Donanemab: a novel treatment to slow early Alzheimer’s disease
Donanemab, a humanised monoclonal antibody targeting aggregated beta-amyloid in the brain, is the first disease-modifying, amyloid-targeting therapy for Alzheimer’s disease to be approved in Australia. GPs play a pivotal role in guiding diagnosis, co-ordinating multidisciplinary care and mitigating treatment risks.
- Donanemab is indicated for mild cognitive impairment and mild dementia due to Alzheimer’s disease (AD) in adults who are apolipoprotein E type ε4 heterozygotes with evidence of brain beta-amyloid pathology.
- Monthly intravenous donanemab infusions have been shown to significantly reduce brain beta-amyloid and modestly slow cognitive decline and clinical progression over 18 months. Sustained benefits up to 36 months in extension studies suggest a disease-modifying effect.
- Although approved by the TGA, donanemab (and the newly approved lecanemab) are not currently subsidised under the PBS, with total treatment costs estimated to exceed $80,000 per patient.
- Common adverse effects include infusion-related reactions and amyloid-related imaging abnormalities, which are mild in most cases but can be serious or fatal in rare instances.
- GPs play a crucial role in identifying eligible patients, understanding safety and monitoring requirements and supporting shared decision-making with specialist teams.
- Beyond pharmacological therapy, it is also vital that GPs implement best practice in the management of modifiable risk factors, support participation in research, offer nonpharmacological management options and address social and caregiver needs.
Dementia is the leading cause of death in Australia, and the most common cause of disease burden for Australian women and people older than 80 years of age. Over 400,000 people live with dementia in Australia today, with Alzheimer’s disease (AD) being the most common cause – it is believed to contribute to 60 to 80% of cases.1
AD is characterised by the gradual accumulation of extracellular beta-amyloid plaques and, subsequently, evidence of intraneuronal neurofibrillary tangles of hyperphosphorylated tau, neuroinflammation, synapse loss and brain atrophy. These features are associated with symptoms of progressive cognitive and functional impairment, and ultimately, dependency and death.2
Dementia claims a great personal cost to patients and caregivers, as well as representing a substantial burden on health and aged care services. An estimated $3.7 billion was spent on dementia diagnosis, treatment and care in 2020 to 2021.1
Recent years have seen an exponential growth in our understanding of the pathophysiology and diagnostic and therapeutic opportunities for dementia due to AD, culminating in the approval of three amyloid-lowering monoclonal antibodies by the US Food and Drug Administration: aducanumab, lecanemab and donanemab.3 Clinical trial evidence shows that this class of medications can significantly lower brain beta-amyloid, modulate downstream pathological processes (e.g. inflammation and abnormal tau deposition) and slow clinical progression in people with AD.3-5
Donanemab is a humanised monoclonal antibody that binds to and clears beta-amyloid plaques. It was approved for clinical use in Australia by the TGA in May 2025.6 Subsequently, lecanemab, a second amyloid-targeting therapy (ATT), was approved by the TGA in September 2025; this will be the subject of a separate, subsequent review. Neither drug has been approved for subsidy under the PBS.
The approved label and appropriate use recommendations for donanemab dictate important eligibility considerations, including the clinical severity of AD, requirement for demonstration of beta-amyloid burden, apolipoprotein E (APOE) type epsilon-4 allele (ε4) status and absence of exclusionary findings on MRI (e.g. baseline markers of severe cerebrovascular disease and cerebral amyloid angiopathy, including microbleeds and superficial siderosis).6,7
This class of medications also has important side effects for GPs to be alert to, including infusion-related reactions (IRRs) and amyloid-related imaging abnormalities (ARIA). These are commonly mild in nature, but serious adverse events can occur; therefore, careful patient selection and rigorous safety monitoring are crucial.7
GPs play a role in discussing treatment options with patients, considering eligibility, co-ordinating shared care with specialists and monitoring for clinical changes and potential treatment toxicities. This article outlines what GPs should know about the evidence and approval details for donanemab, including its indication and eligibility criteria, safety monitoring and common side effects, as well as important general treatment considerations for all patients with cognitive concerns, including those who may not be eligible for donanemab therapy.
What is donanemab?
