Medical therapies for microscopic colitis – a practical update

Microscopic colitis is a prevalent yet easily overlooked cause of chronic watery diarrhoea. This article serves as a practical primer for microscopic colitis, discussing risk factors, clinical presentation and diagnostic work-up. It provides practical treatment strategies for primary care physicians, covering both initial and more advanced management options. The goal is to enhance the speed and precision of diagnosis and to improve care for patients with microscopic colitis.
- Microscopic colitis is a common cause of chronic, profuse, watery, nonbloody diarrhoea that often affects older women and is frequently misdiagnosed as irritable bowel syndrome.
- Diagnosis relies on colonoscopy and biopsy. Biopsies from both the right and left colon are crucial for identifying the histological changes characteristic of lymphocytic and collagenous subtypes of microscopic colitis.
- Numerous risk factors, including medications (proton pump inhibitors, NSAIDs and selective serotonin reuptake inhibitors), smoking and coeliac disease, are associated with microscopic colitis. Removing and treating these underlying risk factors is important for disease remission.
- Budesonide is an effective and generally well tolerated first-line treatment for both subtypes of microscopic colitis. It is recommended for induction and sometimes maintenance therapy, with an induction course of typically six to eight weeks, followed by maintenance treatment if persistent symptoms recur.
- Relapse is common when budesonide is stopped, and strategies for managing relapse include maintenance therapy or intermittent retreatment with budesonide.
- Biologic and small-molecule agents are treatment options for patients who do not respond to budesonide and experience severe symptoms.
GPs are at the forefront of managing most patient presentations involving diarrhoea. Australian general practice data from 1998 to 2004 indicate that diarrhoea is a reported reason for patient encounters in 1.3 per 100 visits.1 Patients will often self-treat based on initial advice sought from various sources, including pharmacists, health websites and, increasingly, artificial intelligence-powered tools, before presenting to a GP.
Despite these initial treatment efforts, some patients develop chronic, watery, nonbloody diarrhoea. In these cases, it is important to consider microscopic colitis. This condition, encompassing lymphocytic and collagenous subtypes, can progress between subtypes and may represent a spectrum of disease, although it rarely advances to inflammatory bowel disease (IBD).2 Diagnosis relies on characteristic histological findings from colonic biopsies. Microscopic colitis is frequently misdiagnosed as diarrhoea-predominant irritable bowel syndrome (IBS), leading to delays in diagnosis and treatment.
Microscopic colitis also has a significant economic impact, driven by high societal costs from work loss, especially in those with active disease.3 High readmission rates and healthcare utilisation further contribute to the associated economic strain.4 This article aims to enhance awareness of microscopic colitis, to ensure its timely diagnosis and effective management.
Epidemiology and pathophysiology
