A clinical approach to managing obesity in adults

Obesity is a complex chronic disease that is strongly associated with an increased risk of all-cause mortality. Therapies for optimising weight and metabolic health should be guided by an individual’s body mass index (with consideration of body composition), comorbidities, and the presence and severity of obesity-associated complications.
- Obesity is a complex chronic disease associated with increased morbidity and mortality.
- Routine screening is required to assess the complications of obesity.
- Preconception counselling is crucial as many women are unaware of the potential adverse effects of obesity on both maternal and offspring outcomes.
- Even small reductions in body weight can reduce the complications of obesity.
- For many individuals, lifestyle interventions alone will not achieve the required amount of weight loss, and pharmacotherapy with or without metabolic surgery should be considered.
What are the health risks from obesity?
Obesity is a complex chronic disease that is strongly associated with an increased risk of all-cause mortality as well as cardiovascular and cancer mortality.1-3 Importantly, the longer the duration of obesity, the greater its impact on mortality, which is an important consideration in young adults with obesity.4
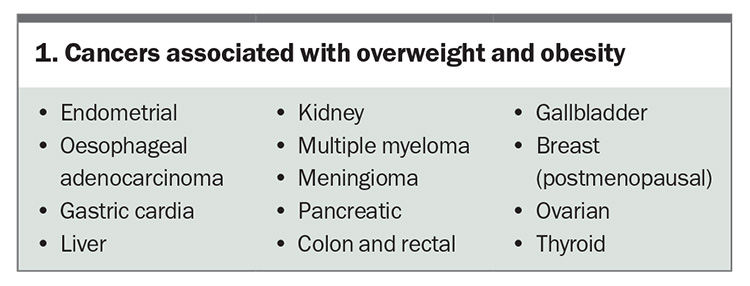
Excess weight is directly linked to various cardiovascular risk factors.5 Obesity is associated with an increased risk of various diseases including type 2 diabetes mellitus (T2DM), cardiovascular disease (CVD), chronic kidney disease and metabolic-associated fatty liver disease.6 The physical burden of carrying excess body weight results in mechanical complications including obstructive sleep apnoea (OSA), urinary incontinence, osteoarthritis and low back pain.6-8 Obesity is also a state of chronic low-grade inflammation and a major risk factor for at least 13 different types of cancer (Box 1) and dementia.9,10
In women of reproductive age, obesity is associated with menstrual disturbance, impairment of oocyte development and quality, anovulation, delayed conception, as well as worsening of the clinical features of polycystic ovary syndrome (PCOS).11,12 Depression appears to be more common amongst people with obesity compared to those of a healthy weight, although the direction of causality is not clear.13,14 Women tend to experience more mental health complications associated with obesity than men, which can be exacerbated by discrimination against individuals with a higher body mass index (BMI).15,16 This can lead to a strong dissatisfaction with one’s body weight, shape, or both, which is a risk factor for the development of comorbid disordered eating behaviours.17,18 The psychological impact of weight stigmatisation can perpetuate weight gain and lead to the development of other chronic diseases.19
Why is weight loss important?
Even modest weight loss significantly reduces the complications of obesity. A 5% reduction in total body weight can prevent the development of T2DM in individuals with prediabetes, improve glycaemic control in individuals with T2DM, reduce liver fat, lower systolic blood pressure and triglyceride levels, and improve physical disability amongst patients with knee osteoarthritis.20-25 Greater weight loss (≥10%) results in additional health benefits including remission of T2DM, improvements in OSA, reductions in liver inflammation and injury, and a lower risk of cardiovascular events and mortality.26-30 In women with PCOS, lifestyle interventions improve hyperandrogenism even if weight loss is minimal, although greater weight loss from metabolic surgery also results in improved menstrual regularity and can result in remission.12,31
In women with obesity and subfertility, lifestyle interventions have a positive effect on pregnancy and natural conception rates, although the effect on live birth rates remains uncertain.32 Concerns about the presence of concomitant disordered eating behaviours may caution clinicians against recommending weight management. However, weight management approaches (including medically supervised moderate and severe caloric restriction in combination with behavioural weight loss therapy) do not induce binge eating in overweight adults without pretreatment binge eating, and can reduce binge eating in those with pretreatment binge eating behaviours.33,34
When and how do you screen for secondary causes of obesity?
