Glaucoma eye drops make you cry or dry – and here’s why

Glaucoma treatment can lead to ocular surface disease, a condition that can affect treatment adherence and worsen outcomes. This article discusses identification, diagnosis and treatment of ocular surface disease, as well as advanced glaucoma management. It highlights the importance of effective collaboration between GPs, optometrists, ophthalmologists and pharmacists to protect patients’ vision and support long-term management.
- Glaucoma requires long-term medical treatment (topical eye drops) to control intraocular pressure and prevent vision loss, but this treatment can lead to or exacerbate ocular surface disease (OSD) in up to 60% of patients, affecting quality of life and treatment adherence.
- GPs play an essential role in identifying, managing and referring patients with glaucoma eye drop-related OSD, especially between ophthalmology reviews.
- The symptoms of OSD (e.g. eye redness, irritation, watering or dry eye) closely resemble those of allergic reactions, making accurate differentiation essential.
- Stepwise treatment for OSD includes preservative-free lubricants, glaucoma eye drops without preservatives, ocular surface immunomodulating agents, steroid eye drops, punctal plugs, blepharitis and meibomian gland dysfunction treatment, glaucoma laser and intraocular surgery.
- Traditional glaucoma filtration surgical options are available for advanced glaucoma management, including trabeculectomy and tube shunt surgery.
- Collaborative care between GPs, optometrists, ophthalmologists and pharmacists is crucial for accurate prescribing, early intervention, improved treatment adherence and prevention of vision loss in glaucoma patients.
Glaucoma is a chronic eye disease that requires long-term management with medications, primarily eye drops, to control intraocular pressure (IOP) and prevent vision loss. However, these medications can lead to or exacerbate ocular surface disease (OSD) in up to 60% of patients.1
Ocular surface disease
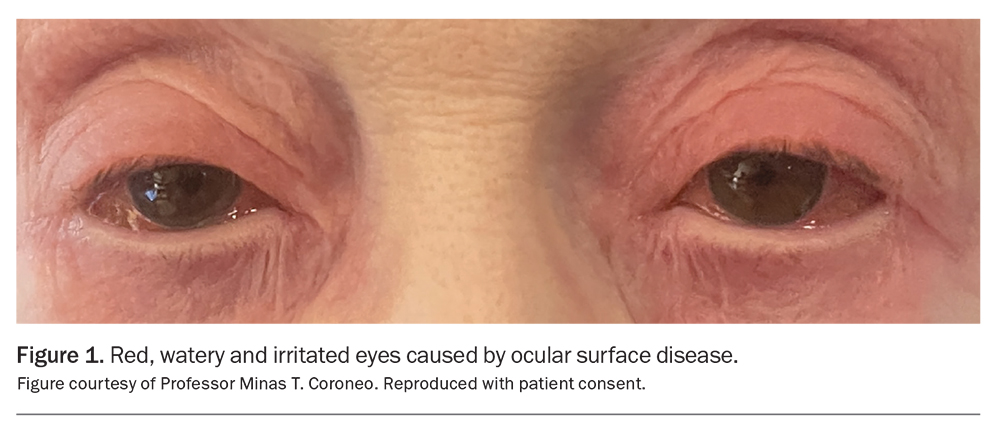
The major impacts of OSD are reduced quality of life, which is well documented in several studies and, most devastatingly, poor adherence to treatment.2,3 Unfortunately, most ophthalmologists have encountered a patient who, after being lost to glaucoma follow up for years, returns with central vision-threatening visual field loss. This is often attributable to discontinuation of eye drops resulting from side effects such as ocular irritation, redness, tearing, dry eye or a decline in visual acuity and quality of vision (Figure 1). Managing an ‘invisible’ disease with an uncomfortable treatment regimen remains a major challenge in glaucoma care, despite all the advances that have occurred in this field.
GPs are often the doctors prescribing medications for repeat prescription in between ophthalmology reviews, so they are well placed to diagnose and differentiate eye drop-related OSD from other common ocular irritations and infections. They can commence treatment or refer back to the patient’s ophthalmologist for an earlier reassessment of the ocular surface and glaucoma management.
