Giant cell arteritis: timely diagnosis, targeted therapy

Giant cell arteritis is the most common vasculitis in older adults, causing significant morbidity from both disease and treatment. Prednisolone, often with corticosteroid-sparing agents such as tocilizumab, is the mainstay of treatment but carries substantial adverse effects and a high relapse risk. Prompt patient referral to a specialist enables timely diagnosis and helps minimise complications such as visual loss.
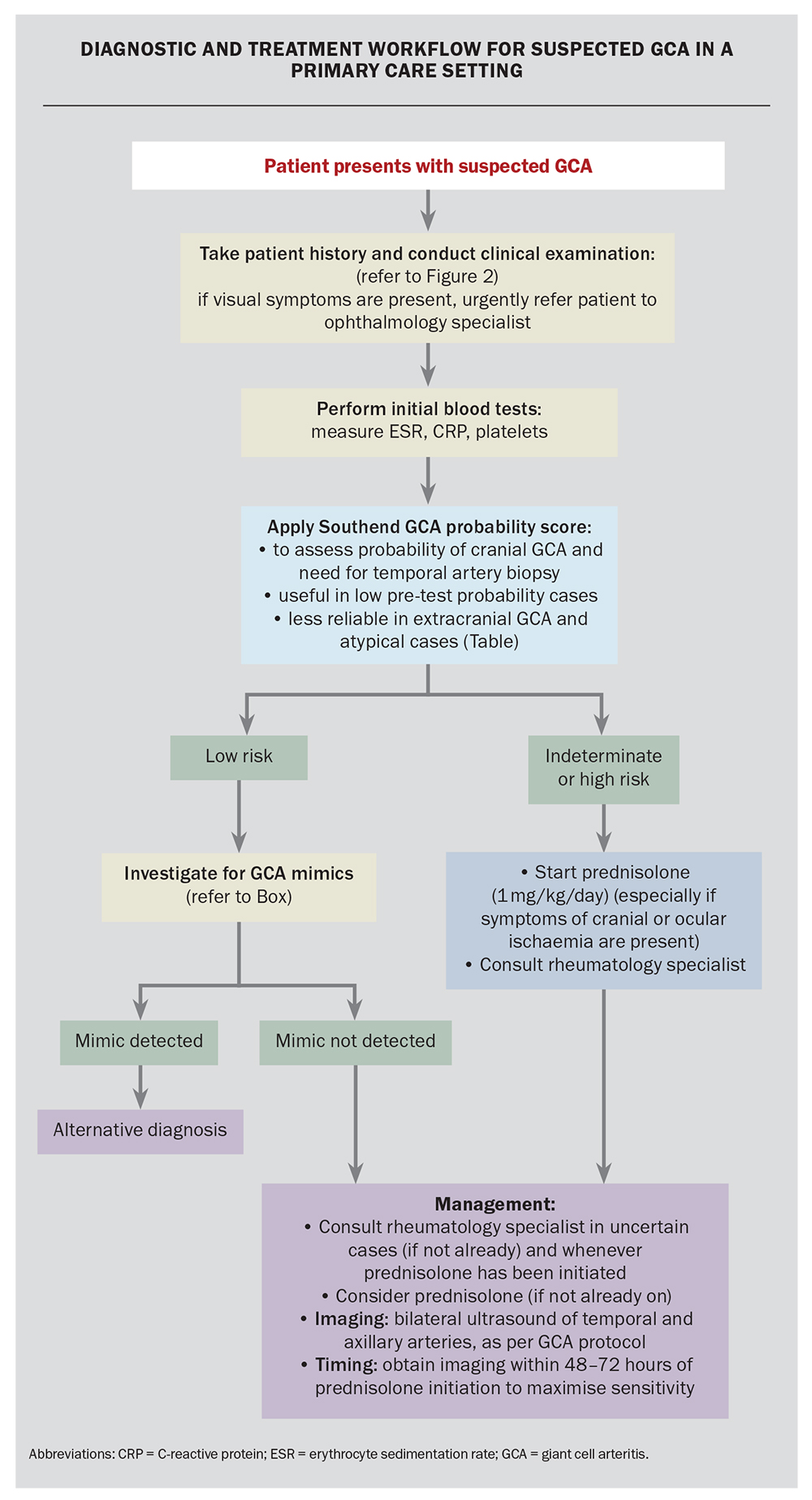
- The diagnosis of giant cell arteritis (GCA) can be challenging. New or severe headache, jaw claudication and visual disturbance remain key features of cranial disease, whereas systemic symptoms are prominent in extracranial involvement.
- The Southend pre-test probability score, validated retrospectively in specialist centres, helps reliably exclude low-risk patients and may reduce unnecessary investigations, the need for prednisolone and specialist referrals. However, it has not yet been validated for use in primary care.