Obesity is a chronic and complex disease influenced by many factors including a genetic predisposition to obesity, work and social environments that promote the consumption of convenient highly processed foods and sedentary behaviour, metabolic adaptations defending against weight loss, as well as other psychosocial and economic drivers.35 For most individuals, their risk for obesity will be conferred by numerous variants in a number of genetic drivers, that is, polygenic obesity. However, screening for rarer monogenic subtypes of obesity should be considered if extreme obesity occurs at a young age (<5 years of age), particularly when accompanied by clinical features of genetic obesity syndromes (such as extreme hyperphagia), a family history of extreme obesity, or both.36
A relatively sudden increase in weight may suggest an endocrine cause for obesity, and screening for causes such as hypothyroidism and Cushing’s syndrome (including iatrogenic cortisol excess from exogenous glucocorticoids) should be considered, particularly if suggestive clinical features are present. For instance, if the patient reports dry skin, cold intolerance or other features to suggest hypothyroidism, check the thyroid stimulating hormone level. If there are clinical features of Cushing’s syndrome (e.g. easy bruising, facial plethora, proximal myopathy or wide purple striae), measure the midnight salivary cortisol, 24-hour urinary free cortisol, or arrange a 1 mg overnight dexamethasone suppression test (the diagnostic approach will depend on the pretest probability). PCOS should be considered if there are clinical features of insulin resistance (acanthosis nigricans), hyperandrogenism (acne and hirsutism), or oligo-ovulation or anovulation (irregular menstrual cycles).
Review the use of medications associated with weight gain such as antidepressants (e.g. mirtazapine, amitriptyline, sertraline, fluoxetine, paroxetine), antipsychotics (e.g. olanzapine, clozapine, quetiapine, risperidone), anticonvulsants (e.g. valproate, carbamazepine, gabapentin) and treatments for diabetes (e.g. insulin, sulphonylureas, thiazolidinediones), and consider switching to less obesogenic medications if possible.37 If there is diagnostic uncertainty, consider referral to a specialist service.
How do you assess for obesity-associated complications?
The following routine assessments should be performed in all adults with overweight and obesity:38
- weight, height and BMI
- waist circumference
- blood pressure measurement (using an appropriately sized arm cuff)
- assessment of fasting glucose levels (repeated regularly according to local guidelines)39
- fasting lipid profile
- liver function tests and screening tools such as calculation of the FIB-4 score (https://liver.org.au/health-professionals/fib-4-calculator/)
- screening for OSA (e.g. STOP-BANG questionnaire)
- screening for depression and anxiety (e.g. K10 screening tool or Patient Health Questionnaire [PHQ]-9)
- screening for disordered eating (e.g. Eating Disorder Examination Questionnaire [EDE-Q])
- screening for CVD (e.g. www.cvdcheck.org.au) for all adults aged 45 years and older (or 30 years and older for Aboriginal and Torres Strait Islander people) without existing CVD or not already known to be at increased risk of CVD
- age-appropriate cancer screening (e.g. bowel, breast, cervix and prostate)
- in women, screening for clinical signs of hyperandrogenism (e.g. hirsutism, acne, male pattern balding).
How do you identify candidates for weight loss interventions?
Treatments for obesity should be guided by an individual’s BMI and waist circumference, and the presence and severity of obesity-associated complications. It is imperative that lifestyle modifications are included in the treatment pathway with goals focused on reducing energy intake, optimising diet quality and increasing energy expenditure. Antiobesity pharmacotherapies are indicated for any individual with a BMI of 30 kg/m2 or more, or a BMI of 27 kg/m2 or more plus the presence of at least one weight-associated comorbidity (see the case study in Box 2). Consider commencing antiobesity pharmacotherapies when lifestyle modifications alone have been unsuccessful in achieving desired weight loss, or to maintain weight loss and prevent weight regain. Metabolic surgery should be considered for adults with a BMI of 40 kg/m2 or more, or a BMI of 30.0 to 39.9 kg/m2 with comorbidities that may improve with weight loss.
