Episodic migraine in women

Hormonal changes experienced by women at various stages of life can affect the frequency and duration of migraine. Treating pregnant or breastfeeding women with migraine can pose a challenge for GPs.
- There are significant associations between migraine activity and hormonal fluctuations throughout a woman’s life.
- Migraine with aura increases the risk of stroke from a very low baseline in women of childbearing age. Smoking and use of oral contraceptives both increase the risk further. In any individual, the risks must be balanced against the benefits of taking the oral contraceptive pill.
- Treatment options for menstrual migraine include standard acute and prophylactic treatments, hormonal manipulation and short-term prophylactic regimens at the vulnerable time (‘mini-prophylaxis’).
- Migraine may worsen or improve in the first trimester, but generally improves significantly for the remainder of the pregnancy.
- Management of migraine during pregnancy is difficult because of warnings about the use of many relevant drugs during pregnancy and lactation.
Migraine is a common neurological disorder affecting about 17% of women and 6% of men. It is characterised by episodic disabling headaches, with the features outlined in Box 1.1 The onset of migraine increases from the first to fourth decade of life and then declines, with the prevalence in postmenopausal women falling to a similar rate to that in men. There are significant associations between migraine activity and hormonal fluctuations throughout the life of a women such as at menarche, during menstruation, during pregnancy, when breastfeeding and during the postpartum period, during perimenopause, at menopause and when using hormonal modulators, such as the oral contraceptive pill (OCP) and menopausal hormone therapy (MHT).
These factors should be considered in the assessment of women with migraine, because they may help target more effective treatment strategies.
Migraine and cardiovascular risk
The cardiovascular risk of OCP use in women with migraine is a common concern for practitioners treating young women with migraine. This risk must be considered bearing in mind that the baseline incidence of stroke is low in young women, which means that any increase in relative risk is a minimal increase in an individual’s absolute risk for stroke. The incidence of stroke varies with gender and ethnicity, but is about 4.5 per 100,000 women aged 25 to 35 years, compared with about 200 per 100,000 in the general population.2 In five meta-analyses, the relative risk for ischaemic stroke varied between 1.56 and 2.41 for migraine with aura, and between 1.02 and 1.83 for migraine without aura compared with those without migraine.3 There is an increased risk of angina, myocardial infarction and cardiovascular death, especially with migraine with aura.3 Furthermore, increases in risk were seen in women older than 45 years of age, smokers and those on the OCP.4 Men with migraine have a relative risk for cardiovascular disease of 1.24 (95% confidence interval, 1.0 to 1.5).5
In addition to stroke and acute myocardial ischaemia, patients with migraine are at increased risk for peripheral vascular disease (claudication) and retinal microvascular disease. The underlying biology of these associations remains unclear and is further clouded by an association between migraine and the metabolic syndrome (obesity, hypertension, hyperlipidaemia and insulin resistance).
The background cardiovascular risk in young women is relatively low, so the presence of migraine is not an absolute contraindication to OCP use. Rather, careful individual assessment of risk is important when considering the risk of OCP use, with evaluation of the patient’s overall cardiovascular risk including body mass index, lipid profile, blood pressure, age, presence of insulin resistance and smoking status (Box 2). Due to an increased thrombotic risk, the use of the combined OCP is at least relatively contraindicated in women with migraine who are smokers, are over the age of 35 years, have other cardiovascular risk factors or valvular heart disease, or have migraine with aura. The level of concern increases considerably when a combination of these factors is present. Ultimately, in any individual, the risks must be balanced against the benefits of taking the OCP.
