Managing obesity – looking beyond lifestyle interventions
Most people in Australia are living with overweight or obesity. Obesity and its complications are associated with excess morbidity and mortality, reduced quality of life and substantial financial costs to both individuals and the healthcare system. Effective treatment options for obesity are available, and several promising therapies are currently being investigated in clinical trials.
- Almost one in three adults in Australia is living with obesity.
- Effective treatments for obesity are available that reduce morbidity and mortality, and improve health and quality of life.
- When treatment goals are not met or are unlikely to be met by lifestyle interventions alone, medications and surgical management should be considered.
- GPs play a key role in supporting patients to manage obesity.
Obesity increases the risk of ill health and is associated with other conditions, such as type 2 diabetes (T2DM), cardiovascular disease and hypertension. This article outlines current and emerging pharmacological and surgical approaches to the management of obesity.
What is obesity?
Obesity is characterised by abnormal or excessive fat accumulation that presents a risk to a person’s health.1 A body mass index (BMI) greater than 30 kg/m2 is commonly used to define obesity, although the BMI alone does not reliably indicate health status and may both under- and overestimate adiposity.2 Obesity is a complex chronic condition that develops in genetically predisposed individuals in response to several (predominantly environmental) factors.3
Why is it important to treat obesity?
Obesity affects almost one-third (31%) of adults in Australia and is the second largest contributor to the national burden of disease.4,5 This is due to both its direct health impact and its role in the development of numerous other chronic diseases.
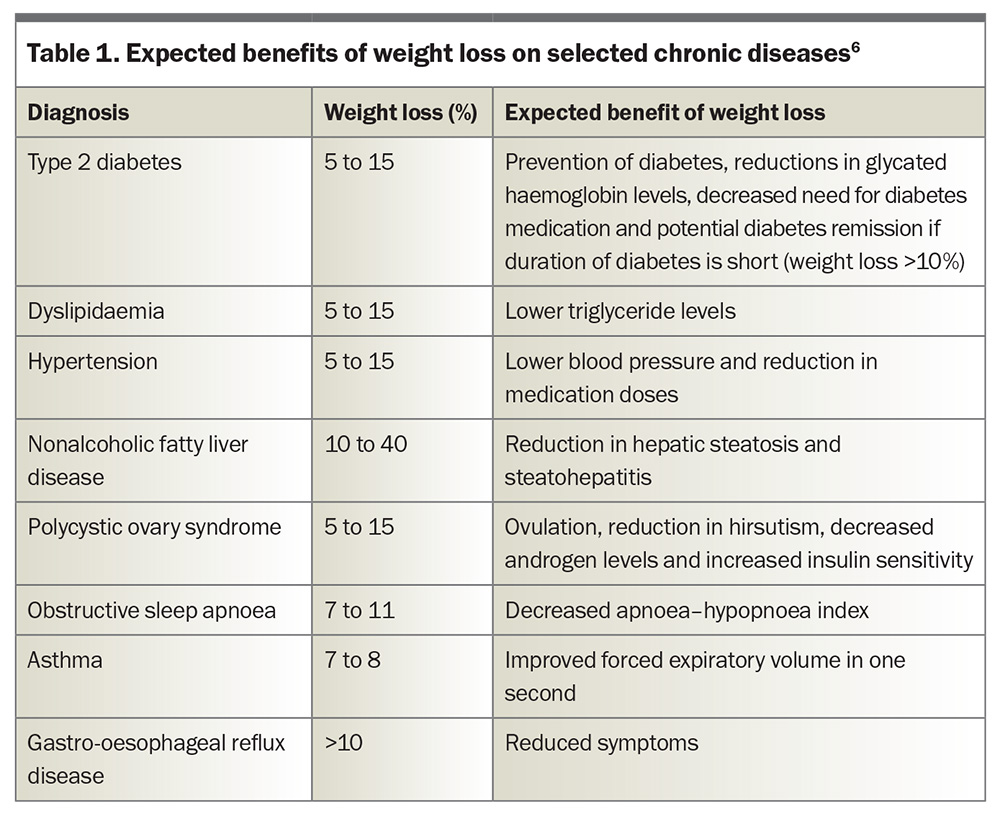
Weight loss can significantly reduce the adverse impact of obesity on health and quality of life; hence, effective treatment is critical (Table 1).6 A reduction of just 5% in original body weight reduces the risk of progression to T2DM by more than 50% in people with impaired glucose tolerance, and also improves blood pressure and triglyceride levels.7 Greater weight loss (10 to 25%) offers progressive benefits, including improvements in the control (and potentially remission) of T2DM, steatotic liver disease, obstructive sleep apnoea, stress urinary incontinence and symptoms of osteoarthritis, as well as improvements in health-associated quality of life.8 Until recently, most nonsurgical interventions for obesity were expected to achieve sustained weight loss of less than 5%. However, increasingly effective nonsurgical treatments are now becoming available.9
What role do GPs play?