Special considerations in women of childbearing age
Many women planning to conceive are unaware of the potential adverse effects of obesity on both maternal and offspring outcomes. Obesity is associated with reduced fertility and oocyte quality, and also adversely impacts the quality and early development of the embryo.40,41 Obesity is associated with increased risks in the antenatal, intrapartum and postpartum periods, as well as increased anaesthetic risk.42 There is a direct relationship between the class of obesity and the likelihood of serious adverse outcomes.42 A large retrospective cohort study found that when compared with normal-weight women, the hazard ratio for stillbirth was 1.36 for overweight women, 1.71 for women with class I obesity, 2.00 for women with class II obesity, 2.48 for women with class III obesity and 3.16 for women with a BMI of 50 kg/m2 and above.43 Perinatal exposure to maternal obesity is also associated with cardiometabolic morbidity in the offspring.44,45
Height, weight and BMI should be measured at preconception appointments. The risks of overweight and obesity on fertility and pregnancy outcomes need to be discussed using a sensitive and person-centred approach. The Royal Australian and New Zealand College of Obstetricians and Gynaecologists Management of Obesity in Pregnancy Best Practice Statement recommends starting folic acid 5 mg daily (due to the increased risk of neural tube defects) and iodine 150 mcg daily (unless contraindicated) in the preconception period for women with obesity.46
Ideally, weight and metabolic health should be optimised prior to conception to reduce the risk of pregnancy-associated complications and to minimise exposure of the foetus to an adverse metabolic environment. A multifaceted and holistic approach to weight management is best provided by an experienced healthcare team. At present, there is a lack of conclusive randomised trial data regarding the optimal weight loss interventions for pre-pregnancy use.46 Women should be advised to use reliable forms of contraception while working on weight loss, particularly when using antiobesity pharmacotherapy. Medications for weight loss should be avoided during the time of conception, during pregnancy and while breastfeeding for safety reasons. Usually, antiobesity pharmacotherapy should be ceased for a period of at least six weeks prior to attempting to conceive (the time period will vary depending on the agent used); the manufacturers of semaglutide have indicated that this agent should be discontinued at least two months before a planned pregnancy due to the medication’s long half-life.47
Women who have undergone metabolic surgery require additional nutritional supplementation and careful monitoring for micronutrient deficiencies in the preconception period. It is recommended that conception is avoided for 12 to 18 months following metabolic surgery, particularly when there is rapid weight loss or nutritional deficiencies.48
Overview of lifestyle interventions and the role of behaviour therapy
Lifestyle interventions to reduce energy intake, increase energy expenditure and improve nutritional quality are the cornerstone of weight management. A network of experienced clinicians providing support and supervision is key to the success of lifestyle interventions. Please refer to the Australian Dietary Guidelines and the Australian Guide to Healthy Eating for general dietary advice.49,50 Involving an experienced multidisciplinary team (including an accredited practicing dietitian and exercise physiologist) is helpful and creates a network of support around the patient. Behaviour therapy is key to helping people make long-term changes in the way they respond to stimuli that trigger eating. Behaviour modification programs offered by psychologists and other suitably experienced clinicians can result in more weight loss and less weight regain.51 If there is any concern for a binge eating disorder, consider referral to a health practitioner with expertise in eating disorders.
Options for reducing energy intake include:
- Reduced Energy Diet (RED): modest energy deficit of 2000 to 4000 kJ/day (energy deficit of 480 to 960 kcal/day)
- Low Energy Diet (LED): aim to reduce total daily energy intake to 4200 to 5000 kJ (1000 to 1200 kcal/day)
- Very Low Energy Diet (VLED): aim to reduce energy intake to less than 3300 kJ/day (800 kcal/day) by substituting meals with formulated meal replacements. Medical supervision and regular clinical review are helpful when using VLEDs, as there are contraindications and precautions.
Those with a BMI of 30 to 39.9 kg/m2 and no obesity-associated complications could trial RED or LED initially, and then consider VLED if weight loss is inadequate.52 People with a BMI of 30 to 39.9 kg/m2 and obesity-associated complications, and those with a BMI of 40 kg/m2 and above should generally commence VLED directly.52 Please refer to the latest Australian Obesity Management Algorithm for further details such as contraindications to VLED, and the use of VLED in special groups (e.g. chronic kidney disease, people with diabetes and those using sodium-glucose cotransporter-2 inhibitors or warfarin).52
Regular physical activity is a key component of weight management and exercise programs should be titrated to individual needs. For instance, people with musculoskeletal problems may benefit from choosing aquatic activities, and those with cardiovascular or respiratory diseases will likely require a gentler program as tolerated. An exercise physiologist will be able to provide tailored advice on suitable exercise programs for the patient. For general advice on physical activity, please refer to the Physical Activity and Exercise Guidelines produced by the Australian Government.53
What are the pharmacotherapies available for weight loss?
