Concurrent management of type 2 diabetes and obesity

Type 2 diabetes is prevalent in Australia, with most diagnosed patients also being affected by comorbid obesity. Weight loss can mitigate otherwise substantial risk in people with obesity and diabetes. Strategies for the management of weight in patients with type 2 diabetes include lifestyle modifications, pharmacotherapy and surgery.
Note: This is an online update of the original version of this article that was published in the April 2023 issue of Endocrinology Today. This update was prepared for World Obesity Day 2026.
- The prevalence of both diabetes and obesity is rapidly increasing in Australia.
- Weight loss improves the core metabolic features common to both conditions and reduces morbidity and mortality.
- Lifestyle interventions and pharmacotherapy should be introduced early.
- Glucagon-like peptide-1 receptor agonists and dual incretin agonists are among the most effective medications to manage type 2 diabetes and obesity concurrently.
- Surgical intervention is a treatment option for the minority of people with type 2 diabetes and obesity.
Diabetes occurs in 6.6% of people in Australia, with 1.3 million people living with the condition in 2022, the majority of whom have type 2 diabetes (T2DM).1 Diabetes contributed to 11% of all deaths in 2022.2 Obesity is one of the leading risk factors for developing T2DM, and almost 80 to 90% of people with T2DM have overweight or obesity.3 Obesity has become an increasing problem worldwide, with 66% of adults in Australia having a body mass index (BMI) in either the overweight (25 to 29.9 kg/m2) or obesity (≥30 kg/m2) range.4 As the prevalence of obesity increases, there is also a rise in obesity-associated complications, leading to further morbidity and mortality. These complications include hypertension, hyperglycaemia, cardiovascular (CV) disease, T2DM, dyslipidaemia, airway disease, obstructive sleep apnoea, metabolic dysfunction-associated steatohepatitis (MASH), gastro-oesophageal reflux disease, urinary stress incontinence and osteoarthritis.5-7
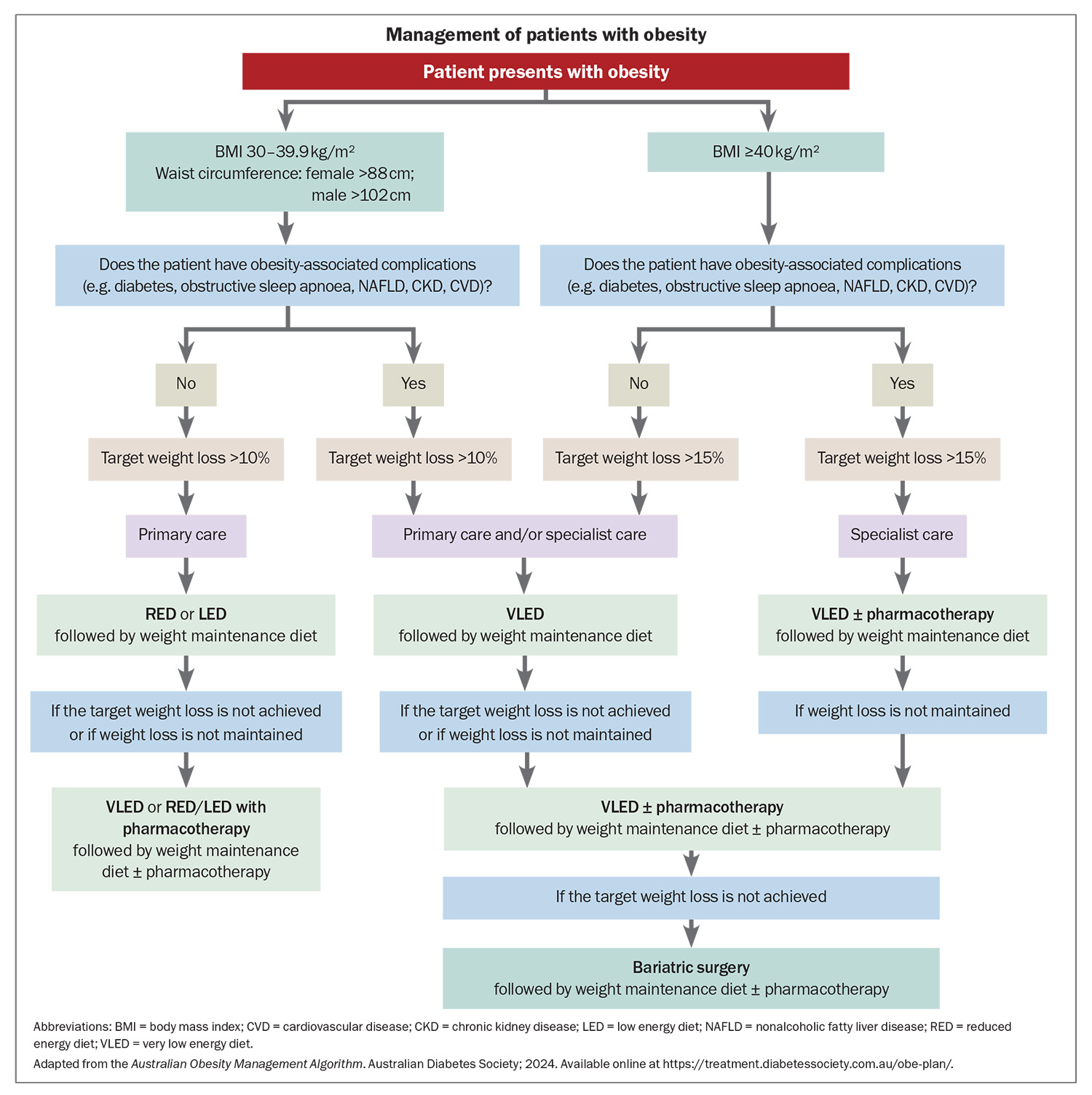
Compared with people who have a BMI within a healthy range, those with obesity have a significantly increased risk of obesity-associated comorbidities. Women have a 12.4-fold increased risk of developing T2DM, almost double the risk seen in men.8 In the context of obesity, women also have a higher risk of developing coronary artery disease, which is especially concerning given CV disease is the leading cause of death in both men and women.6 The rate of other complications is just as alarming, with more than one in 20 cancer cases directly associated with overweight or obesity.7 Obesity is the second most common modifiable risk factor (after smoking) for developing cancer. Therefore, maintaining a healthy weight could be crucial for the primary prevention of a large disease burden in the future (Flowchart).