- Imaging, particularly colour Doppler ultrasound of the temporal and axillary arteries, is increasingly used as a first-line diagnostic tool.
- Rapid access GCA clinics are the gold standard for timely review and management, reducing the risk of visual loss substantially.
- Glucocorticoids remain the cornerstone of treatment, but corticosteroid-sparing agents such as tocilizumab help reduce prednisolone burden and relapse risk. Newer biologic therapies, including the Janus kinase inhibitor upadacitinib (pending PBS approval), are emerging.
- Despite therapeutic advances, relapse remains common (about 50%), underscoring the need for ongoing vigilance and regular review.
Giant cell arteritis (GCA) is a chronic, inflammatory, autoimmune disease targeting large and medium-sized arteries. It is the most common vasculitis in older adults, with a prevalence of 0.5% in men and 1% in women aged older than 50 years, with a female preponderance (roughly 2:1 ratio).1 Classic cranial manifestations include headache, scalp and temporal artery tenderness and jaw claudication, with systemic symptoms (including fevers, night sweats, anorexia, unintentional weight loss and fatigue) and limb claudication representing extracranial disease manifestations. The most feared complications are visual loss (20%), stroke (up to 7%) and aortic involvement, leading to dissection (up to 6%) or dilatation (up to 33%).2,3 GCA-attributable mortality typically arises from cardiovascular causes (45%), infection (17%) and cancer (17%), although the overall mortality is not increased compared with the general population.4
The diagnosis of GCA integrates clinical, biochemical, histological and imaging parameters. Temporal artery biopsy (TAB) remains the diagnostic gold standard; however, diagnosis is often problematic because of variable clinical presentations and inflammatory responses.5 Furthermore, the ‘skip lesion’ nature of arterial inflammation can cause false-negative TABs in up to 44% of cases.5 Variable imaging quality and reporting also contribute to diagnostic uncertainty. A significant concern is that about 90% of patients with GCA experience at least one serious glucocorticoid-related adverse event, including major osteoporotic fractures (50% mortality risk), infections, metabolic side effects (weight gain, type 2 diabetes, hypertension, accelerated heart disease) and cataracts.1 The average glucocorticoid therapy duration is two to three years, which compounds these risks.1 Furthermore, the relapse rate is estimated at 50%, leading to further glucocorticoid use.6 Glucocorticoid-sparing treatment options have diversified in recent years including the hallmark introduction of tocilizumab following the 2017 landmark paper on the Giant-Cell Arteritis Actemra (GiACTA) trial, as well as the US Food and Drug Administration-approved upadacitinib; however, much is unknown about the long-term outcomes these options offer.7 Similarly, predicting relapse and tailoring treatment to individual patients remains challenging. Ongoing research aims to improve our ability to forecast disease trajectory and ultimately provide more targeted treatment options.
When to worry about a headache
