Continuing care of chronic schizophrenia: many pieces in the jigsaw

Schizophrenia is a chronic mental illness requiring ongoing care. Continued antipsychotic treatment, co-ordinated GP involvement and psychosocial interventions support recovery, maintain connections with family and the community, and reduce disability.
- Effective care for schizophrenia requires psychosocial interventions that help support community engagement and reduce disability.
- The GP plays an important role as the central co-ordinator for the array of services needed, many of which are now delivered by non-government organisations.
- People with schizophrenia can benefit from supports available through the National Disability Insurance Scheme; these complement, but do not replace, comprehensive medical care.
- Continued treatment with antipsychotic medication is essential for most people with chronic schizophrenia to maintain stability and prevent relapse.
- Recovery in mental health is a multidimensional process, encompassing improvements across physical, psychological, social and cultural domains, as defined by the individual.
The unusually frank discharge letter outlined in Box 1 asks the GP to take over the care of a person with a complex and chronic mental illness. However, good quality care requires a system of services to address the biopsychosocial problem of a severe mental illness and its resultant disability. Few areas of Australia have access to a coherent and effective mental health system. Instead, a network of services exist that may address a range of issues but seldom comprehensively or cohesively. This fragmentation of care is worsened by the confusion of funding between state and federal governments, which has led to the development of a poorly co-ordinated set of services that differ from region to region. What is consistent for almost all people living with schizophrenia is the trusted and ongoing role of GPs. For almost all people with schizophrenia, their GP remains a key point of continuity, even when specialist and community services are fragmented or unavailable.
This article aims to provide GPs with a deeper understanding about the continued management of people with schizophrenia. Although medication is essential, psychosocial interventions are also important to improve long-term outcomes. This article should be read as a companion to the article published in Medicine Today in July 2025 on the initial care of a person presenting with schizophrenia.1
Recovery
For most people, schizophrenia is a chronic, long-term mental illness. Therefore, care should be planned to be long term and ongoing, providing a stable foundation from which the person can recover after the destabilising effects of an acute psychosis, which may remove control of their lives, undermine key relationships, interrupt work or education, and potentially disconnect them from stable housing and the community.
The concept of recovery places the individual within a broader psychosocial and cultural context, in which they regain control of their life, rebuild connections with family and the community, and create a life with meaning for them.2 Good quality medical care is essential, but on its own is insufficient, because it frequently does not address the psychosocial dimensions of care. Recovery requires many partners, but the direction taken is decided by the individual at the centre.
Treatment: more than medication alone
Disability in people with schizophrenia
In Australia, chronic schizophrenia is associated with high levels of disability, low rates of employment and high rates of welfare dependency. This is often accompanied by social isolation and marginalisation. Many people with schizophrenia have few friends and often depend on ageing parents for connection.3 These difficulties are largely due to the negative symptoms (such as poverty of thought and content of thought, blunted affect, amotivation and asociality) and cognitive deficits (such as problems with attention, concentration, memory, planning, speed of processing, emotion recognition and theory of mind – the ability to imagine the world from the perspective of another person).4,5
Although medications can reduce negative symptoms, antipsychotics provide little relief for cognitive impairments and the older first-generation antipsychotics, such as chlorpromazine, haloperidol and zuclopenthixol, may even exacerbate them.6,7 Cognitive enhancing medication, such as acetylcholinesterase inhibitors or memantine, are also ineffective in the treatment of cognitive symptoms in schizophrenia.8 Treatments, therefore, depend on psychosocial interventions, which show small to moderate benefits and are difficult to access in our current healthcare systems.
Connection with care
People with chronic schizophrenia often drop out of clinical care and, critically, out of our communities. Keeping them engaged and integrated in their network is important. Traditionally, this has been achieved through the allocation of a case manager from a community mental health team. There is good evidence that assertive community case management by a team of mental health workers (nurses, social workers, psychologists and a psychiatrist) with a low case to worker allocation decreases rehospitalisation and improves retention to care for people with a serious mental illness who are acutely unwell.9 The benefit of this intensive model lessens as the person’s condition improves. Lower intensity community case management with a higher case load does have a small positive effect on hospitalisation and quality of life.10 This benefit to clinical and personal recovery is matched by that seen with care provided by peer workers (people with a lived experience of serious mental illness working in a community team) providing psychoeducation and support.11 In summary, the evidence supports an assertive community care for people with an active psychotic illness, but also less intensive forms of community care provided by a case manager or peer worker via a community mental health team or a non-government organisation for people who are not severely unwell but who do have a serious mental illness.
