A man with a widespread pruritic skin eruption
Test your diagnostic skills in our regular dermatology quiz. What is this itchy eruption, which has not responded to topical corticosteroid treatment?
Case presentation
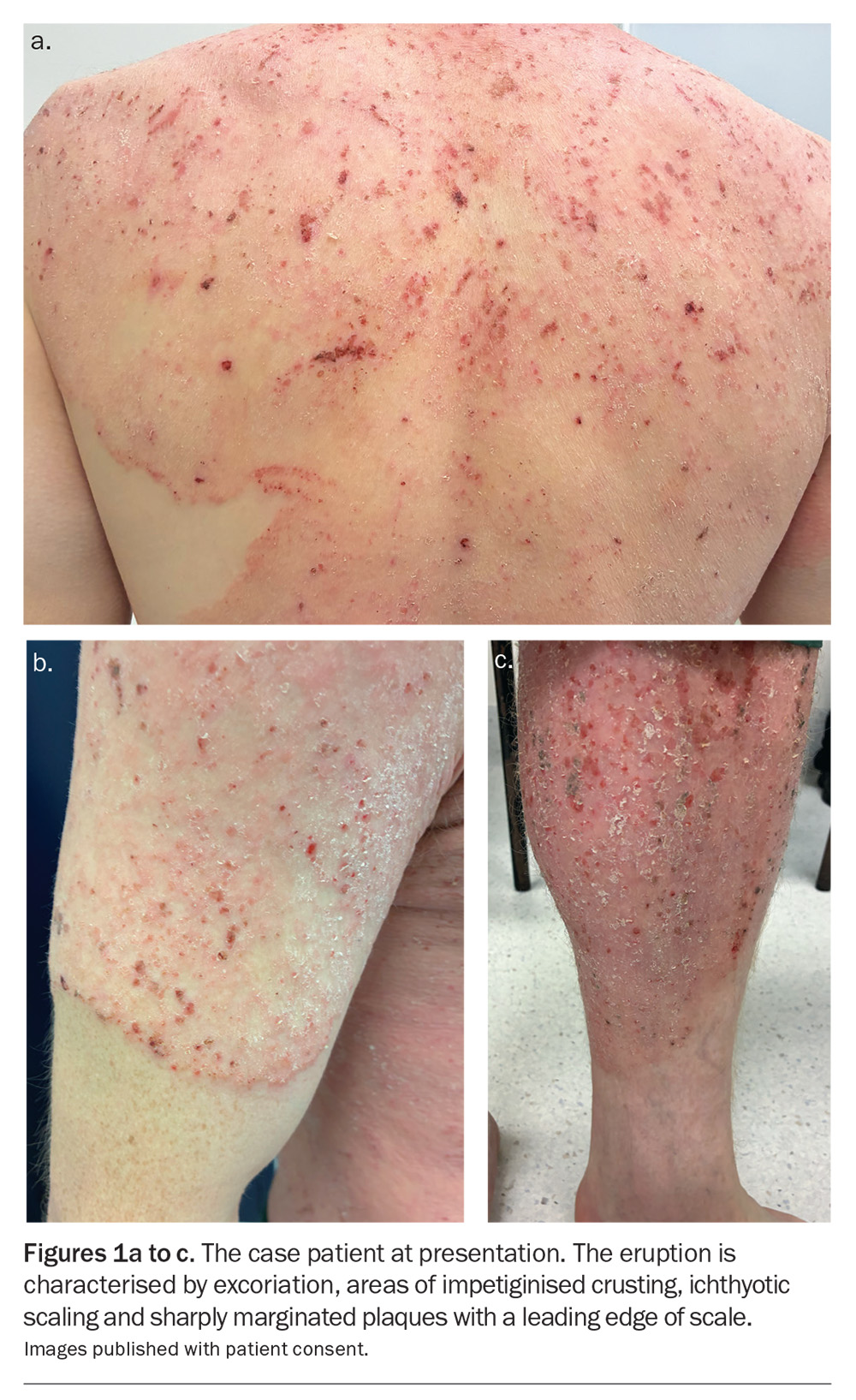
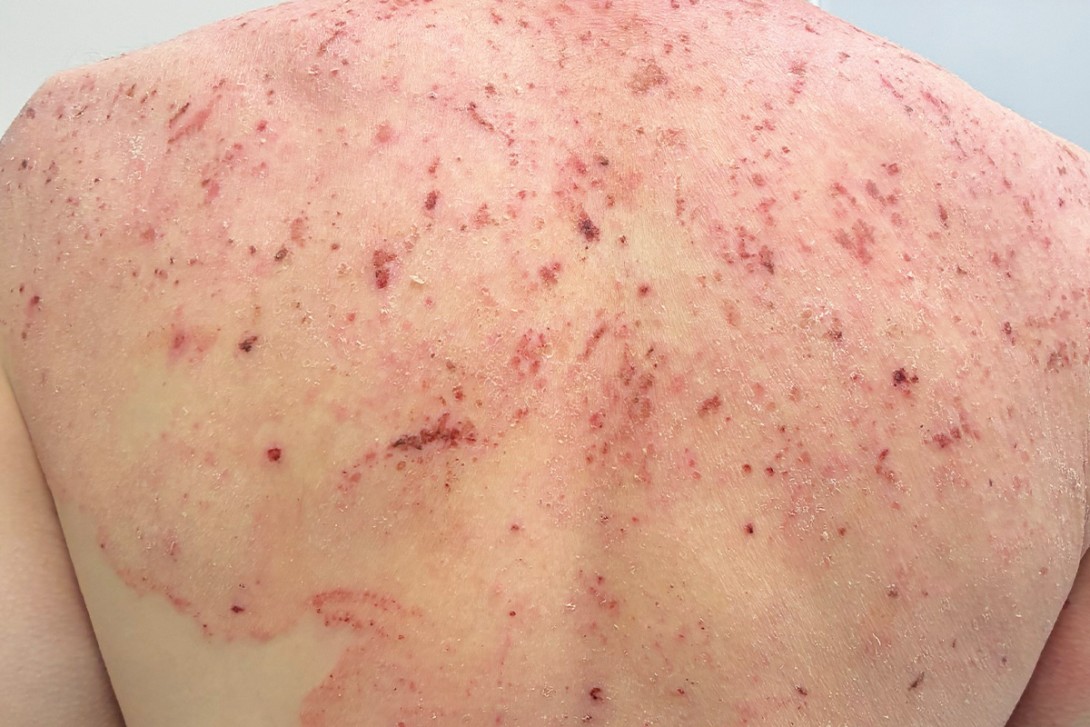
A 37-year-old man presents with a widespread, intensely pruritic eruption (Figures 1a to c). He reports a three-year history of recurrent eruptions, which began as a small patch on the legs that progressively extended. He has been applying calcipotriol 0.005% with betamethasone dipropionate 0.05% ointment daily, along with emollients.
The patient has a history of asthma. His medications include a regular fluticasone propionate and salmeterol combination inhaler, and he uses a salbutamol inhaler and loratadine as needed. He has no reported allergies or hay fever. There is a family history of eczema but no other dermatological conditions.
On examination, an erythematous eruption is observed that involves the patient’s face, trunk and both upper and lower extremities. The eruption is characterised by excoriation, impetiginised crusting, ichthyotic scaling and sharply marginated plaques with a leading edge of scale. The distal skin beyond these lesions appears spared. No nail or mucosal involvement is noted.
Differential diagnoses
Conditions to consider among the differential diagnoses include the following.
Plaque psoriasis
Psoriasis is a chronic immune-mediated inflammatory condition that affects 2 to 3% of the population, most commonly people under the age of 50 years.1 It has a strong genetic predisposition and may be triggered or exacerbated by infections, trauma, stress or medications (e.g. lithium, beta blockers).1,2 The classic form presents as well-demarcated, erythematous plaques covered by silvery-white scale that typically occur on extensor surfaces (elbows, knees), scalp and lower back. Nail involvement (pitting, onycholysis or subungual hyperkeratosis) is common and can serve as a diagnostic clue.1,3 Although lesions are often symmetrical in distribution, there are differences in morphology and distribution among various subtypes (e.g. guttate, flexural, pustular).1 Psoriasis tends to respond to appropriate therapy with potent topical corticosteroids.4
For the case patient, the erythematous and scaly appearance of the lesions meant psoriasis was an important consideration. However, the progression of the rash despite corticosteroid application and its distribution (including sparing of extensor surfaces) made this a less likely diagnosis.