The annual incidence of microscopic colitis is estimated to be between one and 25 cases per 100,000 people.5-7 Its overall prevalence ranges between 50 and 200 cases per 100,000 people. Microscopic colitis predominantly affects adults aged over 50 years, with a median age at diagnosis of 60 years, and is more common in women, with a female-to-male ratio of about 2:1 (77% female for collagenous colitis, 68% for lymphocytic colitis).8-10 A rising incidence of microscopic colitis is partly attributable to increased awareness and improved diagnostics. However, a genuine increase likely exists, potentially driven by the greater use of medications such as proton pump inhibitors (PPIs), NSAIDs and selective serotonin reuptake inhibitors (SSRIs), which are all implicated in the condition’s development.7,11
The exact pathogenesis of microscopic colitis remains unclear. However, likely contributors include luminal factors, such as abnormal microbiota, bile acids and dietary antigens, abnormal mucosal immune responses and altered collagen metabolism, leading to epithelial barrier dysfunction in genetically susceptible individuals.12,13 Emerging evidence suggests a potential role of the gut microbiome in microscopic colitis. Studies have shown decreased Akkermansia bacteria, which maintain gut mucus, and increased Veillonella bacteria in some patients.14 Some microbiome changes resemble those seen in other inflammatory bowel conditions, although overall bacterial diversity results are mixed.13 These findings hint at the microbiome’s involvement, but further research is needed to determine whether these changes are causative or secondary to microscopic colitis. Understanding this relationship could pave the way for novel treatments, such as targeted probiotics.14
Risk factors
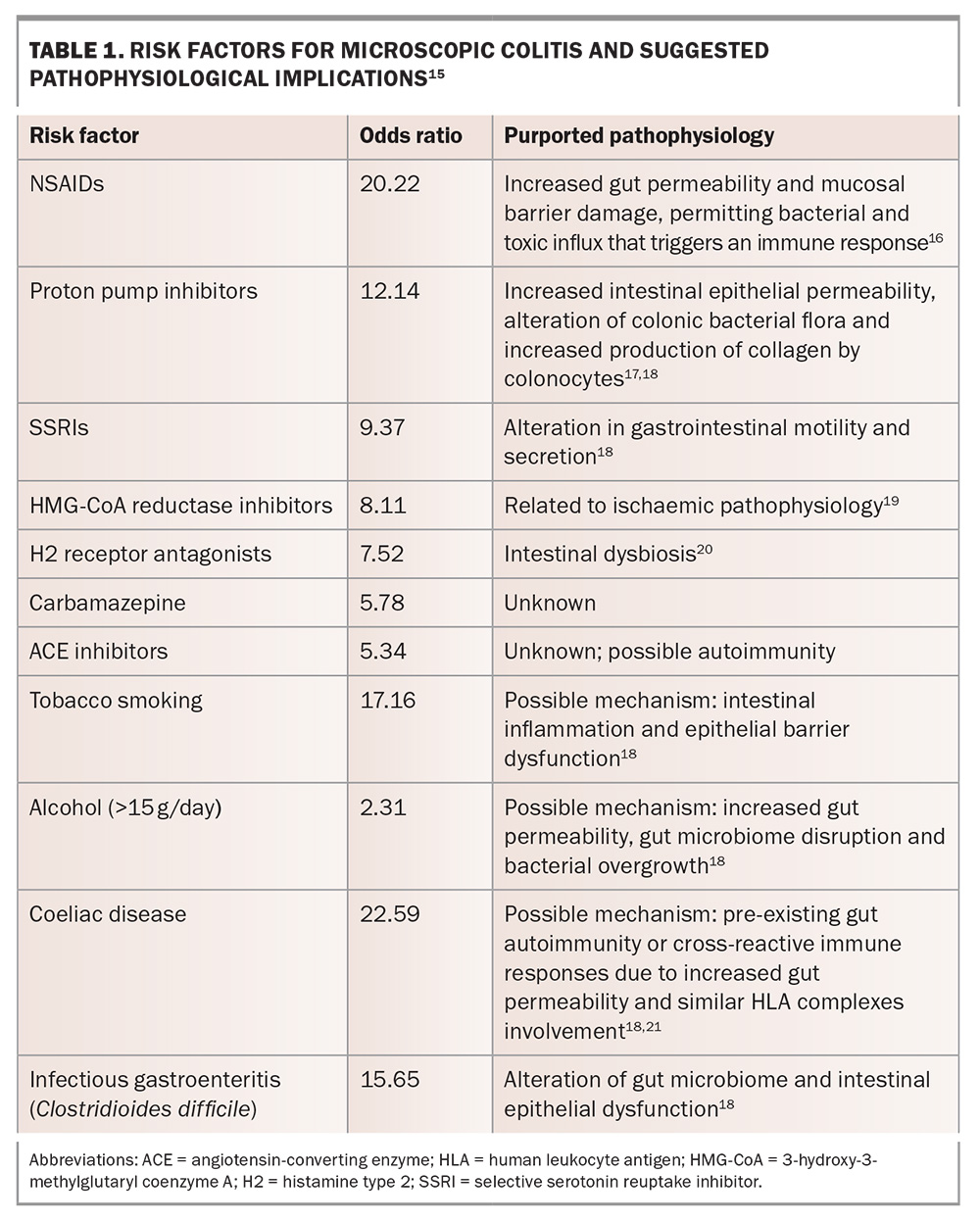
Long-term use of implicated medications, tobacco smoking and coeliac disease are the primary risk factors associated with microscopic colitis.9 Among these, medications and smoking play a crucial role, as they are both significant and modifiable risk factors. NSAIDs, PPIs and SSRIs have been strongly linked to microscopic colitis development (Table 1).15-21 However, recent evidence from a large nationwide study in Sweden challenges this association, suggesting that the reported links between these medications and microscopic colitis may be less definitive than previously thought.22 Although this study provides valuable insights, its limitations highlight the need for further research to fully clarify the role of medications in the development of microscopic colitis. Other important risk factors include alcohol consumption, older age, infectious gastroenteritis (particularly Clostridioides difficile infection) and autoimmune conditions, with coeliac disease being the most common associated condition.9,23
Presentation
Microscopic colitis typically presents as chronic, profuse (large-volume), watery, nonbloody diarrhoea. Bowel movement frequency ranges from four to nine watery stools daily, sometimes exceeding 15.24 Associated symptoms include faecal urgency, potential incontinence, nocturnal diarrhoea and abdominal discomfort. Weight loss may occur as a result of fluid loss and decreased appetite. The profuse and nonbloody nature of microscopic colitis-related diarrhoea helps distinguish it from other causes of diarrhoea in older adults. The diarrhoea may resolve spontaneously within weeks, although relapses are common.