Menstrual migraine
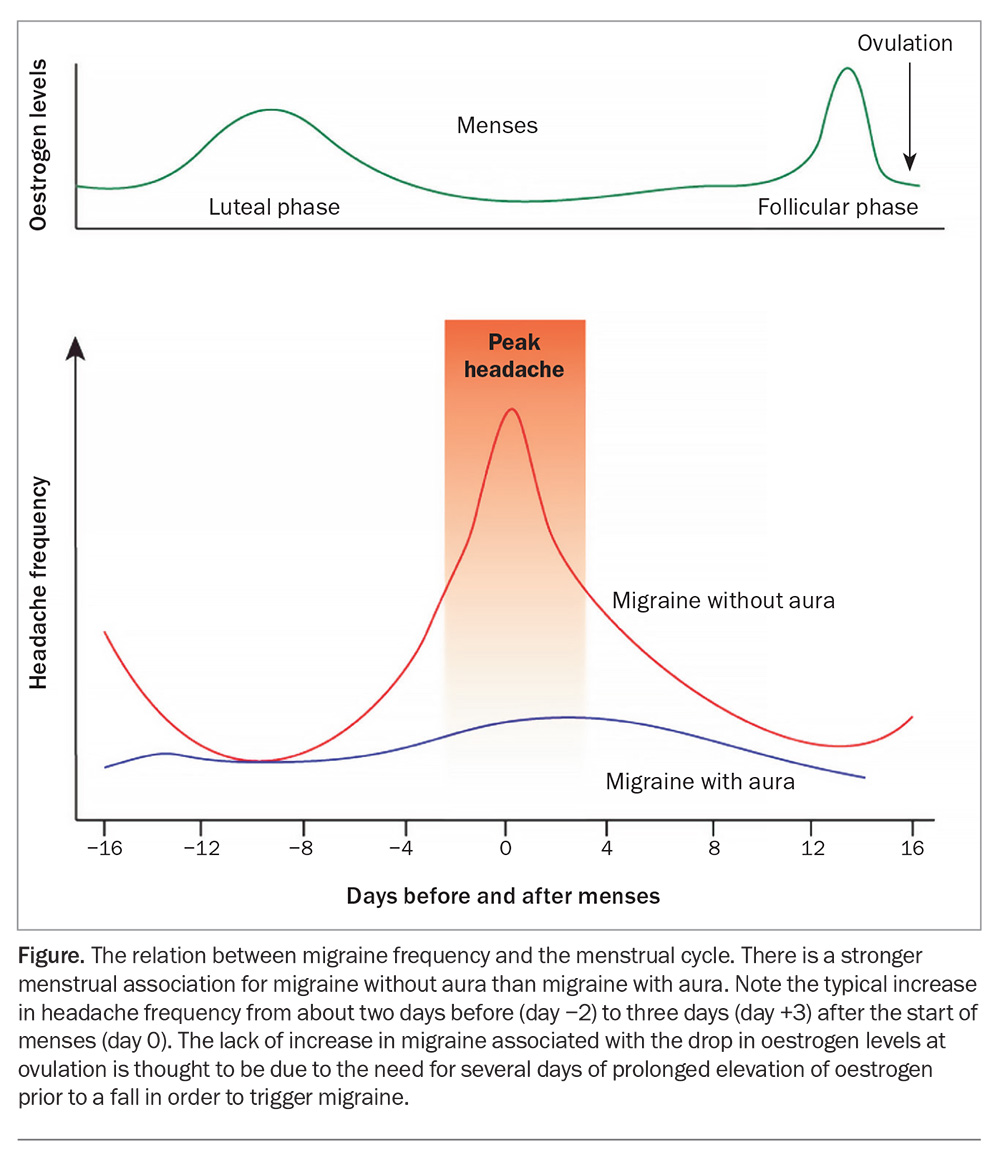
Migraine may be associated with menses, either occurring only at this time or with increased frequency and severity from two days before to three days after the onset of menses. Pure menstrual migraine is defined by the International Headache Society as migraine attacks occurring exclusively on day one of menstruation (± two days) in at least two out of every three menstrual cycles and at no other times of the cycle.1 A headache diary recording the menstrual cycle is often crucial in identifying women with menstrually-related migraines or pure menstrual migraine. Sixty percent of women have an increased frequency of migraine perimenstrually, whereas 14% have migraine exclusively in this period (true menstrual migraine). The trigger is the drop in oestrogen levels in the luteal phase of the menstrual cycle (Figure); however, no increase in migraine frequency is associated with the drop in oestrogen at ovulation. Studies have demonstrated that a persistent elevation in oestrogen levels for several days followed by a drop in levels is needed to trigger a migraine in susceptible individuals. In many patients, triggers are cumulative, so particular attention to good sleep and avoiding other triggers such as stress, alcohol and missed meals at the time of menses may be useful.
Pharmacological treatment for women with menstrual migraine, as for those with other types of migraine headache, may be acute or preventive, with the decision of which mode to employ being guided by the severity and frequency of the migraines and the patient’s response to treatment.