Donanemab is a humanised immunoglobulin G1 monoclonal antibody that targets the N-terminus of beta-amyloid, preferentially binding to aggregated plaques. It received regulatory approval from the Australian TGA in May 2025, which was based on data from the Study of Donanemab (LY3002813) in Participants With Early Alzheimer's Disease (TRAILBLAZER-ALZ 2 [TBZ-ALZ 2]), a global phase 3 trial including 1736 people (aged 60 to 85 years; mean age, 73 years) with early AD (mild cognitive impairment [MCI] due to AD and early dementia due to AD).5,6 To be eligible, participants needed to have demonstrated evidence of brain beta-amyloid with low/medium or high pathological tau accumulation on positron emission tomography (PET) imaging. These categories of progression of tau pathology are reflective of the different biological stages of Alzheimer’s disease and are generally closely linked with symptom severity.5
Details of key study
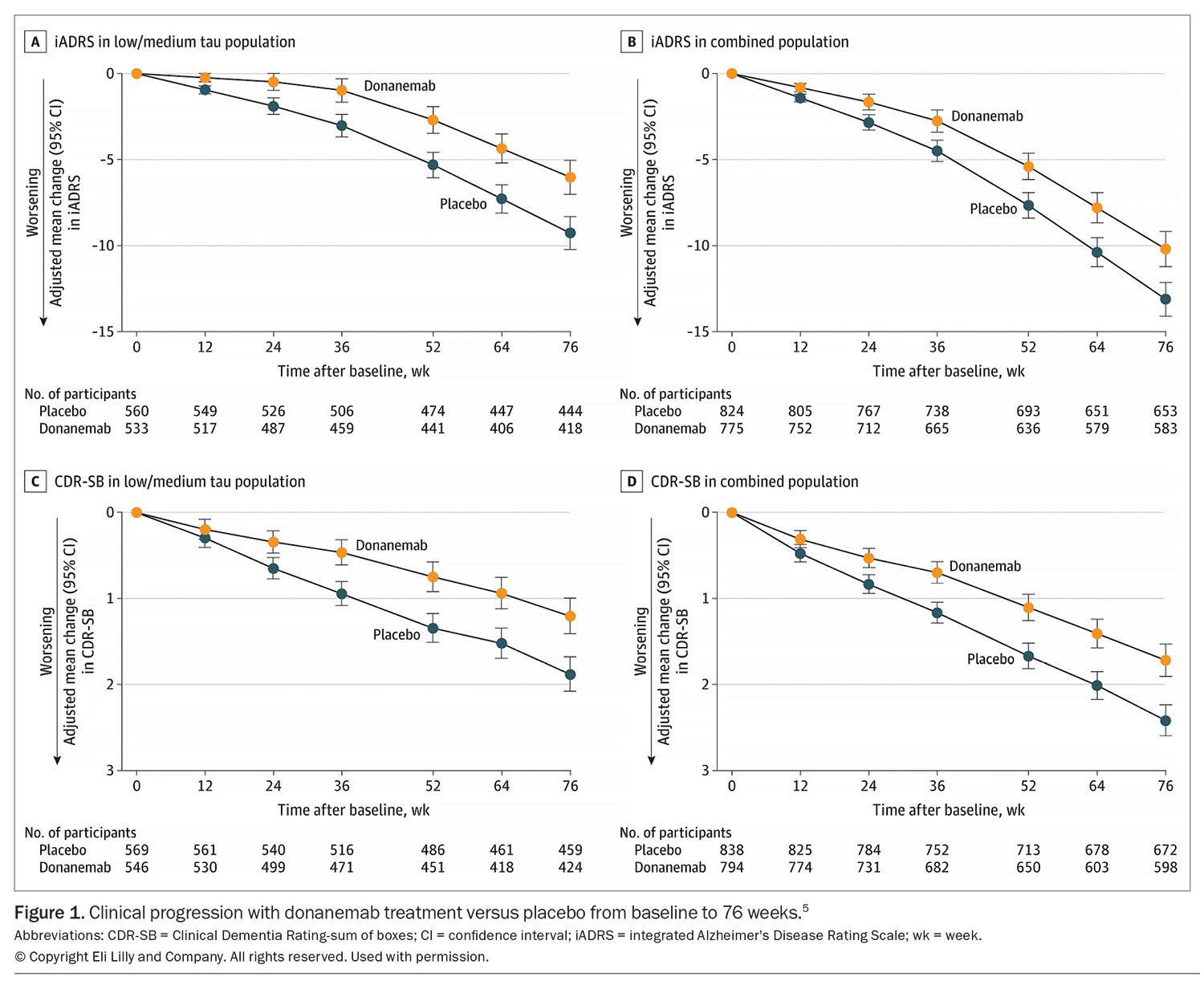
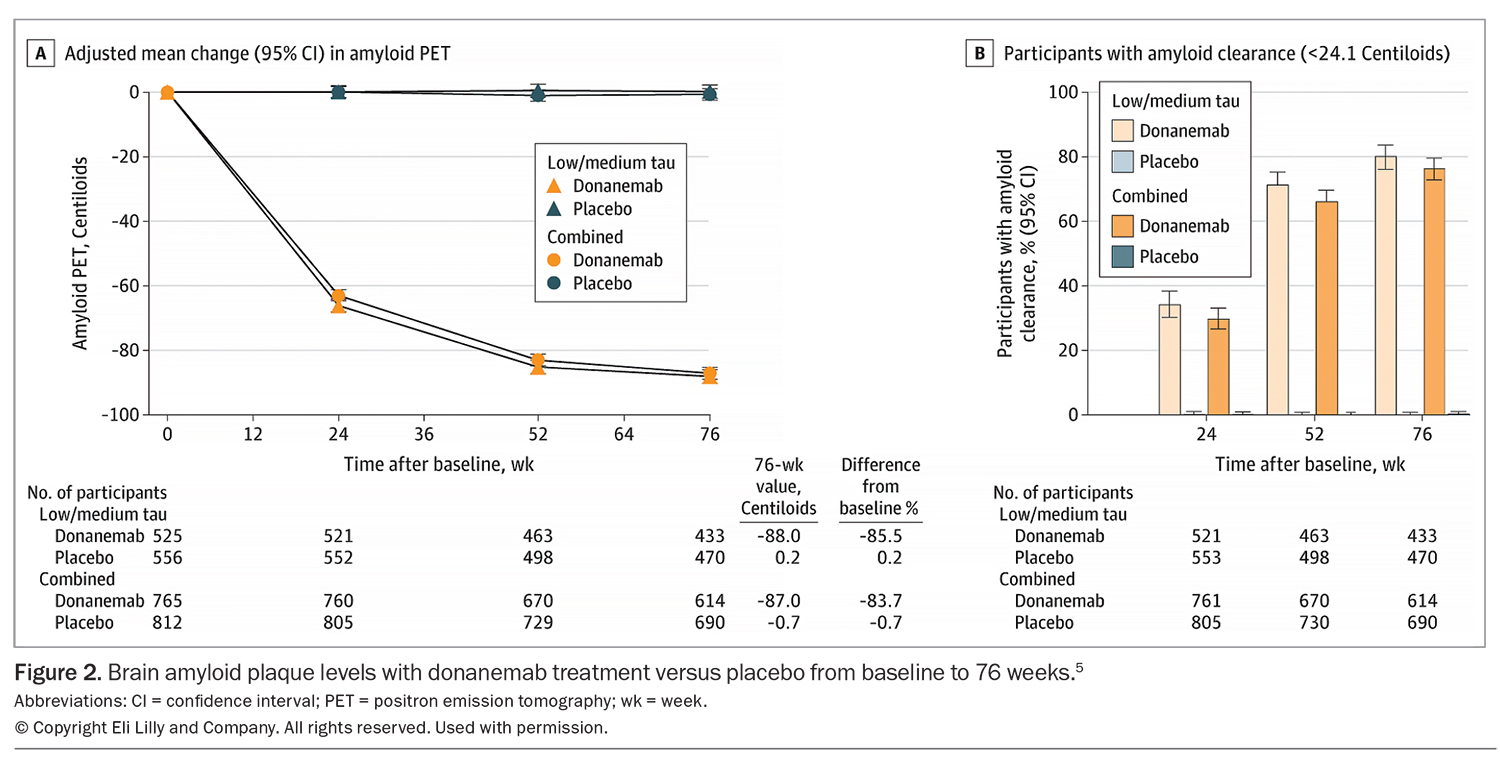
In the study, the participants received monthly intravenous infusions of donanemab or placebo over 18 months. There was also the opportunity to switch to placebo (in a blinded fashion) if there was clearance of beta-amyloid at six or 12 months.5 The primary endpoint was the integrated Alzheimer’s Disease Rating Scale (iADRS) score, which is an assessment of cognition and function, combining scores from the 13-item cognitive subscale of the Alzheimer’s Disease Assessment Scale and the Alzheimer Disease Cooperative Study – Instrumental Activities of