It is essential for GPs to be integrated into the overall care provided. The GP’s role varies from administering long-acting injectable (LAI) antipsychotics to being the central point of co-ordination between services funded under mental health care plans. A key step is identifying case managers or peer workers and establishing open lines of communication. Unfortunately, with the high turnover of case managers, maintaining these networks can be challenging. In practice, many people with schizophrenia are discharged back to the care of their GP with minimal ongoing support from community mental health teams or non-government organisations, particularly if they are considered ‘settled’ but decline engagement. This makes it even more important that GPs are familiar with the range of services that may be required if they become willing to engage again.
Psychoeducation about mental illness
Psychoeducation is one of the simplest yet practical interventions. Ideally delivered over several structured sessions, it can also be provided in a single session if necessary. It provides the individual and their family with information about the nature and effects of their illness, available treatments (both pharmacological and psychosocial), relapse prevention strategies and the roles of different professionals involved in care (e.g. what is the difference between a psychiatrist and a psychologist?). It can also help clarify common concerns, such as whether hospitalisation under the Mental Health Act creates a criminal record. Providing this information has been shown to decrease rehospitalisation and improve satisfaction with care.12
Family work
The onset of schizophrenia in a child or sibling is profoundly challenging for most families. Despite education and reassurance, feelings of guilt about causing the illness can remain. This guilt is often mixed with frustration with the apathy and poor motivation associated with the negative symptoms of the illness, as well as fear. This combination often leads to conflict within the household.
From the perspective of the person with schizophrenia, the family environment may feel overly critical and lacking in understanding. Family therapy aims to increase knowledge about schizophrenia, build understanding of each family member’s perspective, improve communication and develop problem-solving skills. Family therapy has a long history in the management of schizophrenia with evidence supporting its effectiveness in reducing relapse and improving family interactions.13 A diverse range of family therapy techniques are effective; however, access to trained family therapists remains limited. Family therapy should not be offered to families with no significant issues, as it may be counterproductive.14
Cognitive behavioural therapy for psychosis
Cognitive behavioural therapy for psychosis applies the same principles used in other mental health disorders, exploring links between thoughts, emotions and behaviours. Individuals are encouraged to monitor their symptoms, identify the faulty reasoning that led them to their beliefs in delusions or hallucinations, and test alternative explanations. Evidence supports the effectiveness of cognitive behavioural therapy for psychosis in reducing symptom intensity and improving quality of life, particularly in the short to medium term.15 However, therapy usually requires 16 or more sessions, which may be difficult to access under the Better Access Medicare scheme.
Cognitive remediation therapy
Cognitive remediation therapy targets the neurocognitive and social cognitive deficits that are common in people with schizophrenia, including impairments in concentration, memory and planning, emotion recognition, thinking from another person’s perspective and understanding social norms.16-18 These deficits typically present at the time of first presentation to services and remain relatively stable over time,17 making it difficult for the individual to function effectively in the community. Cognitive remediation therapy involves structured cognitive exercises and the development of strategies to transfer these skills into real-world scenarios. Cognitive remediation therapy provides an excellent foundation when combined with other psychosocial treatments.19
Supported employment
Re-engagement in work is a significant achievement offering financial independence, reconnection with a broader community and improved self-esteem. Supported employment (also known as Individual Placement and Support) has the best results in returning people with schizophrenia to work.20 This approach involves placing the person directly into the workplace and providing tailored ongoing support. Several disability employment services across Australia practice this approach with funding linked to assessment and successful placement in employment.
Role of antipsychotic medications
Antipsychotic medication remains central to the long-term control of the positive and negative symptoms of schizophrenia. Stopping antipsychotic medication is associated with relapse, even after many years of treatment.21,22 Continued antipsychotic treatment not only reduces the risk of relapse but increases the likelihood of returning to work and improves quality of life and social functioning.23,24 Even a reduction in dose is associated with an increased risk of relapse,25 although this is frequently negotiated between the doctor and patient. A reduced dose of medication remains better than no medication at all.
