Lipid-lowering therapy after acute coronary syndrome: new developments
Lowering LDL-cholesterol levels early after an acute coronary syndrome (ACS) is crucial to reducing recurrent cardiovascular events. Combination oral therapies and proprotein convertase subtilisin/kexin type 9 inhibitors have emerged as important additions to lipid-lowering therapies, particularly for high-risk patients. This article covers current guidelines and PBS criteria and includes a case study of lipid-lowering therapies post-ACS.
Correction
A correction for this article appears in the August 2025 issue of Medicine Today and is available here. The online version and the full text PDF of this article (see link above) have been corrected.
- Achieving LDL-cholesterol (LDL-C) targets following an acute coronary syndrome (ACS) is a significant challenge, necessitating early and aggressive lipid-lowering strategies to reduce cardiovascular disease risk.
- High-dose statin therapy is recommended as first-line treatment post-ACS, with early in-hospital initiation improving outcomes.
- Only 18 to 20% of people achieve LDL-C targets on statins alone, therefore, a multidrug regimen is also needed; ezetimibe combined with statins reduced LDL-C levels in multiple studies.
- Icosapent ethyl is a new option for patients with well- controlled LDL-C levels while on statins but with elevated triglycerides, offering cardiovascular benefits.
- Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors (such as evolocumab and inclisiran) have emerged as an important addition to lipid-lowering therapies, although evidence for use in acute settings remains incomplete.
- Current consensus supports a sequential approach to lipid-lowering therapies post-ACS, prioritising statins and adding oral therapies before proceeding to PCSK9 inhibitors, with ongoing research likely to refine future recommendations.
Despite currently available therapies, achieving target LDL-cholesterol (LDL-C) levels after an acute coronary syndrome (ACS) remains a significant challenge. Early and aggressive LDL-C management is associated with greater reductions in cardiovascular disease risk.3 Combination oral therapies and agents targeting proprotein convertase subtilisin/kexin type 9 (PCSK9) have emerged as potent additions to lipid-lowering therapies in ACS, particularly in patients who have not adequately met treatment targets.
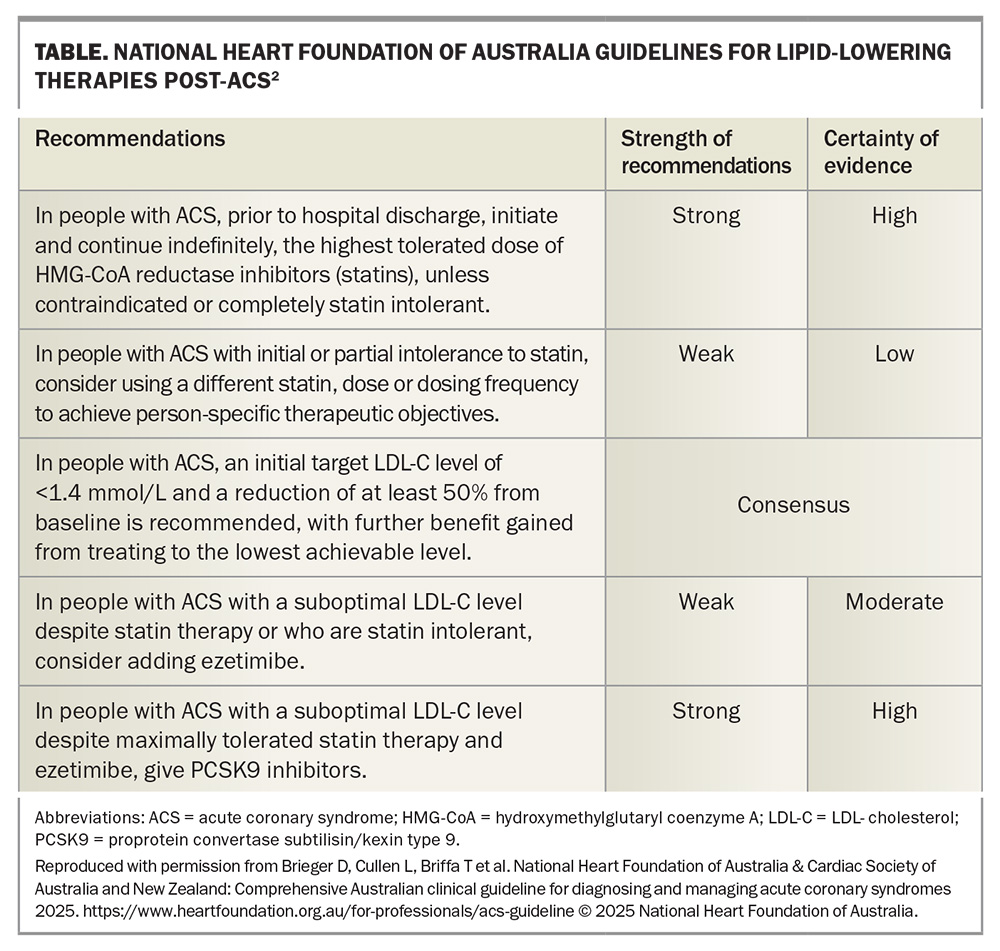
The latest National Heart Foundation of Australia guidelines on management following ACS place a strong focus on initiating an early therapeutic regimen that will aggressively address lipid levels, aiming for a 50% reduction in LDL-C from baseline and a level ideally below 1.4 mmol/L.2 A high-dose statin is recommended as first-line therapy, with evident benefits in the short term, alongside persistence of these benefits with time. Early initiation of high-dose statin therapy during hospital admission is recommended, irrespective of the baseline lipid profile. Routine monitoring of the lipid profile should be carried out four to eight weeks post-initiation.3 In practice, lipid profiles can subsequently be monitored every six to 12 months after an initial coronary event. A case study of lipid-lowering therapies post-ACS is presented in Box 1.