Daily Living scale (Figure 1). Secondary endpoints included the Clinical Dementia Rating (CDR)-sum of boxes, a global measure of severity of cognitive and functional change, as well as beta-amyloid plaque reduction (on amyloid PET), tau PET changes, volumetric MRI changes and safety outcomes (including IRRs and ARIA) (Figure 2). Additional exploratory outcomes included plasma P-tau217, progression risk and clinical progression delay based on the iADRS score, CDR-sum of boxes and CDR-global score.5 In addition, A Study of Different Donanemab (LY3002813) Dosing Regimens in Adults With Early Alzheimer's Disease (TRAILBLAZER-ALZ 6 [TBZ-ALZ 6]) assessed the impact of different dosing regimens on important safety outcomes, including ARIA.8
Cognitive and functional outcomes
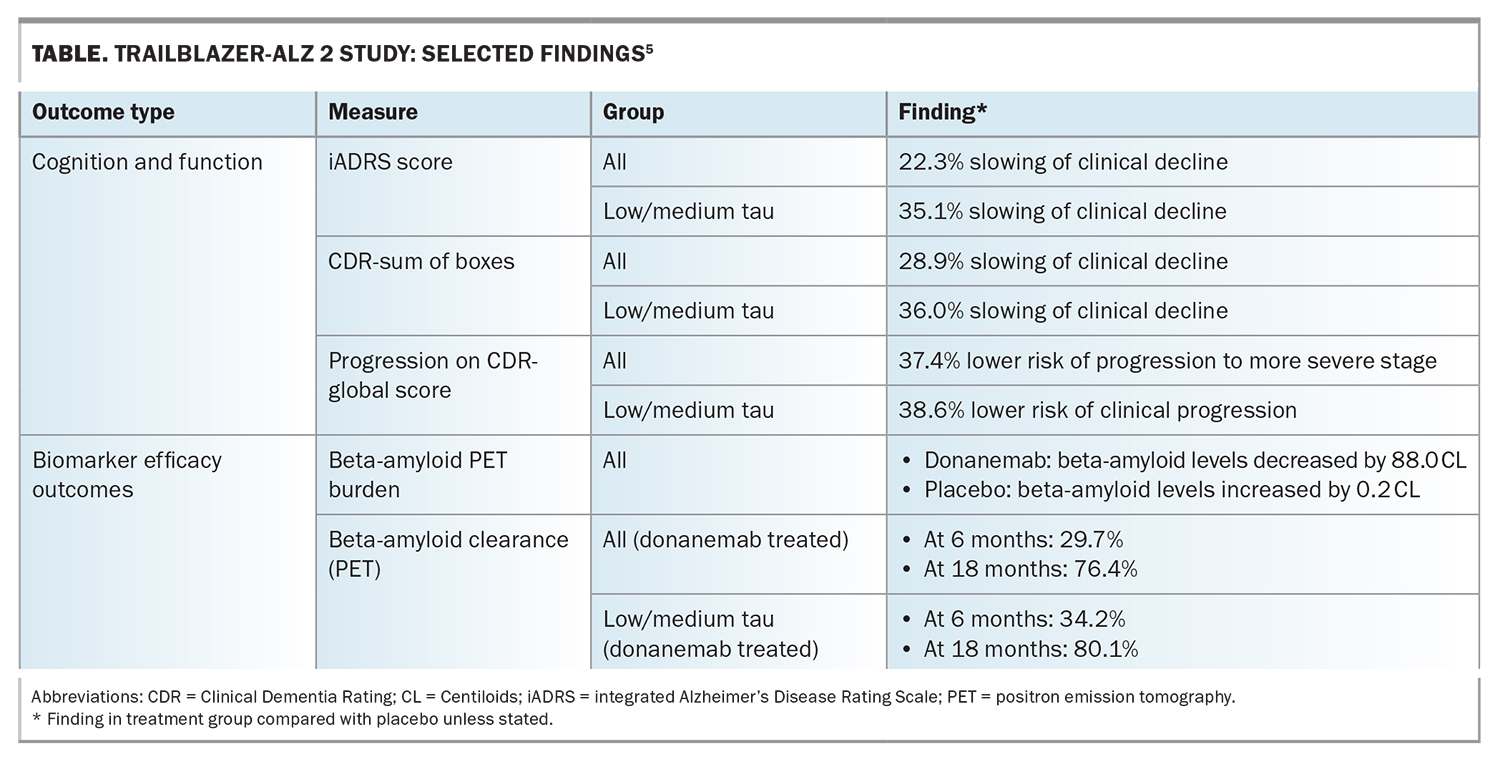
The TBZ-ALZ 2 study demonstrated significant slowing of cognitive decline in the primary endpoint (iADRS) and secondary endpoint (CDR-sum of boxes) in both the low/medium tau group and the combined (low/medium and high tau) group, compared with placebo (Table).5 There was also a significantly lower risk of advancement to a more severe disease stage or ‘clinically meaningful worsening’ of symptoms with donanemab treatment compared with placebo. In the long-term extension study, at 36 months of follow up, the treatment benefit increased even after the infusion regimen was completed. The ‘early start’ donanemab group showed a 27% reduced risk of progression to the next stage of disease compared with the ‘delayed start’ group, which commenced following the 18-month placebo-controlled period.9