Identification
As GPs play a critical role in the ongoing management of patients with glaucoma, recognising medication-induced OSD and distinguishing it from other conditions is crucial.2,3 Early diagnosis and appropriate management can significantly improve patient outcomes and quality of life.
Impact on quality of life
OSD can severely affect a patient’s quality of life and their adherence to glaucoma treatment. Symptoms such as redness, irritation, watering eyes or dry eye can deter patients from regular use of treatment, leading to disease progression and vision loss.4
Pathogenesis
The development of OSD from glaucoma eye drops is a vicious cycle. The disruption of the tear film stability and ocular surface irritation triggers the release of inflammatory cytokines and mediators. This inflammation reduces tear production and alters the tear film quality, perpetuating the cycle of irritation and instability. Chronic inflammation can cause ocular surface, scleral and episcleral scarring, reducing aqueous drainage and impacting IOP.5 Several pre-existing patient factors can predispose individuals to developing OSD.6-10 These include but are not limited to those outlined in Box 1.
Patients can develop sensitivity or allergies to eye drops at any point, even after long-term use. Certain eye drops, such as brimonidine, are more likely to cause allergic reactions, but any eye drops can potentially trigger sensitivity. The most common cause of allergic reactions is the preservative benzalkonium chloride, which is used in most formulations.
