Melanoma: current perspectives on diagnosis and management
Australia has one of the highest melanoma incidence rates worldwide. Accurate risk assessment, early detection, appropriate staging and effective surveillance are essential to improving outcomes. Advances in surgical management, imaging, immunotherapy and targeted therapies continue to reshape melanoma care across all disease stages.
- Melanoma is the third most common cancer in Australia, with risk influenced by ultraviolet exposure, age and family history.
- Breslow thickness, ulceration and sentinel lymph node involvement are key prognostic indicators.
- Dermoscopy, total body photography and mole mapping enhance early detection, particularly in high-risk patients.
- Melanoma comprises multiple subtypes, including superficial spreading melanoma, lentigo maligna melanoma, nodular melanoma, acral lentiginous melanoma, desmoplastic melanoma, and mucosal and ocular melanoma.
- A 2 mm margin excisional biopsy is recommended for accurately diagnosing melanoma and guiding subsequent management decisions.
- Immunotherapy and targeted therapies have significantly improved outcomes in advanced and metastatic disease.
Cutaneous melanoma poses a significant health burden in Australia, which has one of the highest incidence rates of melanoma globally.1 Melanoma develops from malignant melanocytes located in the basal layer of the epidermis. Undetected, melanoma has the potential for metastasis, representing the cause of 2.7% of all cancer deaths in Australia in 2025.2
Tumour thickness, ulceration and the presence of melanoma in the sentinel lymph nodes (SLN) are collectively the most accurate measures of prognosis.3 Patients with thin melanomas of less than 1 mm demonstrate five-year melanoma-specific survival rates reaching 100%. However, survival rates are much lower, at about 60%, for patients with melanoma diagnosed at an advanced stage and tumours thicker than 4 mm.4-6 Early detection and diagnosis of melanoma are therefore paramount.
Epidemiology
Melanoma is the third most common cancer in Australia, with an estimated 17,443 new cases diagnosed in 2025.2 Diagnosis rates show considerable state-wide variation in Australia, with those residing in Queensland at highest risk of developing melanoma.7 In recent years, the incidence of melanoma diagnosed in those below the age of 40 years has reduced, largely attributed to cancer awareness programs that have increased public awareness of melanoma and its prevention.6