Biomarker outcomes
Brain beta-amyloid plaque levels decreased significantly with donanemab treatment, whereas they increased with placebo (Table).5 The proportion of donanemab-treated patients that became beta-amyloid-negative was 34.2% at six months and 80.1% at 18 months in the low/medium tau group, and 29.7% at six months and 76.4% at 18 months in the combined group. Overall, 47% of participants achieved beta-amyloid clearance at six or 12 months and were switched to placebo during the study. Donanemab treatment was also associated with significant lowering of plasma P-tau217 compared with placebo.5 In the extension study, for participants who completed the treatment at 12 months, reaccumulation of beta-amyloid was slow (2.4 Centiloids/year), with mean beta-amyloid levels remaining in the ‘negative’ range (<24.1 Centiloids) at 36 months.9
When and in whom is donanemab used?
Donanemab is indicated for adults with early symptomatic AD: those with MCI or mild dementia, with confirmed beta-amyloid pathology via PET or cerebrospinal fluid testing, who are APOE ε4 heterozygotes or noncarriers.6,7
Subgroup analysis of the TBZ-ALZ 2 study suggests that people with milder symptoms and less advanced brain beta-amyloid and tau pathology appear to benefit more from the treatment than those with more advanced disease.5 Treatment is initiated by a specialist experienced in the diagnosis and treatment of AD, with co-ordination by a multidisciplinary team with the capacity to detect and manage adverse effects.