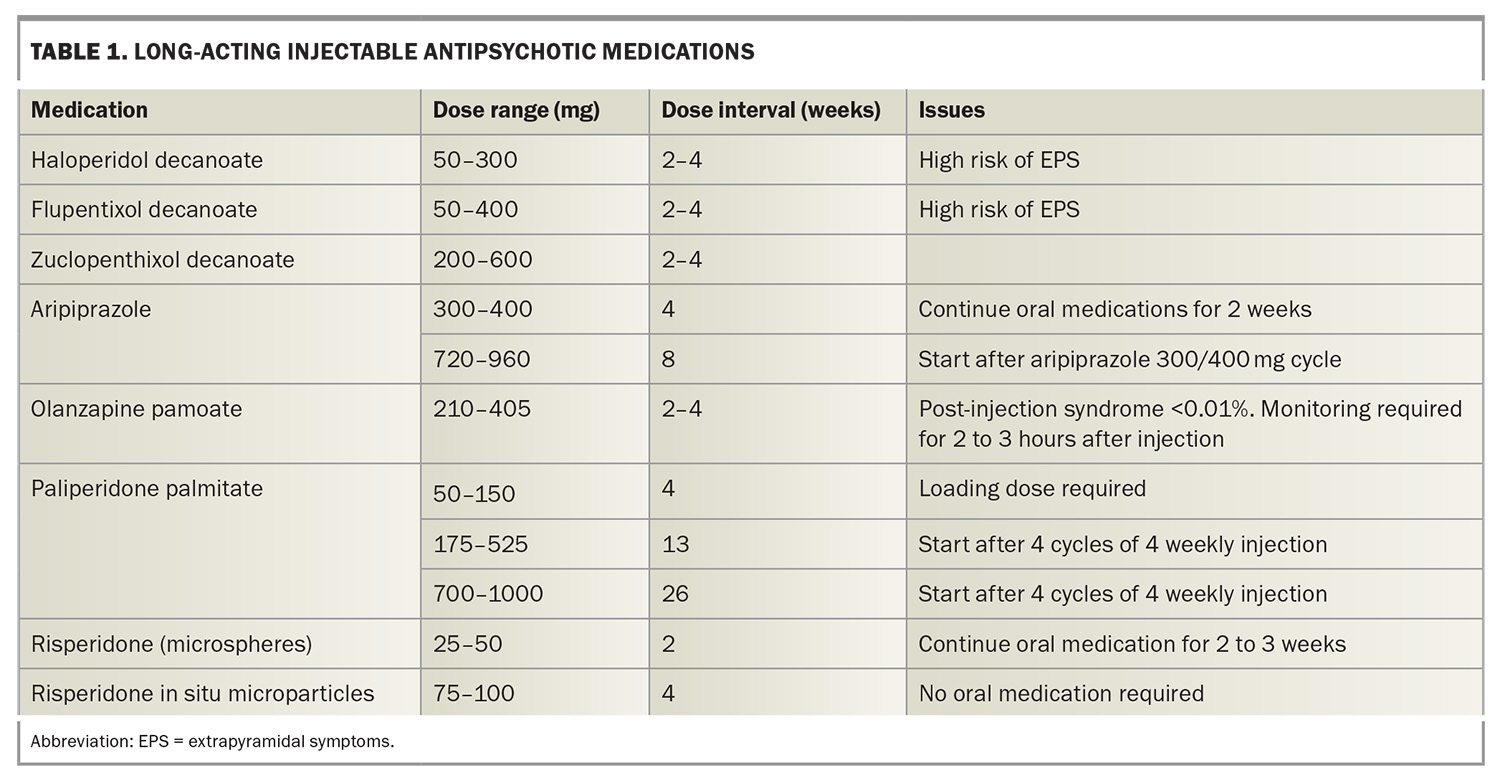
An underutilised way of enhancing medication adherence is the use of LAI preparations. Many, although not all, antipsychotic medications are available as LAI preparations. These preparations can provide therapeutic doses for up to six months. Compared with oral antipsychotic medication, they are associated with reduced risk of relapse and rehospitalisation and improved quality of life.26 They can also reduce conflict at home over whether the oral medication has been taken. Unfortunately, in Australia, LAIs are often linked with community treatment orders mandating compulsory treatment, which has not enhanced their use outside of this restrictive practice.
