Cardiovascular protection in chronic kidney disease: considerations for pre- and post-transplant care
Patients who receive immunosuppression after kidney transplantation or to minimise kidney injury from other systemic immune disorders are at high risk of cardiovascular disease. GPs are likely to care for an increasing number of these patients, whose cardiovascular risk profile needs to be carefully managed.
- Chronic kidney disease (CKD) and kidney transplantation place patients at the highest risk of cardiovascular disease and major adverse cardiovascular events, independent of traditional risk factors. Immunosuppression compounds this risk by promoting metabolic changes.
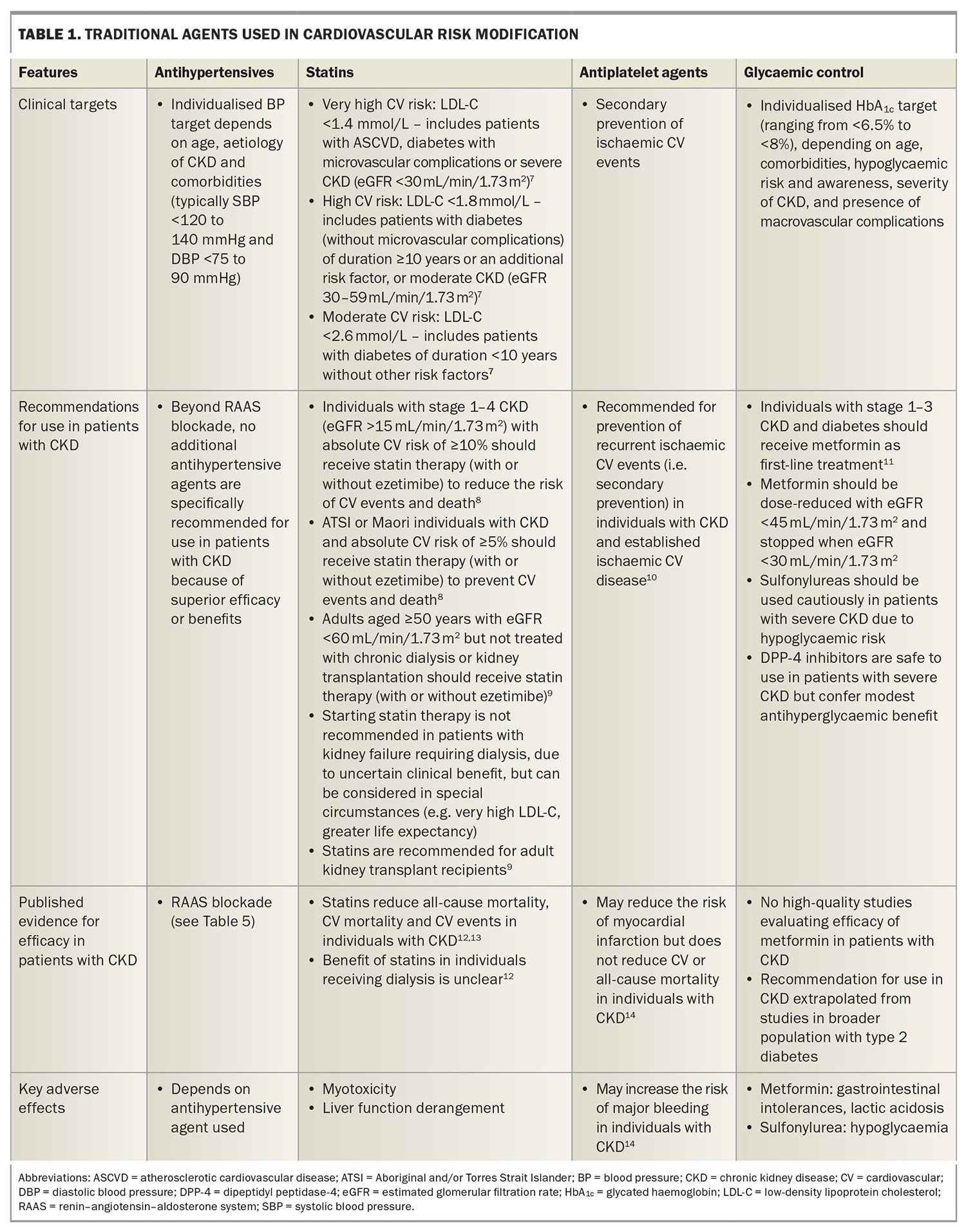
- Standard cardiovascular risk factors, such as hypertension, dyslipidaemia and diabetes, should be rigorously managed in patients with CKD or immunosuppression through lifestyle modification and appropriate pharmacological treatment.
- CKD-associated cardiovascular disease is typically driven by medial arterial calcification and left ventricular remodelling, which are poorly captured by standard diagnostic tools.
- The emergence of newer pharmacological agents, such as sodium-glucose cotransporter-2 inhibitors, mineralocorticoid receptor antagonists and glucagon-like peptide-1 receptor agonists, marks a shift towards treatment options that may modify both kidney and cardiovascular disease progression.
- Co-management frameworks that involve cardiology and nephrology can minimise critical gaps in management to address the complex pathophysiology and treatment needs of patients with CKD.