Donanemab is contraindicated in people with:
- hypersensitivity to donanemab or its excipients
- a baseline presence on MRI of prior intracerebral haemorrhage, more than two microbleeds, superficial siderosis or vasogenic oedema
- severe white matter disease on MRI
- an inability to participate in safety MRI surveillance scans.
Additionally, although not strictly contraindicated, caution is recommended when considering prescribing antithrombotic or thrombolytic medications to patients treated with donanemab because intracerebral bleeding events have been observed in this cohort.6,7
Carriage of the APOE ε4 allele is associated with higher rates of ARIA (particularly for homozygotes). As such, APOE ε4 homozygotes are excluded from treatment with donanemab under the TGA approval, although this is not exclusionary in other countries.
How is donanemab used?
The dosing regimen is 350 mg for the first dose, 700 mg for the second dose and 1050 mg for the third dose, followed by 1400 mg every four weeks. This dosing regimen is based on the TBZ-ALZ 6 study, which compared dosing regimens, finding superior safety and comparable biomarker efficacy with gradual uptitration compared with the regimen used in TBZ-ALZ 2.8 Treatment should continue until beta-amyloid plaques are cleared (using a validated method) for up to a maximum of 18 months.
The drug is diluted in 0.9% NaCl, administered as an intravenous infusion over approximately 30 minutes every four weeks. Donanemab should be administered in a setting with the capacity to manage adverse events such as IRRs.6
Possible side effects
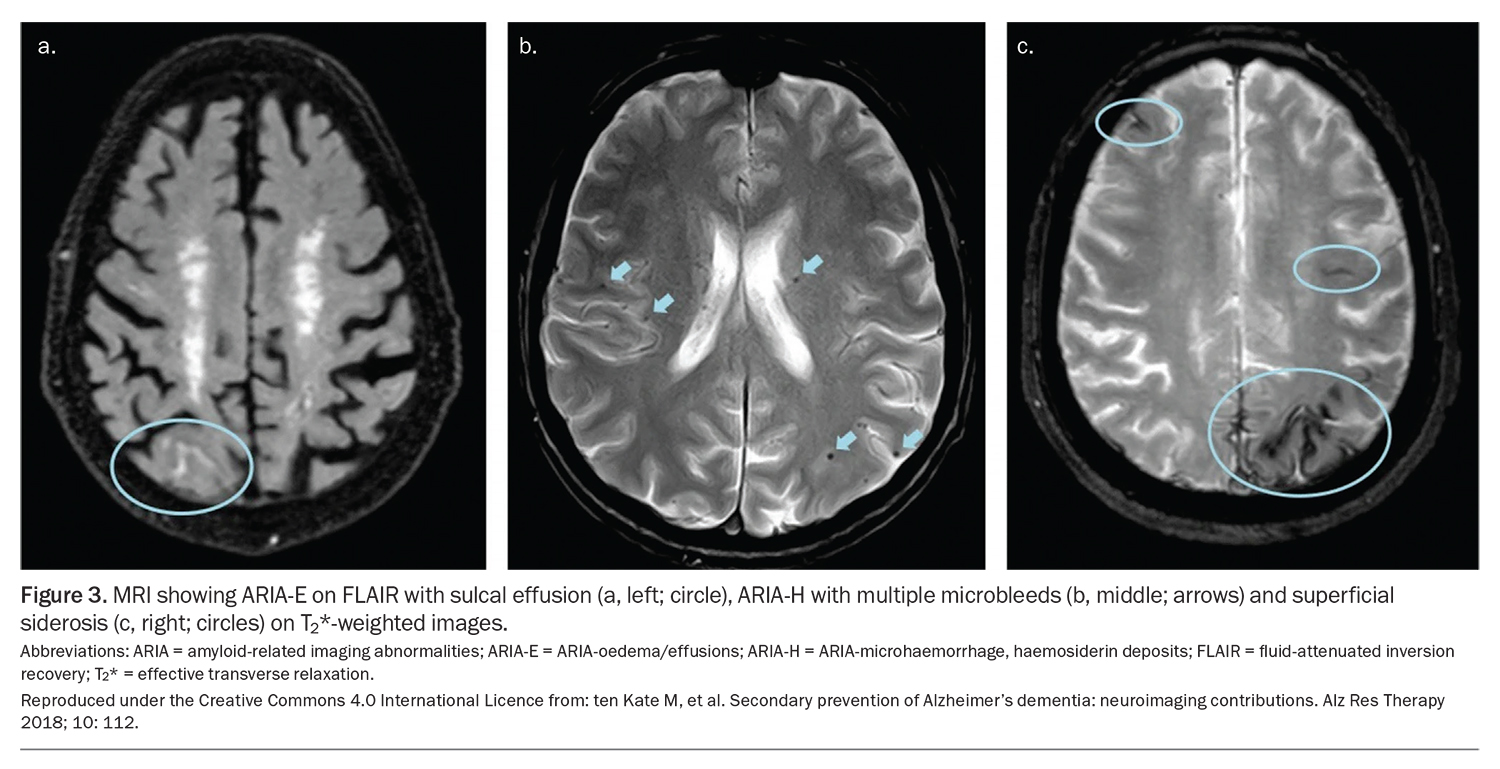
The possible side effects include ARIA and IRRs. ARIA-oedema/effusions (ARIA-E) and ARIA-microhaemorrhage, haemosiderin deposits (ARIA-H) are imaging changes detectable on MRI, believed to occur because of transient alterations in vascular permeability, as seen with ATTs. They can also occur spontaneously in people with cerebral amyloid angiopathy or hypertensive vasculopathy, albeit at a much lower frequency (Figure 3).10