Benefits of weight loss
Weight loss can mitigate otherwise substantial risk in people with obesity and T2DM, with even modest reductions in weight (<5%) leading to improvements in hypertension and hyperglycaemia.9 A 5 to 10% weight loss is associated with a reduction of intrahepatic lipids in metabolic-associated fatty liver disease; improvement in forced expiratory volume at one second in those with asthma and airway disease; reduction in triglyceride levels, increase in high-density lipoprotein cholesterol and reduction in nonhigh-density lipoprotein cholesterol; improvements in ovulation and regularisation of menses, particularly in women with polycystic ovary syndrome; and prevention of T2DM.9,10 A 10% weight loss also results in a reduction in all-cause mortality.11 More progressive weight loss (>10%) significantly improves metrics associated with osteoarthritis (including pain, walking distance and quality of life scores), gastro-oesophageal reflux disease, obstructive sleep apnoea, nonalcoholic steatohepatitis and heart failure with preserved ejection fraction (HFpEF).
Furthermore, achieving greater than 10% weight loss is associated with reductions in CV disease mortality and increased rates of T2DM remission, especially when the duration of T2DM is short (Box).9-13
Therapeutic strategies for diabetes and obesity management
Lifestyle intervention
The Diabetes Remission Clinical Trial (DiRECT) demonstrated that lifestyle intervention implemented in general practice for people with T2DM can lead to meaningful weight loss and disease remission.9 After an initial three-month period of a reduced energy diet (about 850 kcal/day) and a further six-week food reintroduction phase, participants were randomised to receive either standard care or a structured program providing individualised dietary advice and an exercise program. At one year, 46% of participants in the intervention arm were in remission (defined as a glycated haemoglobin [HbA1c] <6.5% without medications) and 24% had achieved the target weight loss of 15 kg or more. Although the number of participants who maintained sustained weight loss reduced over the following year, there was still evidence of a benefit, with 36% of those in the intervention arm remaining in T2DM remission. This pivotal trial provides evidence that, beyond weight reduction, lifestyle intervention can facilitate remission of T2DM.
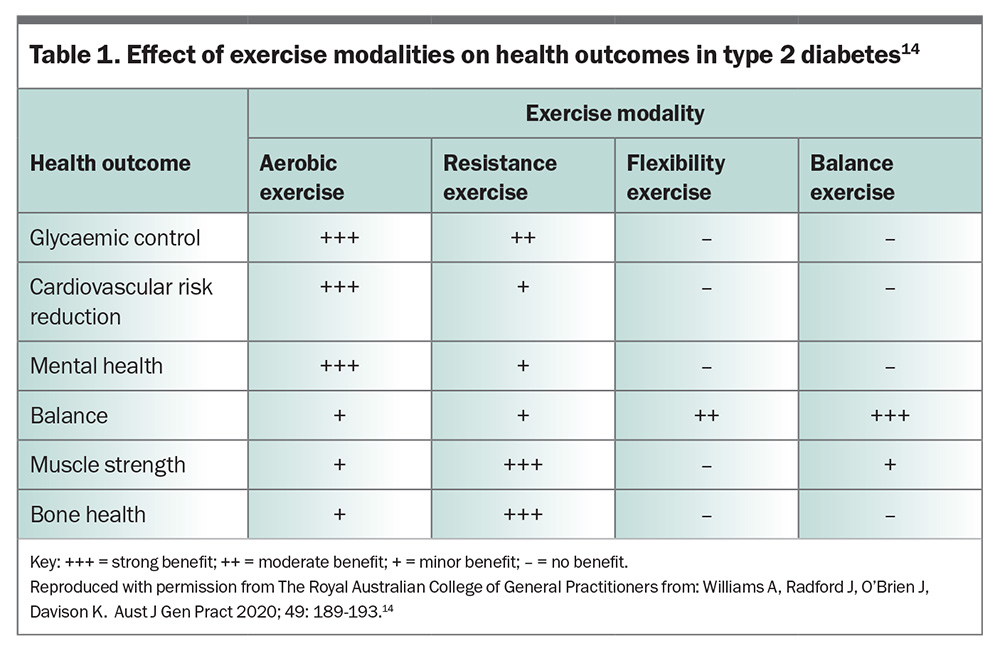
Physical activity should always be encouraged in conjunction with dietary interventions, with key objectives tailored to individuals and available resources. Standard recommendations include 150 minutes of moderate-to-vigorous intensity exercise per week. However, exercise physiologists advise against rigid prescriptions of physical activity or setting unachievable goals, and instead recommend focusing on activities the patient enjoys, such as dancing, yoga or swimming. There is strong evidence linking physical inactivity with an increased risk of many adverse health conditions. Globally, physical inactivity is associated with 9% of premature mortality, meaning over five million deaths per year could be prevented.14 In people with obesity and T2DM, studies of physical activity interventions have shown that early intervention can prevent or ameliorate weight gain and its health consequences, while also improving insulin sensitivity and lowering blood glucose levels (Table 1).15 For those on insulin, basal requirements can reduce as much as 30% after high-intensity interval exercise. As such, close glucose monitoring is required to make appropriate treatment adjustments when physical activity is planned.16
