but seek relief from disruptive night sweats. Nighttime administration of oxybutynin (0.5 to 1 mg) or gabapentin (300 to 900 mg) is often effective for these individuals, although both are prescribed off-label.
Hormone receptor-negative breast cancers
Breast cancers classified as triple-negative may exhibit undetectable hormone receptor levels at diagnosis or may develop hormone receptors during metastasis.39 Therefore, nonhormonal options are typically first-line treatments. However, women with triple-negative breast cancers tend to be younger and there is a low threshold for prescribing systemic MHT if needed. Studies have not demonstrated an increased risk of cancer recurrence associated with MHT in this group.19 In general, women who have had breast cancer should be referred to a specialised menopause service.
Vaginal symptoms
Topical vaginal oestrogen is generally considered safe to use in women with a history of hormone receptor-positive breast cancer if nonhormonal moisturisers are not effective. Although a meta-analysis showed that local oestrogen in women receiving aromatase inhibitor therapy was not associated with systemic absorption of sex hormones, it is recommended to use the lowest effective dose.37,40,41 Aromatase inhibitors are not oestrogen receptor blockers; instead, they work by suppressing systemic oestrogen production. Some oncologists remain concerned that even minimal systemic absorption of vaginal oestrogens could potentially increase the risk of recurrence in patients with a history of ER+ breast cancer.
Intravaginal prasterone can also be considered for off-label usage as studies using mass spectrometry have not detected oestrogenic nor androgenic molecules in the serum of patients using this product; however, this has not been studied extensively in post-cancer populations.37 It may be wise to periodically test oestrogen serum levels using mass spectrometry in patients who are receiving an aromatase inhibitor and using vaginal DHEA.
Vaginal CO2 laser has been explored as a nonhormonal treatment of vaginal atrophy, but studies have not shown a benefit.42
Testosterone
Off-label use of systemic testosterone for low libido is contentious following hormone receptor-positive breast cancer because of concerns around peripheral aromatisation into oestrogen.43 However, there is evidence that androgens have a suppressor role on ER+ breast cancer, and androgens were often used as endocrine therapy before tamoxifen.44,45 Nonetheless, this discussion should be referred to a specialist menopause service.
Treatment of menopause symptoms after endometrial cancers
Endometrial cancer is the sixth most common cancer worldwide and usually affects women around or after the time of menopause.14 At an early stage, survival rates are excellent with a five-year survival rate of up to 97%.15 Treatment normally involves removal of the uterus, fallopian tubes and ovaries. This generates an abrupt iatrogenic menopause.
Some types of endometrial cancer cells may have oestrogen receptors, and therefore there may be reservations about prescribing systemic hormone replacement.15 However, most women with early-stage endometrial cancer will not have any residual cancer cells following surgery.15 Therefore, MHT can be prescribed as unopposed oestrogen for women with stage 1 or 2 disease if nonhormonal treatment options are inadequate. This process should take place in a specialist menopause service.
Conclusion
With increasing cancer survivorship, a growing number of women are living long enough to reach menopause or experience menopausal symptoms as a consequence of their treatment. Management can be complex, particularly when hormonal therapies are contraindicated. GPs can refer women with complex and refractory symptoms to a specialist menopause service. ET
COMPETING INTERESTS: Professor Eden: None. Dr Brown has received payment for lectures by HealthEd.
References
1. Henze M, Stuckey BGA. Endocrine consequences of breast cancer therapy and survivorship. Climacteric 2024; 27: 333-339.
2. Lei S, Zheng R, Zhang S, et al. Global patterns of breast cancer incidence and mortality: a population-based cancer registry data analysis from 2000 to 2020. Cancer Commun (Lond) 2021; 41: 1183-1194.
3. Deli T, Orosz M, Jakab A. Hormone replacement therapy in cancer survivors – review of the literature. Pathol Oncol Res 2020; 26: 63-78.
4. Agency for Clinical Innovation. About the Menopause Initiative. Sydney: Agency for Clinical Innovation; 2025. Available online at: https://aci.health.nsw.gov.au/networks/menopause/about (accessed July 2025).