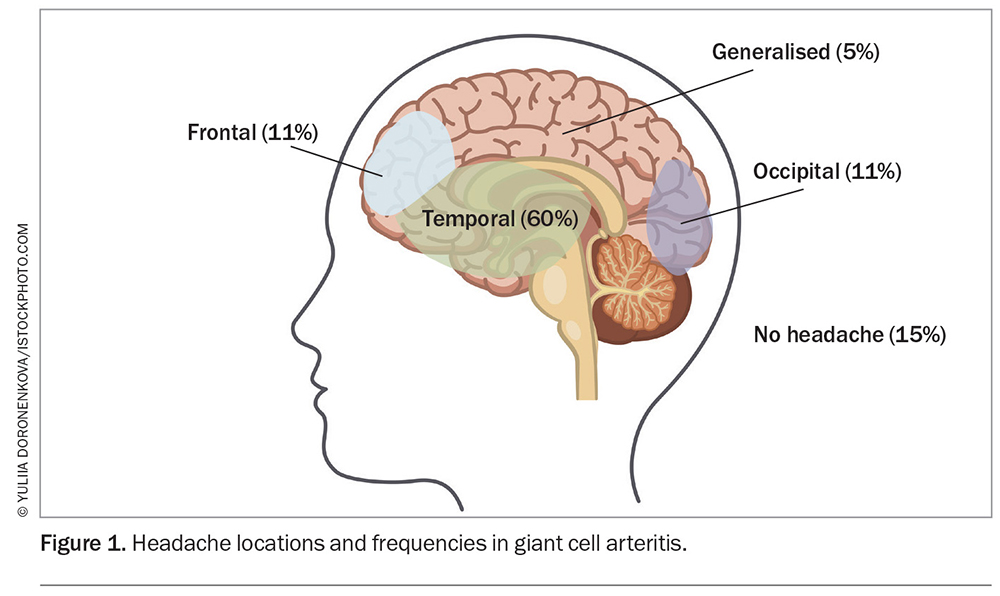
New-onset severe headache in patients aged older than 50 years warrants vigilance, as it is a key symptom of GCA in about 67% of cases.8 The headache has been reported as continuous in 60% of patients and is most often temporal (60%), but can also be occipital or frontal (11% each), generalised (5%) or absent (15%).9 In those with a pre-existing headache disorder, the headache should differ from their usual pattern and be more severe.10
Overall, the most common headache pattern is continuous, unilateral and temporal (Figure 1).11 The International Headache Society definition of a GCA headache requires a minimum of two of the three following characteristics:
- headache has developed in temporal relation to other symptoms or clinical or biological signs of GCA onset, or has led to the diagnosis of GCA
- either or both of the following:
– headache has significantly worsened in parallel with worsening of GCA
– headache has significantly improved or resolved within three days of high-dose steroid treatment - headache is associated with scalp tenderness or jaw claudication.12
Other symptoms
Other significant symptoms include prior or current polymyalgia rheumatica (PMR), involving morning predominant bilateral shoulder or hip girdle aches and stiffness lasting for more than 45 minutes with elevated acute phase reactants (erythrocyte sedimentation rate [ESR] and C-reactive protein [CRP]) with or without systemic symptoms. Up to 50% of patients with GCA experience PMR features. Additionally, around 25% of patients with PMR are either diagnosed with GCA, or have evidence of subclinical GCA; thus, a PMR background significantly increases the pre-test probability of a GCA diagnosis in the right clinical context.13
Systemic complaints and jaw claudication (jaw or masseter muscle pain or fatigue on chewing or prolonged talking, which resolves at rest) each also occur in roughly 50% of patients with GCA; limb claudication (often affecting the upper rather than lower limbs) may occasionally be associated. Importantly, visual manifestations occur in 20%, with high vigilance warranted to prevent permanent visual loss.
Visual complaints must never be ignored during workup for suspected GCA, as this may be the only presenting feature (in up to 20% of cases).14 The most common visual symptoms include blurred vision, visual loss (which can be transient, such as amaurosis fugax, or permanent) or complete field or monocular visual loss, and diplopia.
In a 2020 retrospective study of 100 patients with GCA, 53% of patients had visual manifestations at presentation including blurred vision (30%), diplopia (16%), amaurosis fugax (14%) and blindness (19%).15
Once appropriate high-dose glucocorticoid treatment has been initiated, new or further visual events are rare (about 4%); however, ocular recovery following permanent visual loss is disappointingly low (only 15 to 20%).16 Rare presentations of GCA can include cranial nerve palsies, stroke and tongue and scalp necrosis.17
Examination
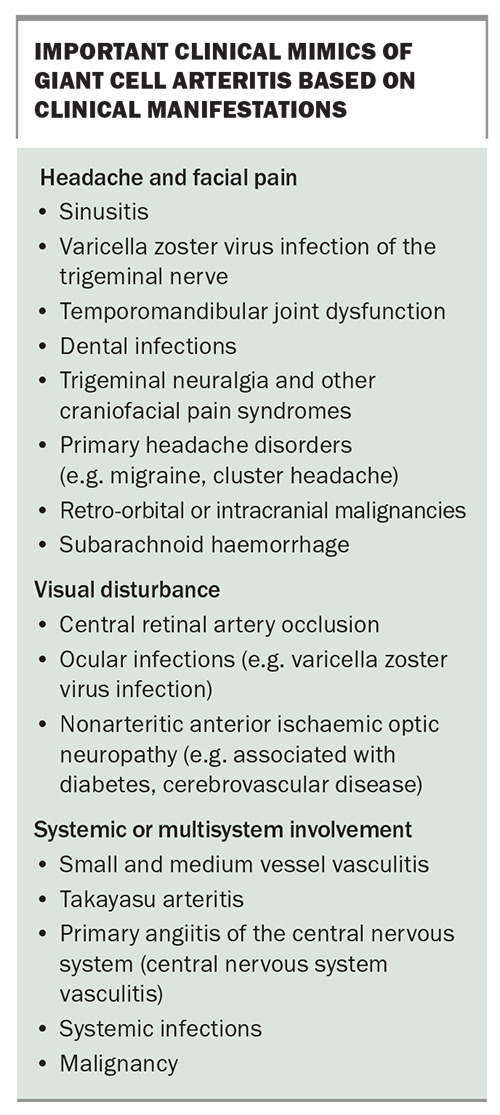
Examination should aim to support a diagnosis of GCA, identify complications and exclude mimics.