Although lifestyle interventions have demonstrated benefits in morbidity and mortality, the LookAHEAD trial highlights the complexity of relationships among weight loss, glycaemic control and CV outcomes for individuals with T2DM.17 Despite achieving significant and sustained weight loss through caloric restriction and increased physical activity over an almost 10-year period, trial participants did not have a statistically significant reduction in CV events. However, the intervention did lead to improved glycaemic control, blood pressure and lipid levels, as well as a higher rate of achieving and maintaining clinically significant weight loss. Some of these findings may be attributable to the unblinded nature of the study and potential selection bias favouring the inclusion of more health-conscious patients. Regardless, these findings underscore the intricate interplay of factors influencing CV outcomes within the context of lifestyle interventions for people with T2DM.
Very low energy diets
Although the sustained rate of T2DM remission in DiRECT was reassuring, there was a trend towards weight regain during the follow-up period. An analysis of eight high-quality weight loss studies showed that, without continued intervention, weight regain occurred in most people.18 Strategies that are easy to implement and support ongoing low caloric intake help reduce weight regain. Very low energy diets (VLEDs) are useful in this context by providing 800 calories per day while ensuring the patient still has adequate intake of essential vitamins, minerals and amino acids. The low carbohydrate content of VLEDs induces a mild ketosis after two to three days.
Despite their efficacy, the practical implementation of VLEDs necessitates careful consideration of individual circumstances and challenges; a substantial proportion of patients face difficulties in adhering to and managing VLEDs. Reduced energy diets and low energy diets are available as milder alternatives to VLEDs. Certain patients may be unsuitable for VLEDs, including those at risk of malnutrition or ketoacidosis (e.g. individuals prescribed a sodium-glucose cotransporter-2 [SGLT-2] inhibitor), individuals with a history of eating disorders and pregnant or lactating women. Patients on VLEDs may experience fluctuations in glucose levels, requiring careful monitoring and potential adjustments to medication.
Despite these considerations, the benefits of the appetite-suppressing effects of VLEDs make them a noteworthy consideration in managing T2DM and obesity. VLEDs should be considered as an initial weight loss strategy if supervised lifestyle interventions have been unsuccessful in reducing weight, or if rapid weight loss is required (e.g. prior to bariatric or general surgery conditional on weight loss). Physical activity should be encouraged alongside VLEDs, which can be safely used in conjunction with other weight loss strategies. Intermittent use may also assist with long-term weight management.
Pharmacotherapy
Although the above lifestyle strategies can help to lower body weight and induce T2DM remission, maintaining long-term weight loss with lifestyle intervention alone is difficult.19 Pharmacotherapy can assist patients with weight loss and maintenance. A significant barrier to ongoing pharmacotherapy use in Australia is cost, as the long-term use of weight loss medications remains expensive and often prohibitive for many people. Unfortunately, obesity management is strongly dictated by affordability.