Patients with chronic kidney disease (CKD) experience a high burden of cardiovascular disease (CVD), such that major adverse cardiovascular events (MACE) constitute the leading cause of mortality in this group.1 Although this can be partly explained by traditional risk factors, such as diabetes, hypertension and dyslipidaemia, there are nontraditional risk factors, such as anaemia, metabolic bone disease and accumulating uraemic toxins, that are highly relevant to patients with CKD and that further amplify this risk. Both reduced kidney function, manifested as a low glomerular filtration rate (GFR), and albuminuria are independently associated with cardiovascular mortality, and this risk increases with disease progression.2,3
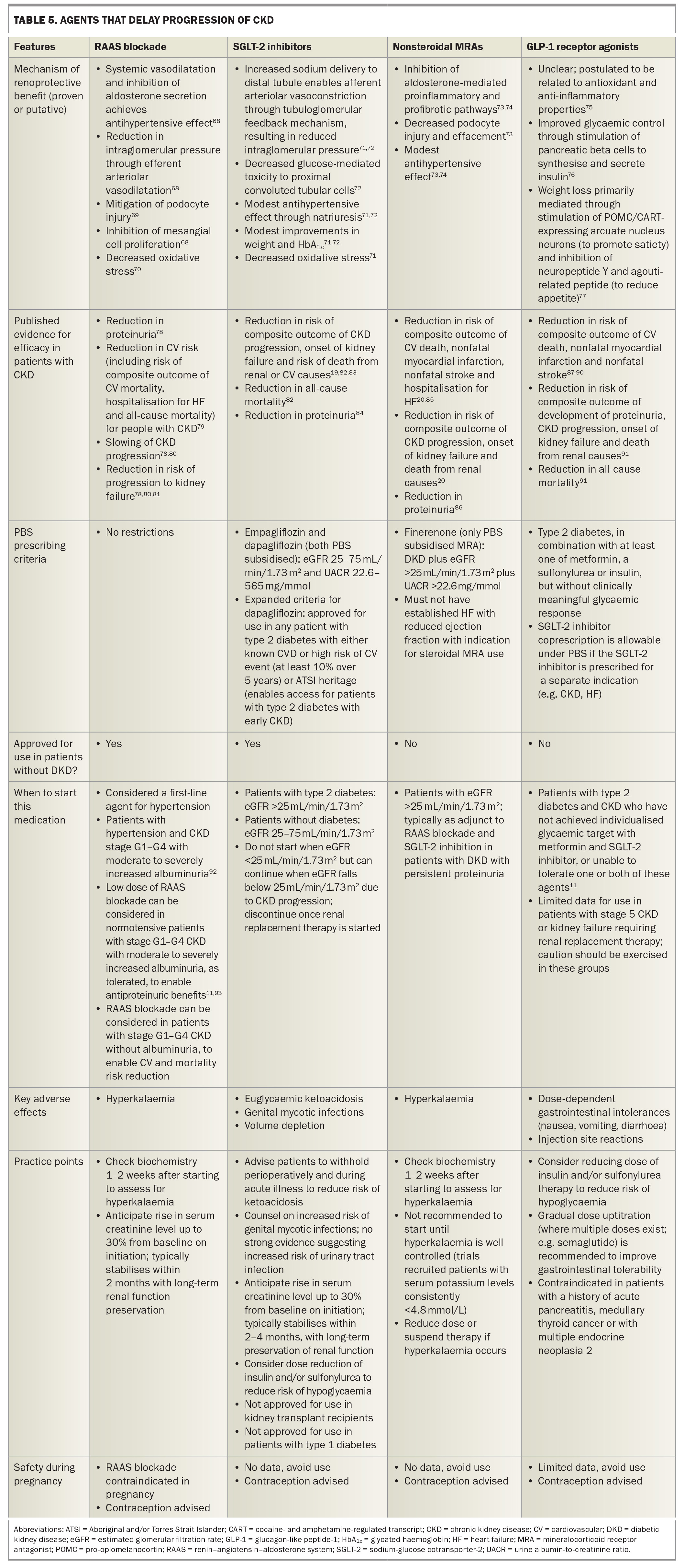
Traditional risk factor modification with lipid-lowering therapy, insulin-sensitising agents and antihypertensives provide modest (or no) cardiovascular benefit in patients with CKD,4-6 although these are valuable in the general population (Table 1).7-14 Furthermore, patients with CKD have been excluded from more than 80% of clinical studies;15-18 although this trend has reversed somewhat with the advent of sodium-glucose cotransporter-2 (SGLT-2) inhibitors, nonsteroidal mineralocorticoid receptor antagonists (MRAs) and glucagon-like peptide-1 (GLP-1) receptor agonists.19-21
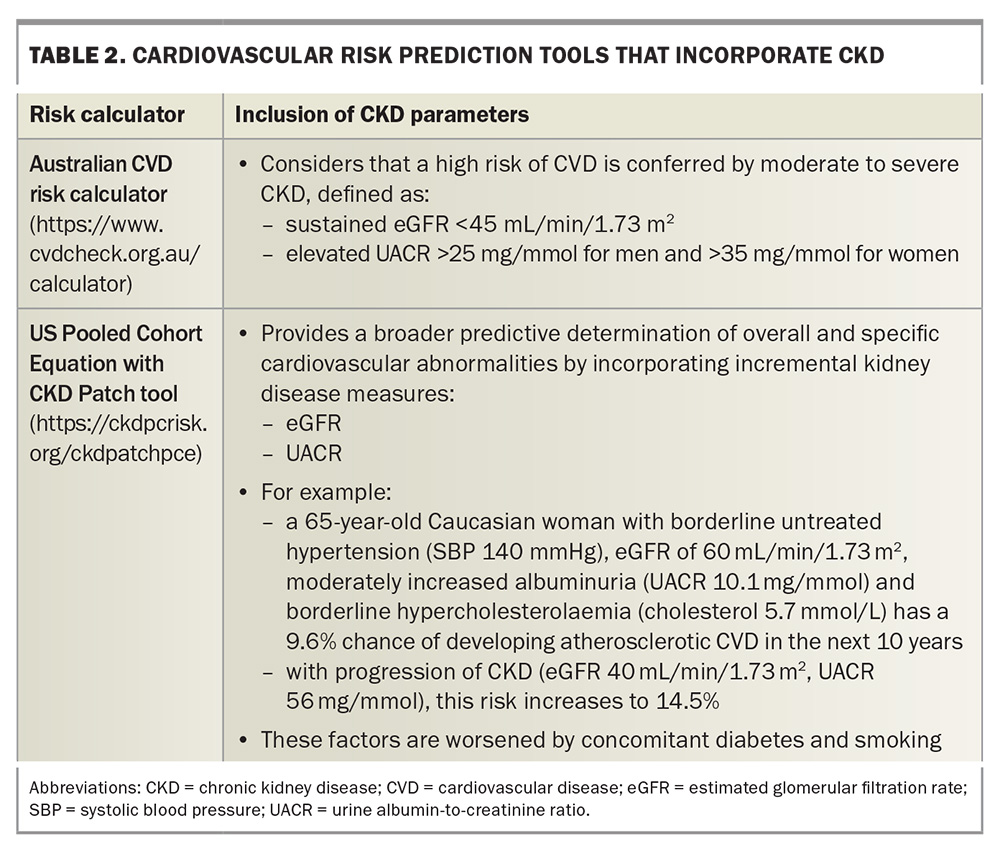
Due to the bidirectional relationship between the heart and kidneys, dysfunction of one organ can contribute to pathological changes in the other – a phenomenon known as cardiorenal syndrome.22 A burgeoning body of evidence links CKD with CVD, and many online cardiovascular risk prediction tools now incorporate parameters relevant to CKD stage (Table 2).