Infected dermatitis
Chronic dermatitis, such as atopic eczema or contact dermatitis, is common and secondary bacterial infection is a frequent complication.5,6 ‘Infected dermatitis’ refers to an eczematous dermatitis complicated by secondary infection, most often by Staphylococcus aureus or Streptococcus pyogenes (impetiginised eczema).5-7 Bacteria colonise the broken skin and can trigger immune reactions that exacerbate inflammation.8
Clinically, infected dermatitis typically presents as an acutely worsening rash characterised by increased erythema, warmth and pain, with pustules or oozing honey-coloured crusts over eczematous patches.8,9 In severe cases, patients may experience systemic symptoms such as fever or malaise.9 It is signs of infection (purulent crusting and tenderness) that make this clinical picture distinct from psoriasis or simple dermatitis.
For the case patient, infected eczematous dermatitis was an important diagnosis to consider because the eruption had features of chronic dermatitis (pruritus, excoriations, erythema and scale) and had responded poorly to topical corticosteroid treatment. However, there were few classic signs of impetiginisation (serous exudate, swelling, honey-yellow crust), and the sharp margins and extensive nature of the eruption also made this diagnosis less likely.
Dermatitis herpetiformis
Dermatitis herpetiformis is a chronic immunobullous dermatosis associated with gluten-sensitive enteropathy. It is a rare autoimmune condition in which IgA antibodies directed against epidermal transglutaminase deposit in the skin.10 Patients classically present with groups of very itchy vesicles and excoriated papules on extensor surfaces, most commonly the elbows, knees and buttocks.10 The rash is usually symmetrically distributed and causes intense pruritus, with patients often showing crusted erosions. Dermatitis herpetiformis tends to affect young and middle-aged adults and is more common in Caucasian men. Patients may have accompanying signs of coeliac disease, such as gastrointestinal symptoms or nutritional deficiencies, although many do not.10,11
The diagnosis of dermatitis herpetiformis is made by skin biopsy with direct immunofluorescence testing that shows granular IgA deposits in dermal papillae. In addition, many patients will have positive serology for coeliac disease (e.g. anti-transglutaminase, antigliadin antibodies, or both).11
The case patient’s rash demonstrated plaques of varying sizes with a truncal distribution, rather than clusters of small vesicles symmetrically distributed on extensor surfaces, which made dermatitis herpetiformis much less likely. In addition, he did not have any history to suggest gluten sensitivity or coeliac disease.
Scabies
Human scabies, an infestation by Sarcoptes scabiei var. hominis, affects over 200 million people worldwide at any one time.12 Patients commonly present with an intensely pruritic, papular eruption, often with excoriations. The lesions are typically small erythematous papules with burrows, invisible to the naked eye, distributed in characteristic areas: interdigital web spaces, wrists, axillae, waist, buttocks and genital regions.13 Infestations that involve the face and scalp are seen in infants or elderly patients but are rare in immunocompetent adults. Outbreaks usually occur in enclosed communities, such as schools or housing units.14 Multiple family members or contacts may have similar itching.
The diagnosis of scabies is based on clinical assessment and laboratory tests. The visual identification of mites, eggs or burrows in skin scrapings or by dermoscopy is confirmatory.14
For the case patient, scabies was an unlikely explanation. The characteristic areas were largely spared, with the eruption involving his face and persisting as a chronic, recurring condition over the past three years. The edge was sharply demarcated, which is less usual in scabies. No mites, eggs or burrows were observed. In addition, his close contacts did not have similar symptoms.
Tinea incognito
This is the correct diagnosis. Tinea incognito is a dermatophyte infection where morphology has been altered by the use of topical or systemic corticosteroids, calcineurin inhibitors or other immunosuppressants.15,16 It is usually caused by Trichophyton rubrum, Trichophyton mentagrophytes or Microsporum canis and less commonly by Epidermophyton floccosum or Trichophyton tonsurans. When these dermatophytes invade keratinised tissues (stratum corneum, hair shafts), they elicit an inflammatory host response that produces the characteristic ring-shaped, scaly, raised border of tinea lesions.17 However, immunosuppression dampens the inflammation, allowing unrestricted fungal growth, leading to loss of the classic annular appearance. The lesions evolve into ill- defined erythematous plaques with minimal scale, serpiginous borders, follicular pustules or even psoriasiform morphology.16-18 The result is an infection that can spread beyond its initial borders and penetrate deeper into hair follicles. When use of the corticosteroid or other immunosuppressant is stopped, the rash rebounds with worsening erythema and extension. This cycle can continue for months, leading to an ‘incognito’ infection that mimics other skin disorders.