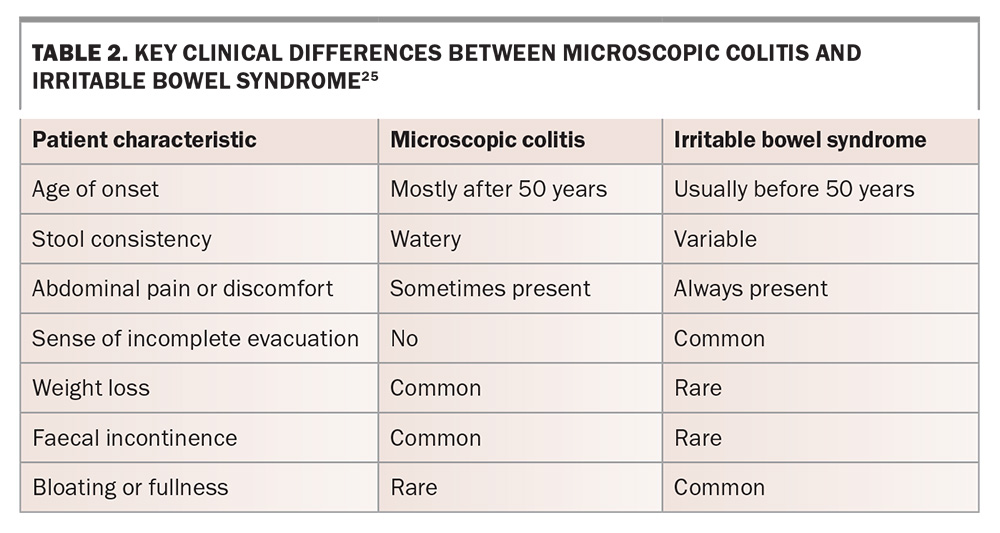
Many of these symptoms are similar to those of IBS,with about one-third to half of patients with microscopic colitis fulfilling the diagnostic criteria for IBS (Table 2).25-27 The resulting hidden prevalence of microscopic colitis presents a significant challenge, with some patients enduring a frustratingly drawn-out journey to the correct diagnosis.
Microscopic colitis significantly impairs health-related quality of life, often causing anxiety, particularly in those with faecal incontinence. This leads to social isolation and reduced participation in leisure activities. Self-esteem may also suffer as patients perceive themselves to be a burden to others.
Diagnostic approach
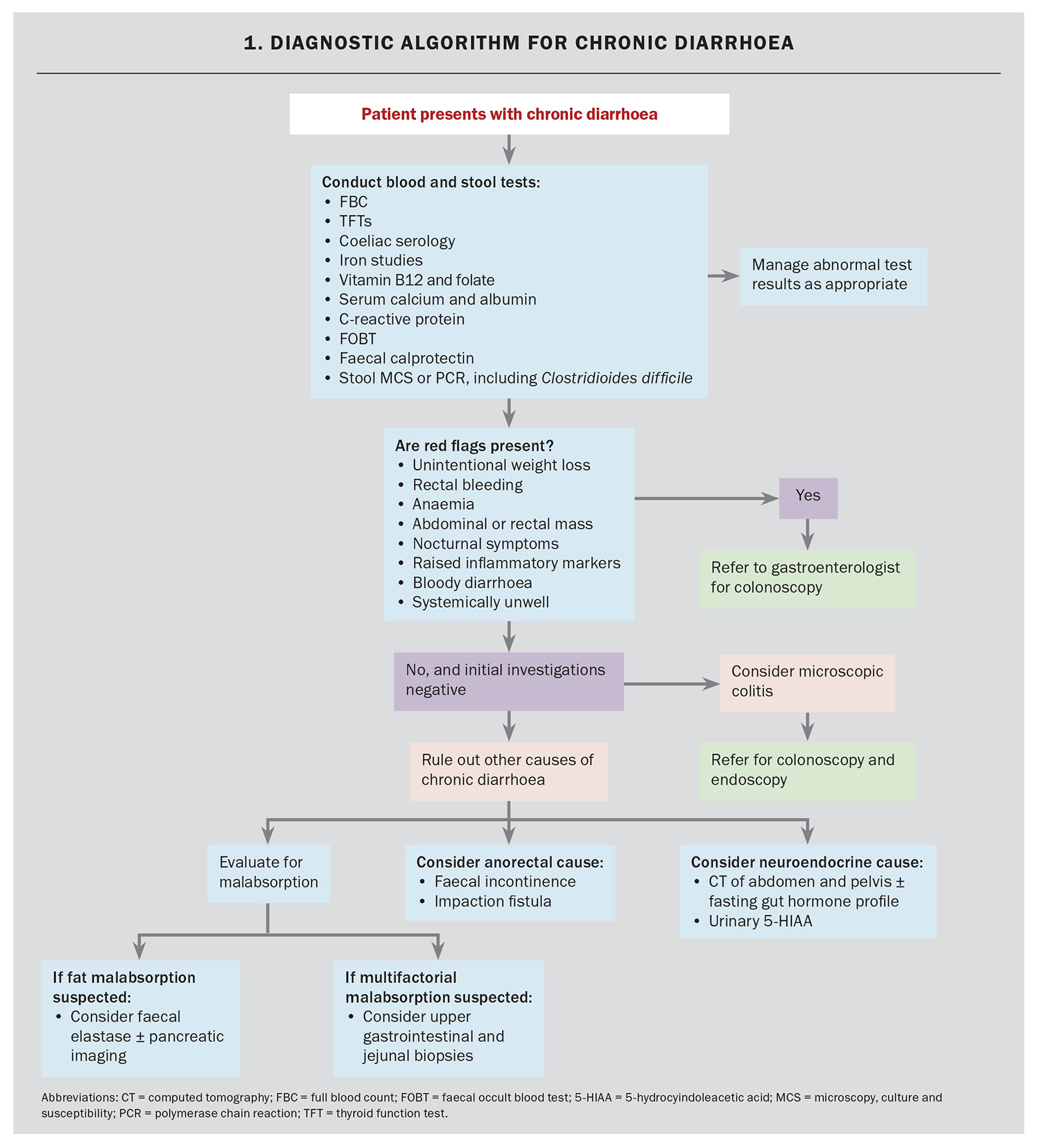
Microscopic colitis should be considered in middle-aged and older adults who present with new chronic, watery, nonbloody diarrhoea. Crucially, GPs should ask patients about all medications and supplements they are taking, as these can be a common cause of diarrhoea; metformin and magnesium are frequent offenders.
Initial investigations should include a full blood count, electrolytes including serum calcium level, serum albumin level, C-reactive protein level, coeliac serology, iron studies, vitamin B12 and folate levels, thyroid function tests, stool tests and a faecal occult blood test (Flowchart 1). Stool investigations should include tests such as polymerase chain reaction or microscopy, culture and susceptibility, and examinations for ova and parasites to exclude infectious causes of diarrhoea. Referral for colonoscopy should occur in the absence of an infectious or other common cause of diarrhoea.
Laboratory findings
In microscopic colitis, laboratory test results are often nonspecific. About half of patients may show mild anaemia and an elevated erythrocyte sedimentation rate.28 If coeliac serology is positive, a gastroscopy should also be performed at the time of colonoscopy to confirm coeliac disease.
Faecal calprotectin levels in patients with microscopic colitis are often elevated, with a mean weighted level of 214.3 mcg/g, compared with 51.0 mcg/g in controls.29 Although testing faecal calprotectin level is a simple method to differentiate patients with IBD from patients with noninflammatory disease, it lacks sufficient sensitivity as a standalone diagnostic and monitoring tool for microscopic colitis.
Endoscopy and biopsy
Definitive diagnosis of microscopic colitis requires colonoscopy, with mucosal biopsy samples taken from both the right and left colon because of lymphocyte variations. Although endoscopic examination typically appears normal, high-definition colonoscopes may show subtle changes, such as oedema or erythema, altered vascular mucosal pattern and mucosal nodularity, especially in collagenous colitis.30,31 Therefore, in patients presenting with persistent diarrhoea, GPs should specifically check whether biopsy samples were taken during any prior colonoscopy, as these are crucial for excluding microscopic colitis.