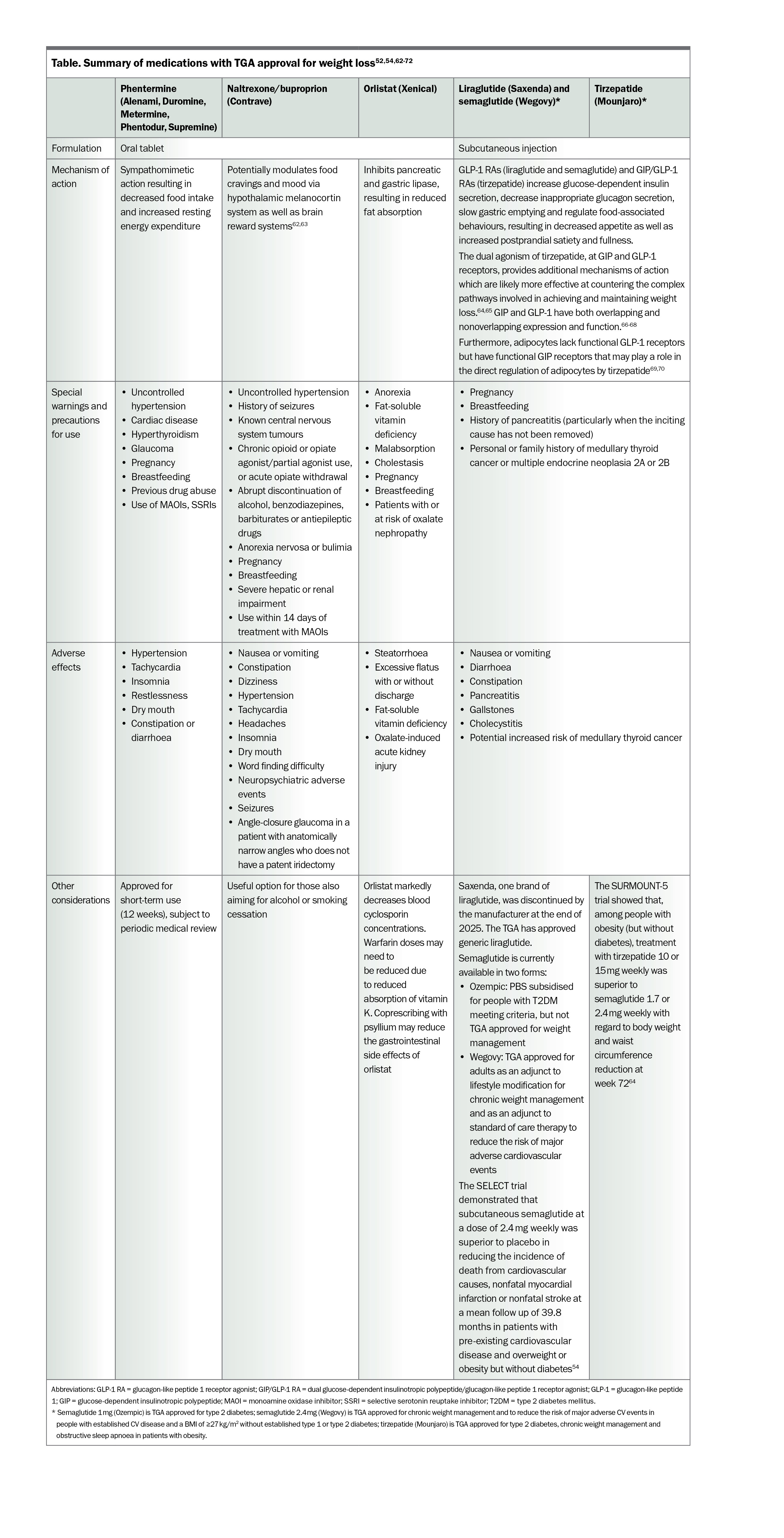
At present, phentermine, naltrexone/buproprion, orlistat, liraglutide, semaglutide (2.4 mg weekly dose) and tirzepatide are approved by the TGA for chronic weight management. Semaglutide (2.4 mg weekly dose) is also is approved by the TGA as an adjunct to standard-of-care therapy to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction or nonfatal stroke) in adults with established cardiovascular disease, with a BMI of 27 kg/m2 and above, and without established type 1 or type 2 diabetes. Topiramate and phentermine-topiramate are frequently used off-label for weight management but are not currently approved by the TGA for this indication.
The choice of pharmacotherapy should be based on a careful assessment of factors such as patient age, comorbidities (e.g. diabetes as well as cardiovascular, chronic kidney or liver diseases), medication side effect profiles and patient preference. For instance, among those with pre-existing cardiovascular disease and overweight or obesity (but without diabetes), subcutaneous semaglutide 2.4 mg weekly was superior to placebo in reducing the incidence of nonfatal myocardial infarction, death from cardiovascular causes or nonfatal stroke (mean follow-up period was 39.8 months).54 Semaglutide 2.4 mg once weekly has also been shown to significantly improve pain associated with knee osteoarthritis and to improve symptoms and physical limitations in individuals with obesity and heart failure with preserved ejection fraction.55,56 In adults with metabolic dysfunction-associated steatohepatitis (MASH) and moderate or advanced liver fibrosis, subcutaneous semaglutide 2.4 mg weekly was shown to result in higher rates of steatohepatitis resolution and greater reduction in liver fibrosis when compared to placebo at interim analysis after 72 weeks of treatment.57 Similarly, in adults with MASH and moderate or severe fibrosis, tirzepatide (5 mg, 10 mg or 15 mg) for 52 weeks was more effective than placebo in achieving resolution of MASH without worsening of fibrosis.58
Tirzepatide has been shown to reduce OSA severity when compared to placebo in adults with OSA and obesity, as well as reduce cardiovascular death and worsening heart failure in people with obesity and heart failure with preserved ejection fraction.59,60
Regular monitoring while on antiobesity pharmacotherapy is crucial to monitor progress and ensure safety. If patients are unable to lose at least 5% of total body weight after 12 to 24 weeks of treatment on the maximum dose of the chosen antiobesity agent, it is important to review medication adherence and individual circumstances, and consider whether a trial of an alternative medication is appropriate.