In men under 55 years of age and women under 60 years of age, the need for further genetic testing for familial hypercholesterolaemias can be stratified via the Dutch Lipid Clinic Network Score. Confirmation of a genetic predisposition can subsequently guide the need for genetic counselling and early statin use among family members.2
Ezetimibe combined with statin therapy
More often than not, patients require a multidrug regimen to meaningfully establish adequate lipid control, with only 18 to 20% of patients reaching the desired LDL-C targets on statins alone.4 Statin intolerance has also emerged as an issue among a sizeable proportion of patients. This has prompted cardiologists to explore additional therapies in the post-ACS setting. The addition of second-line agents such as ezetimibe is now commonly considered in these cases. The evidence supporting this approach was provided by the Improved Reduction of Outcomes: Vytorin Efficacy International Trial (IMPROVE-IT). This randomised controlled trial recruited patients within 10 days of an ACS and demonstrated that ezetimibe combined with a statin reduced LDL-C levels by 24% and decreased the absolute incidence of nonfatal cardiovascular events by 2% over prolonged follow-up, compared with a statin alone.5 Although the magnitude of benefit in this trial was small, subsequent observational analyses have suggested that the benefit may be greater in the real-world setting, including a recent analysis of the Swedish Web-System for Enhancement and Development of Evidence-Based Care in Heart Disease Evaluated According to Recommended Therapies (SWEDEHEART) registry.
The SWEDEHEART registry examined cardiovascular outcomes among ACS patients concurrently treated with ezetimibe and a statin. Among 35,826 patients, groups were categorised based on the timing of ezetimibe initiation: early ezetimibe (within one month of discharge), late ezetimibe or no ezetimibe. A higher proportion of patients in the early ezetimibe group (38.4%) achieved desired LDL-C targets of less than 1.4 mmol/L, compared with those in the late ezetimibe and no ezetimibe groups (28.8% and 25.3%, respectively). Furthermore, over a median follow-up time of 3.96 years, the risk of cardiovascular death was higher in patients receiving late or no ezetimibe therapy (hazard ratio 1.64, 95% confidence interval [CI], 1.15–2.63; hazard ratio 1.83, 95% CI, 1.35–2.69, respectively) compared with patients receiving early additional therapy.6
The availability of ezetimibe as a fixed-dose combination with statins such as atorvastatin and rosuvastatin renders it an attractive therapeutic option, particularly as it can simultaneously address the core issues of suboptimal lipid control and nonadherence.5,7 Although the IMPROVE-IT trial did not commence ezetimibe in hospital, predischarge initiation of this combination therapy is increasingly observed in clinical practice, particularly in patients who have an ACS event while receiving lipid-lowering therapy. This practice is also encouraged in international guidelines. In the Australian setting, use of ezetimibe is facilitated by relaxation of the PBS criteria, such that an authority approval process is no longer required for its subsidised prescription.4
Icosapent ethyl
Icosapent ethyl is a recently available drug for patients with cardiovascular disease and associated hypertriglyceridaemia. In clinical trials, use of icosapent ethyl resulted in a 25% relative improvement in the incidence of cardiovascular outcomes in patients with controlled LDL-C levels while on a statin but with persistently elevated triglyceride levels. Therefore, initiation of icosapent ethyl should be considered if triglyceride levels remain elevated despite optimisation of LDL lowering therapy.8 Of note, use of icosapent ethyl was associated with a significant 1% absolute increase in hospital admissions for atrial fibrillation and a trend towards an increased rate of serious bleeding (p = 0.6).