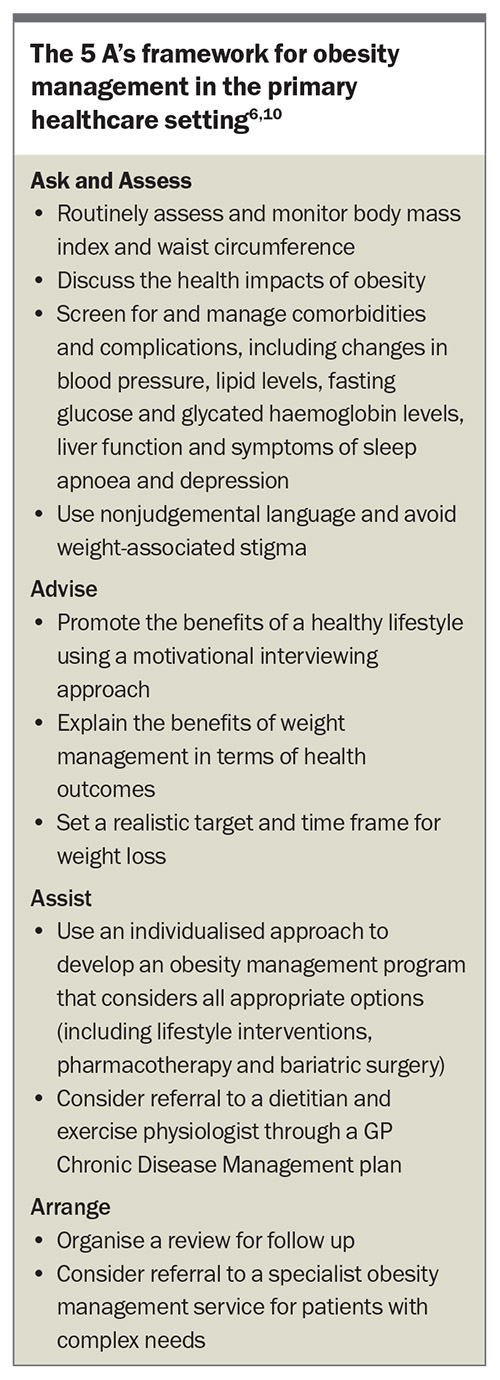
Obesity requires lifelong monitoring and management. GPs are essential in establishing a diagnosis of obesity and in managing obesity at each stage of the treatment pathway. The 5 A’s framework provides a guide to obesity management in the primary healthcare setting (Box).6,10
Specialised obesity services are scarce and often have limited capacity and long waiting times.11 Therefore, obesity often needs to be managed largely or entirely within the primary healthcare setting. With the benefit of an established therapeutic relationship, primary healthcare practitioners are ideally placed to advise and support behaviour change; discuss, initiate and adjust medications when needed; refer to additional clinicians and services (e.g. dietitian, exercise physiologist, psychologist, specialist physician or surgeon) where required; monitor progress; and address challenges that arise during obesity treatment. The Australian Obesity Management Algorithm also provides guidance on the assessment and management of obesity in the primary healthcare setting.12 An update to the National Health and Medical Research Council’s clinical practice guidelines for the management of obesity is expected later this year.
What lifestyle interventions are effective?
The main purpose of lifestyle interventions in obesity management is to reduce energy intake, optimise nutritional quality and increase physical activity. This can be accomplished via a variety of approaches, with no single dietary pattern showing superiority. An individualised regimen supported by a multidisciplinary team with frequent contact (14 or more sessions across six to 12 months) has been shown in clinical trials to be the most effective approach; however, this degree of allied healthcare support is not subsidised by Medicare.10
Most people who lose weight with lifestyle interventions alone will regain weight in the longer term. The misconception that obesity is simply caused by poor lifestyle habits and inadequate motivation for behaviour change is common and leads to stigma. However, weight loss leads to an enduring biological response that results in an increased appetite and a reduction in total energy expenditure (more than expected for the loss of body mass). Interactions between biological, psychosocial and ‘obesogenic’ environmental factors make the maintenance of weight loss challenging for most people.
What pharmacotherapy is available?