Certain patient groups require close monitoring on antiobesity pharmacotherapy. For instance, those with diabetes on glucose-lowering therapy (especially insulin, sulphonylureas or both) require close monitoring of blood glucose levels to avoid hypoglycaemia. Similarly, patients with hypertension require regular blood pressure monitoring with down-titration of antihypertensive therapy as appropriate.
Abrupt discontinuation of antiobesity pharmacotherapy is associated with weight regain and recurrence of weight-associated comorbidities in most cases. For instance, at one year following cessation of semaglutide 2.4 mg once weekly and lifestyle interventions (administered for 68 weeks, including 16 weeks of semaglutide dose escalation), participants in the STEP 1 trial extension regained two-thirds of their prior weight loss. Cardiometabolic improvements observed during treatment (from week zero to week 68) also reverted towards baseline after one year of therapy withdrawal.61
A summary of all the TGA approved medications for weight loss is shown in the Table.52,54,62-72
Overview of metabolic surgery
For individuals with obesity-associated complications, a high BMI or for those in whom lifestyle and pharmacotherapy have not resulted in the required weight loss, metabolic surgery may be considered as part of a comprehensive treatment plan. The potential benefits of metabolic surgery should be carefully weighed against the individual’s risk profile, and a thorough evaluation of mental health and psychosocial factors is critical. Surgery should ideally be performed at high-volume centres with experienced multidisciplinary teams. After metabolic surgery, patients will require long-term monitoring of micronutrient and nutritional status, as well as ongoing review of lifestyle and psychological factors.
The Second Diabetes Surgery Summit, held in 2016 and endorsed by the Australian Diabetes Society, produced guidelines that state that metabolic surgery is recommended for individuals with:
- a BMI of 40 kg/m2 and above, regardless of the level of glycaemic control or complexity of glucose-lowering regimens
- a BMI of 35.0 to 39.9 kg/m2, with inadequate glycaemic management despite lifestyle and optimal medical therapy.73
The American Society for Metabolic and Bariatric Surgery and International Federation for the Surgery of Obesity and Metabolic Disorders Indications for Metabolic and Bariatric Surgery 2022 state that metabolic and bariatric surgery should be:
- recommended for individuals with a BMI of 35 kg/m2 and above, regardless of presence, absence or severity of comorbidities
- considered for individuals with metabolic disease and a BMI of 30 to 34.9 kg/m2
- offered to individuals in the Asian population with a BMI of 27.5 kg/m2 and above, noting that a BMI of 25 kg/m2 and above in this group suggests clinical obesity.74
The American Diabetes Association Standards of Medical Care in Diabetes (2025) recommend that metabolic surgery be considered as a weight and glycaemic management option in people with diabetes with a BMI of 30.0 kg/m2 and above (or ≥27.5 kg/m2 in Asian American individuals) who are otherwise good surgical candidates.75
Ethnicities such as South Asian, East Asian, Southeast Asian and Australian Aboriginal and Torres Strait Islanders have been shown to have higher adiposity and diabetes risk at a given BMI.52,76-79 The Australian Obesity Management Algorithm (updated July 2024) reflects this by adopting lower BMI thresholds for these populations: a BMI of 27.5 to 37.4 kg/m2 is equivalent to a BMI of 30 to 39.9 kg/m2, and a BMI of 37.5 kg/m2 and above is equivalent to a BMI of 40 kg/m2 and above.52
Summary
Therapies for managing obesity should be guided by an individual’s BMI (with consideration of body composition), comorbidities and the presence and severity of obesity-associated complications. A network of experienced clinicians providing support and supervision is crucial to the success of lifestyle interventions. Lifestyle modification, pharmacotherapy and metabolic surgery should be considered as part of a comprehensive treatment plan. ET
COMPETING INTERESTS: Dr Zhen: None. Associate Professor Hocking has received honoraria for lectures and manuscript writing from Lilly Australia, AstraZeneca, Amgen, Sanofi-Aventis, Nestlé Health Sciences, iNova and Servier; support for attending meetings from Lilly Australia, Novo Nordisk, Amgen and CSL Seqirus; participation on Advisory Boards from Lilly Australia, Novo Nordisk, Ethicon and AstraZeneca; is an investigator on pharmaceutical trials of semaglutide and CagriSema (NovoNordisk), tirzepatide and retatrutide (Lilly), BI 456906 (Boehringer Ingelheim) and maridebart/cafraglutide (Amgen); and is President of the National Association of Clinical Obesity Services.
References
1. Global BMI Mortality Collaboration, Di Angelantonio E, Bhupathiraju Sh N, et al. Body-mass index and all-cause mortality: individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 2016; 388: 776-786.