The lifetime risk of acquiring tinea corporis is between 10 and 20%, but the true prevalence of tinea incognito is unknown because lesions are frequently mislabelled as eczema or psoriasis.18-20 It affects people of all ages but is most common in children and young adults.
Predisposing factors for dermatophytosis increase the risk of tinea incognito. Immunocompromised patients are more susceptible to both primary fungal infections and atypical presentations under corticosteroid therapy.21 Chronic dermatophyte infections (e.g. untreated tinea pedis or onychomycosis) can act as reservoirs; corticosteroid application to these sites may trigger tinea incognito. Other risk factors include excessive sweating, occlusive clothing and close contact with infected people or animals.22 It is more prevalent in warmer regions with high humidity.19
Diagnosis
Tinea incognito is a great imitator. The borders of the lesions are smudged, scaling is minimal, colour may be less erythematous or hypopigmented and pustules can develop at the periphery.19 Lesions often spread more extensively than in typical tinea corporis and pruritus is variable – often suppressed during corticosteroid use but flaring on withdrawal.19,21 Clues include satellite lesions, ‘ring-within-a-ring’ patterns, concurrent tinea pedis or onychomycosis, as well as signs of prior corticosteroid use such as atrophy, striae or telangiectasia in sites atypical for eczema.17,20 In practice, it is important to be alert to recurrent or chronic rashes treated repeatedly with corticosteroids (especially in atypical locations for eczema) and patient self-medication with corticosteroid creams. Other sites (feet, nails, scalp, groin) should be examined for fungal reservoirs, and epidemiological clues such as exposure to infected pets or household contacts should be considered.20 Recognising these factors can prevent misdiagnosis and inappropriate corticosteroid use.
Although microscopy and culture of skin scrapings, subungual debris, nail clippings or plucked hair was previously the quickest test to confirm the diagnosis, polymerase chain reaction (PCR) tests are surpassing traditional diagnostic methods.23,24 Dermoscopy may reveal peripheral ‘ruff’ or ‘moth-eaten’ scale, comma or corkscrew hairs. Skin biopsy with periodic acid-Schiff staining is reserved for culture-negative cases with high suspicion.20 An empirical antifungal trial can be diagnostic when testing is delayed, but objective confirmation is preferred to guide treatment and prevent mismanagement.17
Management
Management of tinea incognito encompasses two main principles: discontinuing misuse of topical corticosteroids or immuno-suppressants and eliminating the fungus.
Discontinuing corticosteroids or immunosuppressants
Use of the offending corticosteroid or calcineurin inhibitor should be ceased immediately. Patients should be advised not to apply any corticosteroid creams to the area and made aware that lesions might become temporarily more inflamed or pruritic (a rebound effect as the immune response recovers) after stopping a potent corticosteroid. To manage this, adjunctive measures such as cool compresses can be used. Oral antihistamines can provide relief for severe itch without compromising infection clearance.
Eliminating the fungus
The choice between topical and oral antifungal treatment for tinea depends on the site, severity and response to any previous therapy. Topical agents are suitable for early, localised infections affecting the body, face, groin or web spaces between the fingers or toes.22 All sites of infection should be addressed to prevent recurrence.