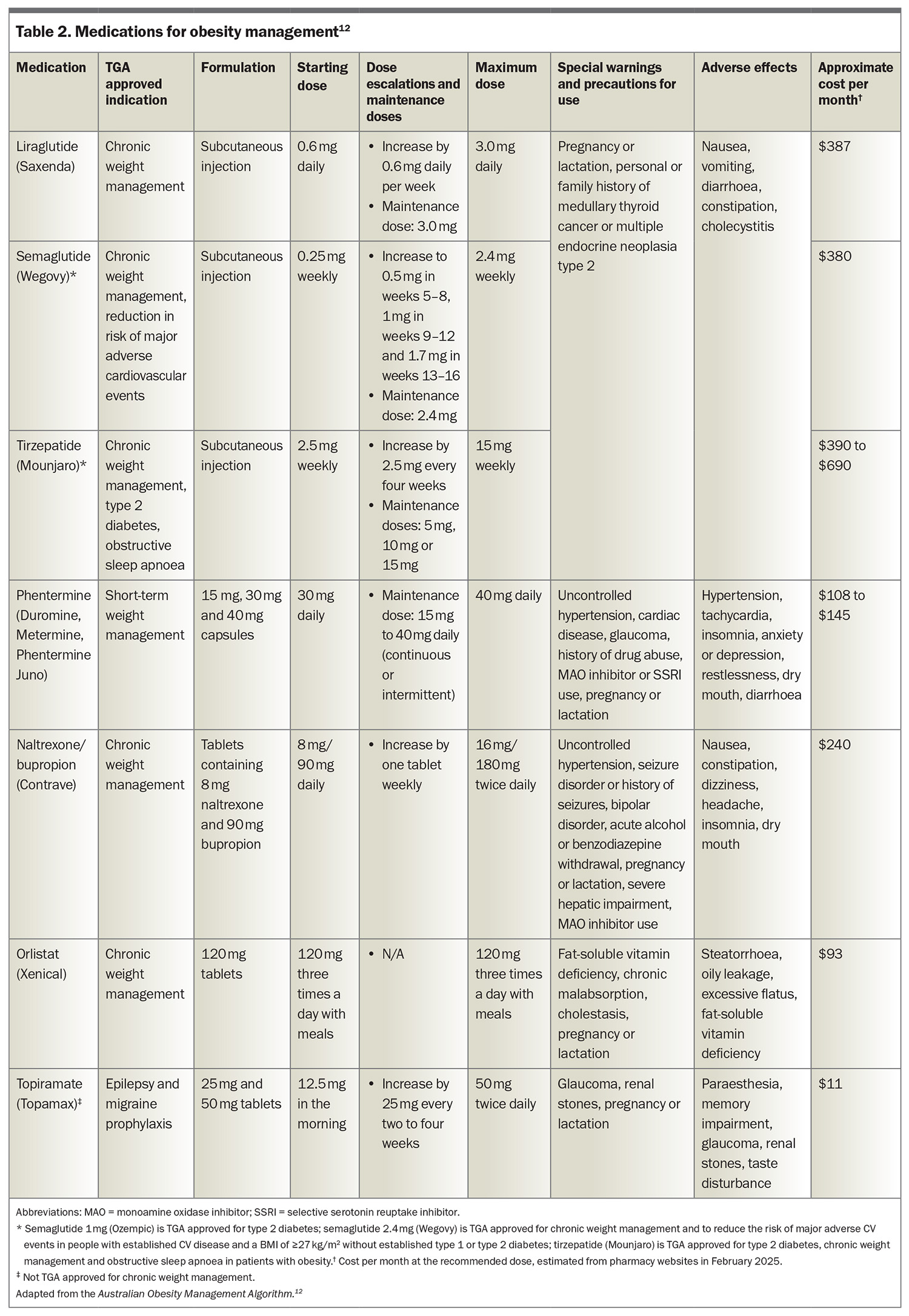
Six medications are approved by the TGA for obesity management: liraglutide, semaglutide (2.4 mg weekly dose), tirzepatide, phentermine, naltrexone/bupropion and orlistat (Table 2).12 Orlistat, liraglutide, naltrexone/bupropion and tirzepatide are indicated for weight management in adults with a BMI 30 kg/m2 or more, or 27 kg/m2 to less than 30 kg/m2 with at least one weight-associated complication. Semaglutide (2.4 mg weekly dose) is indicated for adults with obesity or overweight, and for adolescents aged 12 years and older with obesity and body weight greater than 60 kg. Phentermine is approved for adults and adolescents older than 12 years of age with a BMI of 25 kg/m2 or higher. Currently, no medications are subsidised by the PBS for obesity management.