The impact of CKD on CVD is further compounded by the rising incidence of type 2 diabetes, which is now the third most common chronic disease, affecting 1.5 million Australians and contributing to more than one million hospitalisations each year.23,24 Optimal medical management of diabetes does not provide normal metabolic control, further affecting cardiac and renal function. Diabetes is now the most common cause of CKD and is a comorbidity in more than half of people starting renal replacement therapy (dialysis).25 Despite advances in surveillance and risk factor modification, CVD is the most prevalent cause of morbidity and mortality in people with diabetes and concomitant CKD.26
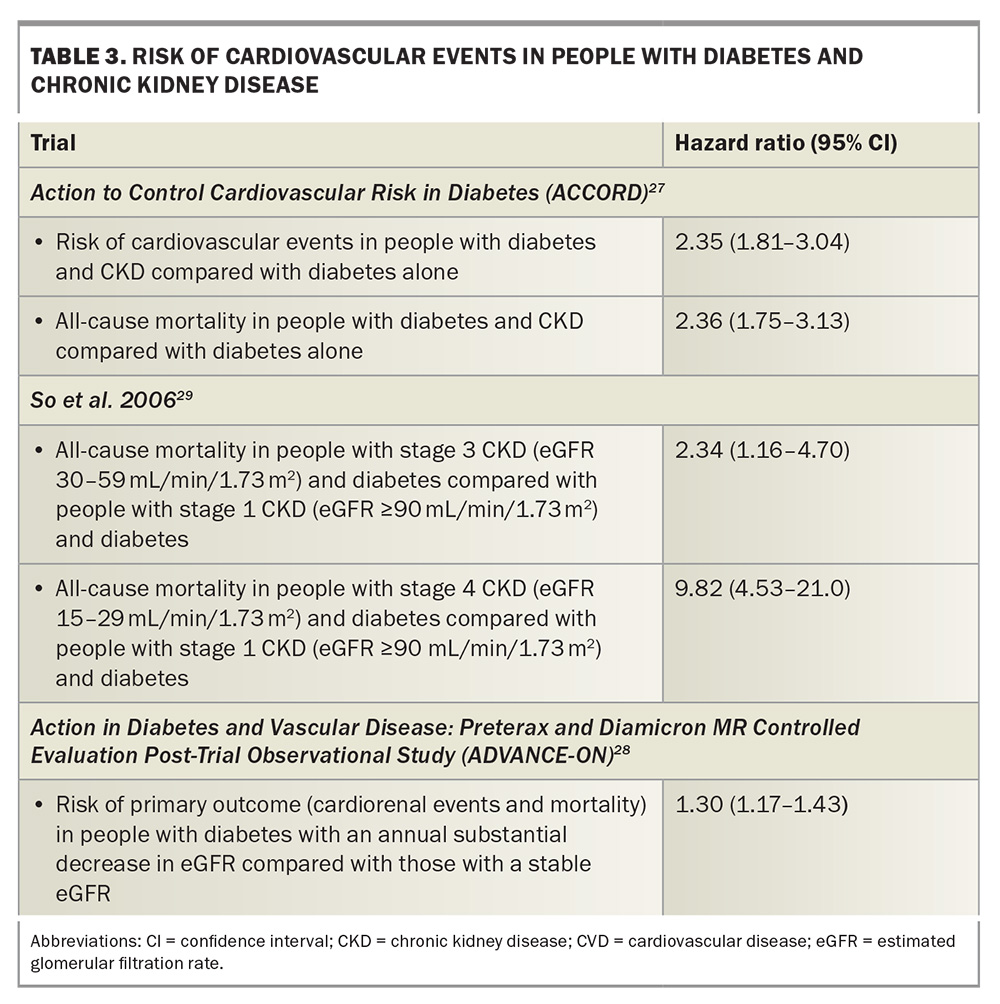
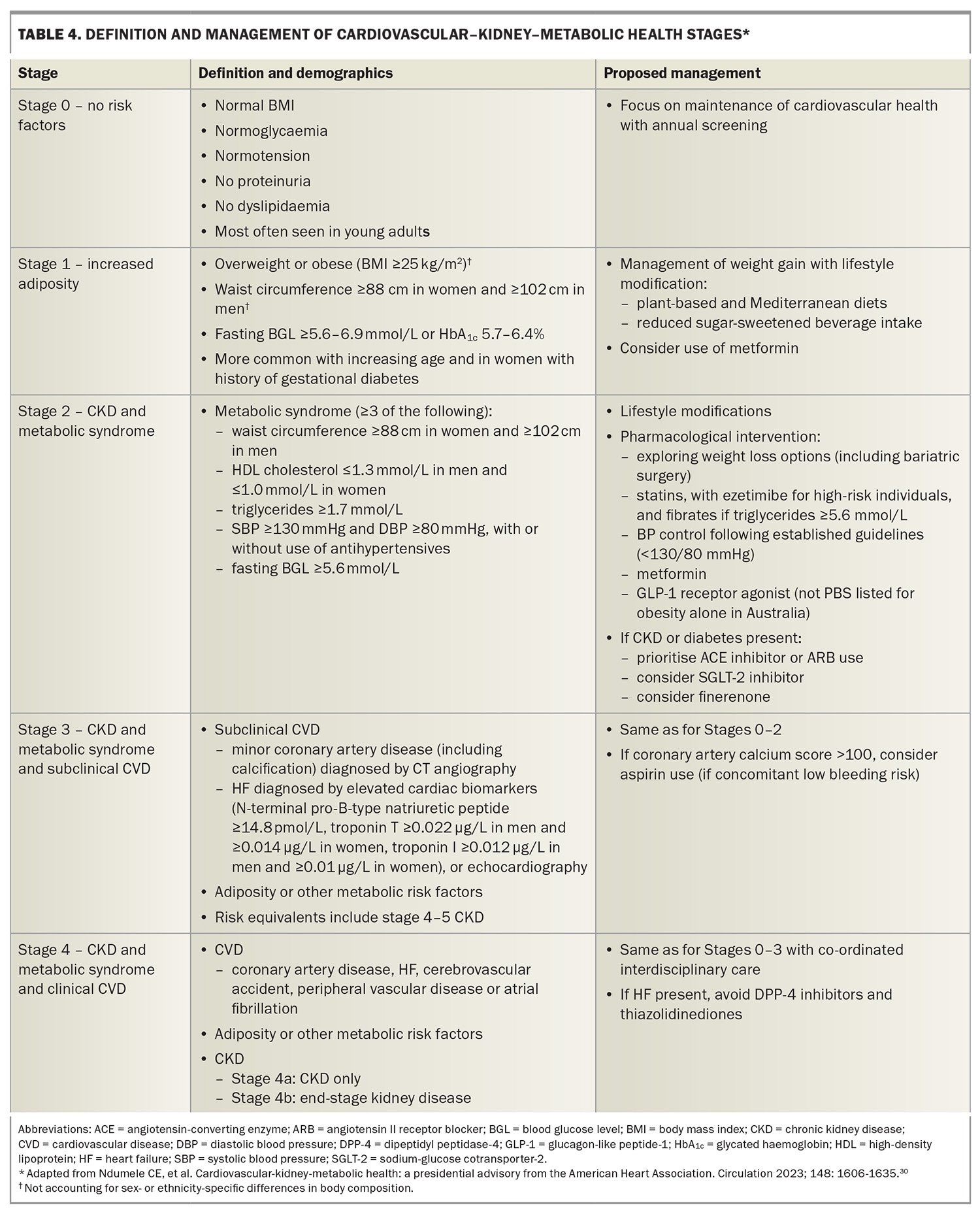
The combination of diabetes and CKD (so-called cardiovascular–kidney–metabolic health) leads to multimorbidity, further accelerates the burden of CVD and multiplies the risk of MACE (Table 3).27-29 People with multimorbidity use more health services, require increased contact with primary and tertiary health care, experience more complex hospitalisations and have poorer outcomes than those with single conditions. An improved recognition of poor cardiovascular–kidney–metabolic health and its intersection with socioeconomic disadvantage, including understanding its prevalence and proactive management strategies, is a centrepiece of the American Heart Association and increasingly recognised by Diabetes Australia and Kidney Health Australia.30-32 The American Heart Association has provided a 10-point recommendation that encompasses risk stratification (Table 4), screening and treatment approaches to facilitate early treatment and interdisciplinary care models.30
Chronic kidney disease-specific considerations when investigating cardiovascular disease
There are limited Australian data on differences in coronary artery disease burden in patients with CKD compared with the general population, with international studies estimating a relative risk of five- to 20-fold and a contribution to nearly 50% of deaths in those receiving dialysis.33 Even patients with CKD who had initially normal coronary angiogram results showed an increased risk of acute myocardial infarction,34 supporting the view of accelerated disease processes and poor outcomes.