As the medication chosen is likely to be used for a prolonged period, the choice should be guided by patient preference, taking into account side effects such as sedation, weight gain and movement disorders (Table 1). LAI preparations are less likely to cause side effects than the oral equivalents.27
To support adherence, practices should implement reminder systems for upcoming injections and have a clear follow-up process for missed appointments, such as phone calls, rescheduling, or liaison with case managers or psychiatrists. Regular injection visits also provide valuable opportunities to review broader aspects of care, including physical health monitoring.
Treatment resistance in schizophrenia
About 20% of people with schizophrenia show treatment resistance from their first presentation with a psychotic illness. This proportion rises to about one-third when including those who relapse and subsequently fail to respond to antipsychotics (see part 2 of the case study in Box 2).28 The treatment of choice for this group is clozapine. About 30 to 40% of people with treatment-resistant schizophrenia will respond to clozapine.29 Clozapine is also helpful for complex clinical problems such as persistent suicidality, substance abuse, aggression and antipsychotic-induced movement disorders.30
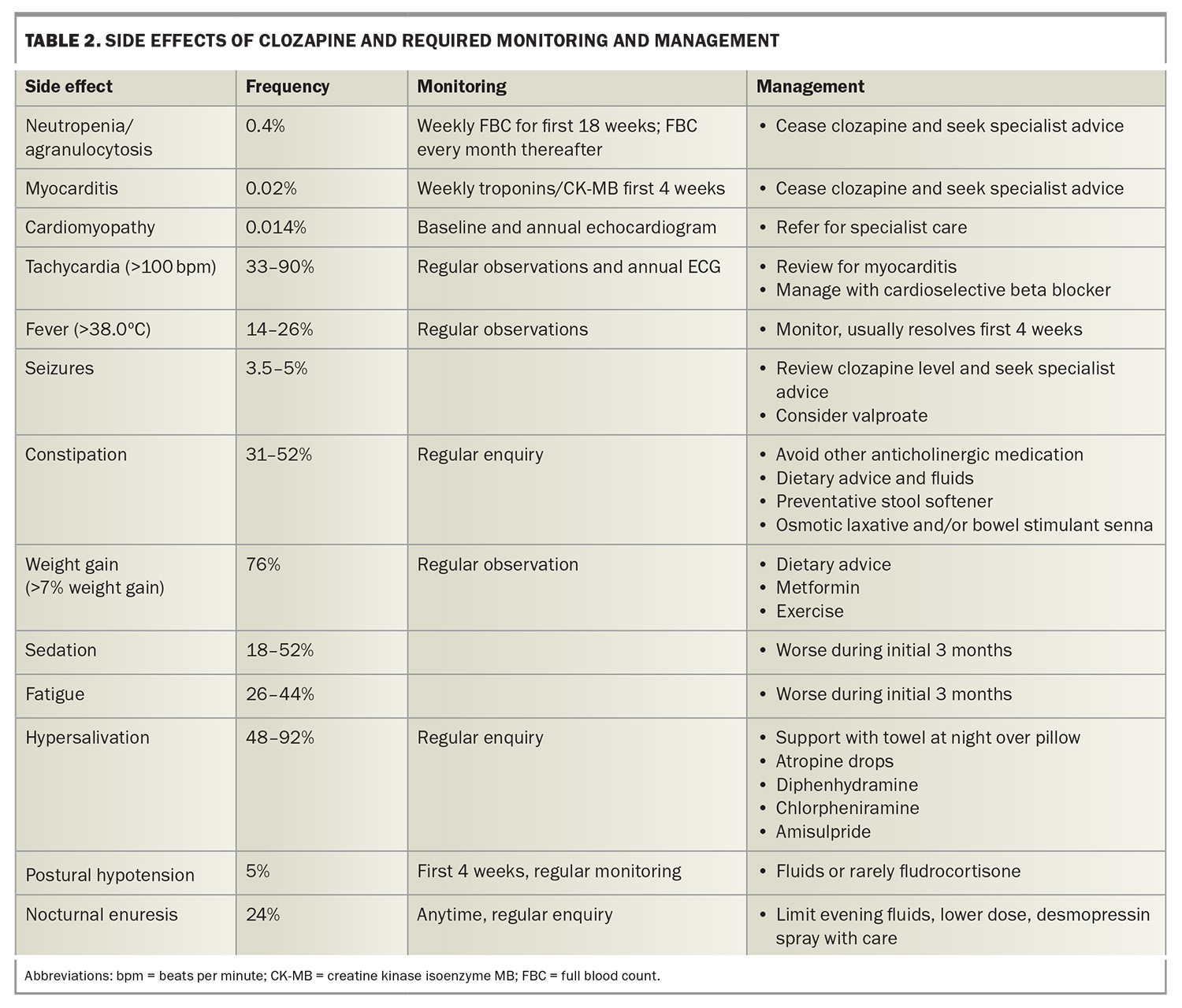
Clozapine initiation is restricted because of significant risks, particularly agranulocytosis and acute cardiomyopathy, and is undertaken in hospital under psychiatric supervision. Eligibility requires nonresponse to at least two antipsychotics, each prescribed for a minimum of six weeks at adequate doses with more than 80% adherence. It is important not to delay the initiation of clozapine as the likelihood of a good response decreases if the person has a period of treatment resistance of more than 2.5 years.31
Clozapine is a S100 medication requiring an authority prescription. Patients are compulsorily entered into a treatment register run by the manufacturers that requires a white cell count (>3.0 × 10⁹/L) and neutrophil count (>1.5 × 10⁹/L) in the 48 hours before clozapine can be dispensed. Clozapine is started by a psychiatrist, and the initial 18 weeks of treatment is usually continued under the care of the specialty team, but thereafter the monitoring responsibility can be transferred to GPs in a shared care arrangement with psychiatrist review every six months. GPs and the dispensing pharmacy also need to be registered with the local clozapine prescribing system. Clozapine is associated with a broad range of side effects and careful monitoring and review are required (Table 2).29,32 Despite these challenges, clozapine remains the most effective treatment for refractory schizophrenia.33 Continued care with clozapine is also associated with an overall reduction in mortality.34
What if my patient wants to stop their antipsychotic?