Topical antifungal therapies
The topical antifungal agents that are used to treat tinea incognito are terbinafine 1% cream and gel and imidazoles (clotrimazole 1%, miconazole 2%, econazole 1%, ketoconazole 2%, bifonazole 1% creams). Terbinafine has fungicidal activity and can often clear dermatophyte infections faster: a 2014 Cochrane review of treatments for tinea corporis and tinea cruris found that both imidazoles and allylamines are effective, with terbinafine achieving slightly higher cure rates and leading to faster symptom resolution in some cases.25 Terbinafine is the preferred topical agent for most superficial fungal infections because of its shorter treatment duration, once-daily application and potential for faster cure as advantages, which can improve patient adherence.22 Terbinafine cream is applied once daily for at least two to four weeks, whereas azoles are usually applied twice daily for four weeks or more.23
Regardless of the topical agent chosen, patients should be advised to apply the cream to the entire lesion and 1 to 2 cm beyond its margin and to continue for at least two weeks beyond visible resolution to ensure full eradication. Longer treatment duration is often needed for patients with tinea incognito. They should also refrain from using corticosteroid–antifungal combination products.23
Oral antifungal therapies
The Australian Therapeutic Guidelines recommend oral therapy for any tinea that has been modified by topical corticosteroid use, as well as for infection that is extensive, persistent despite treatment, recurring quickly or affecting difficult-to-treat areas such as the scalp, palms or soles.22
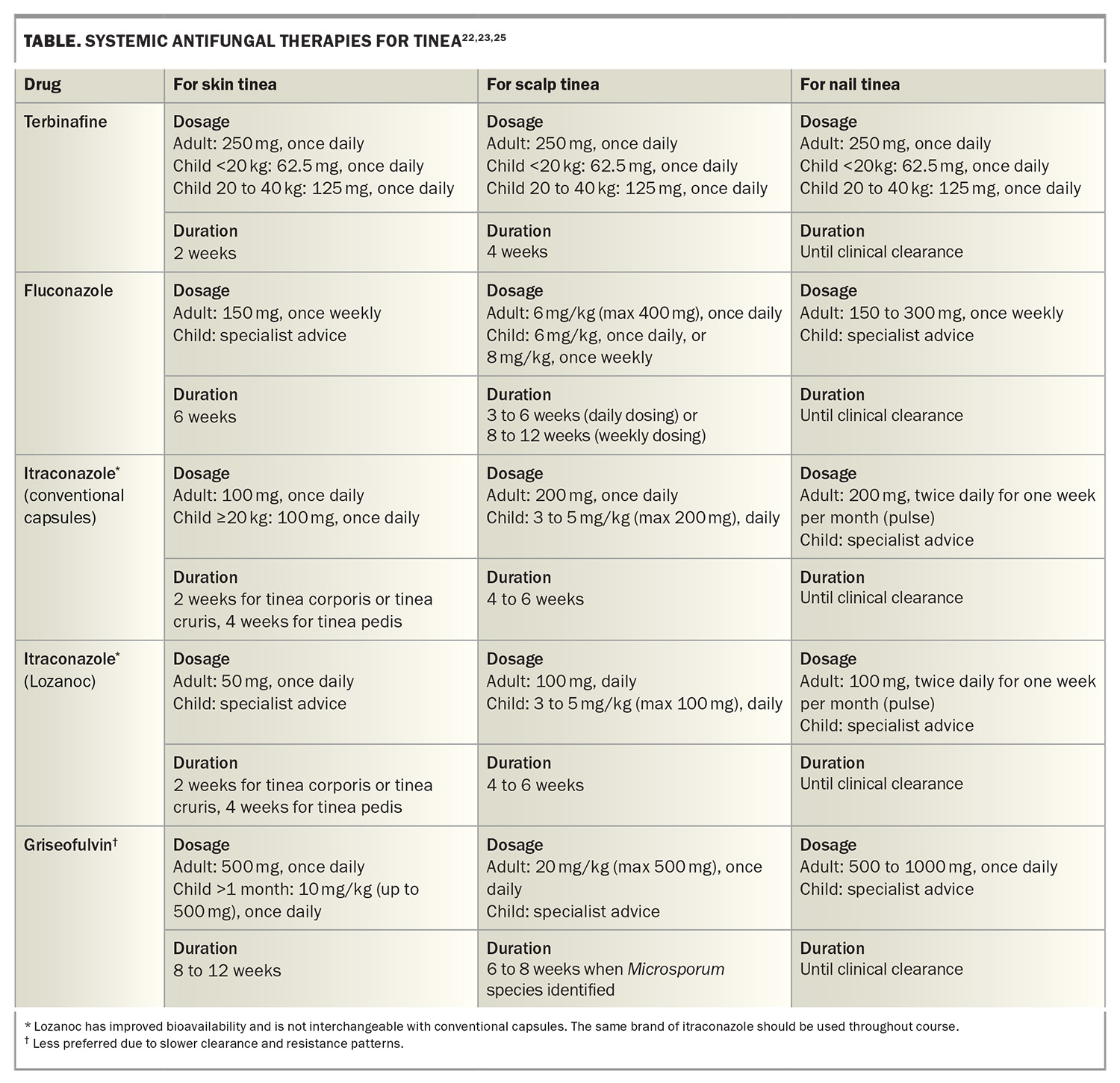
Terbinafine is the first-line oral therapy and is associated with a lower risk of drug interactions compared to azoles (fluconazole and itraconazole) as it causes less inhibition of cytochrome P450 enzymes.22,23 The recurrence rates are similar: 33.3% for terbinafine and 37% for azoles at 10 to 13 months follow up.22 The duration of therapy depends on site – for a typical T. rubrum infection on glabrous skin, a two-week course of terbinafine 250 mg daily is often sufficient (shorter duration than for tinea of nails or hair). Duration for tinea incognito is two to four weeks, with clinical review as a longer treatment course may be required. Use of systemic antifungal therapies is described in the Table.22,23,25
Monitoring requirements differ for oral antifungal therapies. For terbinafine, routine blood monitoring is not required in healthy patients receiving short courses (six weeks or less). Baseline liver function testing and follow-up monitoring should be arranged if treatment is prolonged (over six weeks), if the patient has pre-existing liver disease or risk factors, or if clinical symptoms suggest hepatotoxicity. Itraconazole therapy requires baseline and regular liver function monitoring.22,26 Fluconazole requires baseline liver function testing and a full blood count, but no repeat testing is needed for once-weekly regimens.22,25 Terbinafine should be used cautiously in patients with psoriasis or lupus, as it may exacerbate these conditions.22,23,27 Fluconazole is contraindicated in severe liver disease and may prolong the QT interval, whereas itraconazole should be avoided in heart failure and may be poorly absorbed when coadministered with proton pump inhibitors.22
Other measures