The likely molecular basis for this is medial thickening and calcification (rather than just lesions limited to the intimal layer), in addition to the accumulation of uraemic toxins (even in early-stage CKD) that promote inflammation and oxidative stress. Left ventricular hypertrophy is also prevalent due to increased afterload (systolic hypertension), preload (volume expansion) and systemic inflammatory changes that result in fibrosis and cardiac remodelling, similarly portending a poor prognosis.35,36 Patients with CKD are more likely to present with a cardiac event driven by small vessel disease that presents as unexplained dyspnoea, fatigue or arrhythmia, rather than classic ST-segment electrocardiographic changes.
Echocardiography is a useful initial step to investigate the (potential) burden of left ventricular hypertrophy, subsequent systolic or diastolic dysfunction and risk of sudden death. However, there is a lack of clear guidelines for echocardiographic examination in patients with CKD. Although the Acute Dialysis Quality Initiative working group provided a new classification, it is most relevant to end-stage kidney disease.37 The clinical vignettes in the Box illustrate how CVD can remain hidden in patients with CKD, as well as the limitations related to noninvasive testing for CVD in these patients.
Evaluation of left atrium function
The left atrium (LA) is increasingly recognised as a potential marker of cardiovascular disease and adverse outcomes, and both LA size and strain are relevant measurements.38 Although there is no definitive single marker of LA function, strain (which measures myocardial deformation) and strain rate can be measured using:
- tissue Doppler imaging – where offline measurements are taken from colour tissue Doppler images
- two-dimensional speckle tracking echocardiography – which tracks ‘speckles’ or natural acoustic markers in the two-dimensional ultrasound image
- velocity vector imaging – which combines speckle tracking and endocardial border tracking.
LA strain correlates with diastolic dysfunction.39 LA enlargement is well documented in end-stage kidney disease, as it likely correlates with left ventricular hypertrophy and archetypal risk factors (hypertension and diabetes). CKD itself may affect LA function (particularly strain patterns) and precedes changes in size. This is postulated to occur through mechanisms such as volume expansion due to activation of the renin–angiotensin–aldosterone system (RAAS), reduced vascular compliance due to endothelial dysfunction and medial calcification, chronic inflammation and oxidative stress, and accumulation of uraemic toxins. LA reservoir strain has been shown to independently predict cardiovascular death and MACE in people with CKD, as well as progression to end-stage kidney disease, independent of established clinical risk factors.40,41
Accordingly, routine evaluation of LA function presents an opportunity to improve cardiovascular risk stratification in patients with CKD. However, limitations – such as lack of widespread availability and analytical expertise in such evaluation, and a lack of clinical studies showing that targeting the proposed pathways which promote LA dysfunction mitigates MACE – need to be overcome for this analysis to become embedded in routine clinical practice.
Challenges in screening for coronary artery disease
Screening for coronary artery disease in patients with CKD remains challenging and is typically performed in partnership with nephrologists and cardiologists. Available noninvasive investigations include dobutamine stress echocardiography, myocardial perfusion scintigraphy and coronary CT angiography.
In patients with CKD, dobutamine stress echocardiography has shown widely variable sensitivity (44 to 96%) and specificity (60 to 100%), with an overall diagnostic odds ratio (the odds of a positive test in those with disease relative to the odds of a positive test in those without) of 29.98.42 Myocardial perfusion scintigraphy performed even less robustly (sensitivity of 29 to 100% and specificity of 31 to 88%, diagnostic odds ratio of 6.69). These data suggest moderate diagnostic accuracy, which precludes using either of these tests as a general screening tool. Further, balanced triple vessel disease, submaximal heart rate during testing and subjective interpretation of results also contribute to reduced specificity.
Coronary CT angiography is less useful in patients with CKD than in the general population, as extensive coronary artery calcification does not always correlate with significant stenosis and may lead to gross overestimation of luminal stenosis.43 However, coronary CT angiography, particularly when combined with myocardial perfusion scintigraphy, was found to be useful in ruling out obstructive coronary artery disease and was recommended for potential kidney transplant recipients.44 A recent small meta-analysis found lower sensitivity in patients with CKD compared with the general population.45
Invasive coronary angiography
In patients with CKD presenting with non-ST-elevation acute coronary syndrome, early use of invasive coronary angiography significantly reduced the risk of rehospitalisation but did not substantially reduce risk of death or re-infarction rates.46 The International Study of Comparative Health Effectiveness With Medical and Invasive Approaches-Chronic Kidney Disease (ISCHEMIA-CKD) trial found that an initial invasive strategy (angiography with or without revascularisation) did not reduce the risk of death or nonfatal myocardial infarction compared with medical therapy alone, and this mirrored findings in people with normal renal function.47,48
Furthermore, as CKD progresses, the risks (and potential consequences) of angiography increase. These risks include major and minor bleeding, restenosis and death after percutaneous coronary intervention, and risk of acute kidney injury secondary to contrast exposure, which occurs at a higher rate than in the general population.49,50
Cardiovascular implications of immunosuppression in patients with chronic kidney disease
Kidney transplantation, when compared with alternative renal replacement therapies (dialysis), offers the best cardiovascular and long-term survival outcomes, highlighting the beneficial effects of ameliorating nontraditional cardiovascular risk factors through a functioning renal allograft and ‘normalising’ GFR.51,52 However, a significant and sustained improvement in renal function is only possible through strict adherence to lifelong immunosuppression to prevent allograft rejection. Paradoxically, the very immunosuppression that is directly responsible for achieving improved renal function is associated with adverse cardiometabolic effects that concurrently increase a patient’s cardiovascular risk and expedite development of CVD. This complex balance is highly relevant for kidney transplant recipients, who still experience a higher risk of CVD than the general population and in whom this remains the leading cause of mortality.52-54
Kidney transplant recipients are not the only patients with CKD who require immunomodulation as standard of care. Systemic immune disorders – a consequence of autoimmunity, haematological malignancy or mutations in complement genes – can manifest primarily as glomerular disease, requiring immunosuppression to effectively minimise kidney injury.