The choice of medication requires the consideration of a number of individualised factors, including patient preference, severity of obesity, presence of obesity-related and unrelated medical conditions, and the medication’s efficacy, contraindications, adverse effect profile and cost. All the medications approved for obesity management are contraindicated during pregnancy and lactation.
Glucagon-like peptide-1 receptor agonists
Two glucagon-like peptide-1 (GLP-1) receptor agonists are currently TGA approved for obesity management: liraglutide (dosage of 3.0 mg daily) and semaglutide (dosage of 2.4 mg weekly). Liraglutide (dosage of up to 1.8 mg daily) and semaglutide (dosage of up to 1.0 mg weekly) are also indicated for the treatment of T2DM, as is dulaglutide (dosage of 1.5 mg weekly). Semaglutide (2.4 mg weekly dose) has an additional indication to reduce the risk of major adverse cardiovascular events in people with established cardiovascular disease and a BMI of 27 km/m2 or more with or without diabetes. Saxenda, one brand of liraglutide, will be discontinued by the manufacturer at the end of 2025, although generic versions may become available. In clinical trials for obesity management in people without T2DM, the mean total weight losses were 8% (vs 2.6% in the placebo group) in those taking liraglutide 3.0 mg daily for 56 weeks and up to 16% (vs 5.7% in the placebo group) in those taking semaglutide 2.4 mg weekly for 68 weeks.13,14
GLP-1 receptor agonists act via GLP-1 receptors. In the brain (particularly in the hypothalamus and hindbrain), they act to increase satiety and reduce hunger and food reward. In the pancreas and gastrointestinal tract, they slow gastric emptying, enhance insulin release in the presence of elevated glucose levels and reduce glucagon release.
Their beneficial effects on glycaemia make them particularly suitable for obesity management in people with (or at a high risk of) T2DM.14 At doses used to treat T2DM, some GLP-1 receptor agonists (e.g. dulaglutide, liraglutide and semaglutide) reduce mortality and the risk of cardiovascular and renal disease in patients with T2DM.15 In people with established cardiovascular disease and overweight or obesity but without T2DM, treatment with semaglutide 2.4 mg weekly reduced cardiovascular events (a composite of cardiovascular death, myocardial infarction and stroke) by 20% during 40 months of follow up.16
The adverse effects of GLP-1 receptor agonists include nausea, vomiting, constipation, diarrhoea and an increased risk of cholelithiasis and cholecystitis. Nausea usually improves with continued therapy and can be minimised by starting the medication at a low dosage and gradually uptitrating.
Tirzepatide
Tirzepatide is a dual GLP-1 and glucose-dependent insulinotropic polypeptide receptor agonist. It is TGA approved for the treatment of T2DM, obesity and obstructive sleep apnoea, and is administered weekly at dosages of 5 mg, 10 mg or 15 mg. Tirzepatide is the first medication to show a mean weight loss of more than 20% in clinical trials for obesity management. In a phase 3 clinical trial, participants taking tirzepatide at doses of 5 mg, 10 mg and 15 mg achieved mean weight losses of 15%, 20% and 21%, respectively, after 72 weeks of treatment, compared with 3% in the placebo group.17 In a head-to-head trial over 72 weeks, tirzepatide (10 mg or 15 mg once weekly) resulted in a weight loss of 20%, compared with 14% with semaglutide 2.4 mg once weekly.18 Adverse effects are predominately gastrointestinal, with a profile similar to that of GLP-1 receptor agonists.
Phentermine
Phentermine is a sympathomimetic agent that stimulates the release of noradrenaline, dopamine and serotonin in several areas of the brain to reduce hunger and reward-associated eating. It is indicated as a ‘short-term adjunct’ in a medically monitored obesity management program, with patients requiring medical review within three months. The weight loss within 12 weeks is typically 5 to 10% of initial body weight.19 Common adverse effects include tachycardia, hypertension, insomnia and dry mouth. Phentermine is contraindicated in people with cardiovascular disease or uncontrolled hypertension and those taking monoamine oxidase inhibitors. Phentermine is also not recommended in combination with selective serotonin reuptake inhibitors.
Naltrexone/bupropion
This combination of an antidepressant (bupropion) and an opioid antagonist (naltrexone) acts in the hypothalamus and mesolimbic reward system to reduce hunger and food cravings. In a 56-week randomised trial, naltrexone/bupropion resulted in a mean weight loss of 6.1% (compared with 1.3% in the placebo group).20
The adverse effects include nausea, headache, dizziness, insomnia and dry mouth. Naltrexone/bupropion is contraindicated in patients with a history of seizures or bipolar disorder and in those using opioid medications.