In 2025, the age-standardised incidence rate of cutaneous melanoma was predicted to be 63 cases per 100,000 people, with higher rates observed in men (78 per 100,000) than in women (50 per 100,000).2 Melanoma incidence is projected to increase with age, peaking at 80 to 84 years.2 The estimated lifetime risk of being diagnosed with melanoma by 85 years of age in 2025 was one in 19, with a higher risk in men (one in 16) compared with women (one in 23).2 The rising reported incidence is likely attributable to multiple factors, including an ageing population, cumulative sun exposure, increased awareness and improvements in detection.
Risk factors
Exposure to ultraviolet (UV) radiation is consistently reported as the leading risk factor for the development of both melanoma and nonmelanoma skin cancers.8 However, primary risk factors for cutaneous melanoma include a previous history of melanoma, the presence of multiple naevi and clinically dysplastic naevi.9-11 Other risk factors include a positive family history, which can be attributed to the presence of germline genetic variants of cyclin-dependent kinase inhibitor 2A (CDKN2A), melanocortin 1 receptor (MC1R) or BRCA1-associated protein 1 (BAP1), with or without UV radiation-initiated oncogenic aberrations (e.g. mutations in BRAF, NRAS or KIT genes), or common environmental risk exposures.10
Individuals of Anglo-Celtic origin who have fair skin (skin phototype 1), red hair, blue eyes or freckling on the skin (generally representing MC1R variants) are at increased risk of cutaneous melanoma.9-11 A previous history of keratinocyte skin cancers, immunosuppression, solarium use and childhood sunburns are also considered as secondary risk factors for the development of melanoma.9-12
Diagnosis of melanoma
Clinical assessment and dermoscopic features
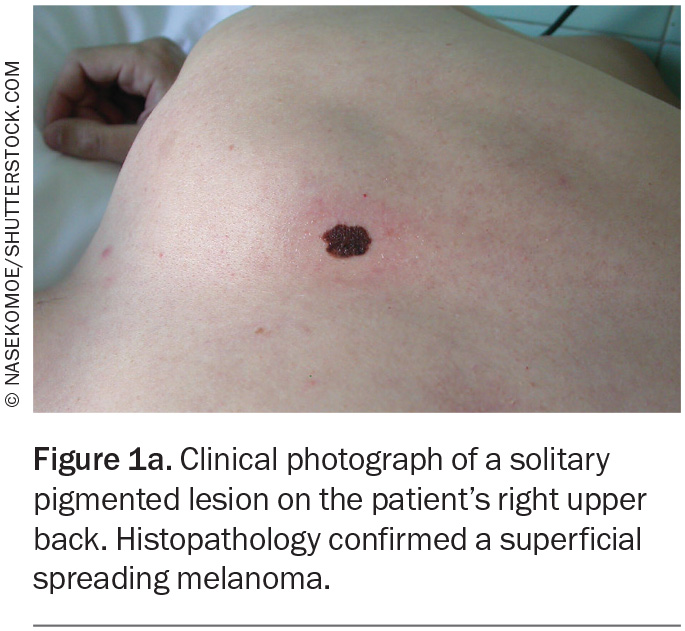
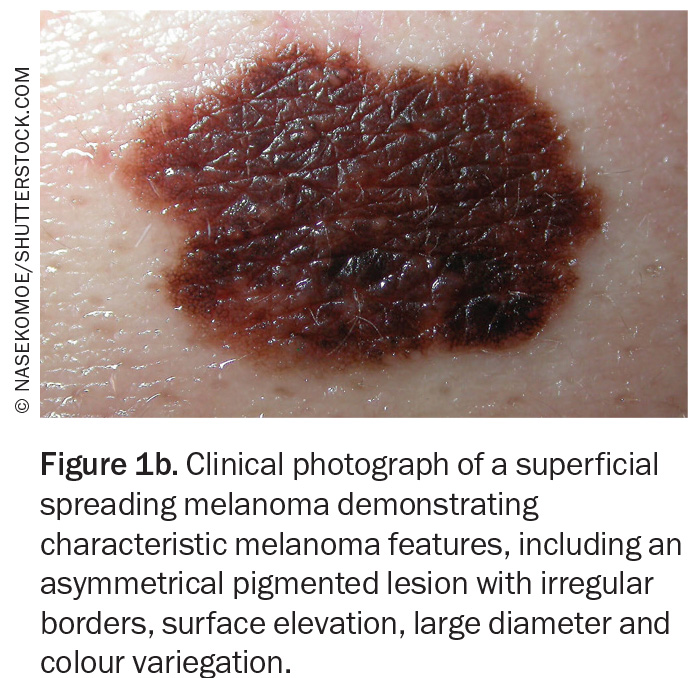
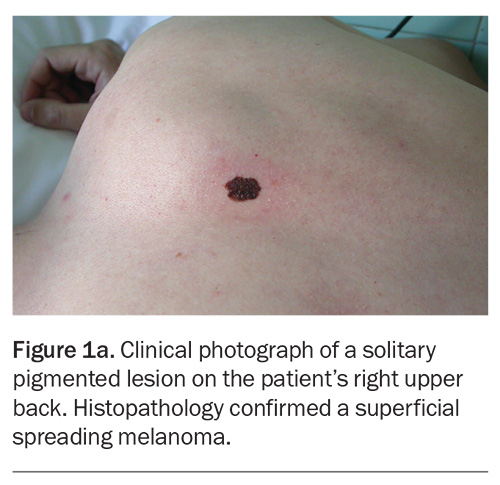
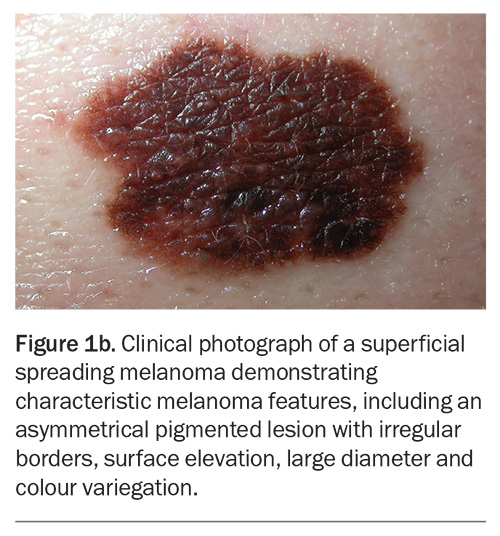
Traditional dermoscopy and sequential digital dermoscopic imaging can both assist with the diagnosis of melanoma in clinical practice.11,12 The ‘ugly duckling’ sign or a lesion that stands out from others can heighten clinical suspicion for melanoma (Figure 1a and Figure 1b).13 Lesion asymmetry, border irregularity, colour variation, large diameter (typically greater than 6 mm) and lesion evolution or change (otherwise known by the acronym ‘ABCDE’) were adopted to facilitate the detection of melanoma on history and physical examination.14
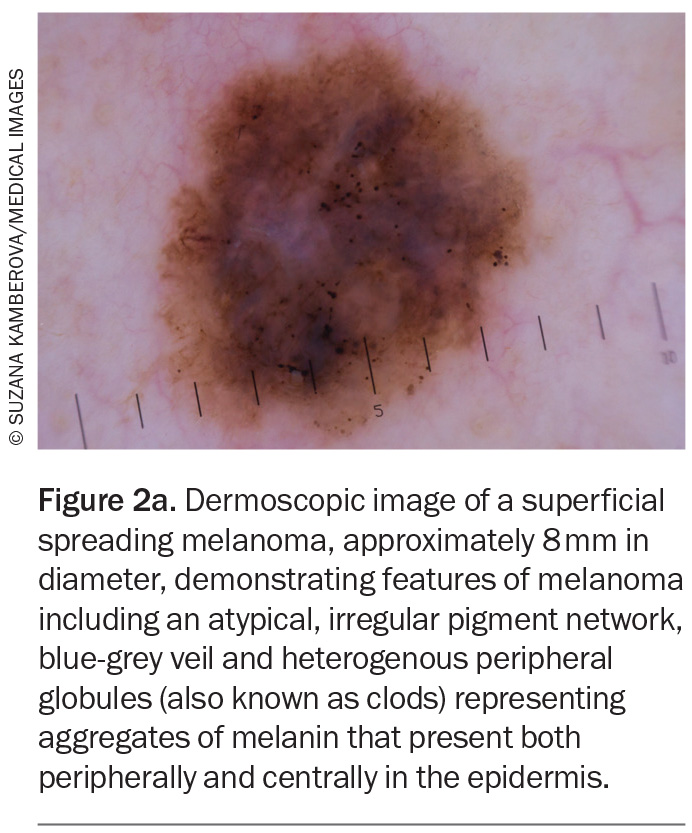
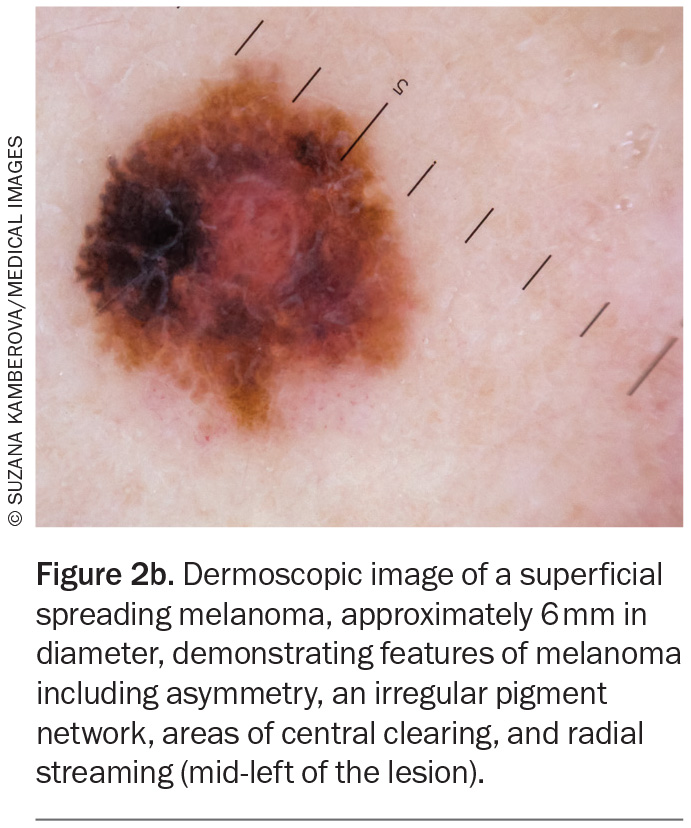
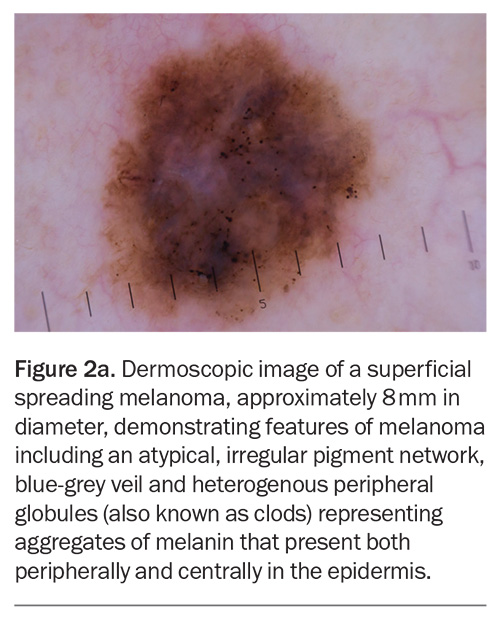