It is common for people with schizophrenia to consider stopping their medication. In one study, only a third of people with schizophrenia said they were happy to continue to take antipsychotic medication in the long term, whereas 31% said they would like to stop medication and 45% said they would like to reduce the dose of medication (overlapping questions, so the total is not 100%).35 The main concerns were side effects, particularly those affecting physical health. Importantly, most participants wanted professional support to guide the process.35
When medication reduction or cessation is being considered, a second opinion or psychiatrist involvement is advisable. After discussing the risks and benefits, a gradual taper should be planned and agreed upon, with regular follow up. Family members or supports should be included, and an action plan established to recognise early relapse signs and what to do if this occurs, including consideration of involuntary care. Relapse may not occur for more than a year after dose reduction, so monitoring over an extended period is essential. Dose reduction should be very gradual (ideally over months to years) in steps no greater than 10% of the baseline dose to avoid relapse secondary to dopaminergic hypersensitivity.36 Despite this, relapse is common and significantly more likely than if the medication dose is maintained.37 The principles for dose reduction are outlined in Box 3.
Physical health of people with schizophrenia
People with a severe mental illness such as schizophrenia die 15 to 20 years earlier than other people in the general population.38 Although there is an increased risk of suicide, especially early in the illness, most premature deaths are due to cardiovascular disease and cancer. Continuing antipsychotic treatment, including clozapine, is associated with lower all-cause mortality despite their side effects.39
People with schizophrenia have high rates of modifiable risk factors, including obesity (45.1% vs 21.0% in the general population), metabolic syndrome (49.1% vs 22.1% in the general population) and tobacco use (71.1% vs 25.3% in the general population).40,3 Poor diet and physical inactivity are also common. Recognition of these issues has improved, but consistent screening, investigation and management remain lacking.41,42 Barriers include socioeconomic disadvantage and therapeutic nihilism among some healthcare professionals.41 GPs play a crucial role in proactively addressing physical health needs, applying the same standards of care as for other patients. Recommended screening intervals were outlined in the first article in this series.1
Social aspects of care
The social aspect of care is fundamental in the treatment of schizophrenia. Without stable housing and financial support, it is very hard to provide clinical care for someone with schizophrenia. Depending on the level of disability, a broad range of interventions may be required, from help with employment to high-level supported accommodation (see part 3 of the case study in Box 4).