Acute treatment options include the use of a triptan. There are many different agents on the market and patients often respond to some triptans better than others, perhaps due to variations in absorption. Therefore, failure of a patient to respond to one agent does not indicate that they will be resistant to other members of the drug class. For patients who are genuinely resistant to all drug classes, subcutaneous sumatriptan is often helpful and is probably underused. Other acute treatment options include NSAIDs (e.g. aspirin 600 to 900 mg, ibuprofen 400 to 800 mg, naproxen 500 to 750 mg [as a single dose, repeated once or twice per day if required]), paracetamol and antiemetics.
Short-term prophylactic regimens (‘mini-prophylaxis’) may be structured to start two to three days before the start of menses and to continue for one to two weeks as needed. Benefit is seen with the use of a long-acting triptan, such as naratriptan 2.5 mg half a tablet twice a day, from three days before to three days after the onset of menses. Another option is naproxen 500 mg three times a day. The oral calcitonin gene-related peptide antagonist, rimegepant, is attractive as an option for mini-prophylaxis because of its long half-life and good side-effect profile. Evidence of its benefit is currently only anecdotal and, as it is not PBS funded, it is expensive. If a woman experiences frequent migraines at other times of the cycle, she should take a preventive agent, the dose of which may be increased perimenstrually if needed. (The choice of preventive agent would be made after considering other comorbidities, previous response and previous adverse reactions to preventives.)
Modulation of migraine by hormonal factors, including OCP use, should be carefully evaluated using a headache and menstruation diary. Both the combined OCP and the progesterone-only pill may ameliorate or worsen migraine headaches, and the effect must be assessed on an individual basis. In addition, the risk of OCP use itself must be considered. In women at lower risk for cardiovascular disease, the risk of OCP use may be minimised by using a low-oestrogen OCP (e.g. a preparation containing 20 mcg ethinyloestradiol). The progesterone-only pill and contraceptive implants do not carry any increased risk of thrombotic disease and may reduce migraine frequency and severity. Some patients with menstrual migraine respond to elimination of the placebo days by continuous cycling (with one withdrawal bleed every three months) and use of one of the mini-prophylactic regimens during the pill-free days. Starting an OCP purely to manage migraine would not be recommended; however, it may be used to maximise benefit and minimise risk in women requiring an OCP for other reasons. Polycystic ovary syndrome and endometriosis confers a different risk–benefit profile.
Migraine in pregnancy
Migraine may worsen or improve in the first trimester, but generally improves significantly for the remainder of the pregnancy. The greatest improvement occurs in women with menstrual migraine or those in whom migraine commenced at menarche. One-quarter of women will not notice any change in migraine frequency during pregnancy. Persistent headaches at the end of the first trimester are associated with a lack of improvement in migraine for the remainder of the pregnancy.
Women with migraine have a greater risk of pregnancy-induced hypertension and pre-eclampsia, as well as other vascular events in pregnancy and peripartum. There is a higher risk of pregnancy-related ischaemic stroke in women with migraines, with an odds ratio of 16.9, on a background risk of about 34 per 100,000 deliveries.6 A population-based cohort study in Denmark showed increased risks of low birthweight, preterm birth, caesarean section delivery, neonatal dispensings, respiratory distress syndrome and febrile seizures (adjusted prevalence ratio 1.2) in women with migraine.7
Other causes of headache in pregnancy must always be considered, including venous sinus thrombosis, pre-eclampsia, stroke, brain tumour, arteriovenous malformation and idiopathic intracranial hypertension. These should be considered particularly in women with new-onset headache, or new aura or a substantial change in their headache phenotype during pregnancy. The preferred method of neuroimaging in pregnant women is MRI, although the use of gadolinium is discouraged because of the limited evidence concerning short- and long-term fetal outcomes, particularly in first trimester use.
The management of women with migraine during pregnancy is difficult because most of the drugs used to treat migraine at other times have warnings about their use in pregnancy and lactation listed in the prescribing information. This leaves the clinician in a dilemma about whether or not to prescribe the drugs; an individual patient’s symptoms may be of such severity that intervention is considered essential despite some risk. In every case, the potential benefits must be balanced against the potential risks.