Several potential drug interactions should be considered when prescribing naltrexone/bupropion, particularly those involving cytochrome P450 (CYP) enzyme activity. These include the need to reduce doses of medications metabolised by CYP2D6 (including selective serotonin reuptake inhibitors, because of CYP2D6 inhibition by bupropion) if prescribed concurrently.
Orlistat
Orlistat reduces gastrointestinal fat absorption by inhibiting pancreatic and gastric lipases. Unlike other obesity medications orlistat is not systemically absorbed, having only local action in the gut lumen. Weight loss of about 5% is expected after one year of treatment.21
The safety and modest efficacy of orlistat have been shown in randomised trials of up to four years’ duration, although the adherence to treatment is poor, largely because of its gastrointestinal adverse effects.21 Common adverse effects include steatorrhoea, oily stools and flatulence. The absorption of fat-soluble vitamins may be reduced by orlistat; therefore, supplementation with a multivitamin should be advised. Orlistat has a weight loss-independent effect on lowering LDL (by 28% compared with lifestyle interventions alone).22
Other agents
Topiramate
Topiramate is approved for the treatment of epilepsy and the prevention of migraine. It is commonly used off-label for obesity management, although the drug’s mechanism of action for weight loss is unclear. In the USA (but not in Australia), a combination of extended-release topiramate and phentermine is approved for obesity management. Meta-analyses of data from randomised controlled trials of topiramate for obesity suggest weight loss of about 5% compared with placebo.23,24 The adverse effects include gastrointestinal disturbances, difficulty concentrating, paraesthesia, depression, teratogenicity and (rarely) closed-angle glaucoma.
Setmelanotide
Setmelanotide is a melanocortin-4 agonist approved in some parts of the world for the management of obesity caused by rare monogenic variants affecting appetite signalling pathways, including those involving leptin, proprotein convertase subtilisin/kexin type 1 or pro-opiomelanocortin. It is not currently TGA approved.
A separate article in this supplement discusses incretin analogues and other novel agents in development for obesity.
When should pharmacotherapy be reviewed?
Patients should be monitored at least monthly for the first three months after initiating pharmacotherapy to assess safety and efficacy and review the need for dose titration.6