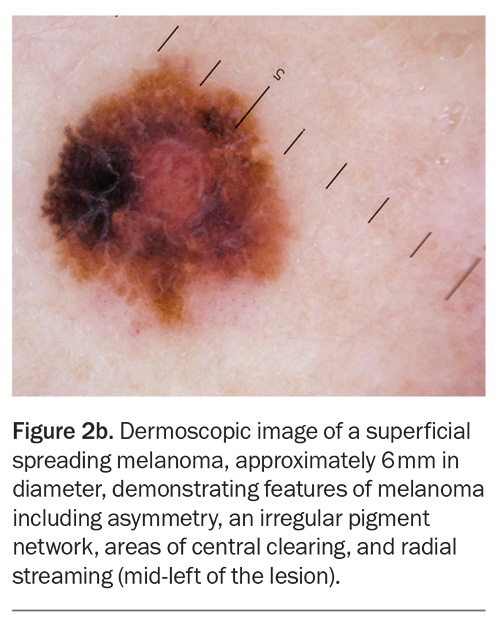
Well-established dermoscopic features of melanoma include an atypical pigment network, blue-grey veil, atypical vascular pattern, irregular or diffuse pigmentation (blotches), radial streaming and irregular dots or globules (also known as clods). Regression-associated features, such as milky-pink structureless areas and shiny white streaks, may also aid in the identification of melanoma (Figure 2a and Figure 2b).15
Imaging-based surveillance
Sequential digital dermoscopic imaging involves the assessment of successive dermoscopic images over time, thereby enhancing the diagnostic accuracy of subtle or featureless melanomas, and reduces the burden of unwarranted biopsies.12 Total body photography and mole mapping can also facilitate the identification of lesions evolving over time, particularly in high-risk patients with multiple dysplastic naevi or a high total melanocytic naevi count.11
Artificial intelligence-assisted detection
Artificial intelligence-generated melanoma tools, such as PanDerm, leverage machine-learning algorithms and have demonstrated significant promise in identifying skin cancer and subtle changes in lesions. These capabilities may facilitate earlier detection of melanoma, particularly in settings with limited access to specialist care, and may serve as an adjunct to support clinician risk stratification and assessment.16,17 Using a deep convoluted neural network, artificial intelligence has been shown to classify skin cancer with a level of accuracy comparable to that of dermatologists.18
Melanoma subtypes
Superficial spreading melanoma
Superficial spreading melanoma (SSM) accounts for about 70% of diagnosed melanomas and is typically seen in young to middle-aged adults (Figure 1a, Figure 1b, Figure 2a and Figure 2b).11,12 The most common site of SSM diagnosis is the trunk in men and the lower legs in women. SSM is generally considered a low cumulative sun damage melanoma and a significant proportion are thought to develop from an existing melanocytic naevus.11,12 In SSM, malignant cells initially remain within the epidermis (in situ) and spread horizontally in a radial growth phase that can persist for months to decades.11,12
Lentigo maligna melanoma