- Temporal arteries: check for prominence, nodularity, tenderness and reduced pulse.
- Vascular signs: listen for carotid and axillary bruits, assess for aortic regurgitation murmur and compare blood pressure in both arms.
- Targeted cranial nerve examination: assess cranial nerves (especially extraocular eye movements), pupils (for a relative afferent pupillary defect, indicating optic neuropathy), visual acuity, visual fields and colour desaturation.
- Scalp and face: check for scalp tenderness (reflecting GCA) and facial tenderness (e.g. reflecting potential sinusitis, which is a common GCA mimic).
Investigations
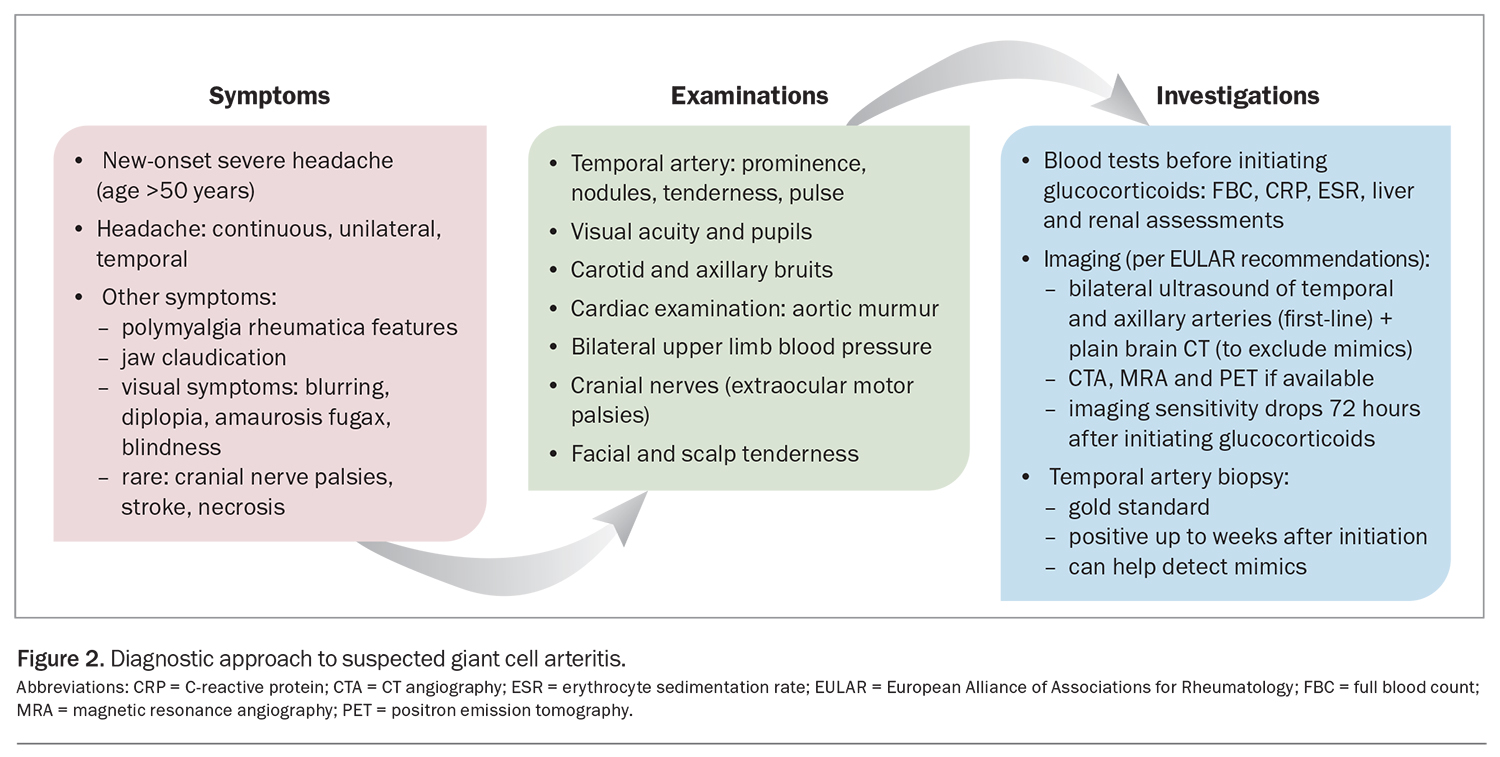
Investigations should include baseline blood tests for a full blood count, CRP levels and ESR, as well as liver and renal function. Overall, a raised CRP level (>20 mg/L) and thrombocytosis (platelet count >300 × 109/L) likely provide the best diagnostic utility with sensitivities of 67% and 71%, respectively, and specificities of 68% and 63%, respectively. The ESR was found to have a sensitivity of 66% and specificity of 57%.18
Blood samples should be taken before initiating prednisolone, on the day of initial consultation. A plain brain CT should also always be considered for new-onset headache in older people to exclude common mimics, as outlined in the Box. In the event of amaurosis fugax, a carotid ultrasound or CT angiography should also be requested to exclude a non-vasculitic cause of stroke or transient ischaemic attack as the underlying cause of visual symptoms. Additional imaging is helpful if readily available, but clinicians are urged to check with the local rheumatology department, as rapid access clinics (including point-of-care ultrasound) may offer a faster diagnostic pathway.