Migraine prophylaxis should ideally be optimised before conception, with a focus on using the safest agents. The use of nonpharmacological techniques such as relaxation, massage, acupuncture, regular exercise, adequate sleep and avoidance of triggers should be encouraged.8 Smoking cessation should also be reinforced, for a variety of health reasons, including reduced headache severity. If possible, women should be weaned off continuous preventives before conception or, if this is not possible, treatment switched to one of the safer prophylactic agents. Supplements such as magnesium 200 to 300 mg twice a day and coenzyme Q10 200 mg a day are likely safe, whereas the safety of riboflavin is less proven. Feverfew should be avoided, as it can induce uterine contractions and spontaneous abortion. Iron deficiency can increase migraine frequency, severity and duration, particularly in women with menstrually-related migraine, and screening and replacement should be considered.
Safer pharmacological options include beta blockers (although they should be tapered four weeks before delivery and their use is associated with a possible risk of growth retardation, especially with atenolol,9 and of fetal and newborn bradycardia) and calcium channel blockers (usually verapamil). Although antidepressants such as amitriptyline and venlafaxine have a lower TGA pregnancy rating (categories C and B2, respectively), they can be highly effective preventives, and have extensive clinical experience of use in pregnancy. They would therefore generally be used ahead of agents such as gabapentin, despite its B1 rating. Other anticonvulsants, particularly topiramate and sodium valproate, should be avoided.
For patients with chronic migraine, there is now some reassuring evidence that onabotulinumtoxinA carries very low risk in pregnancy.10 It may be a very useful option especially when a woman is trying to conceive. Calcitonin gene-related peptide antibodies are not established as safe in pregnancy, and because of their long half-lives need to be withdrawn about five months before conception is attempted.
A particularly difficult time may occur when a woman is trying to conceive. During this time, prophylaxis will usually have been ceased to avoid teratogenesis, but conception may not occur at once, especially in women who used the OCP for contraception. To increase a woman’s chances of achieving a quick conception, a switch from the OCP to barrier contraceptive methods for two or three months before ceasing prophylaxis may be of value.
Options for the treatment of pregnant women with acute migraine include paracetamol 1 g plus metoclopramide 10 mg or, during the second trimester only, NSAIDs. NSAIDs should be avoided in the first trimester because of concerns about an association with miscarriage and in the third trimester because of the risk of premature closure of the ductus arteriosus and oligohydramnios. Triptans may be used to treat pregnant women with moderate to severe migraine symptoms. There is a theoretical risk of uteroplacental vasoconstriction and increased uterotonic activity with their use, but pregnancy registers of sumatriptan (category B3) use are reassuring; sumatriptan is also considered safe to use when breastfeeding. Antiemetics such as prochlorperazine and ondansetron may be helpful both to treat nausea and for their antimigraine effects. Opiates may be used if a woman’s symptoms are severe, but carry a greater risk of inducing medication-overuse headache, as well as a risk of neonatal withdrawal if used near term. Women with prolonged migraines may require intravenous hydration and consideration of magnesium sulfate (1 g intravenous) or high-dose oral prednisolone (up to 75 mg a day for two days) or bilateral greater occipital nerve blocks. Ergotamines are absolutely contraindicated in pregnancy.
Migraine and breastfeeding
Half of women with migraine experience recurrence of headache in the first month postpartum. Most of these will be migraine, but it is worth noting that increased risk of reversible cerebral vasoconstriction syndrome and pre-eclampsia persist for six weeks postpartum.11
Breastfeeding may reduce headache incidence, but the data in this area are conflicting – certainly there is no evidence to suggest worsening of migraine in breastfeeding women.12
During breastfeeding, paracetamol, ibuprofen and sumatriptan may be used acutely. Aspirin is not to be used. Magnesium, riboflavin, beta blockers and verapamil also appear to be safe during breastfeeding. Cyproheptadine and ergotamine should be avoided.
Migraine in the perimenopausal period
The prevalence of migraine decreases with increasing age; however, menopause may increase, decrease or have no effect on migraine frequency. Surgical menopause is more likely to worsen migraine than natural menopause, presumably due to the rapidity of hormonal flux. Evaluation of migraine symptoms should be part of the assessment of menopausal symptoms in all women. When considering the use of MHT, it is useful to know that the oestrogen component of MHT may also worsen migraine, particularly at higher doses or with varying blood levels. This may be minimised by administering continuous, rather than cyclical, MHT, as well as the lowest effective dose of oestrogen by a nonoral route, to achieve more even blood levels. It appears that the transdermal application of hormone therapy is not associated with risk of stroke.13
Other pharmacological agents with evidence for efficacy in both migraine prophylaxis and reduction of hot flushes include the serotonin selective reuptake inhibitors (e.g. fluoxetine and venlafaxine [off-label use]) and gabapentin.