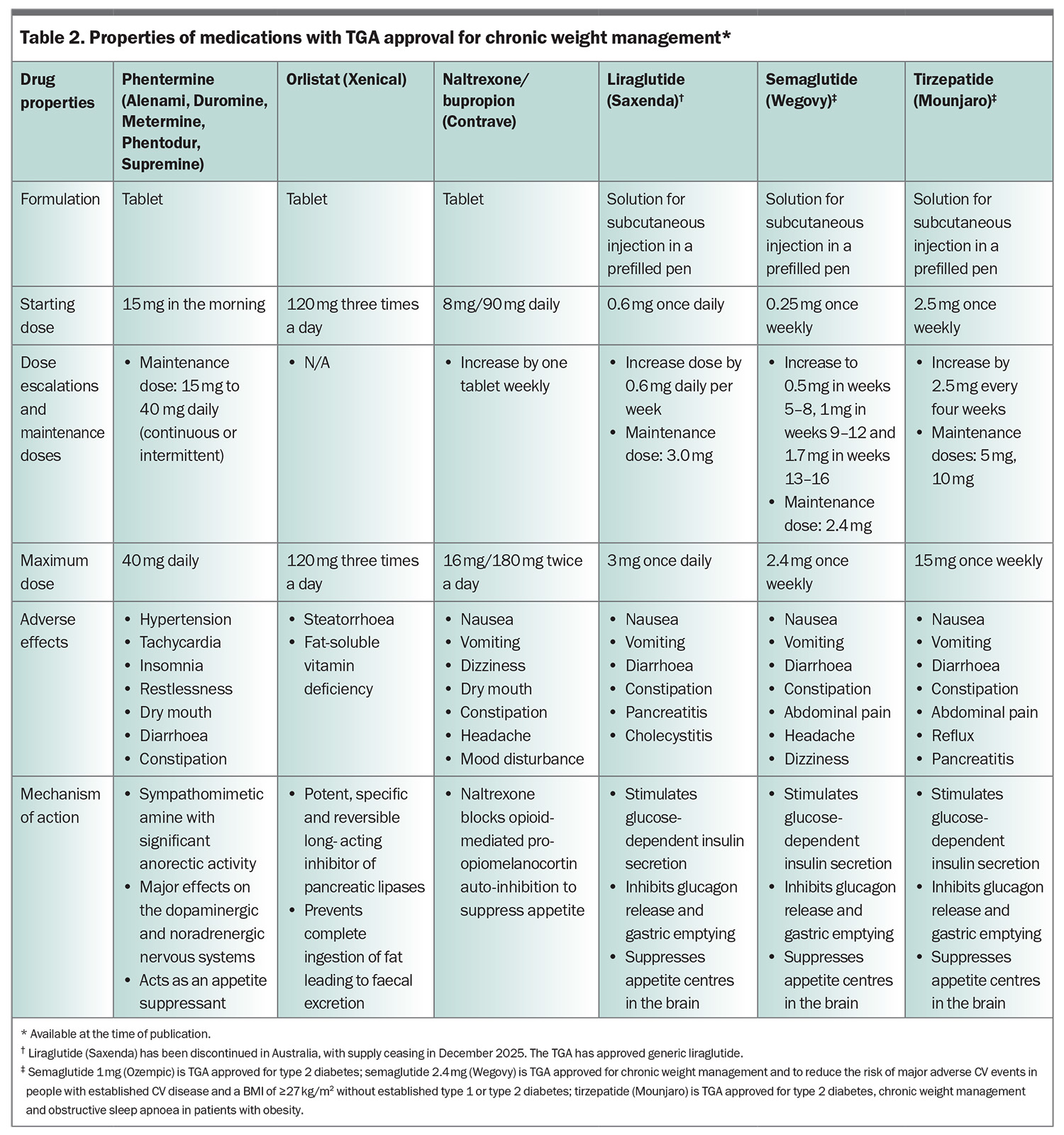
In Australia, six antiobesity medications are currently TGA approved for patients with obesity, or overweight with medical comorbidities: orlistat, phentermine, combination naltrexone/bupropion, liraglutide, semaglutide (2.4 mg weekly dose) and tirzepatide (Table 2).
Orlistat
Orlistat induces weight loss by inhibiting gastric and pancreatic lipases, thereby preventing the hydrolysis of triglycerides and reducing the absorption of free fatty acids. Apart from its beneficial effects on weight, no direct glucose-lowering effects have been recognised with the use of orlistat.
Phentermine
Phentermine is an adrenergic agonist that increases noradrenaline release in the lateral hypothalamus. It is thought to promote weight loss through the inhibition of neuropeptide Y, a key mediator in hunger perception, although its precise mechanism remains unclear. As with orlistat, phentermine does not exhibit direct glucose-lowering effects.
Naltrexone/bupropion
Although the exact neurochemical effects of naltrexone/bupropion are not fully understood, bupropion directly increases pro-opiomelanocortin activity, and naltrexone indirectly increases pro-opiomelanocortin activity by blocking its natural negative feedback loop.20 In mouse models, direct administration into the brain reduces food intake by altering the mesolimbic reward circuit and increases the firing rate of pro-opiomelanocortin neurons that regulate appetite. In people with established T2DM, a 12-month trial of naltrexone/bupropion demonstrated a reduction in HbA1c of 0.6% in participants who achieved a 5% body weight loss in the first 16 weeks, with a 9% body weight loss at 12 months.21
Liraglutide and semaglutide
The Australian Diabetes Society, Australian and New Zealand Obesity Society and Royal Australian College of General Practitioners recommend the early use of glucagon-like peptide-1 receptor agonists (GLP-1RAs), such as liraglutide and semaglutide, for the management of T2DM and obesity.22,23 The primary mechanism of action of GLP-1RAs involves activation of the incretin pathway, which acts on the central nervous system to reduce appetite, leading to reduced food intake and subsequent weight loss.24 Incretin- based therapy with GLP-1RAs also stimulates insulin release from pancreatic beta cells, resulting in reductions in both body weight and blood glucose levels in people with T2DM.25 In patients with obesity, a 56-week treatment with liraglutide 3 mg led to a cumulative reduction in the risk of developing T2DM, as well as a mean body weight loss of 8.4 kg.26 In people with established prediabetes and obesity, three years of liraglutide treatment led to a threefold increase in the likelihood of normoglycaemia.27 After a 30-week treatment course, liraglutide reduced HbA1c by 1.55% and led to a mean body weight loss of 4.53 kg.28 Liraglutide (Saxenda) has now been discontinued in Australia, with supply ceasing in December 2025.