The latest European Alliance of Associations for Rheumatology guidelines recommend bilateral temporal and axillary artery colour Doppler ultrasound as the first-line investigation for GCA, replacing biopsy as the initial test in many cases.19 However, important caveats apply; this approach requires specialised expertise, dedicated equipment and rapid image acquisition, particularly if prednisolone has already been commenced. It is important to note that the sensitivity of all imaging modalities reduces after 72 hours of high-dose prednisolone; however, imaging may still be indicated within one to two weeks of prednisolone initiation.20,21
Other imaging modalities to consider are CT angiography, magnetic resonance angiography and positron emission tomography/CT scans, although availability in a primary care setting, and particularly in rural locations, is likely to be challenging. Thus, our recommendation is to discuss any further investigations aside from blood testing and a plain brain CT with the on-call rheumatologist. The choice of imaging modality will be dictated by the clinical presentation (whether primarily cranial or extracranial or both), and the availability and timing of imaging.
TAB remains important particularly if imaging is not available, the imaging findings may be inaccurate because of corticosteroid use (>3 days) or the imaging findings are negative despite strong clinical suspicion. TABs are performed by a variety of specialists including plastic, vascular and general surgeons and ophthalmologists as a simple procedure under local anaesthesia. Although prednisolone use can potentially reduce the diagnostic yield of a biopsy, this typically occurs more slowly than with imaging. Therefore, a biopsy performed within two to four weeks of prednisolone initiation is still extremely useful. Reports of biopsy positivity several months post-prednisolone commencement have also been demonstrated, including in the authors’ experience. Additional benefits of TABs include gold-standard diagnostic ability and the identification of GCA mimics, including small vessel vasculitis, malignancy (e.g. lymphoma), infection (e.g. varicella zoster virus), calciphylaxis, atherosclerosis and amyloidosis, which are indistinguishable on imaging.
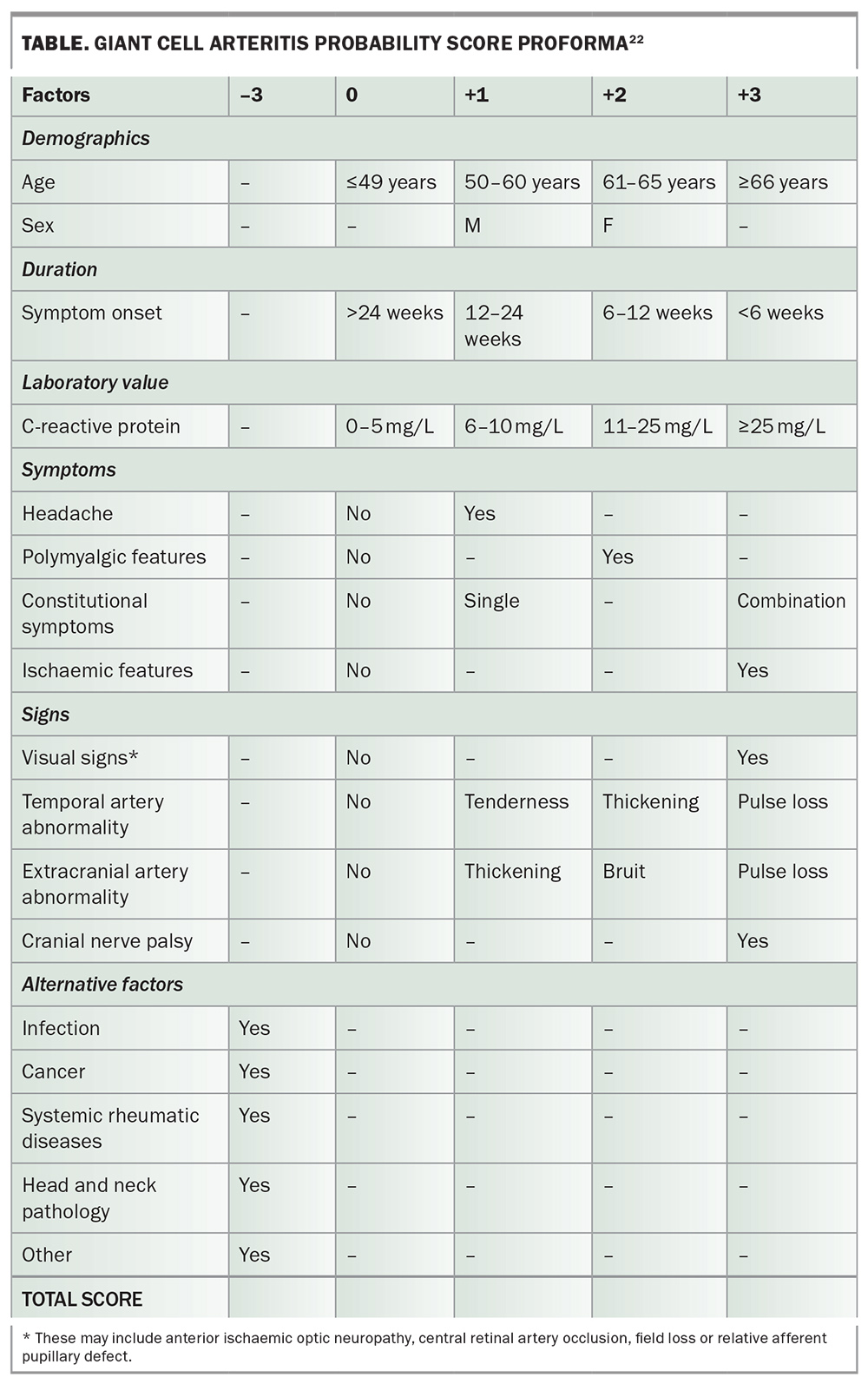
The Southend GCA probability score (GCAPS; Table) is a quick and useful tool combining clinical review and basic bloods to stratify risk.22 Scores less than 9 effectively rule out GCA, with none of these patients in external validation studies having a final diagnosis of GCA.23,24 Scores of 9 or higher warrant specialist referral (9–12 = intermediate risk, >12 = high risk). This score is very useful for excluding low-risk patients, although caution is needed for extracranial disease and atypical presentations. It is important to note that this scoring has not been validated in the primary care setting at this stage. It still a useful tool but should not replace clinical judgement and rapid rheumatology input as required.
The clinical features and investigation approach for GCA are outlined in Figure 2.
Prescribing prednisolone