Symptoms of sensitivity or allergy can mimic OSD, making it crucial to differentiate between the two. The Table lists some nuances that can help with differentiating between these; however, the biggest giveaway lies under the lids.11
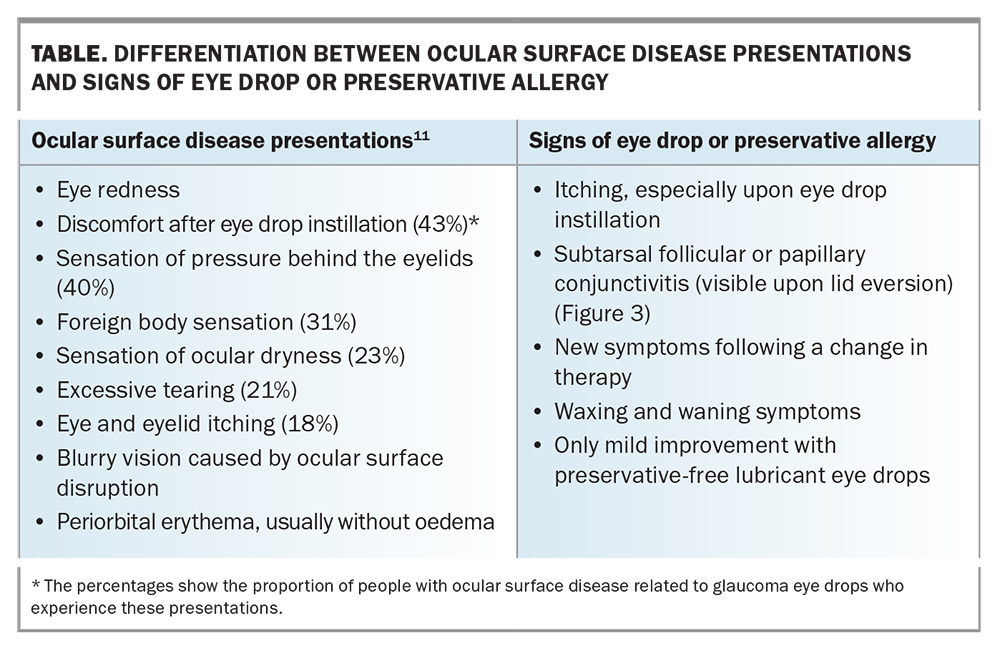
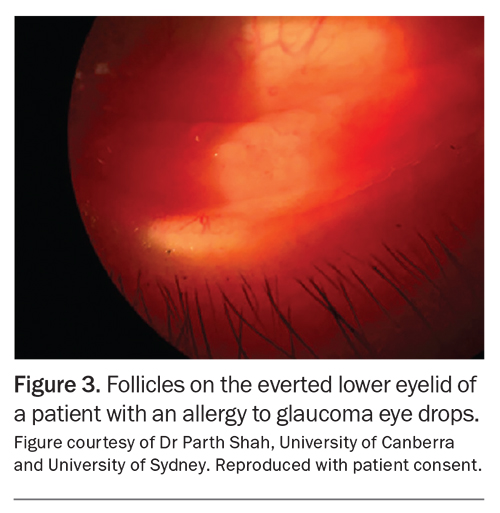
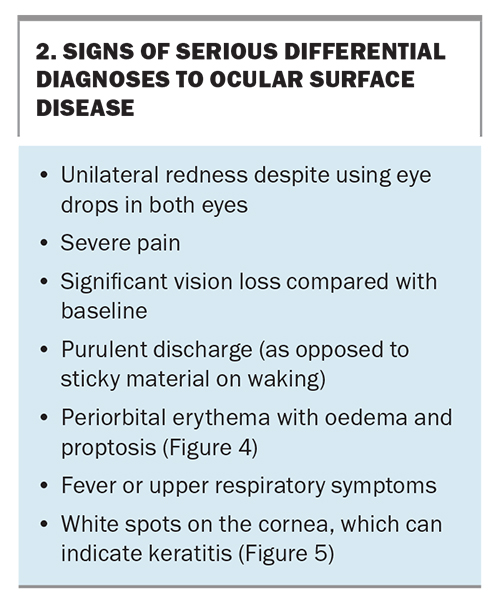
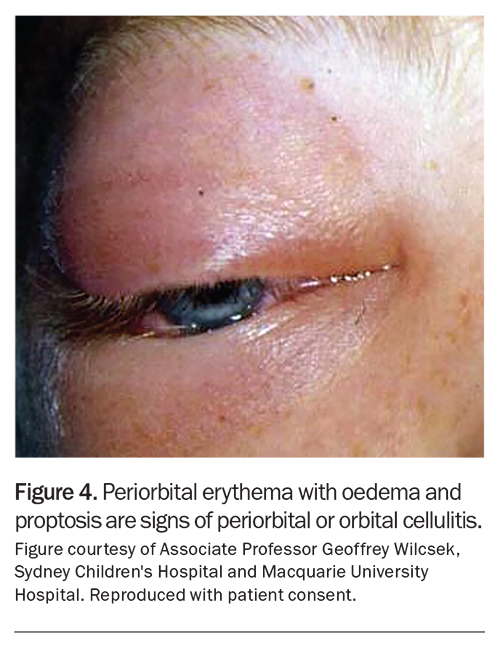
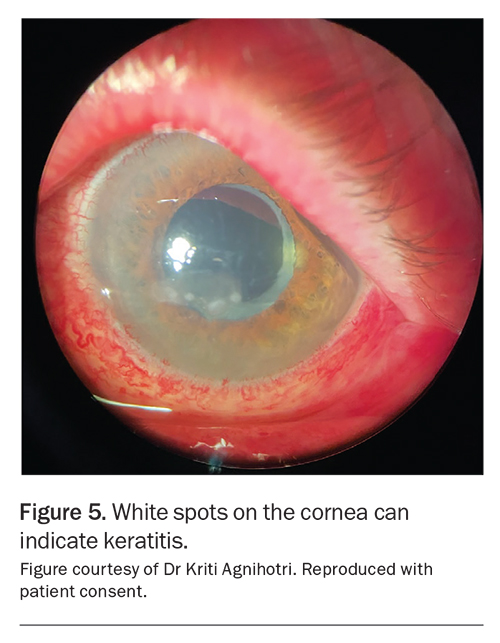
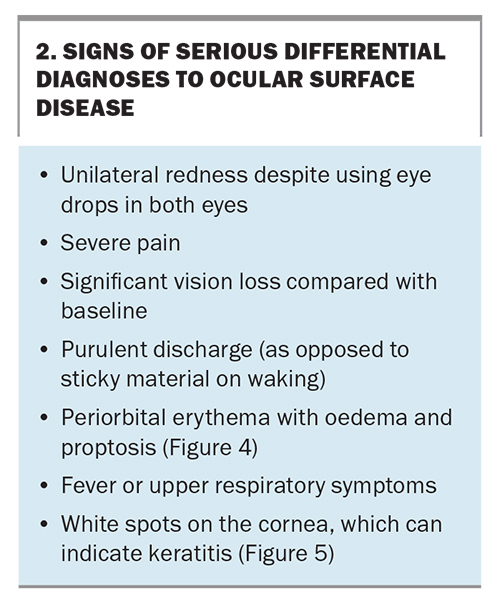
Eyelid eversion (Figure 2) is an easy, noninvasive and important skill for all doctors to master to check for papillae or follicles (Figure 3). Eyelid eversion is also essential for detecting foreign bodies. In papillary conjunctivitis, the conjunctival epithelium covers flat-topped or cobblestone-appearing raised areas with blood vessels. In follicular conjunctivitis, the conjunctival epithelium covers lymphoid follicles, which have a paler centre and darker surrounding area. The presence of papillary or follicular conjunctivitis can suggest that there is an allergic component to the patient’s presentation. Certain symptoms suggest a condition more serious than OSD, warranting urgent review (Box 2). These include periorbital erythema with oedema and proptosis (Figure 4) and white spots on the cornea (Figure 5).