Semaglutide 1.0 mg, administered once weekly by subcutaneous injection, has been shown to lower body weight and sustain body weight reduction over a two-year trial period, with up to two-thirds of patients achieving a clinically meaningful reduction of at least 5% of their initial body weight.28 In addition to weight loss, semaglutide 1.0 mg has demonstrated significant glucose-lowering potential. Head-to-head trials have shown it to be superior to dipeptidyl peptidase-4 inhibitors, SGLT-2 inhibitors and other GLP-1RAs, with a mean HbA1c reduction of 1.5 to 1.8%, and 80% of patients achieving an HbA1c of less than 7%.29-32 Several studies on GLP-1RAs have also demonstrated CV benefits, with up to a 26% reduction in CV risk in high-risk patients.28,33-35 The FLOW trial showed a 24% reduction in the risk of major kidney disease events, an 18% reduction in cardiovascular events, and a 20% reduction in all-cause mortality among participants with T2DM and chronic kidney disease (CKD) treated with semaglutide compared to placebo.36
Semaglutide has also shown promise in treating MASH. In the ESSENCE trial involving participants with biopsy-confirmed MASH and stage 2 or 3 fibrosis, over half of whom had T2DM, interim analysis at 72 weeks showed 63% of those treated with semaglutide 2.4 mg weekly achieved resolution of steatohepatitis without worsening of fibrosis, compared with 34% in the placebo group.37
Semaglutide 2.4 mg reduces body weight by 16% in people with obesity, or with overweight (BMI ≥27 kg/m2) and a medical comorbidity.38 The pivotal SELECT trial demonstrated, for the first time, that a weight loss medication could provide CV benefit – specifically, semaglutide 2.4 mg significantly reduced major adverse CV events by 20% in people with obesity and established CV disease, without diabetes, over a median follow up of 3.3 years.39 Semaglutide (2.4 mg weekly dose) is also TGA approved to reduce the risk of major adverse CV events in people with established CV disease and a BMI of ≥27 kg/m2 without established type 1 or type 2 diabetes. In this population, semaglutide 2.4 mg significantly reduced heart failure symptoms and body weight, while improving physical function, as evidenced by increased six-minute walk test distance.40
Semaglutide is approved for weight management in a 2.4 mg weekly dose formulation, which has TGA approval but is not listed on the PBS. The 1.0 mg weekly dose of semaglutide, indicated for T2DM, has PBS listing under restricted criteria. The Pharmaceutical Benefits Advisory Committee has recently recommended that semaglutide 2.4 mg be listed on the PBS for adults with established CV disease and a BMI of 35 kg/m2 or higher, or 32.5 kg/m2 or higher for Asian, Aboriginal or Torres Strait Islander populations. Specifically, it must be prescribed in combination with at least one of metformin, a sulfonylurea or insulin, and only when glycaemic control remains inadequate despite treatment (with at least one of metformin, a sulfonylurea or insulin). Furthermore, semaglutide (1 mg weekly dose) cannot be coprescribed on the PBS alongside an SGLT-2 inhibitor, dipeptidyl peptidase-4 inhibitor or another GLP-1RA. Due to ongoing supply constraints, regulatory authorities advise that the 1.0 mg formulation should not be prescribed solely for weight loss. To ensure appropriate and equitable access, the 2.4 mg formulation should be prescribed in patients in whom semaglutide is indicated for weight management.
Tirzepatide
Tirzepatide, a dual GLP-1 and gastric inhibitory polypeptide receptor agonist, is TGA approved for the treatment of T2DM, chronic weight management in adults with obesity or overweight with at least one weight-associated comorbidity, and for moderate-to-severe obstructive sleep apnoea in adults with obesity.
In the SURPASS-2 trial, tirzepatide exhibited remarkable efficacy in a head-to-head study in people with T2DM, compared with semaglutide 1 mg once weekly. Over a 40-week duration, tirzepatide 15 mg once weekly resulted in an additional 0.45% reduction in HbA1c beyond that achieved with semaglutide.41 Furthermore, the impact on weight dynamics was equally impressive, with tirzepatide 15 mg once weekly yielding a mean percentage body weight loss of 20.9% after a comprehensive 72-week treatment course in people with obesity and without T2DM.42 The SURMOUNT-5 trial demonstrated that tirzepatide 15 mg facilitated more weight loss compared with semaglutide 2.4 mg.43
Beyond glycaemic control and weight management, clinical trials have unveiled further advantages. A post hoc analysis of the SURPASS-4 trial demonstrated slower progression of CKD among individuals with T2DM and elevated CV risk.44 In the SUMMIT trial, tirzepatide reduced the risk of worsening heart failure and CV death by 38% in people with HFpEF and obesity, almost half of whom also had T2DM.45 Additionally, the phase 2 SYNERGY-NASH trial demonstrated that tirzepatide was superior to placebo in achieving resolution of MASH without worsening of fibrosis, with more than half of patients also showing improvement in fibrosis at 52 weeks.46 The results of the SURMOUNT-MMO study are eagerly awaited to determine the CV protection conferred by tirzepatide (clinical trial registration: NCT05556512). In the future, dual and triple incretin-based therapies are expected to produce more potent weight loss effects.