5. Aromatase inhibitors versus tamoxifen in premenopausal women with oestrogen receptor-positive early-stage breast cancer treated with ovarian suppression: a patient-level meta-analysis of 7030 women from four randomised trials. Lancet Oncol 2022; 23: 382-392.
6. Marino JL, Saunders CM, Emery LI, Green H, Doherty DA, Hickey M. Nature and severity of menopausal symptoms and their impact on quality of life and sexual function in cancer survivors compared with women without a cancer history. Menopause 2014; 21: 267-274.
7. Mauri D, Gazouli I, Zarkavelis G, et al. Chemotherapy associated ovarian failure. Front Endocrinol (Lausanne) 2020; 11: 572388.
8. Hickey M, LaCroix AZ, Doust J, et al. An empowerment model for managing menopause. Lancet 2024; 403: 947-957.
9. Harris PF, Remington PL, Trentham-Dietz A, Allen CI, Newcomb PA. Prevalence and treatment of menopausal symptoms among breast cancer survivors. J Pain Symptom Manage 2002; 23: 501-509.
10. Grossmann M, Ramchand SK, Milat F, et al. Assessment and management of bone health in women with oestrogen receptor-positive breast cancer receiving endocrine therapy: position statement summary. Med J Aust 2019; 211: 224-229.
11. Yoshida Y, Chen Z, Baudier RL, et al. Early menopause and cardiovascular disease risk in women with or without type 2 diabetes: a pooled analysis of 9,374 postmenopausal women. Diabetes Care 2021; 44: 2564-2572.
12. Mension E, Alonso I, Castelo-Branco C. Genitourinary syndrome of menopause: current treatment options in breast cancer survivors – systematic review. Maturitas 2021; 143: 47-58.
13. Rosenberg SM, Partridge AH. Premature menopause in young breast cancer: effects on quality of life and treatment interventions. J Thorac Dis 2013; 5 Suppl 1: S55-S61.
14. Mauri D, Gazouli I, Zarkavelis G, et al. Chemotherapy associated ovarian failure. Front Endocrinol (Lausanne) 2020; 11: 572388.
15. Edey KA, Rundle S, Hickey M. Hormone replacement therapy for women previously treated for endometrial cancer. Cochrane Database Syst Rev 2018; 5: Cd008830.
16. Pearlman MD. Exogenous hormones and breast cancer risk: contraception, menopausal hormone therapy, and breast cancer survivors. Clin Obstet Gynecol 2022; 65: 510-523.
17. Villa P, Bounous VE, Amar ID, et al. Hormone replacement therapy in post-menopause hormone-dependent gynecological cancer patients: a narrative review. J Clin Med 2024; 13: 1443.
18. Hsu LH, Chu NM, Kao SH. Estrogen, estrogen receptor and lung cancer. Int J Mol Sci 2017; 18: 1713.
19. Coronado PJ, Gómez A, Iglesias E, et al. Eligibility criteria for using menopausal hormone therapy in breast cancer survivors: a safety report based on a systematic review and meta-analysis. Menopause 2024; 31: 234-242.
20. Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of venous thromboembolism: nested case-control studies using the QResearch and CPRD databases. BMJ 2019; 364: k4810.
21. Madsen TE, Sobel T, Negash S, et al. A review of hormone and non-hormonal therapy options for the treatment of menopause. Int J Womens Health 2023; 15: 825-836.
22. Dew JE, Wren BG, Eden JA. A cohort study of topical vaginal estrogen therapy in women previously treated for breast cancer. Climacteric 2003; 6: 45-52.
23. Naumova I, Castelo-Branco C. Current treatment options for postmenopausal vaginal atrophy. Int J Womens Health 2018; 10: 387-395.
24. Labrie F, Archer DF, Martel C, Vaillancourt M, Montesino M. Combined data of intravaginal prasterone against vulvovaginal atrophy of menopause. Menopause 2017; 24: 1246-1256.
25. Mension E, Alonso I, Cebrecos I, et al. Safety of prasterone in breast cancer survivors treated with aromatase inhibitors: the VIBRA pilot study. Climacteric 2022; 25: 476-482.