Histologically, both lymphocytic and collagenous colitis are characterised by mononuclear infiltrates within the lamina propria, with a typically sparse presence of neutrophils and eosinophils. The key differentiating feature between the two subtypes lies in the presence in collagenous colitis of a thickened collagen band in the colonic subepithelium, measuring 10 micrometres or more in diameter. In contrast, lymphocytic colitis is defined by an increased number of intraepithelial lymphocytes (20 or more for every 100 surface epithelial cells). A third subgroup, termed incomplete microscopic colitis or not otherwise specified, exhibits increased cellular infiltrate in the colonic lamina propria but does not meet the diagnostic criteria for either collagenous or lymphocytic colitis, highlighting the spectrum of histological presentations (Table 3).
Treatment
The primary goal in managing microscopic colitis is to achieve clinical remission (defined as fewer than three stools per day and no watery stools in one week) and improve quality of life. Effective management alleviates symptoms and improves patients’ psychological wellbeing and social engagement.25
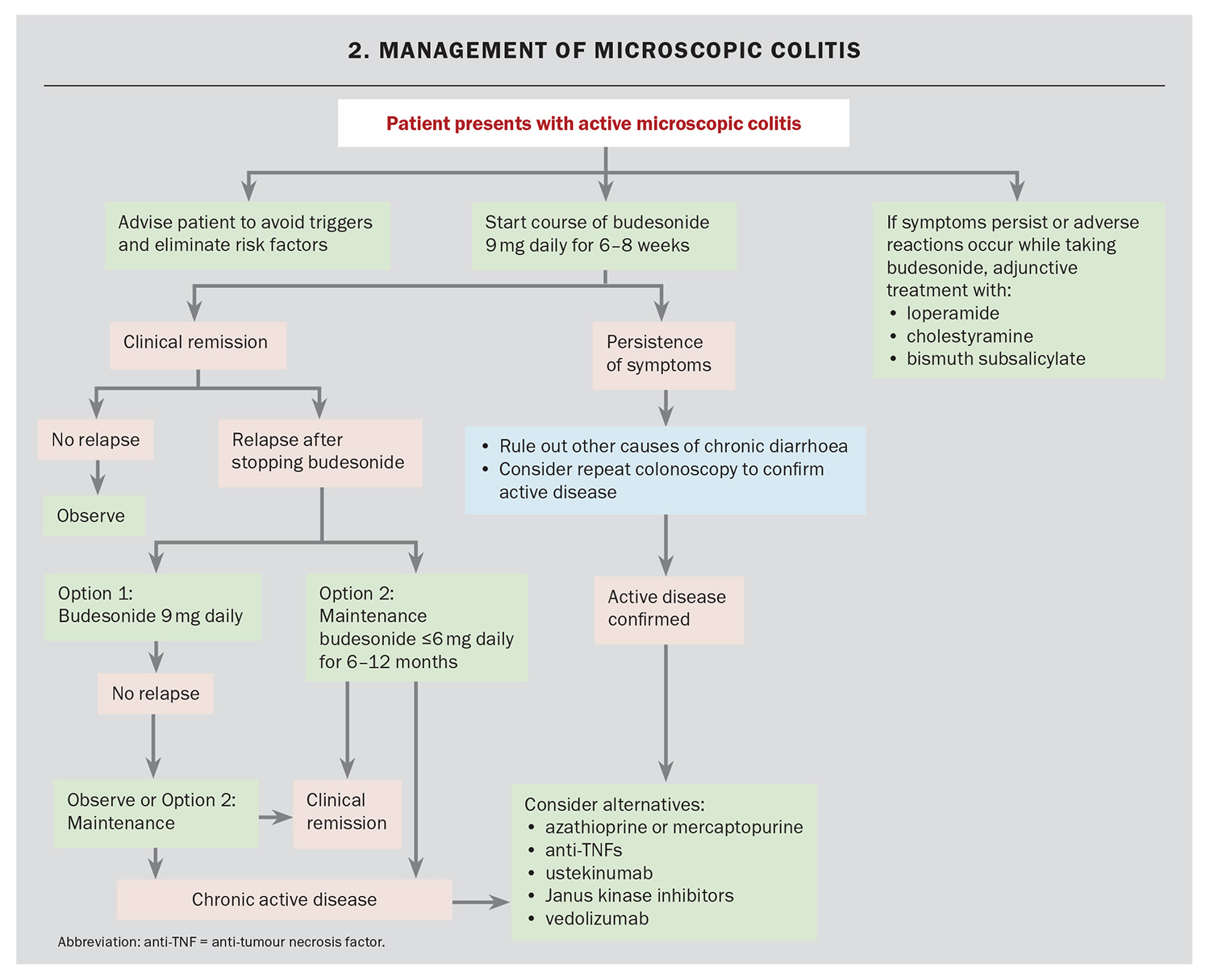
Initial management involves eliminating identifiable and modifiable risk factors, such as smoking, alcohol and relevant medications (PPIs, NSAIDs and SSRIs) (Flowchart 2). Although emerging evidence challenges the strength of the association between these medications and development of microscopic colitis, definitive evidence remains lacking.22 It therefore remains advisable to avoid these medications as part of an overall strategy to minimise potential risk factors. Dietary management and adequate hydration (including reducing caffeine intake) can also aid with managing symptoms.32
First-line medical therapy
Given the significant impact of microscopic colitis symptoms, budesonide is generally recommended as a first-line treatment for both induction and maintenance therapy. The typical regimen involves oral administration of budesonide, with a course usually lasting eight weeks. Several formulations are available, including gastroresistant granules and multimatrix formulations. The cost and availability of budesonide formulations can be a factor in treatment decisions. We recommend GPs refer patients to a gastroenterologist for initiation of budesonide therapy. In Australia, the gastroresistant granule formulations of budesonide are more readily available and have a stronger evidence base for treating microscopic colitis than multimatrix formulations.33 We favour an initial regimen of budesonide 9 mg daily (modified-release gastroresistant granule formulation) for eight weeks, in line with numerous randomised controlled trials. These medications are also included in hospital formularies. Budesonide treatment has shown clinical remission rates of 72 to 91% in patients with lymphocytic colitis and 73 to 100% in those with collagenous colitis.11
Despite budesonide’s initial effectiveness, more than 50% of patients relapse on discontinuation.11 If microscopic colitis flares are more than one year apart, intermittent courses of budesonide are a reasonable treatment approach. If flares occur more than once yearly, maintenance therapy should be considered. In cases of relapse, reinduction followed by maintenance therapy using budesonide (gastroresistant granules) at a dosage of 6 mg daily, tapered to the lowest effective dose, is often an effective strategy.34,35