Lentigo maligna (LM; also known as Hutchinson’s melanotic freckle) is a precursor lesion to lentigo maligna melanoma (LMM), representing about 10% of diagnosed melanomas.11 LM tends to occur on the head and neck of older patients and is associated with severe background solar elastosis.11,12 In LM, atypical or malignant cells are confined to the epidermis, whereas LMM is diagnosed when histopathology demonstrates invasion of malignant cells into the dermis.11,12 Confocal microscopy has shown promise in improving margin evaluation and informing the surgical management of LM and LMM.19 A recent phase 3, randomised trial has demonstrated that both radiotherapy and imiquimod are efficient and well tolerated treatments for LM.20
Nodular melanoma
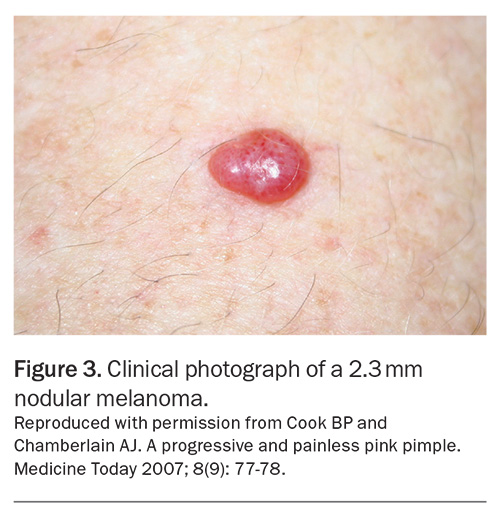
Nodular melanoma can present as a rapidly enlarging nodule with well circumscribed borders and characteristically lacks a radial growth phase (Figure 3). Malignant cells penetrate vertically into deeper layers of the skin and subcutaneous tissue. Nodular melanomas may be amelanotic (often pink or red) and can be associated with ulceration.11,12 These represent about 15% of all melanomas diagnosed.11 Nodular melanoma has been identified as an indicator of poor survival in melanoma and is associated with a greater risk of recurrence, with a five-year survival rate of 53.7% for nodular melanoma compared with 87.3% for SSM.21
Acral lentiginous melanoma
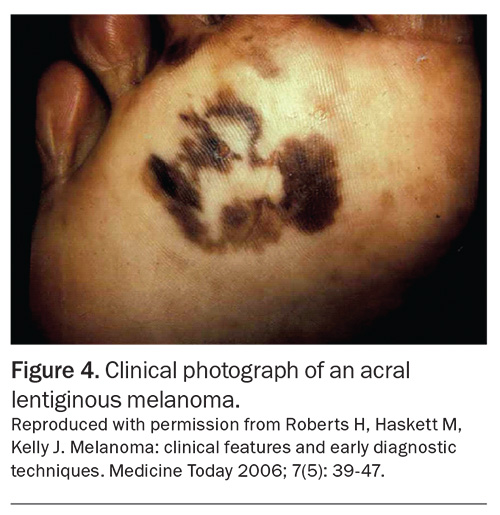
Acral lentiginous melanoma is a subtype of melanoma arising on the palms, soles of the feet or under the nails (Figure 4).22 It is the most common subtype of melanoma in darker skinned individuals. It is generally associated with a poor prognosis and advanced disease at diagnosis.22 Interestingly, acral lentiginous melanomas that develop on the lower limbs are most frequently found on the plantar surface of the foot, whereas those on the upper limbs are primarily linked to the fingernail apparatus.22 Subungual melanoma arises from the nail matrix and is associated with pigmentation with or without ulceration. Pigmentation extending to the proximal nail fold, known as the ‘Hutchinson’s sign’, is highly suggestive of malignant melanoma and warrants urgent management.22
Desmoplastic melanoma
Desmoplastic melanoma accounts for less than 5% of melanomas diagnosed. It most commonly occurs on sun-exposed skin in older adults. Early detection is difficult as desmoplastic melanoma is frequently amelanotic, predominantly located in the dermis and may present as a scar-like plaque with associated fibrosis and perineural invasion.12,23 Desmoplastic melanoma is associated with reduced rates of SLN involvement and demonstrates a comparatively enhanced response to immunotherapy.10
Mucosal and ocular melanoma
Mucosal and ocular melanomas are extremely rare and generally have a poor prognosis, often diagnosed at an advanced stage.