Bempedoic acid
Bempedoic acid is a potent inhibitor within the cholesterol synthesis pathway. Its effect on LDL lowering is comparable to that of statin therapy.9 Among statin-intolerant patients, bempedoic acid was shown to produce a statistically significant relative reduction of about 13% in major adverse cardiovascular events (a composite of cardiovascular death, myocardial infarction, cerebrovascular events and coronary revascularisation) compared with placebo.10 Although bempedoic acid is not yet available for use in Australia, it has been endorsed by international guidelines as a valuable therapeutic option, particularly in patients with statin intolerance.4
Proprotein convertase subtilisin/kexin type 9
PCSK9 is a protein that causes the breakdown of hepatic LDL receptors that facilitate LDL-C elimination. In the clinical context, gain-of-function mutations of PCSK9 can result in the genetic entity of autosomal dominant familial hypercholesterolaemia.11 Conversely, loss-of-function mutations are associated with reductions in LDL-C levels. Thus, pharmacological inhibition of this molecule creates an opportunity for more effective systemic LDL-C clearance. This principle has been a focal point of research, which has resulted in the advent of PCSK9 inhibitors in the realm of ACS treatment. Although PCSK9 inhibition is now established as a cornerstone of secondary prevention in ACS guidelines, its role as a first-line therapy remains less clearly defined.
Evolocumab, inclisiran and alirocumab
In Australia, two PCSK9-targeted therapies (evolocumab and inclisiran) are currently PBS subsidised for the management of familial and non-familial hypercholesterolaemia. A third agent, alirocumab, was previously available. Both evolcumab and inclisiran are administered via subcutaneous injections using prefilled, disposable pen devices. Their dosing regimens are designed to improve adherence by reducing the frequency of administration. Inclisiran is given at baseline and again at three months, followed by six-monthly maintenance doses. Evolocumab, in its currently available formulation, is administered every two weeks, with no subsequent dose reduction. A steady-state concentration is usually achieved for evolocumab after about three months of therapy.12
Reduction of LDL-cholesterol levels
The landmark trial on Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER) showed a 59% reduction in LDL-C levels with evolocumab combined with statin therapy compared with statin therapy alone. Inclisiran shares similar properties to evolocumab and alirocumab in reducing LDL-C levels in patients with cardiovascular disease. The ORION-11 trial focused on a population cohort with atherosclerotic heart disease with persistently elevated LDL-C levels despite maximal statin therapy. In comparison with placebo, it was found that inclisiran reduced LDL-C levels by about 50% (95% CI, 46.6–53.1) over a follow-up period of 18 months.13 A meta-analysis found inclisiran to be therapeutically equivalent to evolocumab and alirocumab in terms of LDL-C reduction.14 However, the impact of inclisiran on long-term cardiovascular outcomes remains a focus of ongoing research.4
In the acute setting, several small studies have shown that early introduction of PCSK9 inhibitors can result in dramatic reduction of LDL-C levels. The Evolocumab for Early Reduction of LDL Cholesterol Levels in Patients With Acute Coronary Syndromes (EVOPACS) study (n = 308) found that 95.7% of patients receiving evolocumab and atorvastatin achieved target LDL-C levels of less than 1.8 mmol/L by eight weeks, compared with 37.6% of patients in the of atorvastatin-only arm.15
Impact on atheroma burden
PCSK9 inhibitors have shown potential to reduce atheroma burden in patients treated following an ACS. In the Effects of the PCSK9 Antibody Alirocumab on Coronary Atherosclerosis in Patients With Acute Myocardial Infarction (PACMAN-AMI) trial of patients with acute myocardial infarction, alirocumab added to high-intensity statin therapy resulted in greater coronary plaque regression in noninfarct-related arteries after 52 weeks, as measured by intravascular ultrasound.16 This difference was quantitatively small, with a percent atheroma volume reduction of about 1%. However, the more recent High-Resolution Assessment of Coronary Plaques in a Global Evolocumab Randomized Study (HUYGENS) demonstrated that the early addition of evolocumab on top of a high-intensity statin in patients with non-ST-elevation myocardial infarction led to a reduction of plaque high-risk features. This study used optical coherence tomography, a better tool than intravascular ultrasound for plaque phenotypic analysis.17
Impact on major coronary events
Current literature for PCSK9 inhibitors have shown promising benefits in the management of patients with coronary artery disease. The FOURIER trial, which evaluated evolocumab in combination with statin therapy, demonstrated a 15% relative risk reduction in the primary composite endpoint of cardiovascular death, myocardial infarction, stroke, hospitalisation for unstable angina or coronary revascularisation compared with statin therapy alone. This was primarily driven by nonfatal events.