Starting patients on high-dose prednisolone should be guided by risk stratification and rheumatology input. Patients may be started immediately if there are visual concerns or there is a high or intermediate pre-test probability based on the GCAPS. However, we still recommend all potential cases be discussed with a rheumatologist regardless of the GCAPS. Once prednisolone is commenced, further imaging or assessment is required as soon as possible. We recommend that any patient with concerning visual features in the context of possible GCA be referred immediately to the nearest ophthalmologist for expert assessment and prompt initiation of intravenous corticosteroids, if indicated. In the absence of expert ophthalmological review opportunity (i.e. in rural settings), patients should be discussed immediately with the on-call ophthalmologist and rheumatologist, and a collaborative decision made on the treatment and further workup.
Rapid access clinic review
Rapid access clinics, utilised ever increasingly in Europe, are considered the gold standard for the rapid assessment and diagnosis of GCA. Rapid access clinics ensure rheumatological GCA expertise, appointment scheduling within 24 hours ideally, expert ultrasonographers with appropriate equipment to facilitate high-quality ultrasound examinations and collaboration with other disciplines as necessary, as well as having the benefit of reducing permanent visual loss.25 In Australia, dedicated rapid access clinics for GCA are limited; however, every tertiary hospital should have a rapid access rheumatology clinic available for assessment in cases of suspected GCA.
Management
Once a diagnosis has been reached, disease activity must be stabilised with high-dose prednisolone and early consideration of glucocorticoid-sparing therapy. This typically involves four weeks of full-dose prednisolone treatment (1 mg/kg, up to a maximum of 80 mg) to stabilise inflammation and ensure biochemical and clinical improvement. A standard 52-week taper will subsequently be initiated, with a rapid 26-week taper if prednisolone is co-prescribed with tocilizumab. Glucocorticoid-sparing agents are being increasingly introduced at diagnosis, based largely on the GiACTA trial, which demonstrated a reduction in the cumulative prednisolone dose and relapse rates with tocilizumab use.7 Despite the significant benefit, post-tocilizumab relapse remains an issue, with rates of relapse of up to 50% at 18 months post-treatment cessation.26 This is particularly pertinent given the PBS only allows for weekly tocilizumab to be prescribed for 12 months. Methotrexate has shown modest benefit and is conditionally recommended for use in the 2021 American College of Rheumatology/ Vasculitis Foundation Guidelines for the Management of GCA and Takayasu Arteritis.27
Other therapeutic targets are in varying stages of investigation and use. Upadacitinib, a Janus kinase inhibitor, is the newest medication to be approved by the US Food and Drug Administration for GCA, after the 2025 phase 3 Study to Evaluate the Safety and Efficacy of Upadacitinib in Participants With Giant Cell Arteritis (SELECT-GCA) trial demonstrated efficacy at a dose of 15 mg daily in combination with a 26-week prednisolone taper, over placebo in combination with a 52-week prednisolone taper.28 Other options including agents targeting interleukin-17 or interleukin-12/23 have not been shown from various trial endpoints to provide benefit; however, with rapid advances in precision medicine and the integration of artificial intelligence, novel therapeutic targets continue to emerge on the horizon.29 Nevertheless, how these emerging treatments might be used optimally and which patient populations might respond most favourably are unknown.