Mucosal melanomas originate from melanocytes within mucosal membranes, namely the respiratory, gastrointestinal and genitourinary tracts. The aetiology and pathogenesis of mucosal melanoma is unclear, and the overall five-year survival rate is less than 20%.24BRAF, NRAS and c-KIT/CD117 gene mutations have been reported in mucosal melanoma. Clinical trials demonstrate reduced responsiveness to immunotherapy.24
Ocular melanomas most commonly arise within the uveal tract, which includes the iris, ciliary body and choroid, and to a lesser extent within the conjunctival membrane. Uveal melanoma is distinguished by an almost complete absence of BRAF mutations, in contrast to their high prevalence in cutaneous melanoma. Mutations in GNAQ and GNA11, BAP1, SF3B1 and EIF1AX are key prognostic determinants.25
Management of melanoma
Diagnostic biopsy and surgical excision
A 2 mm margin excisional biopsy is the recommended technique for accurately diagnosing melanoma and informing its subsequent management.26,27 Shave or punch excision, where the lesion can be completely excised in width and depth, can be considered for lesions with a low clinical suspicion for melanoma; however, this can be associated with positive margins, necessitating further excision to achieve surgical radial margins.28
Partial punch or shave biopsies pose a risk of sampling error and are not recommended as they may fail to capture foci of invasive melanoma, potentially leading to underdiagnosis.29 In line with this, incisional biopsy also carries a similar risk of underdiagnosis but can be considered appropriate by a trained clinician in certain circumstances (e.g. large lesions on the face or acral regions).29
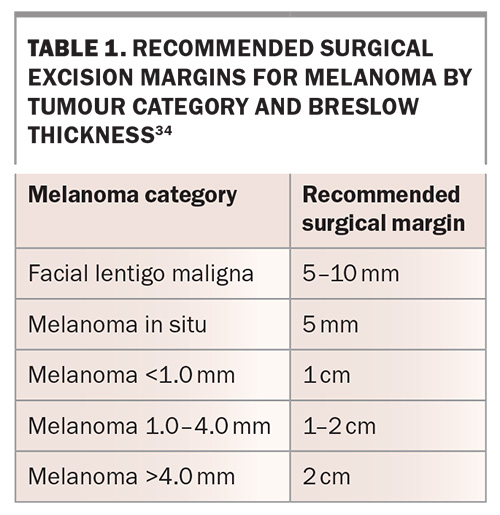
Melanoma confirmed by histopathology warrants wide local excision based on its Breslow thickness, as per the recommended clinical practice guidelines.30 Breslow thickness is a microscopic measurement of the depth of the melanoma, from the stratum granulosum to the deepest point of the tumour. The recommended surgical radial excision margins are presented in Table 1.
Sentinel lymph node biopsy
For melanomas measuring 0.75 to 1.0 mm in thickness, the risk of SLN involvement is about 5 to 10%, and biopsy can be considered if histopathological features such as ulceration or a high mitotic rate are present.27 SLN biopsy is typically advised for patients with melanoma measuring greater than 1 mm in thickness, or 0.8 to 1 mm with high-risk features such as ulceration, in the absence of clinically detectable lymphadenopathy.27 These patients should be referred to a major tertiary multidisciplinary melanoma management unit, as SLN biopsy provides prognostic information by accurately identifying microscopic nodal metastasis – one of the strongest predictors of recurrence risk – and helps guide decisions about adjuvant therapy.