Although most participants had a history of a previous ischaemic coronary event, those with a myocardial infarction within the previous four weeks were excluded, and the majority of the patients in this study were at least three years post-infarct (median 3.4 years in the evolocumab group and median 3.3 years in the placebo group).18
The FOURIER Open-Label Extension (FOURIER-OLE) study reaffirmed a similar extent of benefits over a follow-up period of 8.4 years. Compared with placebo, the use of evolocumab was not associated with an increase in the incidence of myalgias, new-onset diabetes, or cerebrovascular and neurocognitive events over the follow-up period.19
The Evaluation of Cardiovascular Outcomes After an Acute Coronary Syndrome During Treatment With Alirocumab (ODYSSEY OUTCOMES) trial investigated alirocumab, a PCSK9 inhibitor that is no longer available in Australia, and showed a 15% reduction in major cardiovascular events in those who had experienced an ACS in the past one to 12 months.20
Early benefits of proprotein convertase subtilisin/kexin type 9 inhibitors
Although evidence supports the early benefits of PCSK9 inhibitors, this evidence is currently limited and not sufficient to recommend routine use of these therapies in the acute setting.21 Adequately powered studies showing the impact of early initiation of PCSK9 inhibitors on clinical outcomes are awaited. Most consensus guidelines continue to advise sequential therapy for lipid management, with a focus on maximising statin dosage and adding oral therapies such as ezetimibe before proceeding to PCSK9 therapy (Table).2
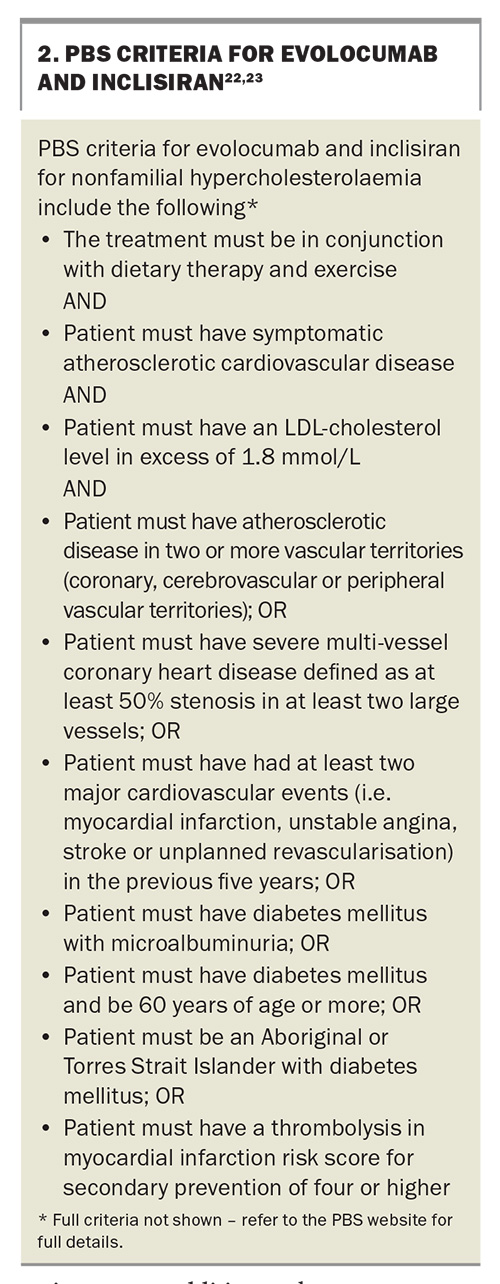
PCSK9 inhibitors are strongly recommended in patients who are intolerant to statin therapy or have persistently elevated lipid profiles despite being on maximally tolerated statin therapy and ezetimibe.3,4 In the Australian context, evolocumab and inclisiran are PBS subsidised for patients with established coronary disease, albeit with stringent criteria (Box 2).22,23
Conclusion
There is general recognition that the greater the reduction in LDL-C levels and the earlier it is achieved, the better the outcomes following an ACS. This is currently achieved by early initiation of high-dose statin therapy, with an increasing trend to commence ezetimibe in hospital in patients who have had cardiovascular events while on statin therapy. PCSK9 inhibitors have emerged as an important addition to the current armamentarium of lipid-lowering therapies. However, based on existing data, initiation of PCSK9 inhibitors at the time of ACS is not currently recommended, except in patients with proven statin intolerance who meet clinical and LDL-C criteria for PBS subsidy. This clinical space remains dynamic and certain to evolve as ongoing trials further strengthen the evidence base for PCSK9 inhibitor use in this setting. MT
COMPETING INTERESTS: None.