Adjuvant therapies
Alongside first-line treatment, several adjuvant therapies can be beneficial.34 Loperamide often provides symptomatic relief for patients with diarrhoea, can be helpful in enabling patients to perform activities of daily living and is safe in patients with microscopic colitis. Cholestyramine is often effective, given it treats bile acid malabsorption, a condition that commonly co-occurs with microscopic colitis. It should be considered in refractory cases. Cholestyramine treatment typically starts between 2 and 4 g daily, with the option to titrate the dosage as needed based on the individual’s response and tolerance. Barriers to cholestyramine use can include palatability, constipation and challenges with the medication timing. Bismuth subsalicylate is another adjunctive option, although its efficacy is generally considered less robust than that of loperamide or cholestyramine.
Alternative therapies for persistent symptoms or frequent relapses
Although budesonide effectively induces remission in most patients with microscopic colitis, a small subset of patients experience persistent symptoms or frequent relapses, necessitating treatment escalation. Prospective clinical trials evaluating advanced therapies in this context are limited, given the relatively low prevalence of microscopic colitis and the typically good initial response to budesonide. Adding to the challenge of conducting trials in this population is the disease’s tendency for a fluctuating course and spontaneous remission.
Despite these limitations, accumulating retrospective evidence suggests that agents used in IBD, such as anti-tumour necrosis factor agents (e.g. infliximab and adalimumab), vedolizumab, ustekinumab and tofacitinib, may be effective in treating microscopic colitis at similar dosing schedules.34,36,37 A multicentre retrospective study evaluating these therapies reported remission rates of 50% with anti-tumour necrosis factor agents, 47% with vedolizumab, 40% with ustekinumab and 79% with Janus kinase inhibitors.37 Thiopurines can be considered in steroid-dependent or persistently symptomatic patients; however, their clinical benefit and safety profile are inferior to those of advanced therapies.37,38 The availability of advanced therapies means that surgical intervention, such as diverting ileostomy or total colectomy,38,39 is now rarely indicated for refractory cases.
Conclusion
Microscopic colitis should be considered in patients with chronic diarrhoea, particularly middle-aged and older people. Recognising its unique clinical features and employing a thorough diagnostic approach enables timely and accurate diagnosis and management. Budesonide is highly effective in reducing symptoms and improving quality of life, although some patients may require maintenance therapy. By enhancing our understanding of microscopic colitis and incorporating it into routine evaluations of chronic diarrhoea, we can improve patient outcomes and support their journey toward effective management. MT
COMPETING INTERESTS: Dr Bhandari and Dr Wilson: None. Dr Elford has received support for travel expenses from Dr. Falk Pharma, Ferring Pharmaceuticals and Galapagos.