2. Whitlock G, Lewington S, Sherliker P, et al. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009; 373: 1083-1096.
3. Chen Y, Copeland WK, Vedanthan R, et al. Association between body mass index and cardiovascular disease mortality in east Asians and south Asians: pooled analysis of prospective data from the Asia Cohort Consortium. BMJ (Clinical research ed) 2013; 347: f5446.
4. Abdullah A, Wolfe R, Stoelwinder JU, et al. The number of years lived with obesity and the risk of all-cause and cause-specific mortality. Int J Epidemiol 2011; 40: 985-996.
5. Powell-Wiley TM, Poirier P, Burke LE, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation 2021; 143: e984-e1010.
6. Kopelman PG. Obesity as a medical problem. Nature 2000; 404: 635-643.
7. Doumouchtsis SK, Loganathan J, Pergialiotis V. The role of obesity on urinary incontinence and anal incontinence in women: a review. BJOG 2022; 129: 162-170.
8. Bliddal H, Leeds AR, Christensen R. Osteoarthritis, obesity and weight loss: evidence, hypotheses and horizons - a scoping review. Obes Rev 2014; 15: 578-586.
9. Lauby-Secretan B, Scoccianti C, Loomis D, et al. Body fatness and cancer–viewpoint of the IARC Working Group. N Engl J Med 2016; 375: 794-798.
10. Alford S, Patel D, Perakakis N, Mantzoros CS. Obesity as a risk factor for Alzheimer’s disease: weighing the evidence. Obes Rev 2018; 19: 269-280.
11. Best D, Avenell A, Bhattacharya S. How effective are weight-loss interventions for improving fertility in women and men who are overweight or obese? A systematic review and meta-analysis of the evidence. Hum Reprod Update 2017; 23: 681-705.
12. Lim SS, Hutchison SK, Van Ryswyk E, Norman RJ, Teede HJ, Moran LJ. Lifestyle changes in women with polycystic ovary syndrome. Cochrane Database Syst Rev 2019; 3: CD007506.
13. Pi-Sunyer X. The medical risks of obesity. Postgrad Med 2009; 121: 21-33.
14. Fulton S, Décarie-Spain L, Fioramonti X, Guiard B, Nakajima S. The menace of obesity to depression and anxiety prevalence. TEM 2022; 33: 18-35.
15. Carpenter KM, Hasin DS, Allison DB, Faith MS. Relationships between obesity and DSM-IV major depressive disorder, suicide ideation, and suicide attempts: results from a general population study. Am J Public Health 2000; 90: 251-257.
16. Blasco BV, García-Jiménez J, Bodoano I, Gutiérrez-Rojas L. Obesity and depression: its prevalence and influence as a prognostic factor: a systematic review. Psychiatry Investig 2020; 17: 715-724.
17. Tronieri JS, Wurst CM, Pearl RL, Allison KC. Sex differences in obesity and mental health. Curr Psychiatry Rep 2017; 19: 29.
18. Camacho-Barcia L, Giel KE, Jiménez-Murcia S, et al. Eating disorders and obesity: bridging clinical, neurobiological, and therapeutic perspectives. Trends Mol Med 2024; 30: 361-379.
19. Tomiyama AJ, Carr D, Granberg EM, et al. How and why weight stigma drives the obesity ‘epidemic’ and harms health. BMC Med 2018; 16: 123.
20. Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002; 346: 393-403.
21. Tuomilehto J, Lindstrom J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001; 344: 1343-1350.
22. Look ARG, Wing RR, Bolin P, et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med 2013; 369: 145-154.
23. Lazo M, Solga SF, Horska A, et al. Effect of a 12-month intensive lifestyle intervention on hepatic steatosis in adults with type 2 diabetes. Diabetes Care 2010; 33: 2156-2163.
24. Wing RR, Lang W, Wadden TA, et al. Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care 2011; 34: 1481-1486.
25. Christensen R, Bartels EM, Astrup A, Bliddal H. Effect of weight reduction in obese patients diagnosed with knee osteoarthritis: a systematic review and meta-analysis. Ann Rheum Dis 2007; 66: 433-439.
26. Lean ME, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet 2018; 391: 541-551.
27. Schauer PR, Bhatt DL, Kirwan JP, et al. Bariatric surgery versus intensive medical therapy for diabetes - 5-year outcomes. N Engl J Med 2017; 376: 641-651.
28. Promrat K, Kleiner DE, Niemeier HM, et al. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010; 51: 121-129.
29. Look ARG, Gregg EW, Jakicic JM, et al. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: a post-hoc analysis of the Look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol 2016; 4: 913-921.
30. Foster GD, Borradaile KE, Sanders MH, et al. A randomized study on the effect of weight loss on obstructive sleep apnea among obese patients with type 2 diabetes: the Sleep AHEAD study. Arch Intern Med 2009; 169: 1619-1626.
