At last, a noninjectable adrenaline device for anaphylaxis management

A needle-free adrenaline nasal spray is now TGA approved for the emergency treatment of anaphylaxis. The new formulation is likely to improve compliance with instructions to administer adrenaline promptly in the event of a severe allergic reaction.
Correction
A correction for this article has been published in the March 2026 issue of Medicine Today and is available here. The online version and the full-text PDF of this article (see link above) have been corrected.
- Adrenaline remains the only recommended first-line treatment for anaphylaxis, yet delayed or absent administration remains common and is associated with poorer outcomes.
- A fear of needles, lack of device availability and incorrect technique are key barriers to the timely use of adrenaline autoinjectors.
- A needle-free intranasal adrenaline spray is now TGA approved and offers a pharmacokinetic and pharmacodynamic profile comparable to that of injectable formulations.
- The nasal spray may be particularly beneficial for children and for patients who are reluctant to carry or use injectable devices.
- By reducing barriers to administration, needle-free adrenaline has the potential to support earlier treatment and improve real-world anaphylaxis management.
Although adrenaline is the only recommended first-line treatment for anaphylaxis management, its underuse is widely reported.1 A number of barriers to its prompt use have been identified. Often, patients do not have the adrenaline autoinjector available to use when they experience an anaphylactic reaction, as many people dislike carrying a needle around. A major barrier is fear of the injection, and so, usage is delayed with detrimental consequences, including increases in biphasic reactions, more prolonged hospitalisations and even death. Incorrect technique is also an issue that has been highlighted.2
A needle-free adrenaline nasal spray for the emergency treatment of anaphylaxis and severe allergic reactions, which has been approved in the USA and Europe, is now TGA approved. The drug consists of three components approved by the Food and Drug Administration:
- adrenaline
- an alkylsaccharide that loosens cell-to-cell connections, resulting in changes to the tight junctions to facilitate absorption
- a unit dose spray that has been shown to be reliable, with a failure rate of less than one in 100,000 users.3
Both the nasal spray device and alkylsaccharide absorption enhancer are already used in a number of intranasally administered medications.
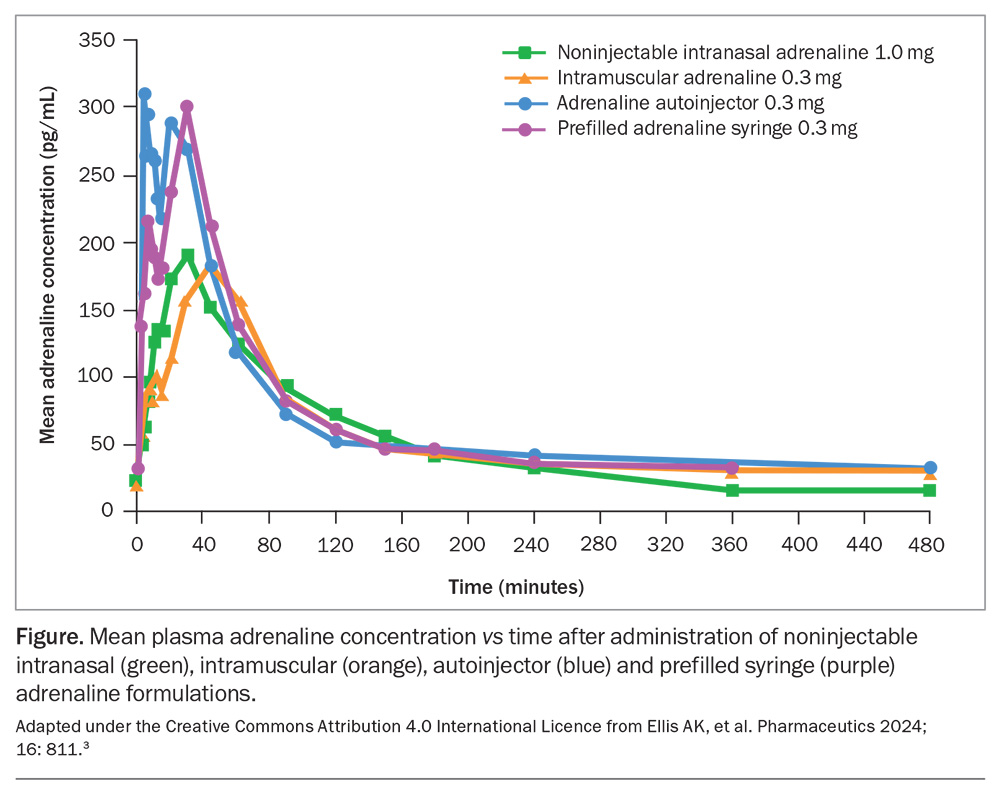
The new needle-free adrenaline spray has demonstrated a pharmacokinetic profile within the range of currently approved injection products. Pharmacodynamic responses are comparable to or better than those seen with injections, with elevations in pulse rate and blood pressure observed within 1 minute of administration, including activation of both the alpha- and beta-adrenergic receptors (Figure). These findings have been reproduced under a variety of nasal conditions, include congestion and rhinorrhoea due to allergic or infectious rhinitis.3 In addition, a dog model of anaphylaxis has demonstrated that intranasal adrenaline is absorbed effectively even in the presence of severe hypotension.4
The needle-free option is particularly attractive for children, as well as for those who fear injections or who wish to travel without the burden of an injectable device. By reducing patients’ hesitation associated with needle-based administration, the nasal spray may encourage earlier use of adrenaline, thereby reducing the risk of progression and complications associated with anaphylaxis.