Staging and surveillance imaging
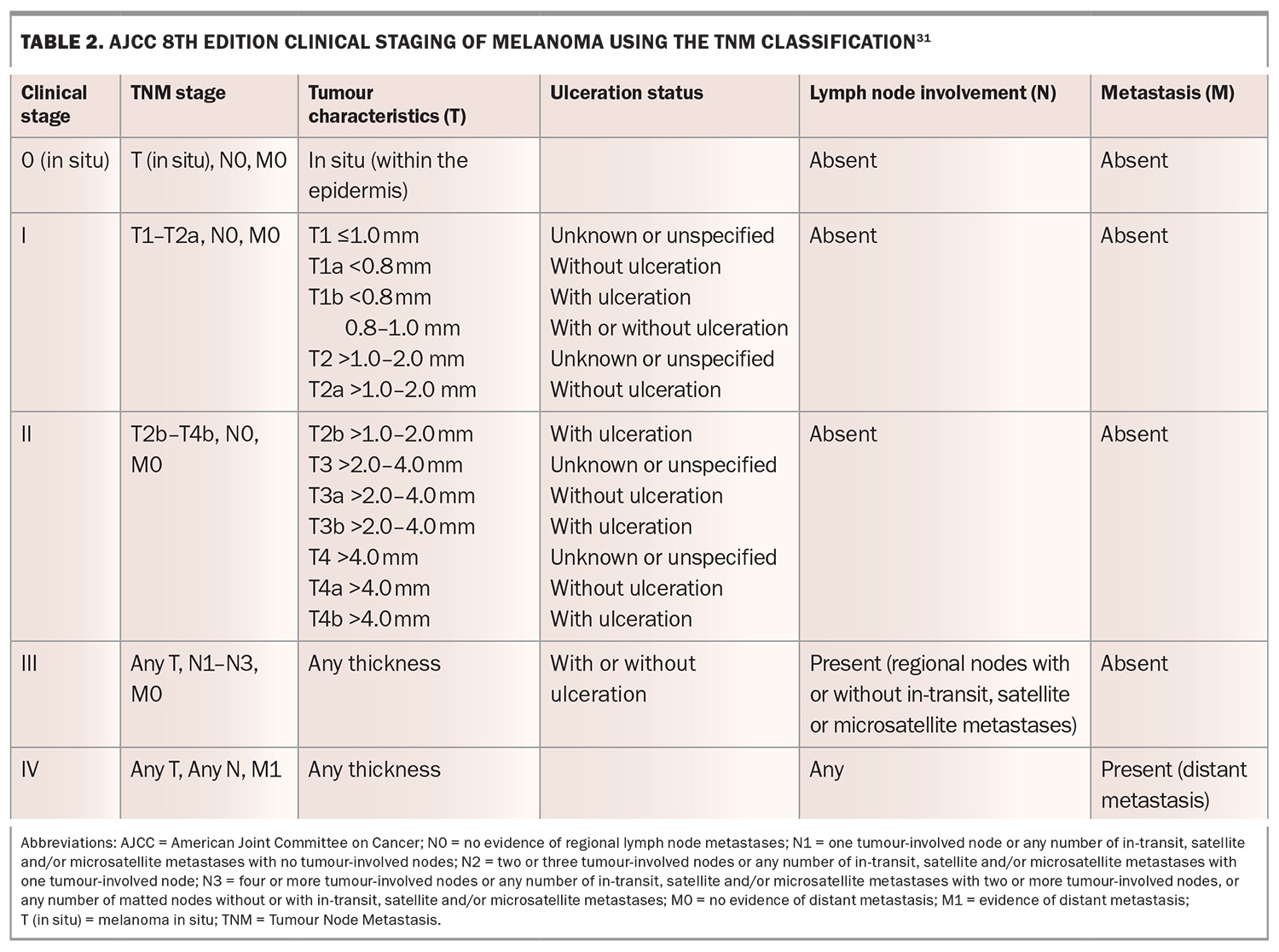
Surveillance imaging including CT and PET scanning may be considered by the treating team to stage disease and further determine patient prognosis.12 Melanoma staging follows the American Joint Committee on Cancer (AJCC) 8th edition staging manual and is based on the Tumour Node Metastasis (TNM) system, incorporating primary tumour (T), regional lymph node involvement (N) and distant metastasis (M). T staging is based on Breslow thickness and ulceration, with mitotic rate being an important prognostic marker. N staging accounts for the number of involved lymph nodes and includes in-transit, satellite and microsatellite metastases.30 M staging accounts for distant metastasis by site (e.g. skin, lung, CNS) and lactate dehydrogenase levels, with the category M1d indicating CNS involvement.30
These components define clinical stage groups from 0 (in situ) to IV (metastatic). Five-year survival rates range from nearly 100% in stage 0 to I, to below 30% in advanced stage IV. This staging system provides prognostic stratification and guides therapeutic decision-making in melanoma management.30 Stages 0 to II melanoma show no lymph node involvement; however, stage II disease is characterised by thicker tumours, with or without ulceration, indicating a higher risk of recurrence (Table 2).31 SLN involvement with or without the presence of satellites upstages disease to stage III, allowing patients to access adjuvant therapies such as immune checkpoint inhibitors (ICIs) or BRAF/MEK inhibitors after wide local excision.12,27
Screening and follow up
The risk of melanoma recurrence is highest during the first three years following diagnosis, with a median interval of 16 months to the development of metastatic disease.12,32 A complete skin examination must be conducted on a routine basis for melanoma surveillance. For patients with stage IIB to III melanoma (Breslow thickness >2 mm or >1 mm with ulceration or nodal metastasis), examinations are recommended every three to four months for the first two years after diagnosis, followed by six to 12 monthly for the next three years. For patients with stage I melanoma, examinations should be performed at least annually.12
Self-examination can also be recommended to patients at three monthly intervals to allow early detection. Studies to date have demonstrated an inverse relationship between tumour thickness and time to recurrence.33 Therefore, long-term follow up of patients with T1 melanoma is essential, as late melanoma-related mortality is more likely in this group, underscoring the importance of regular surveillance.33 A thorough history, including a history of prior melanoma or nonmelanocytic skin cancers, immunosuppression, childhood sunburns, solarium use, occupational history, outdoor hobbies and previous exposure to UVA and UVB will help risk stratify patients.34
As most melanomas are self-detected by patients or their family members, any history of new or evolving lesions warrants careful assessment.11,34 The patient’s skin phototype, the total number of naevi and the number of dysplastic naevi are key features to note on clinical review. Physical examination of melanoma excision scars for evidence of recurrence, assessment of lymph node basins and a full skin examination with dermoscopy should be performed. All areas of the body should be examined, including the scalp, head and neck, ears, trunk, breasts and inframammary region, back, buttocks, upper limbs including axillae, lower limbs including the groin, hands and feet, between the webbing of the toes, palms, plantar surface of the feet, fingernails and toenails, and genitalia if there are specific concerns.11,34
Management of metastatic melanoma
For patients with metastatic melanoma, ICIs that target CTLA-4 (ipilimumab) and PD-1 (nivolumab and pembrolizumab) have shown significant promise.35 Combined anti-CTLA-4 and anti-PD-1 checkpoint inhibition has the highest five-year overall survival rate of any therapies in advanced melanoma.36