A key challenge in GCA is management when there is strong clinical suspicion but negative biopsy or imaging findings. Diagnosis often relies on clinical judgement, with treatment initiated to prevent vision loss despite the risks of overtreatment. In such cases, close monitoring, reassessment of the diagnosis and consideration of adjunctive imaging and investigation are recommended. Advances in biomarkers may improve the diagnostic accuracy in this difficult group in the future but, currently, management is the same for GCA regardless of the modality used for diagnosis or whether the diagnosis was clinical.
Follow up is instigated largely by rheumatologists, with close three-monthly reviews (at minimum) in the first year to ensure satisfactory progress and detect relapses expediently. Routine blood tests (full blood count; electrolytes, urea and creatinine test; liver function tests; measurement of CRP levels and ESR) are conducted every four to six weeks initially and are reduced in frequency depending on the clinical progress and use of glucocorticoid-sparing agents. Glucocorticoid side effects are common (up to 90%) and can include osteoporosis, infections and a heightened cardiovascular risk. Monitoring should include baseline assessments such as dual-energy x-ray absorptiometry scanning, fasting lipid profile, blood pressure measurement and diabetes screening.
If tocilizumab is used, careful monitoring of lipids, infection risk assessment, full blood count and liver function tests are essential throughout treatment. CRP levels and the ESR may be of little value in relapse monitoring with patients on tocilizumab, considering the downstream effects of interleukin-6 inhibition consequently suppressing the hepatic production of CRP and other acute phase reactants (which drive a raised ESR). Thus, a high index of suspicion may be needed in this circumstance (Flowchart).
Patient and clinician resources for GCA, prednisolone and tocilizumab are readily available via the Arthritis Australia and Australian Rheumatology Association websites, including succinct printable handouts summarising key information, toxicities and monitoring requirements.
Conclusion
In conclusion, GCA remains a difficult diagnosis for even the most experienced rheumatologists to confirm and manage. It requires multidisciplinary input for a rapid and effective approach to reduce permanent complications, such as visual loss and stroke. New-onset or severe headaches in those aged older than 50 years, especially with visual symptoms, should prompt urgent rheumatological review. Ultrasound is increasingly replacing biopsy, but TAB still plays a role in confirming the diagnosis and excluding mimics. The introduction of pre-test probability screening and rapid access clinic implementation will reduce unnecessary prednisolone burden and visual loss. The future of management is promising with newer biological therapeutic agents on the horizon; however, despite the widespread uptake of tocilizumab, post-treatment relapse remains a major issue, and more research on relapse prediction and personalised therapeutic approaches is warranted to better stratify patients from the outset. In the meantime, prompt communication with the local rheumatology team remains key to achieving the best outcomes for patients. MT
COMPETING INTERESTS: None.
References
1. Crowson CS, Matteson EL, Myasoedova E, et al. The lifetime risk of adult‐onset rheumatoid arthritis and other inflammatory autoimmune rheumatic diseases. Arthritis Rheum 2011; 63: 633-639.
2. Soriano A, Muratore F, Pipitone N, Boiardi L, Cimino L, Salvarani C. Visual loss and other cranial ischaemic complications in giant cell arteritis. Nat Rev Rheumatol 2017; 13: 476-484.
3. Koster MJ, Matteson EL, Warrington KJ. Large-vessel giant cell arteritis: diagnosis, monitoring and management. Rheumatology 2018; 57: ii32-ii42.
4. Ninan J, Nguyen AM, Cole A, et al. Mortality in patients with biopsy-proven giant cell arteritis: a South Australian population-based study. J Rheumatol 2011; 38: 2215-2217.
5. Davies C, May D. The role of temporal artery biopsies in giant cell arteritis. Ann R Coll Surg Engl 2011; 93: 4-5.
6. De Smit E, Palmer AJ, Hewitt AW. Projected worldwide disease burden from giant cell arteritis by 2050. J Rheumatol 2015; 42: 119-125.