ARIA occurred in 24% of the donanemab- treated patients in the TBZ-ALZ 2 study compared with 2% of placebo-treated patients (the incidence was lower [14%] in the modified titration arm of the TBZ-ALZ 6 study).11 Most participants were asymptomatic, but 6% were symptomatic, with headaches, focal neurological symptoms, seizures and death. Three ARIA-related deaths were reported in the main placebo- controlled TBZ-ALZ 2 study.5 In the TBZ-ALZ 2 extension study, one patient died of intracerebral haemorrhage after receiving a thrombolytic agent for focal neurological symptoms; subsequent MRI demonstrated ARIA as the likely cause for the initial presentation.12
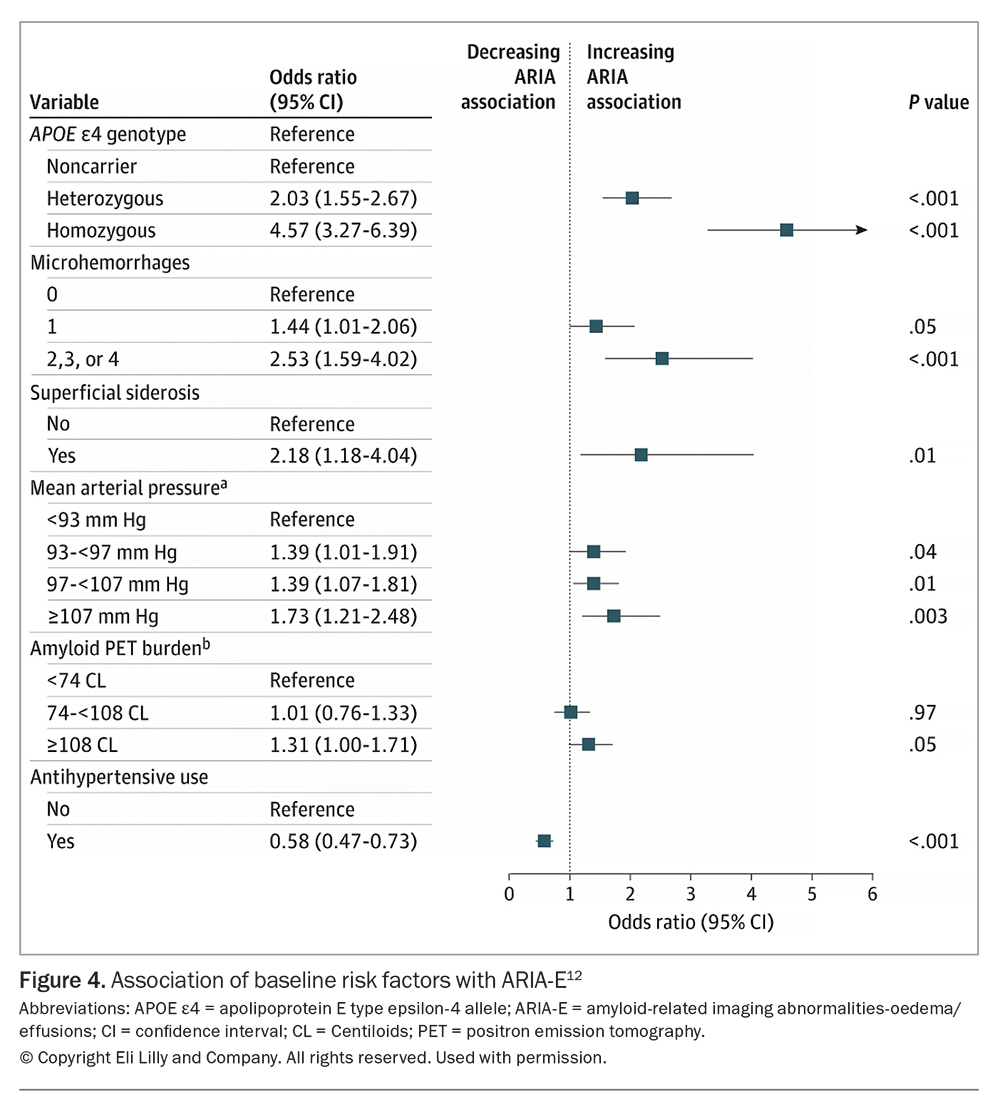
Most ARIA events occur within the first three infusions and are associated with baseline MRI markers (i.e. microbleeds and superficial siderosis) and a higher baseline amyloid PET burden. Other risk factors for ARIA include APOE ε4 carrier status, baseline evidence of cerebral amyloid angiopathy and elevated blood pressure (Figure 4).12 To minimise the risk of severe or symptomatic ARIA, safety MRIs are required prior to the second, third, fourth and seventh infusions.6 Identification of ARIA should prompt a review of the patient’s clinical status and consideration of suspension of dosing until clinical and radiological stabilisation. Symptomatic or severe ARIA needs timely MRI for confirmation and may require hospitalisation for clinical monitoring and consideration of treatment (e.g. corticosteroids).7 The use of antihypertensives is associated with a reduced risk of ARIA.12
IRRs occurred in 8.7% of the donanemab-treated participants; these included chills, rash, fever, nausea, vomiting and fluctuations in blood pressure. Most reactions occurred within 30 minutes of the infusion and were mild to moderate; allergic or hypersensitivity reactions occurred in three (0.4%) participants.5
Important precautions and interactions
- ARIA management. Suspend treatment if symptomatic ARIA or MRI worsens; if mild, asymptomatic or resolved, resume cautiously according to appropriate use recommendations.