Implications for weight management with other hypoglycaemic agents
Sodium-glucose contransporter-2 inhibitors
Although SGLT-2 inhibitors are less effective than GLP-1RAs in reducing body weight, they play an important role in providing substantial CV protection in patients with heart failure and T2DM. SGLT-2 inhibitors have been shown to reduce the risk of composite CV death or first hospitalisation for heart failure by 20%.
Dapagliflozin and empagliflozin are PBS listed as adjunct therapy in patients diagnosed with heart failure (New York Heart Association class II, III or IV), with left ventricular ejection fraction of less than or equal to 40%.47 Dapagliflozin and empagliflozin are also PBS listed as adjunct therapy in patients with T2DM, with CVD, at high risk of a CV event or who identify as Aboriginal or Torres Strait Islander.
In patients with renal impairment (with or without T2DM), the DAPA-CKD trial showed that dapagliflozin reduced the decline in estimated glomerular filtration rate, progression to end-stage CKD and death from renal or CV causes by 39%.48 Similarly, the EMPA-KIDNEY trial showed that empagliflozin significantly reduced the risk of kidney disease progression or CV death in a broad population of patients with CKD, including those without T2DM.49 Both dapagliflozin and empagliflozin are PBS listed for CKD (authority required), in addition to an ACE inhibitor or angiotensin receptor blocker.
Whether using an SGLT-2 inhibitor or a GLP-1RA, metformin should always be considered as first-line therapy for T2DM to reduce insulin resistance. Although metformin is typically listed as a weight-neutral agent in patients with T2DM, it can promote modest weight loss in women with obesity and polycystic ovarian syndrome; however, it is not currently approved by the TGA for this indication.50
Insulin
Ideally, the management of T2DM should address the core metabolic derangements of the disease, namely insulin resistance and reduced insulin secretion. Beta cell loss is an intrinsic component of the pathogenesis of T2DM and, despite the increasing availability of alternative glucose-lowering medications, insulin may still be required to achieve adequate glycaemic control (either via direct insulin injection or through increased insulin production secondary to sulfonylureas). Although insulin can reduce the microvascular and macrovascular complications associated with poor glycaemic control, it often contributes to weight gain. Insulin stimulates lipogenesis, inhibits protein catabolism and slows basal metabolism, thereby promoting fat mass gain. In combination with the abnormal peripheral administration route of insulin, these effects lead to reductions in energy metabolism and the well-known weight gain seen with insulin-dependent diabetes.51 Nonetheless, insulin therapy is often necessary in people with long-standing T2DM to achieve adequate glycaemic control.
Dulaglutide
Dulaglutide, a once-weekly GLP-1 RA, while not TGA approved for obesity treatment, is PBS listed for the adjuvant management of T2DM in patients who meet strict inclusion criteria (including those who are intolerant to, or not achieving a clinically meaningful glycaemic response on, SGLT-2 inhibitors). In the REWIND trial, dulaglutide significantly reduced the risk of major adverse CV events by 12%, including nonfatal myocardial infarction, stroke and cardiovascular death, even among patients without established CV disease.52 Although the weight loss achieved with dulaglutide is modest compared to newer GLP-1RAs, its once-weekly dosing, CV benefit, and PBS accessibility make it a valuable option in the concurrent management of T2DM and obesity.
Bariatric surgery
Weight loss surgery is an important inclusion in the clinician’s armamentarium for the management of obesity and T2DM. Compared with other management strategies, weight loss surgery is associated with long-term reductions in overall mortality, as well as decreased incidences of T2DM, myocardial infarction, stroke and cancer.53 The Australian and New Zealand Obesity Society and the Royal Australian College of General Practitioners recommend considering weight loss surgery for people with a BMI greater than 35 kg/m2 and T2DM, or people with a BMI greater than 40 kg/m2 who are at high risk of developing T2DM when lifestyle interventions and medical therapy have been unsuccessful. The most common types of weight loss surgery in Australia are gastric bypass (mini loop or Roux-en-Y) and sleeve gastrectomy.54 Although frequently performed in previous years, gastric banding is now uncommon due to its high failure rate and requirement for repeat procedures.
Many people with T2DM will experience improvement or normalisation of blood glucose levels after surgery, mandating close monitoring of medications in the postoperative period. These procedures do carry associated surgical and nutritional deficiency risks, with patients needing long-term monitoring of metabolic parameters, nutritional intake and bone health. Many patients who undergo weight loss surgery do experience weight regain and remain at high risk of T2DM recurrence. With the advent of potent incretin-based therapies, it remains unclear whether pharmacotherapy should be mandated prior to consideration for surgery.
Conclusion
Diabetes and obesity are two of the highest contributors to the burden of chronic disease in Australia. A multipronged approach to tackling these inter-related conditions through weight loss can target the core metabolic derangements in both and improve long-term morbidity and mortality. Lifestyle interventions paired with effective pharmacotherapy options have shown significant improvements in inducing weight loss and T2DM control. Such strategies should be considered early; ideally, before the development of comorbidities associated with T2DM and obesity. ET
COMPETING INTERESTS: Dr Weir: None. Professor Glastras has received honoraria and speaker fees, and taken part in advisory boards for Astra Zeneca, Boehringer Ingelheim, Eli Lilly, Novo Nordisk and Sanofi pharmaceutical companies.