BRAF V600I mutations occur in about 40% of melanomas, and oral treatments targeting BRAF and MEK can be considered for patients with BRAF-mutant metastatic melanoma.27 Interestingly, studies to date have demonstrated that in these patients, long-term survival is higher in those treated with ICIs than combined targeted BRAF and MEK inhibition.36 Combined ICI and BRAF–MEK inhibition (triple therapy) is also being explored, although this has been associated with a high toxicity profile in some patients.37
More recently, a phase 3 clinical trial demonstrated that neoadjuvant immunotherapy with ICIs (nivolumab plus ipilimumab) administered before surgery for resectable stage III melanoma improved 12-month event-free survival (84% vs 57% with standard postoperative therapy), establishing a new standard of care.38
Stereotactic radiotherapy might be considered for regional tumour control by tertiary treatment centres or for palliative care when there are multiple brain metastases.39,40
Conclusion
Early diagnosis and management of melanoma are essential to optimise patient prognosis. Novel treatments have revolutionised the management of advanced stage melanoma and improved patient survival outcomes. Further research, integrating the use of immunotherapy and small molecule inhibitors, together with artificial intelligence-generated platforms to facilitate melanoma surveillance, will continue to reduce the public health burden of melanoma. MT
COMPETING INTERESTS: Dr Iyengar, Dr Larney and Associate Professor Shumack: None. Associate Professor Foley has received grants paid to an institution from AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly, Janssen, LEO Pharma, Novartis, Pfizer, Sanofi, Sun Pharma and UCB Pharma; consulting fees from Apogee, Aslan, Boehringer Ingelheim, Eli Lilly, Galderma, GenesisCare, Janssen, LEO Pharma, Mayne Pharma, MedImmune, Novartis, Oruka, Pfizer, Takeda and UCB Pharma; payment or honoraria from AbbVie, Almirall, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly, Janssen, Novartis, Sanofi, Sun Pharma and UCB Pharma; payment for expert testimony from Pfizer; fees for participation on a Data Safety Monitoring Board or Advisory Board from AbbVie, Amgen, Arrotex/Juniper, Aslan, Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly, Galderma, GlaxoSmithKline, Janssen, LEO Pharma, Mayne Pharma, Novartis, Pfizer, Sanofi, Sun Pharma, Takeda and UCB Pharma; and receipt of drugs from AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly, Janssen, LEO Pharma, Mayne Pharma, Novartis, Sun Pharma and UCB Pharma.
References
1. Ferlay J, Ervik M, Lam F, et al. Global Cancer Observatory: Cancer Today. Lyon: International Agency for Research on Cancer; 2020. Available online at: https://gco.iarc.fr/today (accessed January 2026).
2. Cancer Australia. Melanoma of the skin statistics. Sydney: Australian Government; 2026. Available online at: https://www.canceraustralia.gov.au/cancer-types/melanoma-skin/melanoma-skin-statistics (accessed January 2026).
3. Faries MB, Thompson JF, Cochran AJ, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med 2017; 376: 2211-2222.
4. Green AC, Baade P, Coory M, Aitken JF, Smithers M. Population-based 20-year survival among people diagnosed with thin melanomas in Queensland, Australia. J Clin Oncol 2012; 30: 1462-1467.
5. Landow SM, Gjelsvik A, Weinstock MA. Mortality burden and prognosis of thin melanomas overall and by subcategory of thickness, SEER registry data, 1992-2013. J Am Acad Dermatol 2017; 76: 258-263.
6. Australian Institute of Health and Welfare (AIHW). Cancer data in Australia. Canberra: AIHW; 2025. Available online at: https://www.aihw.gov.au/reports/cancer/cancer-data-in-australia/contents/about (accessed January 2026).
7. Lindsay D, Whiteman DC, Olsen CM, Gordon LG. Health care service use by people diagnosed with invasive melanoma in Queensland: a benefit incidence analysis. Med J Aust 2023; 219: 417-422.
8. Elwood JM, Jopson J. Melanoma and sun exposure: an overview of published studies. Int J Cancer 1997; 73: 198-203.
9. Mar V, Wolfe R, Kelly JW. Predicting melanoma risk for the Australian population. Australas J Dermatol 2011; 52: 109-116.
10. Long GV, Swetter SM, Menzies AM, Gershenwald JE, Scolyer RA. Cutaneous melanoma. Lancet 2023; 402: 485-502.
11. Roberts H, Haskett M, Kelly J. Melanoma: clinical features and early diagnostic techniques. Medicine Today 2006; 7(5): 39-47.
12. Mar VJ, Soyer HP, Button-Sloan A, et al. Diagnosis and management of cutaneous melanoma. Aust J Gen Pract 2020; 49: 733-739.