- Caution with other agents. There are limited data on concurrent use with antiplatelets, anticoagulants, steroids, or immunosuppressants – use with caution.
- Pregnancy and lactation. Not studied – refer to specialist advice.
- Drug interactions. No strong metabolic interactions are expected; of note, people in trials were permitted to be on symptomatic AD medications.
- Genetic counselling. APOE genetic testing results should be communicated sensitively; APOE ε4 homozygotes are excluded from donanemab treatment in Australia, and carriage of APOE ε4 is associated with an increased risk of AD in offspring.
The role of the GP
GPs play an essential role in the identification, co-ordination and care for people with cognitive impairment and dementia. Some practical considerations for GPs are listed in the Box.
Awareness and early identification
Timely identification and workup of people with cognitive concerns are crucial to maximise benefits and minimise the risk for ATTs, as commencing treatment in milder stages (e.g. MCI rather than dementia) confers greater benefit than when commencing in more clinically severe or advanced stages of disease.13 In the future, there may also be a role for blood-based biomarker testing for AD by GPs to support eligibility for ATTs.
Monitoring and liaison with specialist centres
Shared-care models enabling both opportunistic and regular scheduled clinical monitoring are important for those receiving treatment. The role of the GP may include consultation for emerging symptoms, including the identification of possible side effects, and escalation of concerns, as well as consideration of risks, benefits and treatment tolerability for people who progress to more severe symptoms.7
Co-ordination of care, health promotion, management of comorbidities and supportive care
For all patients, there is a crucial role for identifying and managing other contributors to brain health, including exercise, mood, sleep and vascular risk factors. Providing the patient and caregivers with strategies to improve brain health is of great importance, particularly as there are likely to be many people living with dementia and cognitive impairment that will be ineligible for ATTs. There is also an increasing need for the ongoing support of patients and caregivers, as well as linkage to services and resources (e.g. MyAgedCare, Dementia Australia), as the cognitive, behavioural and physical symptoms of dementia progress.
Controversies and questions to consider
Donanemab vs lecanemab
A second ATT, lecanemab, was approved by the TGA in September 2025, and will be the focus of a separate, subsequent article. Although the phase 3 Study to Confirm Safety and Efficacy of Lecanemab in Participants With Early Alzheimer's Disease (Clarity AD) demonstrated broadly similar amyloid-lowering and clinical efficacy findings to that of donanemab, there are key differences in the therapeutic target, treatment protocol and certain reported outcomes between the two therapies.4 As a head-to-head study has not been conducted, a direct comparison cannot be drawn between them at this stage.
Are there clinically meaningful benefits?
There has been debate as to whether the cognitive and functional benefits shown in the TBZ-ALZ 2 study, although presented with high statistical significance, translate to clinically meaningful benefits for patients, with some having compared the effect size to those for existing symptomatic treatments. There are challenges with these comparisons, however, with substantial changes in clinical trial screening, recruitment and conduct over recent decades precluding simple comparisons between the groups. Additionally, this approach does not consider the potential for disease modification with ATTs, which is suggested by the widening of the treatment benefit seen in longer-term data to 36 months.9 However, longer follow up will be beneficial to assess this further. People with the mildest stages of disease clinically and biologically (e.g. MCI with low tau) appear to gain greater benefits from treatment, with slower rates of decline to more severe disease stages.