Approach to diagnosis
History and examination
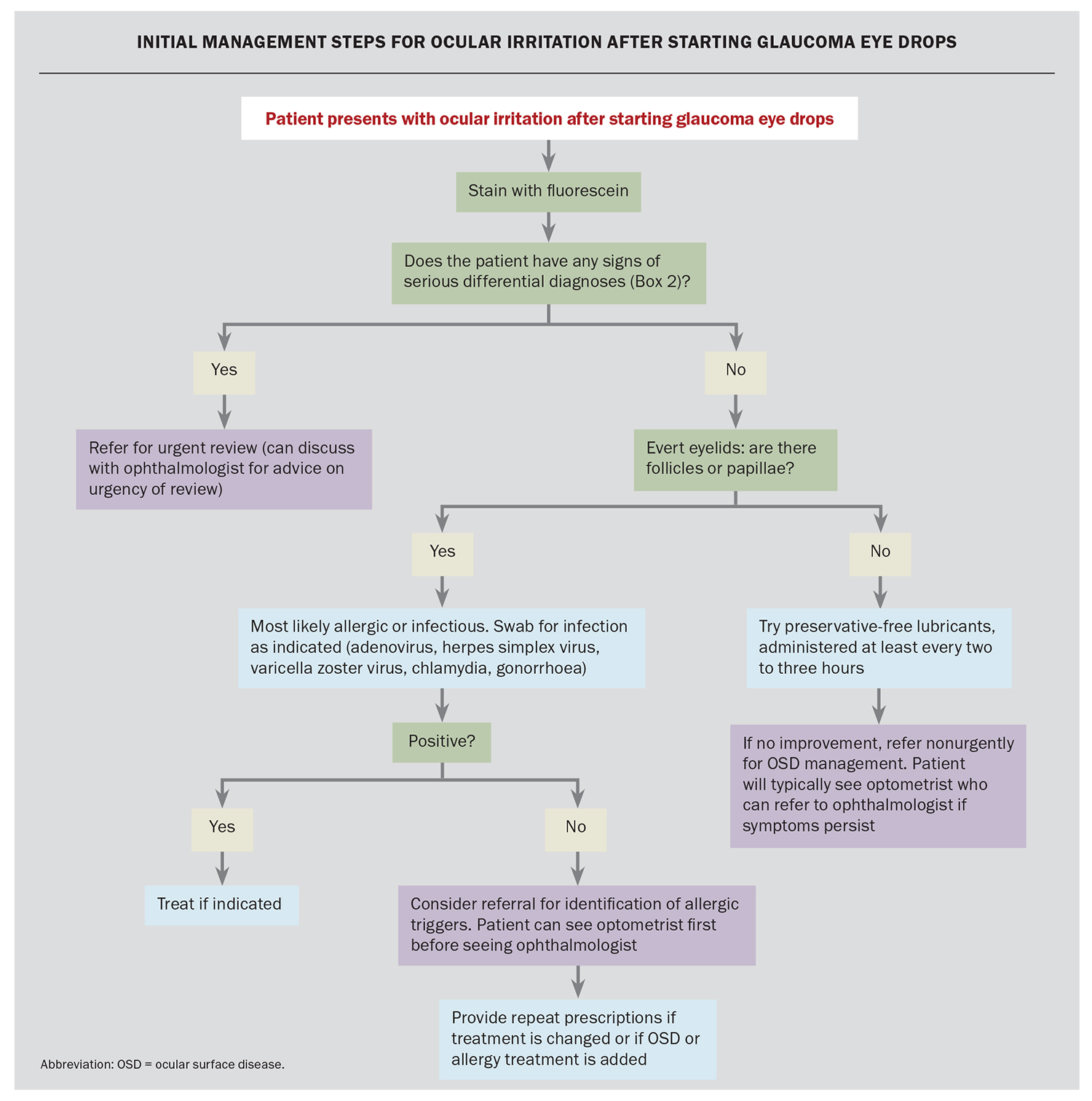
A detailed history and examination, focusing on the signs and symptoms mentioned above, are the first steps in the workup of a patient with suspected OSD. Consideration should be given to any concurrent systemic conditions that may worsen OSD, such as autoimmune diseases, systemic medications or allergies. Initial management steps for ocular irritation after starting glaucoma eye drops are shown in the Flowchart.