31. Skubleny D, Switzer NJ, Gill RS, et al. The impact of bariatric surgery on polycystic ovary syndrome: a systematic review and meta-analysis. Obes Surg 2016; 26: 169-176.
32. Kim SY, Park ES, Kim HW. Effectiveness of non-pharmacological interventions for overweight or obese infertile women: a systematic review and meta-analysis. Int J Environ Res Public Health 2020; 17: 7438.
33. da Luz FQ, Hay P, Gibson AA, et al. Does severe dietary energy restriction increase binge eating in overweight or obese individuals? A systematic review. Obes Rev 2015; 16: 652-665.
34. Yanovski SZ. Eating disorders, race, and mythology. Arch Fam Med 2000; 9: 88.
35. Hruby A, Hu FB. The epidemiology of obesity: a big picture. Pharmacoeconomics 2015; 33: 673-689.
36. Styne DM, Arslanian SA, Connor EL, et al. Pediatric obesity-assessment, treatment, and prevention: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2017; 102: 709-757.
37. van der Valk ES, van den Akker ELT, Savas M, et al. A comprehensive diagnostic approach to detect underlying causes of obesity in adults. Obes Rev 2019; 20: 795-804.
38. Bessell E, Markovic TP, Fuller NR. How to provide a structured clinical assessment of a patient with overweight or obesity. Diabetes Obes Metab 2021; 23 (Suppl 1): 36-49.
39. Bell K, Shaw JE, Maple-Brown L, et al. A position statement on screening and management of prediabetes in adults in primary care in Australia. Diabetes Res Clin Pract 2020; 164: 108188.
40. Snider AP, Wood JR. Obesity induces ovarian inflammation and reduces oocyte quality. Reproduction 2019; 158: R79-R90.
41. Purcell SH, Moley KH. The impact of obesity on egg quality. J Assist Reprod Genet 2011; 28: 517-524.
42. Johansson S, Villamor E, Altman M, Bonamy AK, Granath F, Cnattingius S. Maternal overweight and obesity in early pregnancy and risk of infant mortality: a population based cohort study in Sweden. BMJ 2014; 349: g6572.
43. Yao R, Ananth CV, Park BY, Pereira L, Plante LA. Obesity and the risk of stillbirth: a population-based cohort study. Am J Obstet Gynecol 2014; 210: 457.e1-9.
44. Razaz N, Villamor E, Muraca GM, Bonamy AE, Cnattingius S. Maternal obesity and risk of cardiovascular diseases in offspring: a population-based cohort and sibling-controlled study. Lancet Diabetes Endocrinol 2020; 8: 572-581.
45. Kislal S, Shook LL, Edlow AG. Perinatal exposure to maternal obesity: lasting cardiometabolic impact on offspring. Prenat Diagn 2020; 40: 1109-1125.
46. Royal Australian and New Zealand College of Obstetricians and Gynaecologists (RANZCOG). Management of Obesity in Pregnancy Best Practice Statement. Melbourne: RANZCOG; 2022. Available online at: https://ranzcog.edu.au/wp-content/uploads/Management-Obesity-Pregnancy.pdf (accessed July 2025)
47. Therapeutic Goods Administration (TGA), Ozempic Australian Product Information VV-LAB-098360. Canberra: TGA; 2025. Available online at: https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent=&id=CP-2019-PI-01881-1 (accessed July 2025).
48. Busetto L, Dicker D, Azran C, et al. Obesity Management Task Force of the European Association for the Study of Obesity released "Practical Recommendations for the Post-Bariatric Surgery Medical Management". Obes Surg 2018; 28: 2117-2121.
49. National Health and Medical Research Council (NHMRC). Australian Dietary Guidelines. Canberra: NHMRC; 2013.
50. National Health and Medical Research Council (NHMRC). Australian Guide to Healthy Eating. Canberra: NHMRC; 2013. Available online at: http://www.eatforhealth.gov.au/guidelines/australian-guide-healthy-eating (accessed July 2025).
51. LeBlanc ES, Patnode CD, Webber EM, Redmond N, Rushkin M, O’Connor EA. Behavioral and pharmacotherapy weight loss interventions to prevent obesity-related morbidity and mortality in adults: updated evidence report and systematic review for the US Preventive Services Task Force. Jama 2018; 320: 1172-1191.
52. Markovic TP, Proietto J, Dixon JB, et al. The Australian Obesity Management Algorithm: A Simple Tool to Guide the Management of Obesity in Primary Care. Sydney: Australian Diabetes Society; 2024. Available online at: https://www.diabetessociety.com.au/wp-content/uploads/2024/07/Australian-Obesity-Management-Algorithm-July-2024.pdf (accessed July 2025).