13. Ilyas M, Costello CM, Zhang N, Sharma A. The role of the ugly duckling sign in patient education. J Am Acad Dermatol 2017; 77: 1088-1095.
14. Kopf AW. Prevention of malignant melanoma. Dermatol Clin 1985; 3: 351-360.
15. Argenziano G, Fabbrocini G, Carli P, De Giorgi V, Sammarco E, Delfino M. Epiluminescence microscopy for the diagnosis of doubtful melanocytic skin lesions: comparison of the ABCD rule of dermatoscopy and a new 7-point checklist based on pattern analysis. Arch Dermatol 1998; 134: 1563-1570.
16. Yan S, Yu Z, Primiero C, et al. A multimodal vision foundation model for clinical dermatology. Nat Med 2025; 31: 2691-2702.
17. Wada M, Ge Z, Gilmore SJ, Mar VJ. Use of artificial intelligence in skin cancer diagnosis and management. Med J Aust 2020; 213: 256-259.e1.
18. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature 2017; 542: 115-118.
19. Shah P, Gulati N, Stein J, Polsky D, Lee N, Liebman TN. Utility of confocal microscopy in the management of lentigo maligna and lentigo maligna melanoma. J Am Acad Dermatol 2021; 84: 1736-1737.
20. Hong AM, Lo SN, Fogarty GB, et al. Radiotherapy versus imiquimod for complex lentigo maligna: a phase 3 randomized clinical trial. J Am Acad Dermatol 2025; 93: 1251-1260.
21. Allais BS, Beatson M, Wang H, et al. Five-year survival in patients with nodular and superficial spreading melanomas in the US population. J Am Acad Dermatol 2021; 84: 1015-1022.
22. Howard M, Xie C, Wee E, et al. Acral lentiginous melanoma: clinicopathologic and survival differences according to tumour location. Australas J Dermatol 2020; 61: 312-317.
23. Chen LL, Jaimes N, Barker CA, Busam KJ, Marghoob AA. Desmoplastic melanoma: a review. J Am Acad Dermatol 2013; 68: 825-833.
24. Sergi MC, Filoni E, Triggiano G, et al. Mucosal melanoma: epidemiology, clinical features, and treatment. Curr Oncol Rep 2023; 25: 1247-1258.
25. Lavarone L, Franco R, Zito Marino F, et al. Ocular melanoma: a comprehensive review with a focus on molecular biology. Int J Mol Sci 2025; 26: 9799.
26. Kelly JW, Henderson MA, Thursfield VJ, Slavin J, Ainslie J, Giles GG. The management of primary cutaneous melanoma in Victoria in 1996 and 2000. Med J Aust 2007; 187: 511-514.
27. Bhave P, Wong J, McInerney‐Leo A, et al. Management of cutaneous melanoma in Australia: a narrative review. Med J Aust 2023; 218: 426-431.
28. Matheus GG, Muir J. Shave procedures in the management of skin lesions where melanoma is a differential diagnosis. Aust J Gen Pract 2024; 53: 533-536.
29. Ng JC, Swain S, Dowling JP, Wolfe R, Simpson P, Kelly JW. The impact of partial biopsy on histopathologic diagnosis of cutaneous melanoma: experience of an Australian tertiary referral service. Arch Dermatol 2010; 146: 234-239.
30. Cancer Council Australia Melanoma Guidelines Working Party. Clinical practice guidelines for the diagnosis and management of melanoma. Sydney: Cancer Council Australia; 2021. Available online at: https://www.cancer.org.au/clinical-guidelines/skin-cancer/melanoma (accessed January 2026).
31. Gershenwald JE, Scolyer RA, Hess KR, et al. Melanoma staging: evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J Clin 2017; 67: 472-492.
32. Adler NR, Wolfe R, Kelly JW, et al. Tumour mutation status and sites of metastasis in patients with cutaneous melanoma. Br J Cancer 2017; 117: 1026-1035.
33. Lo SN, Williams GJ, Cust AE, et al. Risk of death due to melanoma and other causes in patients with thin cutaneous melanomas. JAMA Dermatol 2025; 161: 167-174.
34. Lin M, Kelly JW. When is it melanoma? An update on diagnosis and management. Medicine Today 2013; 14(11): 44-53.
35. Carlino MS, Larkin J, Long GV. Immune checkpoint inhibitors in melanoma. Lancet 2021; 398: 1002-1014.
36. Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med 2019; 381: 1535-1546.
37. Lengyel AS, Meznerics FA, Galajda NÁ, et al. Safety and efficacy analysis of targeted and immune combination therapy in advanced melanoma-a systematic review and network meta-analysis. Int J Mol Sci 2024; 25: 12821.
38. Blank CU, Lucas MW, Scolyer RA, et al. Neoadjuvant nivolumab and ipilimumab in resectable stage III melanoma. N Engl J Med 2024; 391: 1696-1708.
39. Kispál M, Jánváry LZ, Balatoni T, et al. The role of stereotactic radiotherapy in the management of melanoma, a retrospective single institute preliminary study of 30 patients. Pathol Oncol Res 2022; 28: 1610550.
40. Thompson JF, Williams GJ, Hong AM. Radiation therapy for melanoma brain metastases: a systematic review. Radiol Oncol 2022; 56: 267-284.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.