Testing
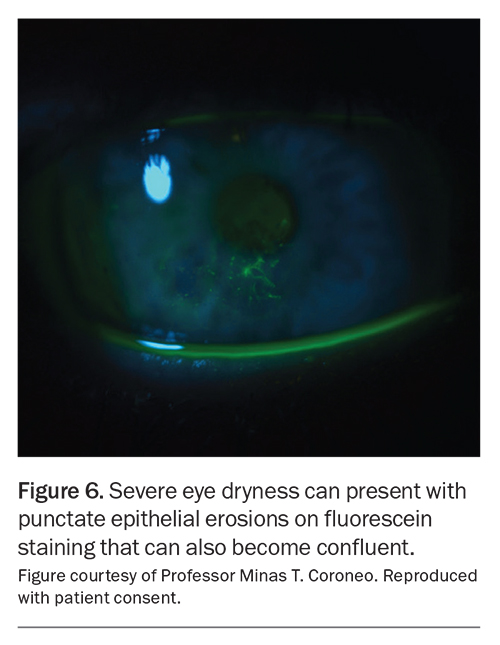
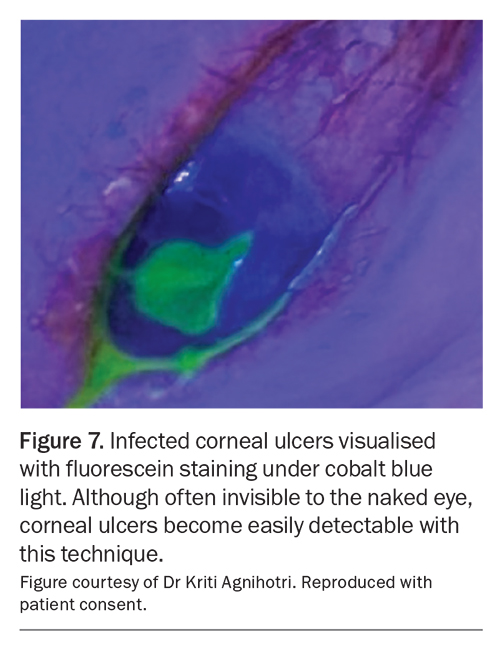
Fluorescein can be used to assess tear break-up time, and corneal staining should be performed with the use of a cobalt blue light to differentiate between severe dryness (Figure 6), corneal ulcers (Figure 7), dendrites and viral conjunctivitis.
Acute red eyes should be swabbed for bacterial culture and sensitivity, and viral polymerase chain reaction testing should be used for adenovirus, herpes simplex virus or varicella zoster virus. Patients with signs of serious differential diagnoses (Box 2) should be promptly referred to an ophthalmologist.
Initial management
Preservative-free lubricating eye drops should be used at least every two to three hours and the condition should be reassessed in a week. Where glaucoma medications are available in both preservative-containing and preservative-free versions, a switch to the latter should be tried. Long-term use of antihistamine eye drops should be avoided; consider mast cell stabilisers such as olopatadine or ketotifen, preferably preservative-free, for true ocular allergies.