References
1. NPS MedicineWise. Statin-associated muscle symptoms (SAMS). Sydney: NPS MedicineWise; 2017. Available online at: https://www.nps.org.au/professionals/managing-lipids/statin-associated-muscle-symptoms-sams (accessed June 2025).
2. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand. Australian clinical guideline for diagnosing and managing acute coronary syndromes 2025. Canberra: National Heart Foundation of Australia; 2025. Available online at: https://www.heartfoundation.org.au/for-professionals/acs-guideline (accessed June 2025).
3. Rao SV, O’Donoghue ML, Ruel M, et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI guideline for the management of patients with acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2025; 151: e771-e862.
4. Banach M, Reiner Ž, Surma S, et al. 2024 Recommendations on the optimal use of lipid-lowering therapy in established atherosclerotic cardiovascular disease and following acute coronary syndromes: a position paper of the international lipid expert panel (ILEP). Drugs 2024; 84: 1541-1577.
5. Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 2015; 372: 2387-2397.
6. Leosdottir M, Schubert J, Brandts J, et al. Early ezetimibe initiation after myocardial infarction protects against later cardiovascular outcomes in the SWEDEHEART Registry. J Am Coll Cardiol 2025; 85: 1550-1564.
7. Chow CK, Archer O, Kritharides L. Should patients with myocardial infarction be started on combination therapy with ezetimibe before hospital discharge? J Am Coll Cardiol 2025; 85: 1565-1567.
8. Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med 2019; 380: 11-22.
9. Lincoff AM, Ray KK, Sasiela WJ, et al. Comparative cardiovascular benefits of bempedoic acid and statin drugs. J Am Coll Cardiol 2024; 84: 152-162.
10. Nissen SE, Lincoff AM, Brennan D, et al. Bempedoic acid and cardiovascular outcomes in statin-intolerant patients. N Engl J Med 2023; 388: 1353-1364.
11. Xia X, Peng Z, Gu H, et al. Regulation of PCSK9 expression and function: mechanisms and therapeutic implications. Front Cardiovasc Med 2021; 8: 764038.
12. Australian Prescriber. New drugs: Evolocumab. Australian Prescriber 2016; 39: 180-182.
13. Ray KK, Wright RS, Kallend D, et al. Two phase 3 trials of inclisiran in patients with elevated LDL cholesterol. N Engl J Med 2020; 382: 1507-1519.
14. Burnett H, Fahrbach K, Cichewicz A, et al. Comparative efficacy of non-statin lipid-lowering therapies in patients with hypercholesterolemia at increased cardiovascular risk: a network meta-analysis. Curr Med Res Opin 2022; 38: 777-784.
15. Koskinas KC, Windecker S, Pedrazzini G, et al. Evolocumab for early reduction of LDL cholesterol levels in patients with acute coronary syndromes (EVOPACS). J Am Coll Cardiol 2019; 74: 2452-2462.
16. Raber L, Ueki Y, Otsuka T, et al. Effect of alirocumab added to high-intensity statin therapy on coronary atherosclerosis in patients with acute myocardial infarction: the PACMAN-AMI randomized clinical trial. JAMA 2022; 327: 1771-1781.
17. Zanda G, Varbella F. Stabilization of vulnerable plaque in the ACS patient: evidence from HUYGENS studies. Eur Heart J Suppl 2023; 25: C106-C108.
18. Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 2017; 376: 1713-1722.
19. O’Donoghue ML, Giugliano RP, Wiviott SD, et al. Long-term evolocumab in patients with established atherosclerotic cardiovascular disease. Circulation 2022; 146: 1109-1119.
20. Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med 2018; 379: 2097-2107.
21. Hao Y, Yang Y-L, Wang Y-C, et al. Effect of the early application of evolocumab on blood lipid profile and cardiovascular prognosis in patients with extremely high-risk acute coronary syndrome. Int Heart J 2022; 63: 669-677.
22. Pharmaceutical Benefits Scheme. Evolocumab. Canberra: Australian Government Department of Health Disability and Ageing; 2025. Available online at: https://www.pbs.gov.au/medicine/item/10958R-11484K-11977J-11985T (accessed June 2025).
23. Pharmaceutical Benefits Scheme. Inclisiran. Canberra: Australian Government Department of Health Disability and Ageing; 2025. Available online at: https://www.pbs.gov.au/medicine/item/14087K-14101E (accessed June 2025).