Early weight loss is predictive of longer-term weight loss; hence, weight loss of less than 5% after three months at the recommended dose requires a change in the treatment strategy.6 It is worth noting that if a medication is initiated to prevent or minimise weight regain (rather than to induce weight loss), weight stability (rather than continued weight gain) may still indicate a beneficial effect. Weight regain is likely after cessation of pharmacotherapy; therefore, long-term treatment is likely to be required (as for most chronic diseases), although data on the long-term efficacy and safety of obesity medications are currently limited.25
What is the role of metabolic and bariatric surgery?
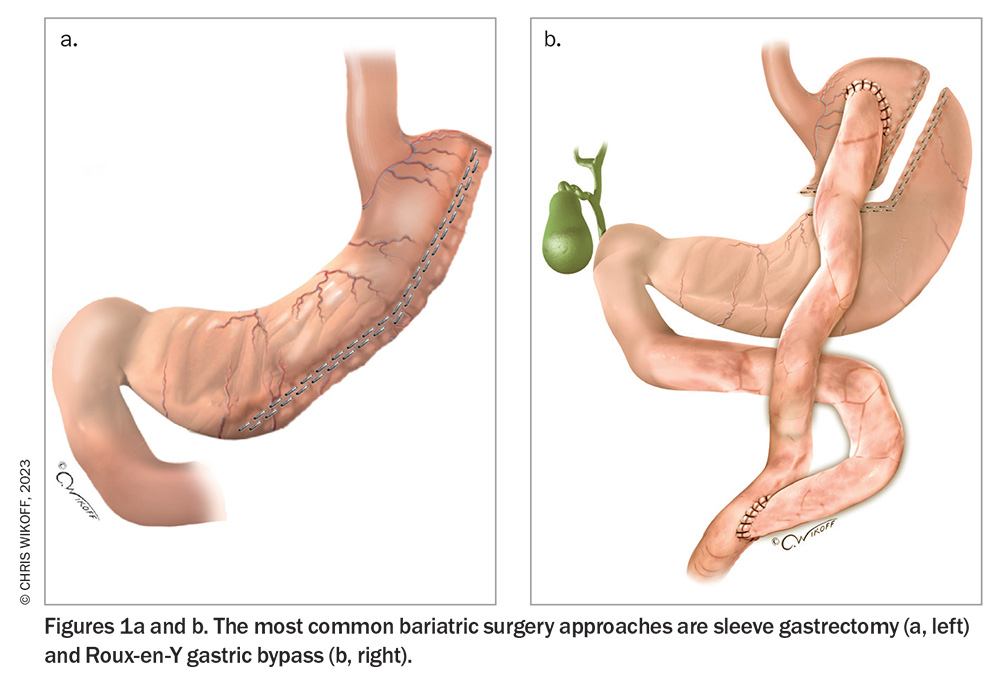
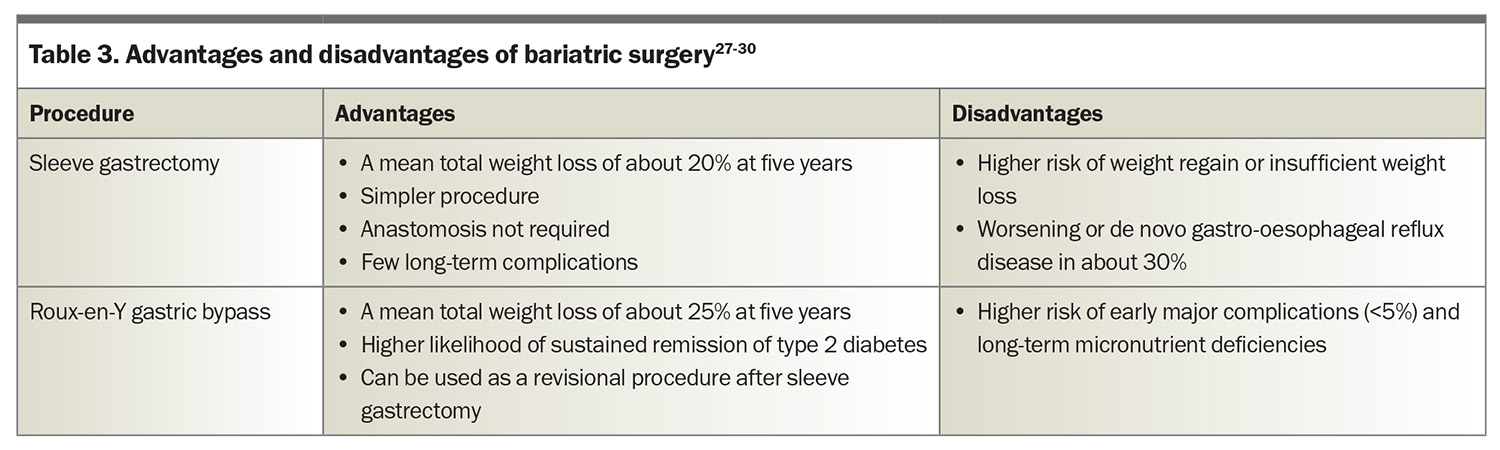
More than 40,000 individuals are estimated to undergo bariatric surgery each year in Australia.26 The most common operations are sleeve gastrectomy and Roux-en-Y gastric bypass (RYGB), both of which help achieve substantial, sustained weight loss (Figure 1 and Table 3).26-29 Compared with sleeve gastrectomy, RYGB results in greater mean weight loss (at five years, around 25% after RYGB and 19% after sleeve gastrectomy in an observational cohort study [n = 65,000]) with a higher risk of major complications (at 30 days, 5.0% after RYGB and 2.6% after sleeve gastrectomy).27 Substantial weight regain, defined as regain to within 5% of preoperative weight, occurs in fewer than 5% of patients after RYGB and in around 10% of patients after sleeve gastrectomy.30
Australian guidelines recommend that bariatric surgery be considered in patients with the following BMIs and presentations:
- BMI of 40 kg/m2 or higher
- BMI of 35.0 to 39.9 kg/m2 and comorbidities that may improve with weight loss
- BMI of 30.0 to 34.9 kg/m2 with suboptimal control of T2DM and increased risk of cardiovascular disease.10
International guidelines were updated in 2022 to recommend bariatric surgery for people with a BMI of 35 kg/m2 or higher, regardless of the presence, absence or severity of obesity-associated conditions.31 Surgery should not be viewed only as a last resort for those in whom other obesity treatments have failed. For example, in patients with suboptimal glycaemic control on maximum medical management of T2DM, early referral to surgery should be considered. Access to bariatric surgery is limited and inequitable because of a lack of services, particularly in the public sector, with about 90% of bariatric surgeries occurring in the private healthcare system in Australia.32,33
A multidisciplinary team approach is imperative to assess the potential benefits and risks of bariatric surgery for each patient before surgery, as well as for preoperative preparation and postoperative follow up. After bariatric surgery, patients require lifelong nutritional supplementation, monitoring and ongoing support, although guidelines differ in terms of specific recommendations.28,34
What nonsurgical procedures are available?