ACKNOWLEDGEMENTS: Dr Elford would like to thank the Australian Government for its support via a Research Training Program Scholarship.
References
1. Britt H, Valenti L, Miller G, Charles J. Presentations of diarrhoea in Australian general practice. Aust Fam Physician 2005; 34: 218-219.
2. Jegadeesan R. Microscopic colitis: is it a spectrum of inflammatory bowel disease? World J Gastroenterol 2013; 19: 4252-4256.
3. Bozorg SR. Epidemiology and health economics in gastrointestinal diseases: real-world evidence from Swedish health registers [thesis]. Stockholm: Karolinska Institutet; 2024. Available online at: https://openarchive.ki.se/articles/thesis/Epidemiology_and_health_economics_in_gastrointestinal_diseases_real-world_evidence_from_Swedish_health_registers/27119406?file=50597619 (accessed July 2025).
4. Contreras CP, Fatuyi M. Rate of 30-day readmission and economic burden in patients with microscopic colitis. In: Proceedings of Digestive Disease Week; 2022 May 21-24; San Diego, USA. Available online at: https://eposters.ddw.org/ddw/2022/ddw-2022/354725/cynthia.contreras.rate.of.30-day.readmission.and.economic.burden.in.patients.html?f=listing%3D0%2Abrowseby %3D8%2Asortby%3D2%2Asearch%3Dmicroscopic (accessed July 2025).
5. Pardi DS, Loftus EV, Smyrk TC, et al. The epidemiology of microscopic colitis: a population based study in Olmsted County, Minnesota. Gut 2007; 56: 504-508.
6. Gentile NM, Khanna S, Loftus EV, et al. The epidemiology of microscopic colitis in Olmsted County from 2002 to 2010: a population-based study. Clin Gastroenterol Hepatol 2013; 12: 838-842.
7. Tong J, Zheng Q, Zhang C, Lo R, Shen J, Ran Z. Incidence, prevalence, and temporal trends of microscopic colitis: a systematic review and meta-analysis. Am J Gastroenterol 2015; 110: 265-276.
8. Rasmussen MA, Munck LK. Systematic review: are lymphocytic colitis and collagenous colitis two subtypes of the same disease – microscopic colitis? Aliment Pharmacol Ther 2012; 36: 79-90.
9. Peery AF, Khalili H, Münch A, Pardi DS. Update on the epidemiology and management of microscopic colitis. Clin Gastroenterol Hepatol 2025; 23: 490-500.
10. Pardi DS, Tremaine WJ, Carrasco-Labra A. American Gastroenterological Association Institute technical review on the medical management of microscopic colitis. Gastroenterology 2016; 150: 247-274.e11.
11. Nielsen OH, Fernandez-Banares F, Sato T, Pardi DS. Microscopic colitis: etiopathology, diagnosis, and rational management. Elife 2022; 11: e79397.
12. Songtanin B, Chen JN, Nugent K. Microscopic colitis: pathogenesis and diagnosis. J Clin Med 2023; 12: 4442.
13. Zabana Y, Tontini G, Hultgren-Hörnquist E, et al. Pathogenesis of microscopic colitis: a systematic review. J Crohns Colitis 2021; 16: 143-161.
14. Garczyk A, Mardas M, Stelmach-Mardas M. Microbiome composition in microscopic colitis: a systematic review. Int J Mol Sci 2023; 24: 7026.
15. Mohammed A, Ghoneim S, Paranji N, Waghray N. Quantifying risk factors for microscopic colitis: a nationwide, retrospective cohort study [abstract S142]. Am J Gastroenterol 2021; 116: S62-S63.
16. van Hemert S, Skonieczna-Żydecka K, Loniewski I, Szredzki P, Marlicz W. Microscopic colitis—microbiome, barrier function and associated diseases. Ann Transl Med 2018; 6: 39.
17. Mori S, Kadochi Y, Luo Y, et al. Proton pump inhibitor induced collagen expression in colonocytes is associated with collagenous colitis. World J Gastroenterol 2017; 23: 1586-1593.
18. Liu Y, Chen M. Insights into the underlying mechanisms and clinical management of microscopic colitis in relation to other gastrointestinal disorders. Gastroenterology Rep (Oxf) 2022; 10: goac011.