References
1. Australian Bureau of Statistics (ABS). Rise in proportion of Australians with diabetes. Sydney: ABS; 2025. Available online at: https://www.abs.gov.au/media-centre/media-releases/rise-proportion-australians-diabetes (accessed February 2026).
2. Australian Institute of Health and Welfare (AIHW). Diabetes: Australian Facts. Canberra: AIHW; 2024. Available online at: https://www.aihw.gov.au/reports/diabetes/diabetes (accessed February 2026).
3. Nianogo RA, Arah OA. Forecasting obesity and type 2 diabetes incidence and burden: the ViLA-obesity simulation model. Front Public Health 2022; 10: 818816.
4. Australian Institute of Health and Welfare (AIHW). Overweight and Obesity. Canberra: AIHW; 2024. Available online at: https://www.aihw.gov.au/reports/overweight-obesity/overweight-and-obesity (accessed February 2026).
5. Yuen M, Earle R, Kadambi N, et al. A systematic review and evaluation of current evidence reveals 195 obesity-associated disorders. New Orleans: The Obesity Society; 2016.
6. GBD 2015 Obesity Collaborators, Afshin A, Forouzanfar MH, et al. Health effects of overweight and obesity in 195 countries over 25 Years. N Engl J Med 2017; 377: 13-27.
7. Whiteman DC, Webb PM, Green AC, et al. Cancers in Australia in 2010 attributable to modifiable factors: summary and conclusions. Aust N Z J Public Health 2015; 39: 477-484.
8. Guh DP, Zhang W, Bansback N, Amarsi Z, Birmingham CL, Anis AH. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health 2009; 9: 88.
9. Lean ME, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet 2018; 391: 541-551.
10. Garvey WT, Mechanick JI, Brett EM, et al. American Association of Clinical Endocrinologists and American College of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pract 2016; 22: 1-203.
11. Ryan D, Yockey S. Weight loss and improvement in comorbidity: differences at 5%, 10%, 15% and over. Curr Obes Rep 2017; 6: 187-194.
12. Benraouane F, Litwin SE. Reductions in cardiovascular risk after bariatric surgery. Curr Opin Cardiol 2011; 26: 555-561.
13. Sundström J, Bruze G, Ottosson J, Marcus C, Näslund I, Neovius M. Weight loss and heart failure: a nationwide study of gastric bypass surgery versus intensive lifestyle treatment. Circulation 2017; 135: 1577-1585.
14. Lee IM, Shiroma EJ, Lobelo F, et al. Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy. Lancet 2012; 380: 219-229.
15. Williams A, Radford J, O’Brien J, Davison K. Type 2 diabetes and the medicine of exercise: the role of general practice in ensuring exercise is part of every patient’s plan. Aust J Gen Pract 2020; 49: 189-193.
16. Lee AS, Way KL, Johnson NA, Twigg SM. High-intensity interval exercise and hypoglycaemia minimisation in adults with type 1 diabetes: a randomised cross-over trial. J Diabetes Complications 2020; 34: 107514.
17. Pi-Sunyer X. The Look AHEAD Trial: a review and discussion of its outcomes. Curr Nutr Rep 2014; 3: 387-391.
18. Nordmo M, Danielsen YS, Nordmo M. The challenge of keeping it off, a descriptive systematic review of high-quality, follow-up studies of obesity treatments. Obes Rev 2020; 21: e12949.
19. Sumithran P, Prendergast LA, Delbridge E, et al. Long-term persistence of hormonal adaptations to weight loss. N Engl J Med 2011; 365: 1597-1604.
20. Billes SK, Sinnayah P, Cowley MA. Naltrexone/bupropion for obesity: an investigational combination pharmacotherapy for weight loss. Pharmacol Res 2014; 84: 1-11.
21. Hollander P, Gupta AK, Plodkowski R, et al. Effects of naltrexone sustained-release/bupropion sustained-release combination therapy on body weight and glycemic parameters in overweight and obese patients with type 2 diabetes [published correction appears in Diabetes Care 2014; 37: 587]. Diabetes Care 2013; 36: 4022-4029.
22. Australian Diabetes Society. Australian Type 2 Diabetes Glycaemic Management Algorithm. Sydney: Australian Diabetes Society; 2024. Available online at: https://www.diabetessociety.com.au/guideline/australian-t2d-glycaemic-management-algorithm-june-2024/ (accessed February 2026).
23. Royal Australian College of General Practitioners (RACGP). Management of Type 2 Diabetes: A Handbook for General Practice. Melbourne: RACGP; 2022. Available online at: https://www.racgp.org.au/clinical-resources/clinical-guidelines/key-racgp-guidelines/view-all-racgp-guidelines/diabetes/introduction (accessed July 2025).
24. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375: 1834-1844.
25. Pulipati VP, Pannain S. Pharmacotherapy of obesity in complex diseases. Clin Obes 2022; 12: e12497.
26. Pi-Sunyer X, Astrup A, Fujioka K, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med 2015; 373: 11-22.
27. Murphy CF, Docherty NG, le Roux CW. Liraglutide: another reason to target prediabetes? Oncotarget 2017; 8: 99203-99204.