Endoscopic insertion of a fluid- or air-filled intragastric balloon is a nonsurgical option for obesity management. This is a temporary procedure, with the balloon removed after around six months (depending on the specific product). This procedure aims to reduce food intake by reducing gastric capacity and delaying gastric emptying. Weight loss achieved in clinical trial settings is about 4% greater than that observed in lifestyle intervention groups after three to six months.35 Longer-term data are scarce. Reported adverse events include nausea, abdominal pain, deflation and (rarely) gastric perforation. Endoscopic sleeve gastroplasty is another minimally invasive option, in which a suturing device is inserted endoscopically and the stomach is sutured to reduce its size. Similarly, long-term data are lacking, and it is not currently widely available in Australia.
Conclusion
The goals of obesity management are to improve a person’s health and quality of life. Many patients will not achieve treatment goals through lifestyle interventions alone. Several pharmacological treatments and bariatric surgical options are available for obesity management. The latest generation of medications is associated with mean weight losses that are close to the magnitude seen with bariatric surgery, as well as improvements in health and quality of life. However, there are inequities in accessibility at every stage of obesity care. ET
COMPETING INTERESTS: Dr Wootton has received a postgraduate scholarship from the National Health and Medical Research Council and funding from the Queensland Technology Future Fund and Royal Australasian College of Physicians for work unrelated to obesity. Associate Professor Sumithran’s institution has received support from the National Health and Medical Research Council. She was a Council Member of the Australian and New Zealand Obesity Society (2017-2022) and a member of the leadership group at the Obesity Collective. She has received honoraria (paid to her institution) for advisory and speaking activities from Novo Nordisk and Eli Lilly, and is a coauthor on manuscripts with medical writing services provided by Novo Nordisk and Eli Lilly.
References
1. Rubino F, Cummings DE, Eckel RH, et al. Definition and diagnostic criteria of clinical obesity. Lancet Diabetes Endocrinol 2025; 13: 221-262.
2. World Health Organization (WHO). Obesity. Geneva: WHO; 2022. Available online at: https://www.who.int/health-topics/obesity (accessed July 2025).
3. Heymsfield SB, Wadden TA. Mechanisms, pathophysiology, and management of obesity. N Engl J Med 2017; 376: 254-266.
4. Australian Bureau of Statistics (ABS). National Health Survey. Canberra: ABS; 2023. Available online at: https://www.abs.gov.au/statistics/health/health-conditions-and-risks/national-health-survey-first-results/latest-release (accessed July 2025).
5. Australian Institute of Health and Welfare (AIHW). Impact of Overweight and Obesity as a Risk Factor for Chronic Conditions. Canberra: AIHW; 2017. Available online at: https://www.aihw.gov.au/reports/burden-of-disease/impact-of-overweight-and-obesity-as-a-risk-factor/summary (accessed July 2025).
6. Schutz DD, Busetto L, Dicker D, et al. European practical and patient-centred guidelines for adult obesity management in primary care. Obes Facts 2019; 12: 40-66.
7. Hamman RF, Wing RR, Edelstein SL, et al. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care 2006; 29: 2102-2107.
8. Tahrani AA, Morton J. Benefits of weight loss of 10% or more in patients with overweight or obesity: a review. Obesity 2022; 30: 802-840.
9. Rucker D, Padwal R, Li SK, Curioni C, Lau DC. Long term pharmacotherapy for obesity and overweight: updated meta-analysis. BMJ 2007; 335: 1194-1199.
10. National Health and Medical Research Council (NHMRC). Clinical Practice Guidelines for the Management of Overweight and Obesity in Adults, Adolescents and Children in Australia. Canberra: NHMRC; 2013. Available online at: https://www.nhmrc.gov.au/about-us/publications/clinical-practice-guidelines-management-overweight-and-obesity (accessed July 2025).
11. Atlantis E, Kormas N, Samaras K, et al. Clinical obesity services in public hospitals in Australia: a position statement based on expert consensus. Clin Obes 2018; 8: 203-210.
12. Markovic TP, Proietto J, Dixon JB, et al. The Australian Obesity Management Algorithm: a simple tool to guide the management of obesity in primary care. Obes Res Clin Pract 2022; 16: 353-363.