26. Ruan X, Mueck AO, Beer AM, Naser B, Pickartz S. Benefit-risk profile of black cohosh (isopropanolic Cimicifuga racemosa extract) with and without St John’s wort in breast cancer patients. Climacteric 2019; 22: 339-347.
27. Hunter MS. Cognitive behavioral therapy for menopausal symptoms. Climacteric 2021; 24: 51-56.
28. Elkins GR, Fisher WI, Johnson AK, Carpenter JS, Keith TZ. Clinical hypnosis in the treatment of postmenopausal hot flashes: a randomized controlled trial. Menopause 2013; 20: 291-298.
29. Loibl S, Schwedler K, von Minckwitz G, Strohmeier R, Mehta KM, Kaufmann M. Venlafaxine is superior to clonidine as treatment of hot flashes in breast cancer patients – a double-blind, randomized study. Ann Oncol 2007; 18: 689-693.
30. Yoon SH, Lee JY, Lee C, Lee H, Kim SN. Gabapentin for the treatment of hot flushes in menopause: a meta-analysis. Menopause 2020; 27: 485-493.
31. Eden J. Endocrine dilemma: managing menopausal symptoms after breast cancer. Eur J Endocrinol 2016; 174: R71-R77.
32. Toulis KA, Tzellos T, Kouvelas D, Goulis DG. Gabapentin for the treatment of hot flashes in women with natural or tamoxifen-induced menopause: a systematic review and meta-analysis. Clin Ther 2009; 31: 221-235.
33. Morga A, Ajmera M, Gao E, et al. Systematic review and network meta-analysis comparing the efficacy of fezolinetant with hormone and nonhormone therapies for treatment of vasomotor symptoms due to menopause. Menopause 2024; 31: 68-76.
34. The 2023 nonhormone therapy position statement of The North American Menopause Society Advisory Panel. The 2023 nonhormone therapy position statement of The North American Menopause Society. Menopause 2023; 30: 573-590.
35. Walega DR, Rubin LH, Banuvar S, Shulman LP, Maki PM. Effects of stellate ganglion block on vasomotor symptoms: findings from a randomized controlled clinical trial in postmenopausal women. Menopause 2014; 21: 807-814.
36. Pistilli B, Paci A, Ferreira AR, et al. Serum detection of nonadherence to adjuvant tamoxifen and breast cancer recurrence risk. J Clin Oncol 2020; 38: 2762-2772.
37. Huguenin A. Menopausal hormone therapy for breast cancer survivors. Curr Opin Obstet Gynecol 2025; 37: 37-41.
38. Sismondi P, Kimmig R, Kubista E, et al. Effects of tibolone on climacteric symptoms and quality of life in breast cancer patients – data from LIBERATE trial. Maturitas 2011; 70: 365-372.
39. van Barele M, Heemskerk-Gerritsen BAM, Louwers YV, et al. Estrogens and progestogens in triple negative breast cancer: do they harm? Cancers (Basel) 2021; 13: 2506.
40. Pavlović RT, Janković SM, Milovanović JR, et al. The safety of local hormonal treatment for vulvovaginal atrophy in women with estrogen receptor-positive breast cancer who are on adjuvant aromatase inhibitor therapy: meta-analysis. Clin Breast Cancer 2019; 19: e731-e740.
41. Cano A, Estévez J, Usandizaga R, et al. The therapeutic effect of a new ultra low concentration estriol gel formulation (0.005% estriol vaginal gel) on symptoms and signs of postmenopausal vaginal atrophy: results from a pivotal phase III study. Menopause 2012; 19: 1130-1139.
42. Buttini MJ, Maher C. The first published randomised controlled trial of laser treatment for vaginal atrophy raises serious questions. Med J Aust 2018; 209: 376-377.
43. Glaser R, Dimitrakakis C. Testosterone and breast cancer prevention. Maturitas 2015; 82: 291-295.
44. Hickey TE, Selth LA, Chia KM, et al. The androgen receptor is a tumor suppressor in estrogen receptor-positive breast cancer. Nat Med 2021; 27: 310-320.
45. Boni C, Pagano M, Panebianco M, et al. Therapeutic activity of testosterone in metastatic breast cancer. Anticancer Res 2014; 34: 1287-1290.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.