All patients should be educated about general skincare measures to help prevent reinfection, with the affected area kept clean and dry. Emollients can help restore barrier function. Loose, breathable clothing (e.g. cotton fabrics) should be encouraged and occlusive attire or footwear that promotes sweating avoided.
If secondary bacterial infection is present then a topical or systemic antibiotic might be needed, but this is case-dependent. Generally, once antifungal therapy is commenced and the corticosteroid is stopped, the skin will become temporarily more inflamed and then progressively improve as the fungus clears. Follow up should be arranged in two to four weeks to ensure the rash is improving, with reduced scaling and erythema and regression of the edges of the lesion.
Outcome
For the case patient, further investigations were undertaken given the extensive distribution of the eruption and poor response to topical corticosteroid therapy. Fungal scrapings and a 4 mm punch biopsy were performed at the active margin of a lesion on the lower back. The diagnosis of tinea incognito was confirmed by culture of Trichophyton rubrum from the scraping. Histopathology demonstrated intracorneal neutrophilic aggregates, a solitary intracorneal fungal hyphal element, mild acanthosis, spongiosis, and superficial perivascular infiltrates composed of eosinophils and neutrophils. These features were consistent with a psoriasiform tissue reaction pattern (resembling psoriasis), but importantly fungal hyphae were identified within the stratum corneum. A superficial swab taken from a wound on the leg with crusting for bacterial microscopy and culture, grew heavy Staphylococcus aureus and Streptococcus dysgalactiae.
The patient commenced treatment with oral terbinafine 250 mg daily as well as a course of cephalexin (500 mg three times daily for 14 days) for the secondary bacterial infection. He was also advised to use emollients. Hepatic function testing was scheduled for eight weeks.
At review three months later, substantial clinical improvement was observed and the patient reported no adverse effects from terbinafine. Examination revealed marked resolution of the eruption, although residual erythematous patches with lichenification and excoriation persisted, particularly in the popliteal fossae and on the trunk. Onychomycosis of the toenails was noted and xerosis remained pronounced, so he was advised to continue oral terbinafine and the need to apply emollients was frequently emphasised. Repeat blood tests were requested and a further review was scheduled in three months for ongoing monitoring.
Key point
The pathogenesis of tinea incognito involves an iatrogenic factor (corticosteroid or immunomodulator treatment) superimposed on a common fungal infection, creating an altered host-pathogen interaction. This mechanism underscores why proper diagnosis of the initial eruption is crucial, as treating an undiagnosed fungal infection with corticosteroids ‘feeds’ the fungus by disarming the skin’s defences. MT
COMPETING INTERESTS: None.
References
1. Feldman SR. Psoriasis: epidemiology, clinical manifestations, and diagnosis. UpToDate; 2025.
2. Lee EB, Wu KK, Lee MP, Bhutani T, Wu JJ. Psoriasis risk factors and triggers. Cutis 2018; 102 (5S): 18-20.
3. Baran R. The burden of nail psoriasis: an introduction. Dermatology 2010; 221 Suppl 1: 1-5.
4. Therapeutic Guidelines (TG). Psoriasis. Melbourne: TG; 2022. Available online at: https://www.tg.org.au (accessed September 2025).