Disability Support Pension
Not all people with schizophrenia require the Disability Support Pension, but a large proportion find competitive employment hard to sustain. Supporting an application for the Disability Support Pension, provided they meet Centrelink’s criteria (i.e. they are unlikely to return to work or study in the coming two years), is important as it provides financial security and access to other services that would otherwise be unavailable. GPs can help by providing medical evidence of diagnosis and likely disability duration.
Non-government organisations
Non-government organisations (e.g. Mind Australia, One Door, MIFWA, Wellways) are increasingly providing community-based support, including transport, group programs, supported employment and housing. They can be accessed via the organisation’s own websites or via primary health networks. However, their ability to deliver these services depend on state, territory and federal government programs that vary between states and territories.
National Disability Insurance Scheme
People with a significant level of disability should be eligible for National Disability Insurance Scheme (NDIS) funding to more intensively support their needs. Unfortunately, people with mental health disorders have a low rate of acceptance into the scheme compared with other areas of disability. GPs are well placed to co-ordinate applications by referring individuals to occupational therapists for functional assessments, together with psychiatric, cognitive and medical reports. Some large NDIS providers offer guidance on preparing applications.
Driving assessments
Fitness to drive assessments vary by state and territory and often require psychiatric input. GPs should consider whether the illness or treatment:
- impairs insight relevant to driving
- causes symptoms, such as auditory hallucinations, that might affect driving
- changes behaviour
- impairs cognitive abilities (e.g. poor concentration, sedation).
If uncertain, a formal assessment of fitness to drive by an occupational therapist should be considered.
Conclusion
Schizophrenia affects one in every 100 to 200 people, making it likely that most general practices will be caring for several people with the disorder. Good quality care requires a team of healthcare professionals, with GPs having a central role in co-ordination and management, particularly given the increased risk of premature death from cardiovascular and oncological diseases. Although access to psychiatry and public mental healthcare services vary, non-government organisations and other community resources are playing an increasing role in psychosocial support. For most patients, continuing antipsychotic medication remains the foundation of recovery. Good quality of life and recovery are possible for most people with schizophrenia, but the complex jigsaw puzzle of services requires skill and experience to piece together. MT
COMPETING INTERESTS: Professor Harris has received industry sponsored trial payment to the Westmead Institute of Medical Research from Alto Neuroscience; has received grants as an Investigator from the Australian Research Council, the Medical Research Futures Fund and the National Health and Medical Research Council; has received payment or honoraria from Servier for educational meetings; is Director of Mind Australia; and is Chair of the Research Trust Fund of the Schizophrenia Fellowship of New South Wales. Dr Cannon has received ongoing grant funding for insomnia research from the Medical Research Futures Fund; has received payment for involvement in educational events from United Clinical Australia and Pfizer; participates on the Migraine advisory board for Pfizer; and is Co-Chair of the Primary Care Council at the Australasian Sleep Association. Dr Shivakumar has received grant funding from the Greek Young Matrons’ Associations. Dr Holdenson Kimura: None.
References
1. Shivakumar G, Cannon B, Holdenson Kimura P, Harris A. The first psychotic episode: navigating early-stage schizophrenia and other psychoses. Med Today 2025; 26(7): 12-21.
2. World Health Organization (WHO). Guidance on community mental health services: promoting person-centred and rights-based approaches. Geneva: WHO; 2021. Available online at: https://www.who.int/publications/b/57927 (accessed September 2025).
3. Morgan VA, Waterreus A, Jablensky A, et al. People living with psychotic illness in 2010: the second Australian national survey of psychosis. Aust N Z J Psychiatry 2012; 46: 735-752.
4. Halverson TF, Orleans-Pobee M, Merritt C, et al. Pathways to functional outcomes in schizophrenia spectrum disorders: meta-analysis of social cognitive and neurocognitive predictors. Neurosci Biobehav Rev 2019; 105: 212-219.