28. Sorli C, Harashima SI, Tsoukas GM, et al. Efficacy and safety of once-weekly semaglutide monotherapy versus placebo in patients with type 2 diabetes (SUSTAIN 1): a double-blind, randomised, placebo-controlled, parallel-group, multinational, multicentre phase 3a trial. Lancet Diabetes Endocrinol 2017; 5: 251-260.
29. Ahrén B, Masmiquel L, Kumar H, et al. Efficacy and safety of once-weekly semaglutide versus once-daily sitagliptin as an add-on to metformin, thiazolidinediones, or both, in patients with type 2 diabetes (SUSTAIN 2): a 56-week, double-blind, phase 3a, randomised trial. Lancet Diabetes Endocrinol 2017; 5: 341-354.
30. Ahmann AJ, Capehorn M, Charpentier G, et al. Efficacy and safety of once-weekly semaglutide versus exenatide ER in subjects with type 2 diabetes (SUSTAIN 3): a 56-week, open-label, randomized clinical trial. Diabetes Care 2018; 41: 258-266.
31. Pratley RE, Aroda VR, Lingvay I, et al. Semaglutide versus dulaglutide once weekly in patients with type 2 diabetes (SUSTAIN 7): a randomised, open-label, phase 3b trial. Lancet Diabetes Endocrinol 2018; 6: 275-286.
32. Lingvay I, Catarig AM, Frias JP, et al. Efficacy and safety of once-weekly semaglutide versus daily canagliflozin as add-on to metformin in patients with type 2 diabetes (SUSTAIN 8): a double-blind, phase 3b, randomised controlled trial. Lancet Diabetes Endocrinol 2019; 7: 834-844.
33. Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375: 311-322.
34. Hernandez AF, Green JB, Janmohamed S, et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (harmony outcomes): a double-blind, randomised placebo-controlled trial. Lancet 2018; 392: 1519-1529.
35. Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 2019; 394: 121-130.
36. Perkovic V, Tuttle KR, Rossing P, et al. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med 2024; 391: 109-121.
37. Newsome PN, Sanyal AJ, Engebretsen KA, et al. Semaglutide 2.4 mg in participants with metabolic dysfunction-associated steatohepatitis: baseline characteristics and design of the phase 3 ESSENCE trial. Aliment Pharmacol Ther 2024; 60: 1525-1533.
38. Wadden TA, Bailey TS, Billings LK, et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: the STEP 3 randomized clinical trial. JAMA 2021; 325: 1403-1413.
39. Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med 2023; 389: 2221-2232.
40. Kosiroborod M, Abildstrøm S, Borlaug B, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med 2023; 389: 1069-1084.
41. Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med 2021; 385: 503-515.
42. Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med 2022; 387: 205-216.
43. Aronne LJ, Horn DB, le Roux CW, et al. Tirzepatide as compared with semaglutide for the treatment of obesity. N Engl J Med 2025; 393: 26-36.
44. Bosch C, Carriazo S, Soler MJ, Ortiz A, Fernandez-Fernandez B. Tirzepatide and prevention of chronic kidney disease. Clin Kidney J 2022; 16: 797-808.
45. Packer M, Zile MR, Kramer CM, et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N Engl J Med 2025; 392: 427-437.
46. Loomba R, Hartman ML, Lawitz EJ, et al. Tirzepatide for metabolic dysfunction-associated steatohepatitis with liver fibrosis. N Engl J Med 2024; 391: 299-310.
47. Vaduganathan M, Docherty KF, Claggett BL, et al. SGLT-2 inhibitors in patients with heart failure: a comprehensive meta-analysis of five randomised controlled trials [published correction appears in Lancet 2023;401: 104]. Lancet 2022; 400: 757-767.
48. Heerspink HJL, Stefánsson BV, Correa-Rotter R, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020; 383: 1436-1446.
49. The EMPA-KIDNEY Collaborative Group, Herrington WG, Staplin N, et al. Empagliflozin in patients with chronic kidney disease. N Engl J Med 2023; 388: 117-127.
50. Harborne LR, Sattar N, Norman JE, Fleming R. Metformin and weight loss in obese women with polycystic ovary syndrome: comparison of doses. J Clin Endocrinol Metab 2005; 90: 4593-4598.
51. Liu CP. Type 1 Diabetes - Complications, Pathogenesis, and Alternative Treatments. London: InTechOpen; 2011. Available online at: http://dx.doi.org/10.5772/1540 (accessed February 2026).
52. Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 2019; 394: 121-130.
53. Sjöström L. Review of the key results from the Swedish Obese Subjects (SOS) trial - a prospective controlled intervention study of bariatric surgery. J Intern Med 2013; 273: 219-234.
54. Australia and Aotearoa New Zealand Bariatric Surgery Registry. The Bariatric Surgery Registry 2022 Annual Report. Melbourne: Central Clinical School, Monash University; 2023. Available online at: https://www.monash.edu/__data/assets/pdf_file/0018/3351042/Bariatric-Surgery-Registry-Annual-Report-2022_web.pdf (accessed February 2026).
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.