5. Coulson I. Dermatitis: types and treatment. DermNet; 2022. Available online at: https://dermnetnz.org/topics/dermatitis (accessed September 2025).
6. Stanway A. Complications of atopic dermatitis. DermNet; 2021 [updated by Rashid A, Jarrett P]. Available online at: https://dermnetnz.org/topics/complications-of-atopic-dermatitis (accessed September 2025).
7. Duncan N, Whittaker L. Bacterial skin infections. DermNet; 2023. Available online at: https://dermnetnz.org/topics/bacterial-skin-infections (accessed September 2025).
8. Tamagawa-Mineoka R, Katoh N. Atopic dermatitis: identification and management of complicating factors. Int J Mol Sci 2020; 21: 2671.
9. Wang V, Boguniewicz J, Boguniewicz M, Ong PY. The infectious complications of atopic dermatitis. Ann Allergy Asthma Immunol 2021; 126: 3-12.
10. Reunala T, Hervonen K, Salmi T. Dermatitis herpetiformis: an update on diagnosis and management. Am J Clin Dermatol 2021; 22: 329-338.
11. Caproni M, Antiga E, Melani L, Fabbri P. Guidelines for the diagnosis and treatment of dermatitis herpetiformis. J Eur Acad Dermatol Venereol 2009; 23: 633-638.
12. Karimkhani C, Colombara DV, Drucker AM, et al. The global burden of scabies: a cross-sectional analysis from the Global Burden of Disease Study 2015. Lancet Infect Dis 2017; 17: 1247-1254.
13. Arora P, Rudnicka L, Sar-Pomian M, et al. Scabies: a comprehensive review and current perspectives. Dermatol Ther 2020; 33: e13746.
14. Engelman D, Yoshizumi J, Hay RJ, et al. The 2020 International Alliance for the Control of Scabies consensus criteria for the diagnosis of scabies. Br J Dermatol 2020; 183: 808-820.
15. Meena S, Gupta LK, Khare AK, et al. Topical corticosteroids abuse: a clinical study of cutaneous adverse effects. Indian J Dermatol 2017; 62: 675.
16. Ive FA, Marks R. Tinea incognito. Br Med J 1968; 3: 149-152.
17. Goldstein A. Goldstein GB. Dermatophyte (tinea) infections. UpToDate; 2025.
18. Holubar K, Male O. Tinea incognita vs. tinea incognito. Acta Dermatovenerol Croat 2002; 10: 39.
19. Zacharopoulou A, Tsiogka A, Tsimpidakis A, Lamia A, Koumaki D, Gregoriou S. Tinea incognito: challenges in diagnosis and management. J Clin Med 2024; 13: 3267.
20. Leung AK, Lam JM, Leong KF, Hon KL. Tinea corporis: an updated review. Drugs Context 2020; 9: 2020-5-6.
21. Oakley A. Tinea incognito. DermNet; 2017 [updated by Gomez J]. Available online at: https://dermnetnz.org/topics/tinea-incognito (accessed September 2025).
22. Kovitwanichkanont T, Chong A. Superficial fungal infections. Aust J Gen Pract 2019; 48: 706-711.
23. Therapeutic Guidelines. Infected skin: tinea. Melbourne: TG; 2022. Available online at: https://www.tg.org.au (accessed September 2025).
24. Jeon JJ, Kim YH, Lim SH, Lee S, Choi EH. Comparing the diagnostic accuracy of PCR-reverse blot hybridization assay and conventional fungus study in superficial fungal infection of the skin: a systematic review. Mycoses 2024; 67: e13678.
25. El-Gohary M, van Zuuren EJ, Fedorowicz Z, et al. Topical antifungal treatments for tinea cruris and tinea corporis. Cochrane Database Syst Rev 2014; (8): CD009992.
26. Gupta AK, Chwetzoff E, Del Rosso J, Baran R. Hepatic safety of itraconazole. J Cutan Med Surg 2002; 6: 210-213.
27. Stolmeier DA, Stratman HB, McIntee TJ, Stratman EJ. Utility of laboratory test result monitoring in patients taking oral terbinafine or griseofulvin for dermatophyte infections. JAMA Dermatol 2018; 154: 1409-1416.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.