5. Kharawala S, Hastedt C, Podhorna J, Shukla H, Kappelhoff B, Harvey PD. The relationship between cognition and functioning in schizophrenia: a semi-systematic review. Schizophr Res Cogn 2022; 27: 100217.
6. Huhn M, Nikolakopoulou A, Schneider-Thoma J, et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet 2019; 394: 939-951.
7. Feber L, Peter NL, Chiocchia Vet al. Antipsychotic drugs and cognitive function: a systematic review and network meta-analysis. JAMA Psychiatry 2025; 82: 47-56.
8. Sinkeviciute I, Begemann M, Prikken M, et al. Efficacy of different types of cognitive enhancers for patients with schizophrenia: a meta-analysis. NPJ Schizophr 2018; 4: 22
9. Burns T, Catty J, Dash M, Roberts C, Lockwood A, Marshall M. Use of intensive case management to reduce time in hospital in people with severe mental illness: systematic review and meta-regression. BMJ 2007; 335: 336-340.
10. Lim CT, Caan MP, Kim, CH, et al. Care management for serious mental illness: a systematic review and meta-analysis. Psychiatr Serv 2022; 73: 180-187.
11. Smit D, Miguel C, Vrijsen JN, Groeneweg B, Spijker J, Cuijpers P. The effectiveness of peer support for individuals with mental illness: systematic review and meta-analysis. Psychol Med 2023; 53: 5332-5341.
12. Bighelli I, Rodolico A, García-Mieres H, et al. Psychosocial and psychological interventions for relapse prevention in schizophrenia: a systematic review and network meta-analysis. Lancet Psychiatr 2011 8: 969-980.
13. Pharoah F, Mari J, Rathbone J, et al. Family intervention for schizophrenia. Cochrane Database Syst Rev 2010; 12: CD000088.
14. Linszen, D, Dinemans P, Van Der Does JW, et al. Treatment, expressed emotion and relapse in recent onset schizophrenia. Psychol Med 1996; 26: 333-342.
15. Solmi, M, Croatto G, Piva G, et al. Efficacy and acceptability of psychosocial interventions in schizophrenia: systematic overview and quality appraisal of the meta-analytic evidence. Mol Psychiatry 2023; 28: 354-368.
16. Heinrichs RW, Zakzanis KK. Neurocognitive deficits in schizophrenia: a quantitative review. Neuropsychology 1998; 12: 426-445.
17. Mesholam-Gately RI, Giuliano AJ, Goff KP, et al. Neurocognition in first-episode schizophrenia. A meta-analytic review. Neuropsychology 2009; 23: 315-336.
18. Savla GN, Vella L, Armstrong CC, et al. Deficits in domains of social cognition in schizophrenia: a meta-analysis of the empirical evidence. Schizophr Bull 2013; 39: 979-992.
19. Vita A, Barlati S, Ceraso A et al. Effectiveness, core elements, and moderators of response of cognitive remediation for schizophrenia: a systematic review and meta-analysis of randomized clinical trials. JAMA Psychiatry 2021; 78: 848-858.
20. Modini M, Tan L, Brinchmann B, et al. Supported employment for people with severe mental illness. Systematic review and meta-analysis of the international evidence. Br J Psychiatry 2016; 209: 14-22.
21. Leucht S, Tardy M, Komossa K et al. Antipsychotic drugs versus placebo for relapse prevention in schizophrenia. a systematic review and meta-analysis. Lancet 2012; 379: 2063-2071.
22. Tiihonen J, Tanskanen A, Taipale H. 20-year nationwide follow-up study on discontinuation of antipsychotic treatment in first-episode schizophrenia. Am J Psychiatry 2018; 175: 707-795.
23. Solmi M, Taipale H, Holm M, et al. Effectiveness of antipsychotic use for reducing risk of work disability: results from a within-subject analysis of a Swedish national cohort of 21,551 patients with first-episode nonaffective psychosis. Am J Psychiatry 2022; 179: 938-946.
24. Ceraso A, Lin JJ, Schneider-Thoma J, et al. Maintenance treatment with antipsychotic drugs in schizophrenia: a cochrane systematic review and meta-analysis. Schizophr Bull 2022; 48: 738-740.