Although antiproliferative agents (e.g. mycophenolate mofetil and azathioprine) are not considered to exacerbate cardiovascular risk, pivotal immunosuppressive agents, such as calcineurin inhibitors (CNIs) and corticosteroids, as well as mammalian target of rapamycin inhibitors (mTORIs) that are designated for use in certain circumstances, are strongly associated with increased cardiovascular risk. The key pathophysiological mechanisms for this increased cardiovascular risk are summarised below.
Diabetes
New-onset diabetes after transplant (NODAT), which affects about 25% of kidney transplant recipients, is a consequential complication of transplantation, leading to an increased risk of CVD, graft failure and infections, and decreased patient survival.55-57 Immunosuppressive therapy significantly contributes to the development of NODAT, with a dose-dependent relationship explaining why most cases of NODAT are diagnosed in the first three months after transplantation.56
Corticosteroids exert diabetogenic effects, including impairing pancreatic beta islet cell secretory function and glucose sensitivity, promoting insulin resistance in peripheral tissues by directly interfering with components of the insulin signalling cascade, and increasing hepatic gluconeogenesis.57,58 CNIs also impair pancreatic beta islet cell growth and function and reduce adipocyte expression of glucose transporter type 4 receptors, which hinder insulin secretion and promote peripheral insulin resistance, respectively.57,59,60 Further, CNIs are directly toxic to pancreatic beta islet cells, with increased beta cell vacuolisation seen in patients with higher CNI trough levels.57 Notably, tacrolimus has been linked with a higher risk of NODAT than cyclosporin. Finally, mTORIs exert toxic effects on pancreatic beta islet cells and impair insulin-mediated suppression of hepatic glucose production.54
Hypertension
Hypertension is highly prevalent in kidney transplant recipients, constituting a leading cause of allograft dysfunction and incident CVD.52 Standard immunosuppressive therapies, such as CNIs and corticosteroids, are key drivers of post-transplant hypertension. Corticosteroids promote hypertension primarily through their modulation of vascular smooth muscle tone, which increases peripheral vascular resistance, with renal sodium retention also thought to play a role.54 CNIs increase blood pressure through multiple mechanisms, including sympathetic nervous system activation, afferent arteriolar vasoconstriction, activation of the RAAS, oxidative stress, and, in the long term, their contribution towards chronic allograft nephropathy.52-54
Dyslipidaemia
Immunosuppressive agents also contribute to the development of dyslipidaemia after transplantation. CNIs, particularly cyclosporin, interfere with low-density lipoprotein (LDL) cholesterol binding, leading to reduced LDL clearance.54 Corticosteroids promote dyslipidaemia through multiple mechanisms, including increased hepatic synthesis of very-low-density lipoprotein (VLDL) cholesterol, increased conversion of VLDL to LDL cholesterol, increased activity of 3-hydroxy-3-methylglutaryl coenzyme A (which drives cholesterol synthesis) and impaired triglyceride clearance through reduced lipoprotein lipase activity. mTORIs are strongly linked with dyslipidaemia through reduced lipoprotein lipase activity, hepatic overproduction of lipoprotein and increased secretion of VLDL cholesterol.54 Transition from a CNI to an mTORI, typically done to avoid CNI toxicities, is associated with a two- to threefold increase in the risk of dyslipidaemia.61
Management considerations
Immunosuppression
Recognition of the significant cardiovascular sequelae of immunosuppression has inspired attempts to implement CNI-free or corticosteroid-free regimens; however, these have been unsuccessful owing to an unacceptable risk of acute allograft rejection.62 Consideration of a patient’s individualised cardiovascular risk when selecting and reviewing an immunosuppression regimen and appropriately reducing the doses of immunosuppression – particularly CNIs and corticosteroids – in patients considered to have a lower risk of rejection are important ways in which clinicians can identify, and reduce, iatrogenic cardiovascular risk.
Modification of traditional cardiovascular risk factors
Aggressive modification of traditional cardiovascular risk factors in patients with CKD, and particularly kidney transplant recipients, requires greater attention (Table 1).63 Almost one-third of kidney transplant recipients do not meet the blood pressure target of less than 130/80 mmHg, and one-fifth have borderline or elevated LDL cholesterol levels.64
With respect to hypertension management, dihydropyridine calcium channel blockers have been identified as a preferred antihypertensive due to their attenuation of CNI-induced vasoconstriction.52 Calcium channel blockers have shown greater preservation of renal function when compared with placebo, as well as RAAS blockade.65
Additionally, initiation of statin therapy to treat elevated LDL cholesterol levels has been shown to be beneficial in kidney transplant recipients, with a randomised controlled trial finding that moderate- to high-dose fluvastatin achieved a 38% reduction in risk of cardiovascular mortality and a 32% reduction in risk of nonfatal myocardial infarction, despite not significantly reducing the risk of MACE.66 However, caution must be exercised when coadministering statins with CNIs because of the risk of increased systemic statin exposure and possible myotoxicity from the competitive inhibitory effect of cyclosporin on cytochrome P450 3A4, impairing statin catabolism.67
Pharmacotherapy to slow progression of chronic kidney disease
Newer pharmacotherapeutic agents – SGLT-2 inhibitors, GLP-1 receptor agonists and MRAs (finerenone) – have all been shown to play a central role in slowing the progression of CKD (Table 5).68-94 These agents complement the pre-existing treatment rationale of using RAAS blockers, but patients remain at higher risk of disease progression and MACE, suggesting that the therapeutic ceiling has not yet been reached. The nephroprotective effects of these agents (irrespective of diabetes status) reflect haemodynamic, metabolic and anti-inflammatory changes that have been poorly characterised in human studies but robustly supported by many clinical trials and meta-analyses.68-91 Treatment strategies must be tailored to a patient’s CKD stage, comorbidities and risk of adverse events (e.g. persistent deterioration in eGFR or hyperkalaemia).