Referral
Patients with mild symptoms can be referred to an optometrist initially. A sample ophthalmologist referral template is presented in Box 3 and key information to communicate to the patient for suspected OSD is presented in Box 4.
Medications to avoid
Even though they are readily available and are often over-the-counter medications, broad-spectrum antibiotics such as chloramphenicol should be avoided without consulting an ophthalmologist as these can exacerbate OSD and preservative allergy. Steroids should only be prescribed under the instruction of an ophthalmologist, as they can worsen infections and significantly aggravate conditions like glaucoma with medium- to long-term use by increasing the IOP.
Primary care doctors should recognise that bilateral bacterial conjunctivitis in this setting is rare. If conjunctivitis is infective, it is usually viral and does not typically require treatment with antibiotic eye drops.
Management of eye drop intolerance
As GPs are not expected to measure IOP, any decision to stop glaucoma eye drops or start steroids should be preceded by an early review by the ophthalmologist within the week. Any patient using medium- to long-term steroid eye drops should have their IOP monitored regularly.
Ophthalmic management of ocular surface disease associated with glaucoma eye drops
It is pertinent to understand that chronic ocular surface inflammation plays a critical role in OSD, and this is exacerbated by the proinflammatory effect of topical glaucoma medications and their excipients and preservatives. Untreated chronic inflammation can cause ocular surface, scleral and episcleral scarring, which can also reduce intraocular aqueous drainage and impact IOP. Therefore, modulating or minimising this inflammation both improves patient symptoms, and, in effect, compliance with glaucoma medication, improving IOP by more than one mechanism.9
The stepwise management of OSD from glaucoma treatment involves two principles of intervention:
- early recognition and management of OSD
- minimising the effect of glaucoma eye drops on the surface of the eye.
The sequence of treatments is shown below. With encouragement and regular follow up, initially weekly, ocular surface irritation can be reversed and patient comfort can be improved.
Sequence of treatments
Step 1. Preservative-free lubricants
The first step can be commenced in the GP office, with authority prescriptions enabling pensioners to access additional repeats of preservative-free lubricants at PBS subsidised rates. Preservative-free lubricants should be used every few hours and the patient should be referred to the ophthalmologist for review within one week. It may take weeks for regular use of these eye drops to take effect.
Step 2. Glaucoma eye drops without preservatives
A straightforward next step is to switch to a preservative-free version of the patient’s current eye drop medication, available either in single-dose vials or specially designed bottles that maintain sterility without the need for additives. However, not all glaucoma medications are available in preservative-free forms in Australia.
Glaucoma eye drops should not be changed without consulting the patient’s ophthalmologist, as the preserved and preservative-free formulations can have different effects on IOP, and the patient may require an IOP assessment. In addition, some eye drops may be contraindicated or should be avoided in certain patients, including those with specific conditions or risk factors, such as:
- beta blockers (e.g. timolol) in patients with asthma
- carbonic anhydrase inhibitors (e.g. acetazolamide) in individuals with a sulfonamide allergy
- alpha agonists (e.g. brimonidine) in children under 6 years of age
- prostaglandin analogues during pregnancy.
It may take up to six weeks for the effects of the eye drops to ‘wash out’ from the ocular system, so the new eye drops do need to be persevered with. Additionally, patients may inform their GP whether they have started the new eye drops as advised, or if they have encountered any issues, such as cost, difficulty of use or wanting to finish their supply of previous eye drops first. Nonadherence to eye drop treatment because of medication cost, frequency of reviews and the associated cost of reviews are significant and ongoing barriers to glaucoma care. A collaborative approach between the GP, optometrist, ophthalmologist and pharmacist may help address these challenges for this invisible yet debilitating disease.
Step 3. Ocular surface immunomodulating agents
Ocular surface immunomodulating agents such as ciclosporin or lifitegrast may help minimise long-term chronic changes to the ocular surface and treat OSD.12 Further information about ocular surface immunomodulating agents is presented in Box 5.13
Step 4. Steroid eye drops
Steroid eye drops should be used with caution and only for short durations in cases of significant allergy, as they can cause a marked increase in IOP, potentially leading to permanent glaucomatous damage.