Conclusion
In all women, standard principles of migraine treatment apply. Women with infrequent headaches with a good response to acute treatment may be managed with intermittent acute therapies. Women with headaches occurring on more than four to five occasions per month may benefit from the addition of a preventive agent. Use of acute migraine treatments (triptans and NSAIDs) should be limited to two to three days a week to reduce the risk of transformation to chronic migraine and acute medication overuse.
Hormonal considerations in women may suggest the use of mini-prophylaxis around the time of menses, as well as guiding decisions about OCP use and awareness of the possible exacerbation of migraine perimenopausally. Prepregnancy counselling and planning in women with migraines is also of great importance.
Lifestyle factors remain important in the management of migraine in women, with the focus on limiting variations in daily routine that may trigger migraine. These include ensuring regular adequate sleep, regular healthy meals, identification and avoidance of any potential food triggers, avoidance or moderation of alcohol use, regular moderate exercise and the use of relaxation and stress management techniques. MT
COMPETING INTERESTS: Dr Hutton has served on advisory boards for Sanofi-Genzyme, Novartis, Teva, Eli Lilly, Allergan, Lundbeck, been involved in clinical trials sponsored by Novartis, Teva, Xalud, Cerecin, and has received payment for educational presentations from Allergan, Teva, Eli Lilly and Novartis. Dr Cheng has received lecture fees from Pfizer. Professor Stark has served on advisory boards for Teva, Eli Lilly, AbbVie, Viatris and Lundbeck; and received payment for educational presentations from AbbVie, Teva, Eli Lilly and Viatris.
References
1. Headache Classification Committee of the International Headache Society. The International Classification of Headache Disorders, 3rd ed. Cephalalgia 2018; 38(Suppl 1): 1-211.
2. Putaala J, Metso AJ, Metso TM, et al. Analysis of 1008 consecutive patients aged 15 to 49 with first-ever ischemic stroke: the Helsinki young stroke registry. Stroke 2009; 40: 1195-1203.
3. Kalkman D, Couturier E, Bouziani A, et al. Migraine and cardiovascular disease: what cardiologists should know. Eur Heart J 2023; 44: 2815-2828.
4. MacClellan LR, Giles W, Cole J, et al. Probable migraine with visual aura and risk of ischemic stroke: the stroke prevention in young women study. Stroke 2007; 38: 2438-2445.
5. Kurth T, Gaziano J, Cook NR, et al. Migraine and risk of cardiovascular disease in men. Arch Intern Med 2007; 167: 795-801.
6. James AH, Bushnell CD, Jamison MG, Myers ER. Incidence and risk factors for stroke in pregnancy and the puerperium. Obstet Gynecol 2005; 106: 509-516.
7. Skajaa, N, Szepligeti S, Xue F, et al. Pregnancy, birth, neonatal and postnatal neurological outcomes after pregnancy with migraine. Headache 2019; 59: 869-879.
8. Mauskop A. Nonmedication, alternative, and complementary treatments for migraine. Continuum (Minneap Minn) 2012; 18: 796-806.
9. Lydakis C, Lip GY, Beevers M, Beevers DG. Atenolol and fetal growth in pregnancies complicated by hypertension. Am J Hypertens 1999; 12: 541-547.
10. Wong HT, Khan R, Buture A, Khalil M, Ahmed F. OnabotulinumtoxinA treatment for chronic migraine in pregnancy: An updated report of real-world headache and pregnancy outcomes over 14 years in Hull. Cephalalgia 2025; 45: 3331024251327387.
11. Burch R. Epidemiology and treatment of menstrual migraine and migraine during pregnancy and lactation: a narrative review. Headache 2020; 60: 200-216.
12. Kvisvik EV, Stovner LJ, Helde G, Bovim G, Linde M. Headache and migraine during pregnancy and puerperium: the MIGRA-study. J Headache Pain 2011; 12: 443-451.
13. Lokkegaard E, Nielsen L, Keidine N. Risk of stroke with various types of menopausal hormone therapies: a national cohort study. Stroke 2017; 48: 2266-2269.