25. Højlund M, Kemp AF, Haddad PM, Neill JC, Correll CU. Standard versus reduced dose of antipsychotics for relapse prevention in multi-episode schizophrenia: a systematic review and meta-analysis of randomised controlled trials. Lancet Psychiatry 2021: 8: 471-486.
26. Kishimoto T, Hagi K, Kurokawa S, Kane JM, Correll CU. Long-acting injectable versus oral antipsychotics for the maintenance treatment of schizophrenia: a systematic review and comparative meta-analysis of randomised, cohort, and pre-post studies. Lancet Psychiatry 2021; 8: 387-404.
27. Wang D, Schneider-Thoma J, Siafis S, et al. Efficacy, acceptability and side-effects of oral versus long-acting- injectables antipsychotics: systematic review and network meta-analysis. European Neuropsychopharmacology 2024; 83: 11-18.
28. Siskind, D, Orr S, Sinha S, et al. Rates of treatment-resistant schizophrenia from first-episode cohorts: systematic review and meta-analysis. Br J Psychiatry 2022; 220: 115-120.
29. Siskind D, Siskind V, Kisely, S. Clozapine response rates among people with treatment-resistant schizophrenia: data from a systematic review and meta-analysis. Can J Psychiatry 2017; 62: 772-777.
30. Galletly, C, Castle D, Dark F, et al. Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for the management of schizophrenia and related disorders. Aust N Z J Psychiatry 2016; 50: 410-472.
31. Yoshimura B, Yada, Y, So R, Takaki M, Yamada, N. The critical treatment window of clozapine in treatment-resistant schizophrenia: secondary analysis of an observational study. Psychiatry Research 2017; 250: 65-70.
32. Winckel K, Siskind D. Clozapine in primary care. Aust Pres 2017; 40: 231-236.
33. O’Donoghue B, Piacenza F, Plapp H et al. Response rates to sequential trials of antipsychotic medications according to algorithms or treatment guidelines in psychotic disorders. A systematic review and meta-analysis. Schizophr Res 268, 193-204.
34. Tanskanen A, Tiihonen J, Taipale, H. Mortality in schizophrenia: 30-year nationwide follow-up study. Acta Psychiatrica Scandinavica 2018; 138: 492-499.
35. Crellin N, Priebe S, Morant N et al. An analysis of views about supported reduction or discontinuation of antipsychotic treatment among people with schizophrenia and other psychotic disorders. BMC Psychiatry 22: 185.
36. Horowitz MA, Jauhar S, Natesan S, Murray RM, Taylor D. A method for tapering antipsychotic treatment that may minimize the risk of relapse. Schizophr Bull 2021; 47: 1116-1129.
37. Moncrieff J, Crellin N, Stansfeld, J, et al. Antipsychotic dose reduction and discontinuation versus maintenance treatment in people with schizophrenia and other recurrent psychotic disorders in England (the RADAR trial): an open, parallel-group, randomised controlled trial. Lancet Psychiatry 2023; 10: 848-859.
38. Correll CU, Solmi M, Croatto G, et al. Mortality in people with schizophrenia: a systematic review and meta-analysis of relative risk and aggravating or attenuating factors. World Psychiatry 2022; 21: 248-271.
39. Taipale H, Tanskanen A, Mehtälä J, Vattulainen P, Correll CU, Tiihonen J. 20-year follow-up study of physical morbidity and mortality in relationship to antipsychotic treatment in a nationwide cohort of 62,250 patients with schizophrenia (FIN20). World Psychiatry 2020; 19: 61-68.
40. Cameron AJ, Magliano DJ, Zimmet PZ, Welborn, T, Shaw JE. The metabolic syndrome in Australia: prevalence using four definitions. Diabetes Res Clin Pract 2007; 77: 471-478.
41. Firth J, Siddiqi N, Koyanagi A, et al. The Lancet Psychiatry Commission: a blueprint for protecting physical health in people with mental illness. Lancet Psychiatry 2019; 6: 675-712.
42. Mitchell AJ, Lord O, Malone D. Differences in the prescribing of medication for physical disorders in individuals with v. without mental illness: meta-analysis. Br J Psychiatry 2012; 201: 435-443.