Conclusion
The benefits and cardiovascular side effect profile of immunosuppression after kidney transplantation pose a complex clinical dilemma. Clinicians are tasked with striking a balance between avoiding under-immunosuppression – to successfully prevent allograft rejection, preserve renal function and ultimately reduce a patient’s cardiovascular and mortality risk – while avoiding over-immunosuppression and its anticipated cardiovascular sequelae. To improve cardiovascular outcomes for kidney transplant recipients and other patients who require immunosuppression for preservation of native kidney function, frequent evaluation of the immunosuppression regimen, with individualised modification of immunosuppression dosing where appropriate, should complement traditional cardiovascular risk reduction measures. A focus on cardiovascular–kidney–metabolic health, with increasing clinician and consumer awareness, multidisciplinary care, appropriate screening and pharmacological intervention, is an important consideration for integration into clinical care. MT
COMPETING INTERESTS: None.
References
1. Ravid JD, Kamel MH, Chitalia VC. Uraemic solutes as therapeutic targets in CKD-associated cardiovascular disease. Nat Rev Nephrol 2021; 17: 402-416.
2. Fox CS, Matsushita K, Woodward M, et al. Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: a meta-analysis. Lancet 2012; 380: 1662-1673.
3. Mahmoodi BK, Matsushita K, Woodward M, et al. Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without hypertension: a meta-analysis. Lancet 2012; 380: 1649-1661.
4. Strippoli GF, Navaneethan SD, Johnson DW, et al. Effects of statins in patients with chronic kidney disease: meta-analysis and meta-regression of randomised controlled trials. BMJ 2008; 336: 645-651.
5. Chan DT, Watts GF, Irish AB, Dogra GK. Rosiglitazone does not improve vascular function in subjects with chronic kidney disease. Nephrol Dial Transplant 2011; 26: 3543-3549.
6. Zannad F, Kessler M, Lehert P, et al. Prevention of cardiovascular events in end-stage renal disease: results of a randomized trial of fosinopril and implications for future studies. Kidney Int 2006; 70: 1318-1324.
7. Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 2020; 41: 111-188.
8. Cashmore B, Tunnicliffe DJ, Palmer S, et al. Australian and New Zealand living guideline cholesterol-lowering therapy for people with chronic kidney disease (CARI guidelines): reducing the evidence-practice gap. Nephrology (Carlton) 2024; 29: 495-509.
9. Wanner C, Tonelli M, Kidney Disease: Improving Global Outcomes Lipid Guideline Development Work Group Members. KDIGO clinical practice guideline for lipid management in CKD: summary of recommendation statements and clinical approach to the patient. Kidney Int 2014; 85: 1303-1309.
10. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int 2024; 105(4S): S117-S314.
11. Kidney Disease: Improving Global Outcomes (KDIGO) Diabetes Work Group. KDIGO 2022 clinical practice guideline for diabetes management in chronic kidney disease. Kidney Int 2022; 102(5S): S1-S127.
12. Palmer SC, Craig JC, Navaneethan SD, Tonelli M, Pellegrini F, Strippoli GF. Benefits and harms of statin therapy for persons with chronic kidney disease: a systematic review and meta-analysis. Ann Intern Med 2012; 157: 263-275.
13. Upadhyay A, Earley A, Lamont JL, Haynes S, Wanner C, Balk EM. Lipid-lowering therapy in persons with chronic kidney disease: a systematic review and meta-analysis. Ann Intern Med 2012; 157: 251-262.
14. Natale P, Palmer SC, Saglimbene VM, et al. Antiplatelet agents for chronic kidney disease. Cochrane Database Syst Rev 2022; 2(2): CD008834.
15. Pickup L, Law JP, Townend J, Ferro CJ. Cardiorenal medicine: an emerging new speciality or a need for closer collaboration? Br J Cardiol 2020; 27: 77-78.
16. Zannad F, Rossignol P. Cardiovascular outcome trials in patients with advanced kidney disease: time for action. Circulation 2017; 135: 1769-1771.
17. Konstantinidis I, Nadkarni GN, Yacoub R, et al. Representation of patients with kidney disease in trials of cardiovascular interventions: an updated systematic review. JAMA Intern Med 2016; 176: 121-124.
18. Charytan D, Kuntz RE. The exclusion of patients with chronic kidney disease from clinical trials in coronary artery disease. Kidney Int 2006; 70: 2021-2030.
19. The EMPA-KIDNEY Collaborative Group, Herrington WG, Staplin N, et al. Empagliflozin in patients with chronic kidney disease. N Engl J Med 2023; 388: 117-127.
20. Bakris GL, Agarwal R, Anker SD, et al. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med 2020; 383: 2219-2229.
21. Perkovic V, Tuttle KR, Rossing P, et al. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med 2024; 391: 109-121.
22. Ronco C, McCullough P, Anker SD, et al. Cardio-renal syndromes: report from the consensus conference of the acute dialysis quality initiative. Eur Heart J 2010; 31: 703-711.
23. Zimmet PZ. Diabetes and its drivers: the largest epidemic in human history? Clin Diabetes Endocrinol 2017; 3: 1.
24. Australian Institute of Health and Welfare. Diabetes: Australian facts. Canberra; AIHW; 2023. Available online at: https://www.aihw.gov.au/reports/diabetes/diabetes/contents/summary (accessed July 2025).
25. Australia and New Zealand Dialysis and Transplant Registry (ANZDATA). Chapter 1: Incidence of kidney failure with replacement therapy. In: 44th Annual ANZDATA report 2021. Adelaide: ANZDATA; 2021. Available online at: https://www.anzdata.org.au/report/anzdata-44th-annual-report-2021-data-to-2020/ (accessed July 2025).
26. Marx N, Federici M, Schutt K, et al. 2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes. Eur Heart J 2023; 44: 4043-4140.
27. Branch M, German C, Bertoni A, Yeboah J. Incremental risk of cardiovascular disease and/or chronic kidney disease for future ASCVD and mortality in patients with type 2 diabetes mellitus: ACCORD trial. J Diabetes Complications 2019; 33: 468-472.
28. Oshima M, Jun M, Ohkuma T, et al. The relationship between eGFR slope and subsequent risk of vascular outcomes and all-cause mortality in type 2 diabetes: the ADVANCE-ON study. Diabetologia 2019; 62: 1988-1997.
29. So WY, Kong AP, Ma RC, et al. Glomerular filtration rate, cardiorenal end points, and all-cause mortality in type 2 diabetic patients. Diabetes Care 2006; 29: 2046-2052.
30. Ndumele CE, Rangaswami J, Chow SL, et al. Cardiovascular-kidney-metabolic health: a presidential advisory from the American Heart Association. Circulation 2023; 148: 1606-1635.