Indications and administration
The new needle-free adrenaline spray is indicated for prompt treatment of type I allergic reactions, including anaphylaxis, in adults and children. The 2 mg device is recommended for those weighing 30 kg or more, whereas the 1 mg device is recommended for children at least 4 years of age and weighing 15 to 30 kg. Two nasal sprays should be carried at all times. Each carton contains two devices, each individually packaged in blister packs with instructions for use.
As with adrenaline autoinjectors, the needle-free nasal spray should be delivered into one nostril immediately at the first signs of a reaction, and a second dose administered into the same nostril after 5 minutes if symptoms continue or worsen. Emergency medical help should be actioned following use. Visual instructions for use are available in the TGA Product Information.
Side effects
Common side effects of the needle-free adrenaline spray can include headache, nasal congestion, throat irritation and nasal discomfort.
Prescribing information
The needle-free adrenaline spray is TGA approved but not PBS listed at the time of writing this article. The manufacturer has applied for PBS reimbursement for both strengths of the drug under the same criteria as for other prescribed adrenaline products in Australia. The application is currently undergoing review by the Pharmaceutical Benefits Advisory Committee.
In the interim, the drug can be prescribed privately with similar pricing to adrenaline autoinjectors. Prices can vary depending on individual pharmacy price mark-ups from the wholesaler cost. Provision of a specially designed action plan should accompany a prescription.
Conclusion
The needle-free adrenaline nasal spray will be incorporated into anaphylaxis information materials developed by the Australasian Society of Clinical Immunology & Allergy (ASCIA), Allergy and Anaphylaxis Australia and National Allergy Campaign. These include ASCIA anaphylaxis action and first aid plans, ASCIA anaphylaxis guidelines, ‘how-to-use’ instructional videos and other patient resources. By reducing barriers associated with injectable devices, this needle-free adrenaline formulation offers a promising advance that may support improved patient confidence and earlier treatment of anaphylaxis. MT
COMPETING INTERESTS: Professor Katelaris has received consulting fees from Seqirus and participated on the advisory board for Neffy®.
This article is for general information purposes only, and the full Product Information should be consulted before prescribing any of the mentioned medications.
References
1. Prince B, Mikhail I, Stukus DR, et al. Underuse of epinephrine for the treatment of anaphylaxis: missed opportunities. J Asthma Allergy 2018; 11: 143-151.
2. Turner PJ, Jerschow E, Umasunthar T, et al. Fatal anaphylaxis: mortality rate and risk factors. J Allergy Clin Immunol Pract 2017; 55: 1169-1178.
3. Ellis AK, Kasali TB, Callana M, et al. Development of neffy and epinephrine nasal spray for severe allergic reactions. Pharmaceutics 2024; 16: 811.
4. Tanimoto S, Kaliner M, Lockey RF, et al. Pharmacokinetic and pharmacodynamic comparison of epinephrine, administered intranasally and intramuscularly – an integrated analysis. Ann Allergy Asthma Immunol 2023; 130: 508-514.
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.