System readiness
There are concerns that our current systems of care are not prepared nor equipped to deliver some of the requirements of treatment with donanemab at scale, and costs could widen the existing inequities across demographics and regions. Treatment with ATTs such as donanemab requires the demonstration of the presence of beta-amyloid using one of two approved biomarker tests (in Australia, these include cerebrospinal fluid testing and PET), which are not currently funded by the Medicare Benefits Schedule and are not widely available. APOE genetic tests are not currently rebated. Requirement for specialist review, and multiple MRI scans for screening and surveillance will place pressure on existing services, which may already be stretched, particularly in regional and remote areas. MRI findings need to be reviewed by neuroradiologists trained in the detection and reporting of ARIA, using specific sequences and, ideally, consistent reporting templates.14 Treatment is delivered at supervised infusion centres, which require dedicated staffing and infrastructure.15 Emergency department clinicians need to be prepared for the management of complications of treatment, including IRRs and ARIA.
Cost and reimbursement
As the Pharmaceutical Benefits Advisory Committee decided not to subsidise donanemab, the medication and ancillary costs (investigations and surveillance) for a course of donanemab treatment are suggested to be well in excess of $80,000.15 Barriers of cost and limited access to diagnostic and imaging services are likely to exacerbate the existing disparities in clinical outcomes between advantaged and disadvantaged demographic groups.
Conclusion
The approval of donanemab for the treatment of MCI and mild AD opens a new treatment paradigm for AD in Australia. It confers significant slowing of cognitive and functional decline, but there are well-documented risks and precautions. GPs and patients need to ensure that thorough screening, individualised shared decision-making and careful surveillance are conducted to ensure safe and appropriate delivery of this important therapy. Where possible, policymakers and service providers should consider and address the existing system disparities to improve access to diagnosis and care for this and future advances in dementia care. MT
COMPETING INTERESTS: Associate Professor Yates has received payment for advisory board participation and presentations from Eli Lilly, Eisai and Novo Nordisk.
This article is for general information purposes only, and the full Product Information should be consulted before prescribing any of the mentioned medications.
References
1. Australian Institute of Health and Welfare (AIHW). Dementia in Australia. Canberra: AIHW; 2024. Available online at: https://www.aihw.gov.au/reports/dementia/dementia-in-aus (accessed October 2025).
2. Long JM, Holtzman DM. Alzheimer disease: an update on pathobiology and treatment strategies. Cell 2019; 179: 312-339.
3. Kim BH, Kim S, Nam Y, Park YH, Shin SM, Moon M. Second-generation anti-amyloid monoclonal antibodies for Alzheimer’s disease: current landscape and future perspectives. Transl Neurodegener 2025; 14: 6.
4. Van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early alzheimer’s disease. N Engl J Med 2023; 388: 9-21.
5. Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 2023; 330: 512-527.
6. Therapeutic Goods Administration (TGA). Australian Product Information – KISUNLA donanemab 350 mg/20 mL concentrated solution for intravenous infusion vial (420194). Canberra: Australian Government Department of Health, Disability and Ageing, TGA; 2025. Available online at: https://www.tga.gov.au/resources/artg/420194 (accessed October 2025).
7. Rabinovici GD, Selkoe DJ, Schindler SE, et al. Donanemab: appropriate use recommendations. J Prev Alzheimers Dis 2025; 12: 100150.
8. Wang H, Nery ESM, Ardayfio P, et al. The effect of modified donanemab titration on amyloid-related imaging abnormalities with edema/effusions and amyloid reduction: 18-month results from TRAILBLAZER-ALZ 6. J Prev Alzheimer’s Dis 2025; 12: 100266.
9. Mintun MA. Donanemab in early symptomatic alzheimer’s disease: efficacy and safety from the TRAILBLAZER‐ALZ 2 long-term extension [Conference presentation]. Toronto, Canada: Alzheimer’s Association International Conference (AAIC); 2025. Available online at: https://medical.lilly.com/us/science/conferences/ineuroscience/aaic2025 (accessed October 2025).
10. Sperling R, Salloway S, Brooks DJ, et al. Amyloid-related imaging abnormalities in patients with Alzheimer’s disease treated with bapineuzumab: a retrospective analysis. Lancet Neurol 2012; 11: 241-249.
11. Wang H, Serap Monkul Nery E, Ardayfio P, et al. Modified titration of donanemab reduces ARIA risk and maintains amyloid reduction. Alzheimers Dement 2025; 21: e70062.
12. Zimmer JA, Ardayfio P, Wang H, et al. Amyloid-related imaging abnormalities with donanemab in early symptomatic Alzheimer disease: secondary analysis of the TRAILBLAZER-ALZ and ALZ 2 randomized clinical trials. JAMA Neurol 2025; 82: 461-469.
13. Low LF, Daly S, Long M, et al. Mild cognitive impairment and dementia: postdiagnostic and ongoing care. Med Today 2025; 26(5 Suppl): 7-12.
14. Cogswell BPM, Barakos JA, Barkhof F, et al. Amyloid-related imaging abnormalities with emerging Alzheimer disease therapeutics: detection and reporting recommendations for clinical practice. Am J Neuroradiol 2022; 43: 9.
15. Maher S, Pulle RC. Are Australians living with dementia ready for new therapies? Intern Med J 2025; 55: 1059-1062.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.