31. Kwok R, MacIsaac R, Ekinci E. Change the future: saving lives by better detecting diabetes-related kidney disease. Canberra: Diabetes Australia; 2023. Available online at: https://www.diabetesaustralia.com.au/wp-content/uploads/2023-Diabetes-Related-Kidney-Disease-Report-1.4-DIGITAL.pdf (accessed July 2025).
32. White SL. Make the link: kidneys, diabetes & heart. Chronic kidney disease, diabetes & cardiovascular disease: evidence report 2021. Melbourne: Kidney Health Australia; 2020. Available online at: https://assets.kidney.org.au/resources/KHA-Report-Make-the-link-kidneys-diabetes-heart.pdf (accessed July 2025).
33. Ohtake T, Kobayashi S, Moriya H, et al. High prevalence of occult coronary artery stenosis in patients with chronic kidney disease at the initiation of renal replacement therapy: an angiographic examination. J Am Soc Nephrol 2005; 16: 1141-1148.
34. Herzog CA, Ma JZ, Collins AJ. Poor long-term survival after acute myocardial infarction among patients on long-term dialysis. N Engl J Med 1998; 339: 799-805.
35. Xu C, Tsihlis G, Chau K, Trinh K, Rogers NM, Julovi SM. Novel perspectives in chronic kidney disease-specific cardiovascular disease. Int J Mol Sci 2024; 25: 2658.
36. Hillege HL, Nitsch D, Pfeffer MA, et al. Renal function as a predictor of outcome in a broad spectrum of patients with heart failure. Circulation 2006; 113: 671-678.
37. Chawla LS, Herzog CA, Costanzo MR, et al. Proposal for a functional classification system of heart failure in patients with end-stage renal disease: proceedings of the Acute Dialysis Quality Initiative (ADQI) XI workgroup. J Am Coll Cardiol 2014; 63: 1246-1252.
38. Gan GCH, Ferkh A, Boyd A, Thomas L. Left atrial function: evaluation by strain analysis. Cardiovasc Diagn Ther 2018; 8: 29-46.
39. Gottdiener JS, Kitzman DW, Aurigemma GP, Arnold AM, Manolio TA. Left atrial volume, geometry, and function in systolic and diastolic heart failure of persons ≥65 years of age (the Cardiovascular Health Study). Am J Cardiol 2006; 97: 83-89.
40. Gan GCH, Kadappu KK, Bhat A, et al. Left atrial strain is the best predictor of adverse cardiovascular outcomes in patients with chronic kidney disease. J Am Soc Echocardiogr 2021; 34: 166-175.
41. Gan GCH, Bhat A, Kadappu KK, et al. Usefulness of left atrial strain to predict end stage renal failure in patients with chronic kidney disease. Am J Cardiol 2021; 151: 105-113.
42. Wang LW, Fahim MA, Hayen A, et al. Cardiac testing for coronary artery disease in potential kidney transplant recipients. Cochrane Database Syst Rev 2011; 2011(12): CD008691.
43. Sharples EJ, Pereira D, Summers S, et al. Coronary artery calcification measured with electron-beam computerized tomography correlates poorly with coronary artery angiography in dialysis patients. Am J Kidney Dis 2004; 43: 313-319.
44. Winther S, Svensson M, Jorgensen HS, et al. Diagnostic performance of coronary CT angiography and myocardial perfusion imaging in kidney transplantation candidates. JACC Cardiovasc Imaging 2015; 8: 553-562.
45. Cheng XS, Mohanty S, Turner V, et al. Coronary computed tomography angiography in diagnosing obstructive coronary artery disease in patients with advanced chronic kidney disease: a systematic review and meta-analysis. Cardiorenal Med 2021; 11: 44-51.
46. Charytan DM, Wallentin L, Lagerqvist B, et al. Early angiography in patients with chronic kidney disease: a collaborative systematic review. Clin J Am Soc Nephrol 2009; 4: 1032-1043.
47. Bangalore S, Maron DJ, O’Brien SM, et al. Management of coronary disease in patients with advanced kidney disease. N Engl J Med 2020; 382: 1608-1618.
48. Maron DJ, Hochman JS, Reynolds HR, et al. Initial invasive or conservative strategy for stable coronary disease. N Engl J Med 2020; 382: 1395-1407.
49. Attallah N, Yassine L, Fisher K, Yee J. Risk of bleeding and restenosis among chronic kidney disease patients undergoing percutaneous coronary intervention. Clin Nephrol 2005; 64: 412-418.
50. Prasad A, Rosenthal NA, Kartashov A, Knish K, Dreyfus J. Contemporary trend of acute kidney injury incidence and incremental costs among US patients undergoing percutaneous coronary procedures. Catheter Cardiovasc Interv 2020; 96: 1184-1197.
51. Meier-Kriesche HU, Schold JD, Srinivas TR, Reed A, Kaplan B. Kidney transplantation halts cardiovascular disease progression in patients with end-stage renal disease. Am J Transplant 2004; 4: 1662-1668.
52. Stoumpos S, Jardine AG, Mark PB. Cardiovascular morbidity and mortality after kidney transplantation. Transpl Int 2015; 28: 10-21.
53. Zeier M, Van Der Giet M. Calcineurin inhibitor sparing regimens using m-target of rapamycin inhibitors: an opportunity to improve cardiovascular risk following kidney transplantation? Transpl Int 2011; 24: 30-42.
54. Chakkera HA, Sharif A, Kaplan B. Negative cardiovascular consequences of small molecule immunosuppressants. Clin Pharmacol Ther 2017; 102: 269-276.
55. Kasiske BL, Snyder JJ, Gilbertson D, Matas AJ. Diabetes mellitus after kidney transplantation in the United States. Am J Transplant 2003; 3: 178-185.
56. Solhjoo M, Kumar SC. New onset diabetes after transplant. In: StatPearls. Treasure Island, Fla: StatPearls Publishing; 2025.
57. Chakkera HA, Kudva Y, Kaplan B. Calcineurin inhibitors: pharmacologic mechanisms impacting both insulin resistance and insulin secretion leading to glucose dysregulation and diabetes mellitus. Clin Pharmacol Ther 2017; 101: 114-120.
58. Hwang JL, Weiss RE. Steroid-induced diabetes: a clinical and molecular approach to understanding and treatment. Diabetes Metab Res Rev 2014; 30: 96-102.
59. Peev V, Reiser J, Alachkar N. Diabetes mellitus in the transplanted kidney. Front Endocrinol (Lausanne) 2014; 5: 141.