13. Pi-Sunyer X, Astrup A, Fujioka K, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med 2015; 373: 11-22.
14. Wadden TA, Bailey TS, Billings LK, et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: The STEP 3 randomized clinical trial. JAMA 2021; 325: 1403-1413.
15. Sattar N, Lee MMY, Kristensen SL, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol 2021; 9: 653-662.
16. Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med 2023; 389: 2221-2232.
17. Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med 2022; 387: 205-216.
18. Aronne LJ, Horn DB, le Roux CW, et al. Tirzepatide as compared with semaglutide for the treatment of obesity. N Engl J Med 2025; 393: 26-36.
19. Kang JG, Park CY, Kang JH, Park YW, Park SW. Randomized controlled trial to investigate the effects of a newly developed formulation of phentermine diffuse-controlled release for obesity. Diabetes Obes Metab 2010; 12: 876-882.
20. Greenway FL, Fujioka K, Plodkowski RA, et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2010; 376: 595-605.
21. Torgerson JS, Hauptman J, Boldrin MN, Sjöström L. XENical in the prevention of diabetes in obese subjects (XENDOS) study: a randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care 2004; 27: 155-161.
22. Shi Q, Wang Y, Hao Q, et al. Pharmacotherapy for adults with overweight and obesity: a systematic review and network meta-analysis of randomised controlled trials. Lancet 2022; 399: 259-269.
23. Kramer CK, Leitão CB, Pinto LC, Canani LH, Azevedo MJ, Gross JL. Efficacy and safety of topiramate on weight loss: a meta-analysis of randomized controlled trials. Obes Rev 2011; 12: e338-e347.
24. Lei XG, Ruan JQ, Lai C, Sun Z, Yang X. Efficacy and safety of phentermine/topiramate in adults with overweight or obesity: a systematic review and meta-analysis. Obesity 2021; 29: 985-994.
25. Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: the STEP 1 trial extension. Diabetes Obes Metab 2022; 24: 1553-1564.
26. Dona SWA, Angeles MR, Nguyen D, Gao L, Hensher M. Obesity and bariatric surgery in Australia: future projection of supply and demand, and costs. Obes Surg 2022; 32: 3013-3022.
27. Arterburn D, Wellman R, Emiliano A, et al. Comparative effectiveness and safety of bariatric procedures for weight loss: a PCORnet cohort study. Ann Intern Med 2018; 169: 741-750.
28. Mechanick JI, Apovian C, Brethauer S, et al. Clinical practice guidelines for the perioperative nutrition, metabolic, and nonsurgical support of patients undergoing bariatric procedures - 2019 update: cosponsored by American Association of Clinical Endocrinologists/American College of Endocrinology, The Obesity Society, American Society for Metabolic and Bariatric Surgery, Obesity Medicine Association, and American Society of Anesthesiologists. Surg Obese Relat Dis 2020; 16: 175-247.
29. International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO). Bariatric surgery. Naples: IFSO; 2018. Available online at: https://www.ifso.com/bariatric-surgery/ (accessed July 2025).
30. Arterburn DE, Telem DA, Kushner RF, Courcoulas AP. Benefits and risks of bariatric surgery in adults: a review. JAMA 2020; 324: 879-887.
31. Eisenberg D, Shikora SA, Aarts E, et al. 2022 American Society for Metabolic and Bariatric Surgery (ASMBS) and International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO): indications for metabolic and bariatric surgery. Surg Obes Relat Dis 2022; 18: 1345-1356.
32. Aly A, Talbot ML, Brown WA. Bariatric surgery: a call for greater access to coordinated surgical and specialist care in the public health system. Med J Aust 2022; 217: 228-231.
33. Australian Institute of Health and Welfare (AIHW). Weight Loss Surgery in Australia 2014–15: Australian Hospital Statistics. Canberra: AIHW; 2017. Available online at: https://www.aihw.gov.au/reports/hospitals/ahs-2014-15-weight-loss-surgery/summary (accessed July 2025).
34. O’Kane M, Parretti HM, Pinkney J, et al. British Obesity and Metabolic Surgery Society Guidelines on perioperative and postoperative biochemical monitoring and micronutrient replacement for patients undergoing bariatric surgery - 2020 update. Obes Rev 2020; 21: e13087.
35. Kotinda APST, de Moura DTH, Ribeiro IB, et al. Efficacy of intragastric balloons for weight loss in overweight and obese adults: a systematic review and meta-analysis of randomized controlled trials. Obes Surg 2020; 30: 2743-2753.