53. Australian Government. Physical Activity and Exercise Guidelines for Adults
(18 to 64 years). Canberra: Australian Government; 2021. Available online at: https://www.health.gov.au/health-topics/physical-activity-and-exercise/physical-activity-and-exercise-guidelines-for-all-australians/for-adults-18-to-64-years (accessed July 2025).
54. Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med 2023; 389: 2221-2232.
55. Bliddal H, Bays H, Czernichow S, et al. Once-weekly semaglutide in persons with obesity and knee osteoarthritis. N Engl J Med 2024; 391: 1573-1583.
56. Kosiborod MN, Abildstrøm SZ, Borlaug BA, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med 2023; 389: 1069-1084.
57. Sanyal AJ, Newsome PN, Kliers I, et al. Phase 3 trial of semaglutide in metabolic dysfunction-associated steatohepatitis. N Engl J Med 2025; 392: 2089-2099.
58. Loomba R, Hartman ML, Lawitz EJ, et al. Tirzepatide for metabolic dysfunction-associated steatohepatitis with liver fibrosis. N Engl J Med 2024; 391: 299-310.
59. Packer M, Zile MR, Kramer CM, et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N Engl J Med 2025; 392: 427-437.
60. Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N Engl J Med 2024; 391: 1193-1205.
61. Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab 2022; 24: 1553-1564.
62. Billes SK, Sinnayah P, Cowley MA. Naltrexone/bupropion for obesity: an investigational combination pharmacotherapy for weight loss. Pharmacol Res 2014; 84: 1-11.
63. Billes SK, Greenway FL. Combination therapy with naltrexone and bupropion for obesity. Expert Opin Pharmacother 2011; 12: 1813-1826.
64. Aronne LJ, Horn DB, Roux CWl, et al. Tirzepatide as compared with semaglutide for the treatment of obesity. N Engl J Med. Epub ahead of print. Doi: 10.1056/NEJMoa2416394.
65. Zhang Q, Delessa CT, Augustin R, et al. The glucose-dependent insulinotropic polypeptide (GIP) regulates body weight and food intake via CNS-GIPR signaling. Cell Metab 2021; 33: 833-44.e5.
66. Samms RJ, Christe ME, Collins KA, et al. GIPR agonism mediates weight-independent insulin sensitization by tirzepatide in obese mice. J Clin Invest 2021; 131: e146353.
67. Adriaenssens AE, Biggs EK, Darwish T, et al. Glucose-dependent insulinotropic polypeptide receptor-expressing cells in the hypothalamus regulate food intake. Cell Metab 2019; 30: 987-96.e6.
68. Seino Y, Fukushima M, Yabe D. GIP and GLP-1, the two incretin hormones: similarities and differences. J Diabetes Investig 2010; 1: 8-23.
69. Regmi A, Aihara E, Christe ME, et al. Tirzepatide modulates the regulation of adipocyte nutrient metabolism through long-acting activation of the GIP receptor. Cell Metab 2024; 36: 1534-49.e7.
70. Kagdi S, Lyons SA, Beaudry JL. The interplay of glucose-dependent insulinotropic polypeptide in adipose tissue. J Endocrinol 2024; 261: e230361.
71. Müller TD, Blüher M, Tschöp MH, DiMarchi RD. Anti-obesity drug discovery: advances and challenges. Nat Rev Drug Discov 2022; 21: 201-223.
72. Garvey WT, Mechanick JI, Brett EM, et al. American Association of Clinical Endocrinologists and American College of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pract 2016; 22 (Suppl 3): 1-203.
73. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care 2016; 39: 861-877.
74. Eisenberg D, Shikora SA, Aarts E, et al. 2022 American Society of Metabolic and Bariatric Surgery (ASMBS) and International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO) indications for metabolic and bariatric surgery. Obes Surg 2023; 33: 3-14.
75. American Diabetes Association Professional Practice Committee. 8. Obesity and weight management for the prevention and treatment of type 2 diabetes: standards of care in diabetes–2025. Diabetes Care 2024; 48: S167-S80.
76. Caleyachetty R, Barber TM, Mohammed NI, et al. Ethnicity-specific BMI cutoffs for obesity based on type 2 diabetes risk in England: a population-based cohort study. Lancet Diabetes Endocrinol 2021; 9: 419-426.
77. Deurenberg P, Yap M, van Staveren WA. Body mass index and percent body fat: a meta analysis among different ethnic groups. Int J Obes Relat Metab Disord 1998; 22: 1164-1171.
78. Razak F, Anand SS, Shannon H, et al. Defining obesity cut points in a multiethnic population. Circulation 2007; 115: 2111-2118.
79. Hsu WC, Araneta MR, Kanaya AM, Chiang JL, Fujimoto W. BMI cut points to identify at-risk Asian Americans for type 2 diabetes screening. Diabetes Care 2015; 38: 150-158.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.