7. Stone JH, Tuckwell K, Dimonaco S, et al. Trial of tocilizumab in giant-cell arteritis. New Engl J Med 2017; 377: 317-328.
8. Tuckwell K, Collinson N, Dimonaco S, et al. Newly diagnosed vs. relapsing giant cell arteritis: baseline data from the GiACTA trial. Semin Arthritis Rheum 2017; 46: 657-664
9. Imai N, Kuroda R, Konishi T, Serizawa M, Kobari M. Giant cell arteritis: clinical features of patients visiting a headache clinic in Japan. Intern Med 2011; 50: 1679-1682.
10. Jones RV. Unbearable headache of temporal arteritis. Br Med J 1977; 1: 1355.
11. Vincenten SC, Mulleners WM. The quest for a headache pattern in giant cell arteritis: a cohort study. Cephalalgia Reports 2021; 4.
12. Mollan SP, Paemeleire K, Versijpt J, et al. European Headache Federation recommendations for neurologists managing giant cell arteritis. J Headache Pain 2020; 21: 28.
13. Hemmig AK, Gozzoli D, Werlen L, et al. Subclinical giant cell arteritis in new onset polymyalgia rheumatica: a systematic review and meta-analysis of individual patient data. Semin Arthritis Rheum 2022; 55: 152017.
14. Hayreh SS, Podhajsky PA, Zimmerman B. Occult giant cell arteritis: ocular manifestations. Am J Ophthalmol 1998; 125: 5216.
15. Dumont A, Lecannuet A, Boutemy J, et al. Characteristics and outcomes of patients with ophthalmologic involvement in giant-cell arteritis: a case-control study. Semin Arthritis Rheum 2020; 50: 335-341.
16. Baalbaki H, Jalaledin D, Lachance C, et al. Characterization of visual manifestations and identification of risk factors for permanent vision loss in patients with giant cell arteritis. Clin Rheumatol 2021; 40: 3207-3217.
17. Jalaledin DS, Ross C, Makhzoum JP. Rare ischemic complications of giant cell arteritis: case series and literature review. Am J Case Rep 2022; 23: 937565.
18. Chan FL, Lester S, Whittle SL, Hill CL. The utility of ESR, CRP and platelets in the diagnosis of GCA. BMC Rheumatol 2019; 3: 1-7.
19. Dejaco C, Ramiro S, Bond M, et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice: 2023 update. Ann Rheum Dis 2024; 83: 741-751.
20. Hauenstein C, Reinhard M, Geiger J, et al. Effects of early corticosteroid treatment on magnetic resonance imaging and ultrasonography findings in giant cell arteritis. Rheumatology (Oxford) 2012; 51: 1999-2003.
21. Taimen K, Salomäki SP, Hohenthal U, et al. The clinical impact of using 18F-FDG-PET/CT in the diagnosis of suspected vasculitis: the effect of dose and timing of glucocorticoid treatment. Contrast Media Mol Imaging 2019; 2019: 9157637.
22. Laskou F, Coath F, Mackie SL, Banerjee S, Aung T, Dasgupta B. A probability score to aid the diagnosis of suspected giant cell arteritis. Clin Exp Rheumatol 2019; 37: 104-108.
23. Sebastian A, Tomelleri A, Kayani A, et al. Probability based algorithm using ultrasound and additional tests for suspected GCA in a fast-track clinic. RMD Open 2020; 6: e001297.
24. Melville AR, Donaldson K, Dale J, Ciechomska A. Validation of the Southend giant cell arteritis probability score in a Scottish single-centre fast-track pathway. Rheumatol Adv Practice 2022; 6: rkab102.
25. Schmidt WA, Czihal M, Gernert M, et al. Recommendations for defining giant cell arteritis fast-track clinics. English version. Zeitschrift für Rheumatologie 2024: 83: 285-288.
26. Matza MA, Dagincourt N, Mohan SV, et al. Outcomes during and after long-term tocilizumab treatment in patients with giant cell arteritis. RMD Open 2023; 9: e002923.
27. Maz M, Chung SA, Abril A, et al. 2021 American College of Rheumatology/Vasculitis Foundation guideline for the management of giant cell arteritis and Takayasu arteritis. Arthritis Rheumatol 2021; 73: 1349-1365.
28. Blockmans D, Penn SK, Setty AR, et al. A phase 3 trial of upadacitinib for giant-cell arteritis. New Engl J Med 2025; 392: 2013-2024.
29. Thiel J. Giant cell arteritis – new treatment targets at the horizon. Semin Arthritis Rheum 2025; 72: 152686.