Newer steroid eye drops that reduce but do not remove the risk of IOP elevation have been developed and these can be a tool in quickly settling inflammation and allergy once the diagnosis has been confirmed by the ophthalmologist.14 Repeat prescriptions of steroid eye drops should not be given unless indicated by the ophthalmologist and there is a plan in place to monitor the IOP regularly.
Step 5. Punctal plugs
Punctal plugs are small devices that are inserted into the punctum, which is the entry of the canaliculus and drains tears from the eye to the nasolacrimal duct. Punctal plugs increase the volume of the natural tear lake by reducing the speed of drainage into the nasolacrimal duct. This also has the dual effect of prolonging the retention of glaucoma medication on the ocular surface and further contributing to the reduction of IOP.15,16
Step 6. Blepharitis and meibomian gland dysfunction treatment
Meibomian gland dysfunction is an uncomfortable eyelid margin inflammation involving the meibomian glands. These glands produce meibum, a wax that is a component of the tear film. As blepharitis and meibomian gland dysfunction can affect up to 80% of glaucoma patients, it is helpful for patients to be reminded by all healthcare professionals, but especially those they see most such as their GP, of the importance of consistent adherence to treatment.17 Treatment includes eyelid hygiene warm compresses, pulsed light therapy and thermal vectored pulsation with or without meibomian gland expression. Warm compresses are highly effective but are often dismissed as they can be inconvenient for patients, especially elderly patients, because of the time taken to use them and the sensation of heat on the face. Thus, they should be encouraged by all healthcare professionals and family members.
Step 7. Glaucoma laser
Glaucoma laser, such as selective laser trabeculoplasty, can be used to reduce eye drop burden.18 This laser procedure is an almost painless, in-office treatment that can be performed by an ophthalmologist to modify the natural aqueous drainage channels of the eye and improve their function in select patients. It may need to be repeated multiple times because its effect can wear off months to years after the first treatment, but it is a low-risk and well-tolerated option.
Step 8. Intraocular surgery
A modern technique known as minimally invasive glaucoma surgery can be used either standalone or combined with cataract surgery. It involves the insertion of various different types and sizes of shunts that can be placed in different positions to increase the drainage of aqueous humour out of the anterior chamber of the eye. Minimally invasive glaucoma surgery is less invasive than traditional glaucoma surgery; however, it may be less effective and is generally not suitable for advanced cases. The lowering of IOP from this procedure may allow for eye drops to be reduced or potentially ceased altogether.19-21
Advanced glaucoma management
For patients who are unable to tolerate glaucoma eye drops, or for whom the eye drops are insufficient to prevent progression, traditional glaucoma filtration surgical options are available.10 These include:
- trabeculectomy, which creates a new drainage pathway for aqueous humour through formation of a conjunctival bleb
- tube shunt surgery, which involves implantation of a tube to drain aqueous humour, reducing IOP.
However, these surgeries can disrupt the ocular surface and tear film stability, potentially complicating the management of OSD, and are therefore not considered first-line treatments.
Communication and collaboration
Good communication between ophthalmologists and GPs and continuity of care are essential in the management of patients with OSD from glaucoma. This helps to ensure accurate prescriptions and allows GPs to seek quick advice or clarification and further information when needed. Verification of ongoing medications is extremely important, especially as some drugs can have similar names but vastly different effects.
Communication with the patient’s pharmacist can be useful too – they can recommend preservative-free formulations and indicate which preservative-free lubricants may be cost effective for the patient. They can also review the patient’s medical history and identify any potential contraindications that may not have been flagged to the ophthalmologist when the eye drops were prescribed.
Conclusion
By staying vigilant and proactive in managing OSD, GPs can help improve patients’ quality of life and adherence to glaucoma treatment, ultimately protecting their vision. Understanding the nuances of glaucoma treatment and its impact on the ocular surface is crucial for effective patient management. With early recognition, appropriate interventions, and ongoing communication and collaboration with specialists, GPs can play a pivotal role in optimising care and advocating for glaucoma patients. Some practice points for GPs are listed in Box 6. MT
COMPETING INTERESTS: Dr Agnihotri and Dr Durai: None. Professor Coroneo is Chairman of the Look for Life Foundation.
References
1. Nijm LM, De Benito-Llopis L, Rossi GC, Vajaranant TS, Coroneo MT. Understanding the dual dilemma of dry eye and glaucoma: an international review. Asia Pac J Ophthalmol (Phila) 2020; 9: 481-490.