60. Pereira MJ, Palming J, Rizell M, et al. Cyclosporine A and tacrolimus reduce the amount of GLUT4 at the cell surface in human adipocytes: increased endocytosis as a potential mechanism for the diabetogenic effects of immunosuppressive agents. J Clin Endocrinol Metab 2014; 99: E1885-E1894.
61. Schena FP, Pascoe MD, Alberu J, et al. Conversion from calcineurin inhibitors to sirolimus maintenance therapy in renal allograft recipients: 24-month efficacy and safety results from the CONVERT trial. Transplantation 2009; 87: 233-242.
62. Haller MC, Royuela A, Nagler EV, Pascual J, Webster AC. Steroid avoidance or withdrawal for kidney transplant recipients. Cochrane Database Syst Rev 2016; 2016(8): CD005632.
63. Kidney Disease: Improving Global Outcomes (KDIGO) Transplant Work Group. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant 2009; 9 Suppl 3: S1-155.
64. Carpenter MA, Weir MR, Adey DB, House AA, Bostom AG, Kusek JW. Inadequacy of cardiovascular risk factor management in chronic kidney transplantation – evidence from the FAVORIT study. Clin Transplant 2012; 26: E438-E446.
65. Cross NB, Webster AC, Masson P, O’Connell P J, Craig JC. Antihypertensives for kidney transplant recipients: systematic review and meta-analysis of randomized controlled trials. Transplantation 2009; 88: 7-18.
66. Holdaas H, Fellstrom B, Jardine AG, et al. Effect of fluvastatin on cardiac outcomes in renal transplant recipients: a multicentre, randomised, placebo-controlled trial. Lancet 2003; 361: 2024-2031.
67. Lemahieu WP, Hermann M, Asberg A, et al. Combined therapy with atorvastatin and calcineurin inhibitors: no interactions with tacrolimus. Am J Transplant 2005; 5: 2236-2243.
68. Kobori H, Mori H, Masaki T, Nishiyama A. Angiotensin II blockade and renal protection. Curr Pharm Des 2013; 19: 3033-3042.
69. Suzuki K, Han GD, Miyauchi N, et al. Angiotensin II type 1 and type 2 receptors play opposite roles in regulating the barrier function of kidney glomerular capillary wall. Am J Pathol 2007; 170: 1841-1853.
70. Izuhara Y, Nangaku M, Inagi R, et al. Renoprotective properties of angiotensin receptor blockers beyond blood pressure lowering. J Am Soc Nephrol 2005; 16: 3631-3641.
71. Abdelrahman AM, Awad AS, Abdel-Rahman EM. Sodium-glucose co-transporter 2 inhibitors: mechanism of action and efficacy in non-diabetic kidney disease from bench to bed-side. J Clin Med 2024;13: 956.
72. Heerspink HJ, Perkins BA, Fitchett DH, Husain M, Cherney DZ. Sodium glucose cotransporter 2 inhibitors in the treatment of diabetes mellitus: cardiovascular and kidney effects, potential mechanisms, and clinical applications. Circulation 2016; 134: 752-772.
73. Barrera-Chimal J, Girerd S, Jaisser F. Mineralocorticoid receptor antagonists and kidney diseases: pathophysiological basis. Kidney Int 2019; 96: 302-319.
74. Kim DL, Lee SE, Kim NH. Renal protection of mineralocorticoid receptor antagonist, finerenone, in diabetic kidney disease. Endocrinol Metab (Seoul) 2023; 38: 43-55.
75. Lee B, Holstein-Rathlou NH, Sosnovtseva O, Sorensen CM. Renoprotective effects of GLP-1 receptor agonists and SGLT-2 inhibitors-is hemodynamics the key point? Am J Physiol Cell Physiol 2023; 325: C243-C256.
76. Zheng Z, Zong Y, Ma Y, et al. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal Transduct Target Ther 2024; 9: 234.
77. Ard J, Fitch A, Fruh S, Herman L. Weight loss and maintenance related to the mechanism of action of glucagon-like peptide 1 receptor agonists. Adv Ther 2021; 38: 2821-2839.
78. Jafar TH, Schmid CH, Landa M, et al. Angiotensin-converting enzyme inhibitors and progression of nondiabetic renal disease. A meta-analysis of patient-level data. Ann Intern Med 2001; 135: 73-87.
79. Mann JF, Gerstein HC, Pogue J, Bosch J, Yusuf S. Renal insufficiency as a predictor of cardiovascular outcomes and the impact of ramipril: the HOPE randomized trial. Ann Intern Med 2001; 134: 629-636.
80. Hsu TW, Liu JS, Hung SC, et al. Renoprotective effect of renin-angiotensin-aldosterone system blockade in patients with predialysis advanced chronic kidney disease, hypertension, and anemia. JAMA Intern Med 2014; 174: 347-354.
81. Xie X, Liu Y, Perkovic V, et al. Renin-angiotensin system inhibitors and kidney and cardiovascular outcomes in patients with CKD: a Bayesian network meta-analysis of randomized clinical trials. Am J Kidney Dis 2016; 67: 728-741.
82. Heerspink HJL, Stefansson BV, Correa-Rotter R, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020; 383: 1436-1446.
83. Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med 2019; 380: 2295-2306.
84. Piperidou A, Sarafidis P, Boutou A, et al. The effect of SGLT-2 inhibitors on albuminuria and proteinuria in diabetes mellitus: a systematic review and meta-analysis of randomized controlled trials. J Hypertens 2019; 37: 1334-1343.
85. Pitt B, Filippatos G, Agarwal R, et al. Cardiovascular events with finerenone in kidney disease and type 2 diabetes. N Engl J Med 2021; 385: 2252-2263.
86. Bakris GL, Agarwal R, Chan JC, et al. Effect of finerenone on albuminuria in patients with diabetic nephropathy: a randomized clinical trial. JAMA 2015; 314: 884-894.
87. Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 2019; 394: 121-130.
88. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375: 1834-1844.
89. Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375: 311-322.
90. Hernandez AF, Green JB, Janmohamed S, et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony Outcomes): a double-blind, randomised placebo-controlled trial. Lancet 2018; 392: 1519-1529.
91. Sattar N, Lee MMY, Kristensen SL, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol 2021; 9: 653-662.
92. Kidney Disease: Improving Global Outcomes (KDIGO) Blood Pressure Work Group. KDIGO 2021 clinical practice guideline for the management of blood pressure in chronic kidney disease. Kidney Int 2021; 99(3S): S1-S87.
93. Kidney Disease: Improving Global Outcomes (KDIGO) Glomerular Diseases Work Group. KDIGO 2021 clinical practice guideline for the management of glomerular diseases. Kidney Int 2021; 100(4S): S1-S276.
94. Kim D, Kotwal S. Reducing cardiovascular risk in people with chronic kidney disease. Medicine Today 2024; 25 (6 Suppl): 3-9.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.