19. Pascua MF, Kedia P, Weiner MG, Holmes J, Ellenberg J, Lewis JD. Microscopic colitis and medication use. Clin Med Insights Gastroenterol 2010; 2010(3): 11-19.
20. Tarar ZI, Farooq U, Gandhi M, et al. Are drugs associated with microscopic colitis? A systematic review and meta-analysis. Diseases 2022; 11: 6.
21. Nimri F, Muhanna A, Almomani Z, et al. The association between microscopic colitis and celiac disease: a systematic review and meta-analysis. Ann Gastroenterol 2022; 35: 281-289.
22. Khalili H, McGee EE, Challa PK, et al. Medications and risk for microscopic colitis: a nationwide study of older adults in Sweden. Ann Intern Med 2025; 178: 1106-1115.
23. Niccum B, Casey K, Burke K, et al. Alcohol consumption is associated with an increased risk of microscopic colitis: results from 2 prospective US cohort studies. Inflamm Bowel Dis 2022; 28: 1151-1159.
24. Münch A, Langner C. Microscopic colitis: clinical and pathologic perspectives. Clin Gastroenterol Hepatol 2015; 13: 228-236.
25. Fedor I, Zold E, Barta Z. Microscopic colitis in older adults: impact, diagnosis, and management. Ther Adv Chronic Dis 2022; 13: 20406223221102821.
26. Abboud R, Pardi DS, Tremaine WJ, Kammer PP, Sandborn WJ, Loftus EV. Symptomatic overlap between microscopic colitis and irritable bowel syndrome. Inflamm Bowel Dis 2013; 19: 550-553.
27. Limsui D, Pardi DS, Camilleri M, et al. Symptomatic overlap between irritable bowel syndrome and microscopic colitis. Inflamm Bowel Dis 2007; 13: 175-181.
28. Hempel KA, Sharma AV. Collagenous and lymphocytic colitis. In: StatPearls. Treasure Island, Fla: StatPearls Publishing; 2022. Available online at: https://www.ncbi.nlm.nih.gov/books/NBK541100/ (accessed July 2025).
29. Songtanin B, Kahathuduwa C, Nugent K. Fecal calprotectin level in microscopic colitis: a systematic review and meta-analysis. Proc (Bayl Univ Med Cent) 2023; 36: 641-646.
30. Koulaouzidis A. Distinct colonoscopy findings of microscopic colitis: Not so microscopic after all? World J Gastroenterol 2011; 17: 4157.
31. Smirnidis A, Trimble KC, Lessells A, Koulaouzidis A. Endoscopic findings in collagenous colitis; not always microscopic [abstract PMO-197]. Gut 2012; 61 Suppl 2: A154.
32. Crohn’s & Colitis UK. Information sheet: microscopic colitis. Hatfield: Crohn's & Colitis UK; 2025. Available online at: https://crohnsandcolitis.org.uk/info-support/information-about-crohns-and-colitis/all-information-about-crohns-and-colitis/understanding-crohns-and-colitis/microscopic-colitis (accessed July 2025).
33. Kumar A, Hiner G, Brookes MJ, Segal JP. Efficacy and safety of medical therapies in microscopic colitis: a systematic review and network meta-analysis. Therap Adv Gastroenterol 2023; 16: 17562848231154319.
34. Miehlke S, Guagnozzi D, Zabana Y, et al. European guidelines on microscopic colitis: United European Gastroenterology and European Microscopic Colitis Group statements and recommendations. United European Gastroenterol J 2021; 9: 13-37.
35. American Gastroenterological Association. AGA Institute guideline on the management of microscopic colitis: clinical decision support tool. Gastroenterology 2016; 150: 276.
36. Esteve M, Mahadevan U, Sainz E, Rodriguez E, Salas A, Fernández-Bañares F. Efficacy of anti-TNF therapies in refractory severe microscopic colitis. J Crohns Colitis 2011; 5: 612-618.
37. Verstockt B, Taelman T, Vavricka SR, et al. Promising efficacy of biologicals and small molecules for microscopic colitis: results from a large real-life multicenter cohort [abstract DOP79]. J Crohns Colitis 2024; 18 Suppl 1: i218-i220.
38. Park T, Cave D, Marshall C. Microscopic colitis: a review of etiology, treatment and refractory disease. World J Gastroenterol 2015; 21: 8804-8810.
39. Datta I, Brar SS, Andrews CN, et al. Microscopic colitis: a review for the surgical endoscopist. Can J Surg 2009; 52: E167-E172.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.