2. DEWS II. The epidemiology of dry eye disease: report of the International Dry Eye Workshop (DEWS II). Ocul Surf 2017; 15: 334-365.
3. Schiffman RM, Walt JG, Jacobsen G, Doyle JJ, Lebovics G, Sumner W. Utility assessment among patients with dry eye disease. Ophthalmology 2003; 110: 1412-1419.
4. Buchholz P, Steeds CS, Stern LS, et al. Utility assessment to measure the impact of dry eye disease. Ocul Surf 2006; 4: 155-161.
5. Moore SG, Richter G, Modjtahedi BS. Factors affecting glaucoma medication adherence and interventions to improve adherence: a narrative review. Ophthalmol Ther 2023; 12: 2863-2880.
6. Nijm LM, Dunbar GE. Understanding the science behind the inflammatory cascade of dry eye disease. US Ophthalmic Rev 2019; 12: 15-16.
7. Coroneo M. High and dry: an update on dry eye syndrome. Medicine Today 2013; 14(1): 53-61.
8. Baudouin C. Detrimental effect of preservatives in eye drops: implications for the treatment of glaucoma. Acta Ophthalmol 2008; 86: 716-726.
9. Pisella PJ, Pouliquen P, Baudouin C. Prevalence of ocular symptoms and signs with preserved and preservative free glaucoma medication. Br J Ophthalmol 2002; 86: 418-423.
10. Mastropasqua R, Agnifili L, Mastropasqua L. Structural and molecular tear film changes in glaucoma. Curr Med Chem 2019; 26: 4225-4240.
11. Iyer JV, Zhao Y, Lim FPM, Tong L, Wong TTL. Ocular lubricant use in medically and surgically treated glaucoma: a retrospective longitudinal analysis. Clin Ophthalmol 2017; 11: 1191-1196.
12. Batra R, Tailor R, Mohamed S. Ocular surface disease exacerbated glaucoma: optimizing the ocular surface improves intraocular pressure control. J Glaucoma 2014; 23: 56-60.
13. Saini M, Dhiman R, Dada T, Tandon R, Vanathi M. Topical cyclosporine to control ocular surface disease in patients with chronic glaucoma after long-term usage of topical ocular hypotensive medications. Eye (Lond) 2015; 29: 808-814.
14. Saini M, Vanathi M, Dada T, Agarwal T, Dhiman R, Khokhar S. Ocular surface evaluation in eyes with chronic glaucoma on long term topical antiglaucoma therapy. Int J Ophthalmol 2017; 10: 931-938.
15. Sheppard JD, Comstock TL, Cavet ME. Impact of the topical ophthalmic corticosteroid loteprednol etabonate on intraocular pressure. Adv Ther 2016; 33: 532-552.
16. Chen M, Yung Choi S. Preliminary outcomes of temporary collagen punctal plugs for patients with dry eye and glaucoma. Med Hypothesis Discov Innov Ophthalmol 2020; 9: 56-60.
17. Sherwin JC, Ratnarajan G, Elahi B, et al. Effect of a punctal plug on ocular surface disease in patients using topical prostaglandin analogues: a randomized controlled trial. Clin Exp Ophthalmol 2018; 46: 888-894.
18. Uzunosmanoglu E, Mocan MC, Kocabeyoglu S, et al. Meibomian gland dysfunction in patients receiving long-term glaucoma medications. Cornea 2016; 35: 1112-1116.
19. Gazzard G, Konstantakopoulou E, Garway-Heath D, et al. Selective laser trabeculoplasty versus eye drops for first-line treatment of ocular hypertension and glaucoma (LiGHT): a multicentre randomised controlled trial. Lancet 2019; 393: 1505-1516.
20. Boussard T, Pershing S. Intraocular pressure changes after cataract surgery in patients with and without glaucoma: an informatics-based approach. Ophthalmol Glaucoma 2020; 3: 343-349.
21. Zhang X, Vadoothker S, Munir WM, Saeedi O. Ocular surface disease and glaucoma medications: a clinical approach. Eye Contact Lens 2019; 45: 11-18.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.